Abstract
Presented is a series of narrative reviews that summarize published information regarding the effect or potential effect of interfering substances on the accuracy of continuous glucose monitoring (CGM) devices. Whilst drawing together what is currently known regarding this topic, the future direction in this field and clinical implications posed by polypharmacy on CGM performance are considered. This fourth and final in this series of articles reviews the following topics: the benefits and drawbacks of conducting interfering substance studies in vitro versus in vivo; the balance between the cost and complexity of undertaking in vitro or in vivo studies to screen for potential interferences versus the clinical relevance of the findings; clinical factors influencing interferent behavior on CGM performance (pharmacokinetics and polypharmacy considerations); the need for standardization, including consideration of the metrological traceability of CGM readings, establishment of CGM analytical performance metrics and standardized CGM assessment procedures for measuring interferents.
CGM Performance With Interferents: In Vitro Versus In Vivo Considerations
Introduction
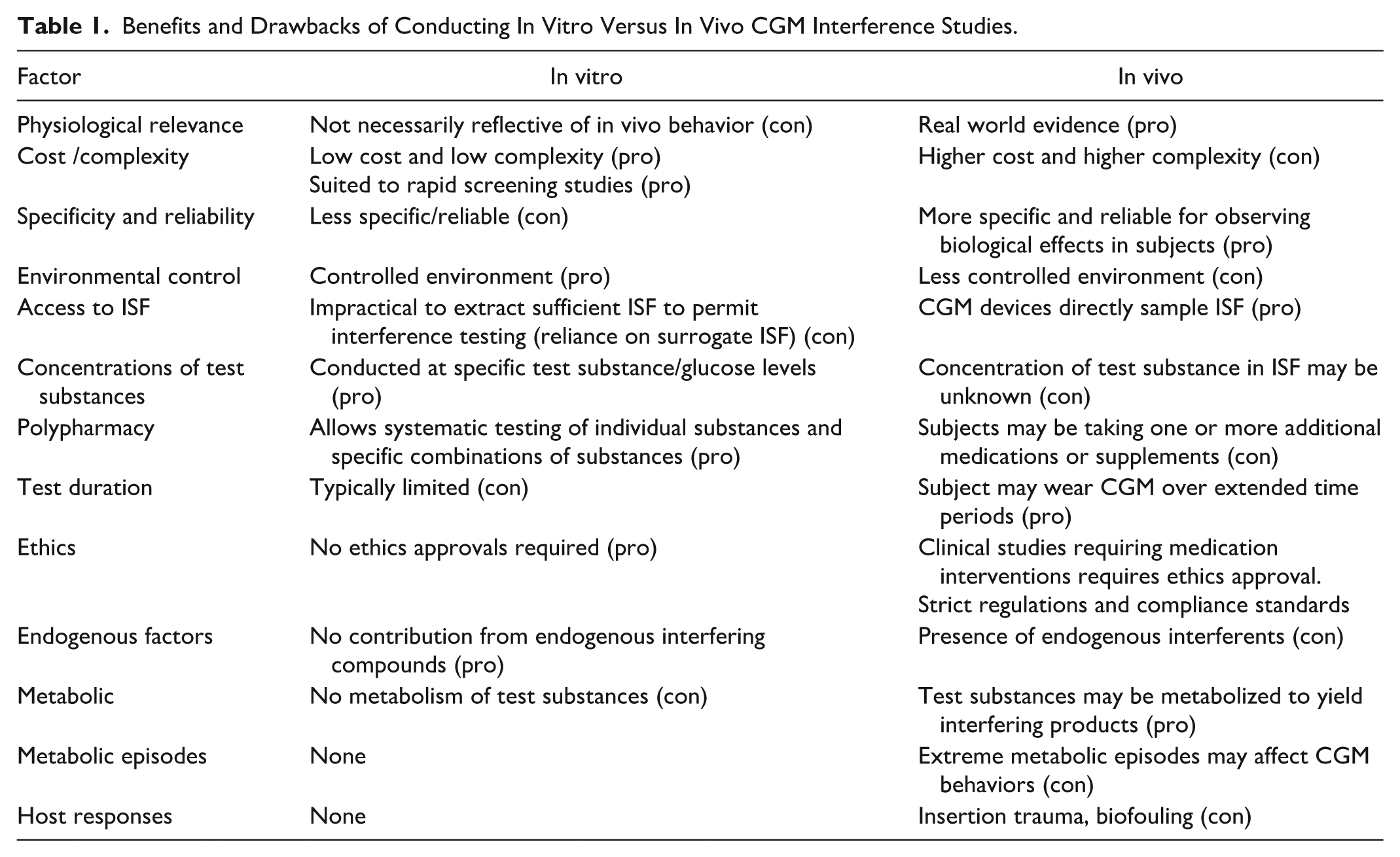
Most in vivo continuous glucose monitoring (CGM) interferent studies to date have compared CGM values, which are measured in interstitial fluid (ISF), with corresponding values from a suitable comparator method, which are measured within extracted blood samples, following administration of a test substance to a subject. Difficulties inherent in studying the in vivo effect of interfering substances on CGM systems are diverse and include challenges in obtaining representative ISF samples for reference glucose and interfering substance measurements, sensor biofouling and host-related factors such as metabolic crises. Consequently, many CGM interferent studies have been conducted in vitro. Whilst such studies are valuable in identifying potential CGM interference risks, their limitations should also be recognized, such as the use of surrogate ISF and questions regarding physiological relevance. Nonetheless, difficulties in obtaining in vivo data means that there is a role for in vitro testing, at least from a perspective of screening and identifying potential interfering substances for further in vivo investigation. Table 1 provides a summary of benefits and drawbacks in conducting interfering substance studies in vitro versus in vivo.
Benefits and Drawbacks of Conducting In Vitro Versus In Vivo CGM Interference Studies.
Practical Limitations in Extracting ISF
Ideally, CGM in vitro assessments should be performed with potential interferents dissolved within an ISF matrix. However, unlike blood glucose monitoring (BGM) studies, in which sufficient volumes of blood may be obtained through venipuncture, extraction of sufficient volumes of ISF remains impractical for conducting comparable bench-based CGM interference studies. 1 Consequently, in vitro CGM testing must rely on surrogate ISF as the test medium, albeit that researchers seek to match key parameters, such as ionic strength and pH with that of native ISF.
Additionally, it would be preferred to measure interfering substances in ISF to ascertain their pharmacokinetic behavior. As described in part 1, ISF is a complex medium, containing multiple components, including solutes, macromolecules and proteins that can readily cross the blood/interstitium barrier.2,3 Consequently, far more is known about the pharmacokinetic behavior of medications and other substances in blood. The in vivo microdialysis procedure of Basu et al 4 for measuring levels of interfering substances in ISF demonstrated significant differences in concentrations and elimination rates of the test substances between blood plasma and ISF (described in part 3 of this review). Thus recommendations provided by regulators or professional bodies, such as CLSI, regarding the recommended maximum therapeutic/test levels of exogenous and endogenous substances in blood may require reassessment to provide appropriate levels for ISF.
There is also potential for substances that do not display interfering effects in vitro to be metabolized in vivo to yield interfering intermediates or end-products. For example, the aspirin (acetylsalicylic acid) metabolites, gentisic acid and salicylic acid, have each been associated with CGM interference effects, as described in part 3 of this review.5-7
Electrochemical Sensor Fouling
There is little known about the extent to which substances can foul or passivate the electrode surfaces of electrochemical CGM systems in vivo. However, it has been observed that some substances can apparently foul or passivate CGM electrode surfaces when evaluated in vitro under specific test conditions. Pfützner et al 5 have reported apparent in vitro electrochemical fouling effects for the G6 system when exposed to dithiothreitol, gentisic acid, L-cysteine and mesalazine. Unlike BGM devices, in which a single-use test strip is exposed to only a one-time interferent challenge, CGM devices are typically designed to work for 7-15 days (365 days in the case of the optical Senseonics Eversense 365 CGM), raising the risk that the repeated dosing of a medication or dietary supplement, or an intermittently or chronically elevated level of an endogenous interferent, may progressively incapacitate the electrode during the sensor wear lifetime. This potentially slower, but cumulative, effect and subsequent loss in CGM sensitivity, if present, may not be apparent to the individual using the system.
As a further complication, many classical bench tests are performed on bare (unmodified) electrodes, where passivation effects may be most pronounced, whereas marketed CGM devices incorporate membranes designed to reduce the flux of potential fouling agents from the surrounding medium into the sensor filament structure, whilst supporting the diffusion of the mediating species from the active reagent layer to the electrode surface (described in part 2 of this review). Thus, in vitro behavior of a passivating agent may not translate into a similar in vivo effect. Consequently, any in vitro test regime for interferents suspected of causing sensor fouling effects, whether electrochemical or otherwise, should consider protocols in which individual CGM designs are repeatedly challenged with these substances to better simulate real world conditions.
Biofouling and Other Host Responses to In Vivo CGM Wear
CGM sensors used in bench studies are not subject to the same conditions as when assessed in vivo. The multi-day or multi-week in vivo wear of systems in which the sensing portion of the device is positioned sub-dermally, makes these devices more prone to localized effects such as tissue trauma exacerbated by continued body movement, localized immune responses and the risk of biofouling/tissue formation over the sensing element that may induce a longer-term glucose-specific signal deterioration. All these factors of have the potential to influence the observed in vivo behaviors of interfering substances (for a more detailed consideration of host-mediated immune responses on CGM accuracy, the reader is referred to Joseph et al 8 ). Senseonics have sought to mitigate inflammation effects by incorporating a dexamethasone eluting silicone collar within their implantable Eversense CGM models.
Electrolyte Related Factors That May Impact CGM Measurements
In classical bench electrochemical studies, the choice and concentration of electrolyte species used in the supporting medium can influence interactions between the analyte and electrode. 9 The choice of supporting medium can also be influential in assays dependent on optical transduction, with electrolyte factors being known to influence the kinetics of analyte-enzyme and analyte-ligand (e.g., boronic acid) interactions. This is relevant in that blood and ISF contain many electrolyte species, including sodium, potassium, chloride, magnesium, calcium, phosphate and bicarbonates, with certain of these species providing buffering capacity to biological fluids.
Referring specifically to minimally invasive electrochemical CGMs, pH (a measure of hydrogen ion activity) has the potential to influence both the kinetics of the enzyme-glucose reaction and the transduction process. For example, the biocatalytic oxidation of glucose by the glucose oxidase enzyme (GOx) yields gluconic acid, resulting in a local pH change, 10 a factor recognized by some CGM manufacturers. For example, Dexcom CGMs incorporate an electrolyte domain adjacent to the electrode, designed to control the movement of ionic species, including hydrogen ions, in the vicinity of the electroactive surface as discussed in part 2 of this review. 11
The multi-membrane design is common to commercial first-generation electrochemical CGMs, as described in part 2 of this review, and serves as a protective environment to the underlying platinum electrode. Toghill and Compton note that under physiological conditions, bare platinum electrodes may be poisoned by a wide range of chemical species. 12 Chloride anions, for example, are known to strongly chemisorb to platinum surfaces, albeit more so under acidic conditions, rendering the surface inaccessible to other substances. 13 The use of charged membranes in an acidic medium has been found to reduce the chloride ion effect by influencing mass transfer rates to the underlying electrode, but proved less effective under conditions of prolonged exposure. 14 Other organic compounds can similarly reduce platinum electroactivity, including uric acid, amino acids, and proteins, as well as many phenolic and thiolated compounds. 12 It is therefore possible that such species may migrate through the multi-layered membrane arrangements typically employed within current CGM designs, leading to signal deterioration. Issues of sensor drift, whether caused by sensor poisoning or other factors, have been recognized by CGM manufacturers and researchers who have proposed algorithmic corrections to minimize impacts on system performance.15-17
Extreme Metabolic Episodes and Potential Impacts on CGM Performance
Extreme metabolic episodes can result in significant changes in the levels and overall balance of electrolytes in the body. Diabetic ketoacidosis (DKA) is a serious condition seen in people with diabetes, characterized by rapid and significant decreases in blood (and hence ISF) pH and bicarbonate levels coupled to osmotic diuresis, in which significant amounts of electrolytes, including potassium, magnesium and phosphate, are excreted in the urine. It is therefore reasonable to consider if such electrolyte imbalances can influence CGM performance. CGM accuracy during episodes of DKA present a mixed picture, with some studies18,19 suggesting no effect on CGM accuracy, but others observing a reduction in accuracy depending on the severity of the DKA episode. 20 No reports to date have been identified regarding CGM performance during episodes of hyperglycemic hyperosmolar syndrome, another acute complication of diabetes characterized by osmotic diuresis and that ultimately results in severe intracellular dehydration. However, glucose levels during such episodes can often exceed 720 mg/dL (40 mmol/L), well above the claimed upper limit of commercial CGM systems. 21
Lactic acidosis, another type of metabolic acidosis, is a condition characterized by a decrease in blood and ISF pH. This condition may be caused by number of disorders relating to the delivery and consumption of oxygen in body tissues. Lactate accumulation can also occur during periods of intense exercise. Whilst several studies have evaluated the general impact of exercise on CGM accuracy, for example, by examining the effect of rapid glucose changes, 22 others have also sought to equate exercise-induced lactate levels with CGM performance. Bally et al, 23 studying the accuracy of the G4 CGM during different exercise regimes in a cohort with type 1 diabetes, found a significant change in both lactate (7.3 vs 2.6 mmol/l and pH (7.27 vs 7.38) in an intermittent high intensity exercise group versus a moderate intensity exercise control group, without a significant change in device accuracy. Similarly, Da Prato et al 22 measured temporal changes in plasma lactate levels within 2 different exercise groups, but did not report any significant correlations to CGM accuracy.
Marics et al, 24 postulating that insufficient peripheral perfusion could lead to temporal shifts between glucose levels in blood and ISF, evaluated the effect of serum pH, selected in this study as a marker of metabolic/respiratory insufficiency, and serum lactate, a marker for tissue hypoxia, on CGM accuracy within a pediatric intensive care unit population. Neither marker affected CGM accuracy, nor did hematocrit, selected as a marker of hemoconcentration/blood viscosity, or serum potassium, selected as a marker for renal hypoperfusion/hypofiltration.
Electrolyte imbalance is not restricted to only metabolic or respiratory abnormalities. As discussed in part 3, Moscardó et al 25 found a significant difference in CGM accuracy during periods of alcohol consumption, an effect attributed to ethanol-induced changes in pH. Extreme dehydration will also affect electrolyte balance. 26
Clinical Implications of Interfering Substances on CGM Performance
Pharmacokinetics
Whilst this review has sought to collate and discuss interfering substances based on their effect upon the in vitro and in vivo performance of the more widely commercially available CGMs, clinically meaningful interpretation of the findings is complicated by many factors. For example, the concentration and distribution of a medication within the human body at a given time will be driven by its dosage, mode of delivery (e.g., oral, intravenous, transdermal) and pharmacokinetic behavior, including the liberation of the active compound from its delivery formulation, its bioavailability and absorption properties, the distribution and metabolism of the substance in the body, and the rate of elimination and excretion from the body (Figure 1).
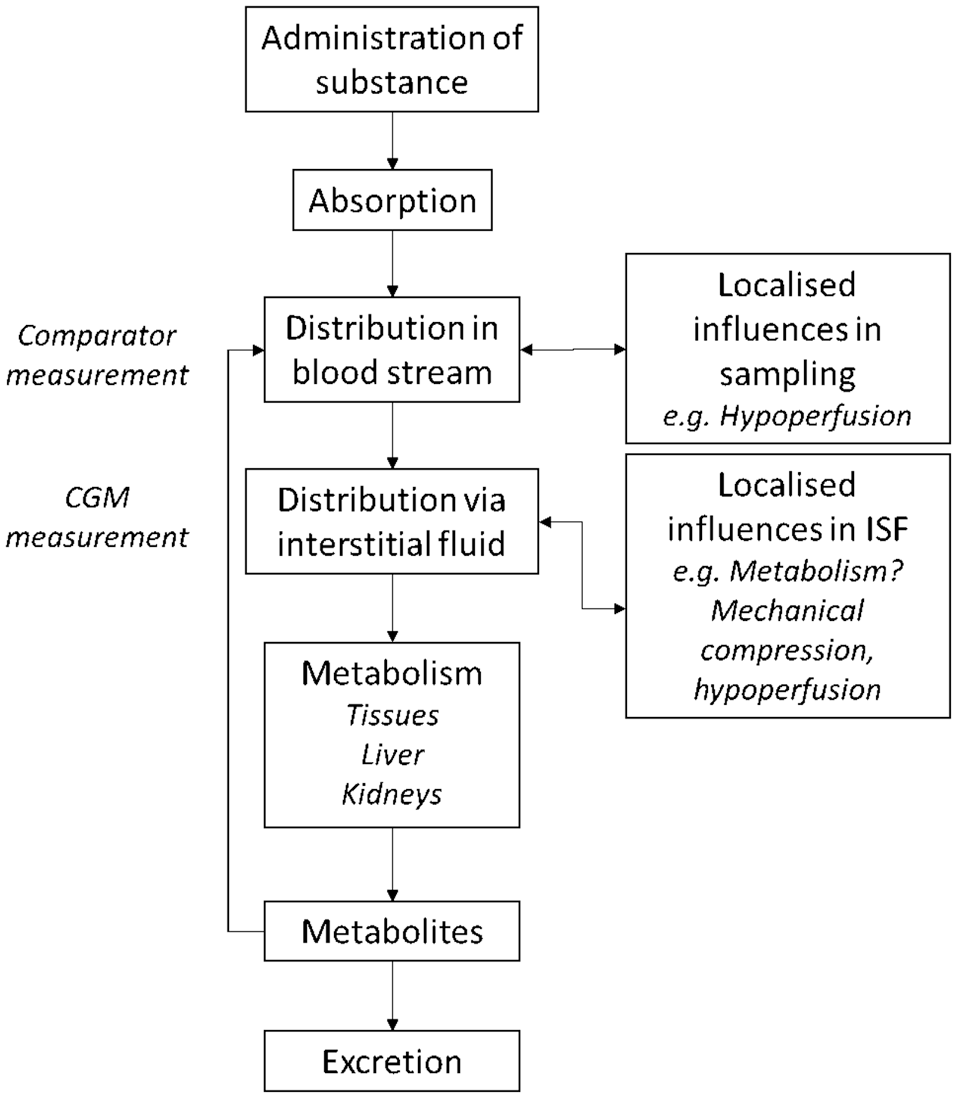
Simplified pharmacokinetic pathway of an administered substance, with distribution via the blood stream and interstitial fluid compartment. Active metabolic products may recirculate before elimination from the body. CGM comparator measurements are blood-based, a distinct compartment to actual CGM measurements, performed in interstitial fluid.
Patient-specific factors may also influence pharmacokinetic behaviors, such as renal/hepatic function, genetics, gender and age. Importantly, for CGM outcome studies and drug studies that use CGM devices to measure glucose changes, clinical study designs typically exclude critically ill or less healthy subjects, including individuals with significant liver or kidney dysfunction.27-30 This leads to the under-representation of subjects who may have complications with respect to the metabolism of medications and other substances. Such morbidities raise the risk of significantly altered pharmacokinetic behaviors within these vulnerable populations, such as prolonged and/or elevated circulatory levels of a substance. This, in turn, raises fundamental questions as to how broadly representative the interference labeling information is for CGM systems.
In summary, as many commercial CGMs operate within the interstitial compartment, the mechanisms and kinetic behaviors that govern transfer and partitioning of interfering substances from the blood to the ISF compartment have the potential to significantly influence ISF-based CGM readings on a temporal basis.
Active Metabolites
Part 3 of this review identified substances that may be metabolized to yield compounds with the potential to interfere with CGM readings. Given the substances identified are electroactive, this issue is most relevant to CGMs based on the first-generation design of electrochemical biosensors. Examples include the metabolism of acetylsalicylate (aspirin) to salicylic acid and gentisic acid. The metabolism of the noncatecholamine atenolol indicates evidence of an in vivo interfering effect not replicated in vitro.4,31 The main catecholamines metabolites may retain their electroactive catechol moiety and thus retain their electroactive nature as they are metabolized in the body.
Polypharmacy
Polypharmacy has been defined as the consumption of multiple medications and/or the excessive use of medications. Evidence would suggest that this issue applies to many people with diabetes who have adopted the use of CGM devices. 32 For example, in 2014, Bauer and Nauck reported that people with type 1 and type 2 diabetes were prescribed an average 5.5 and 8.4 medications per day respectively within a specialized diabetes clinic in Germany. 33 Both Grady et al 34 and Phillips et al, 35 evaluating clinical datasets obtained as part of a manufacturer’s ongoing postmarket surveillance of BGM products, noted an average of 6.5 and 6.9 prescribed medications within populations of mixed type 1/type 2 diabetes. Some subjects were found to be taking >12 medications (830 subjects, 473 different medications; 1023 subjects, 535 medications). Hauss et al, 36 discussing the accuracy of BGM systems, also comment on complications arising from polypharmacy within populations with diabetes, noting that the entry of new diabetes medications to the market increases the likelihood of replacement of current drugs by novel therapeutic options, leading to more unknown interactions.
The concern regarding polypharmacy in CGM users occurs when two or more medications, that individually may be considered noninterfering, when taken together, introduce a more significant systematic bias to CGM readings due to the stacking of the individual substance biases. Whilst this issue is not new to the diabetes management field (being common to BGM systems) the increasing integration of CGMs into automated insulin delivery systems, and the use of apps and algorithms that predict future CGM readings, suggests that focusing only on individual substance specific interference effects may not be sufficiently robust, and evidence on interference stacking should be pursued to identify additional risks of sub-optimal insulin dosing by AID systems, based on inaccurate CGM values.
Interfering behaviors are not just limited to prescribed medications since over-the-counter (OTC) medications (e.g. cold and flu remedies) and nutritional supplements (e.g. vitamin C, antioxidant tablets) are widely used within the diabetes and wider populations. 34 These substances may not necessarily be recorded on a patient’s medical record, further complicating the interpretation of the effect of exogenous substances on the performance of CGM and other devices. As this review has sought to show, an interfering substance may immediately or gradually alter the magnitude of a CGM reading during prolonged sensor exposure. Furthermore, a combination of medications may affect a CGM in an unpredictable way, even within an in vitro setting. Current guidelines, whether BGM or CGM, do not recommend the testing of medication and/or other substance combinations, leaving this area understudied.
Standardization
The Need for Standardization of CGM Performance
The International Federation of Clinical Chemists and Laboratory Medicine (IFCC) working group on Continuous Glucose Monitoring (WG-CGM) have been tasked with proposals for the standardization of CGM performance, traceability of CGM readings to methods of highest metrological order, the establishment of CGM analytical performance metrics and the creation of standardized CGM assessment procedures. 37 The remit of WG-CGM is correspondingly broad and as such does consider general recommendations for reporting clinical CGM performance evaluations, including the need to define inclusion and exclusion criteria for possible interfering substances. 38
Reference Methodology
The WG-CGM has made the case for a more standardized reference method approach, whether to assess clinical accuracy, or to better assess the effect of potential interfering substances. 39 However, due to challenges in extracting ISF in sufficient quantities, it is not possible to produce commutable reference materials and establish direct metrological traceability between CGMs designed to work in ISF and higher order reference measurement procedures (RMPs). Correspondingly, comparator methods used to assess CGM performance are reliant on the measurement of glucose in capillary or venous blood.
Analysers that measure glucose in blood are required to be traceable to higher order RMPs that also measure glucose in blood. This requires the establishment of analytical performance specifications (APSs) which, whilst applicable to laboratory-based analysers, are equally applicable to any device that measures glucose in blood, including BGM systems. Thus, capillary blood BGM measurements have been used to generate supplemental accuracy data outside of the clinic in many CGM studies.40,41 Furthermore, there is also a recent example of the European regulatory clearance of a CGM, the Accu-Chek SmartGuide, in which the manufacturer’s own capillary BGM system served as the sole comparator method for clinical accuracy assessment (which similarly demands the establishment of a robust traceability chain with the same APS requirements as more established comparators, irrespective of whether the measurements are performed in a clinic or nonclinic location). 42 Furthermore, any test method designed to evaluate the performance of a CGM system should be of an appropriate design in terms of rate of change of glucose and claimed glucose range 43 and ideally should itself be minimally impacted by the presence of interfering substances.
From the perspective of quantifying the effects of interfering substances on CGM performance, this disparity in measurement compartments (ISF versus venous or capillary blood) means that CGM devices operate in a matrix of a different compositional make-up to blood-based reference measurements. Therefore, pharmacokinetic factors such as the rate of transfer of interfering substances from blood to ISF and the rate of metabolism/elimination of substances within the different compartments, may impact the observed interfering effects between the CGM and reference method (Figure 2).
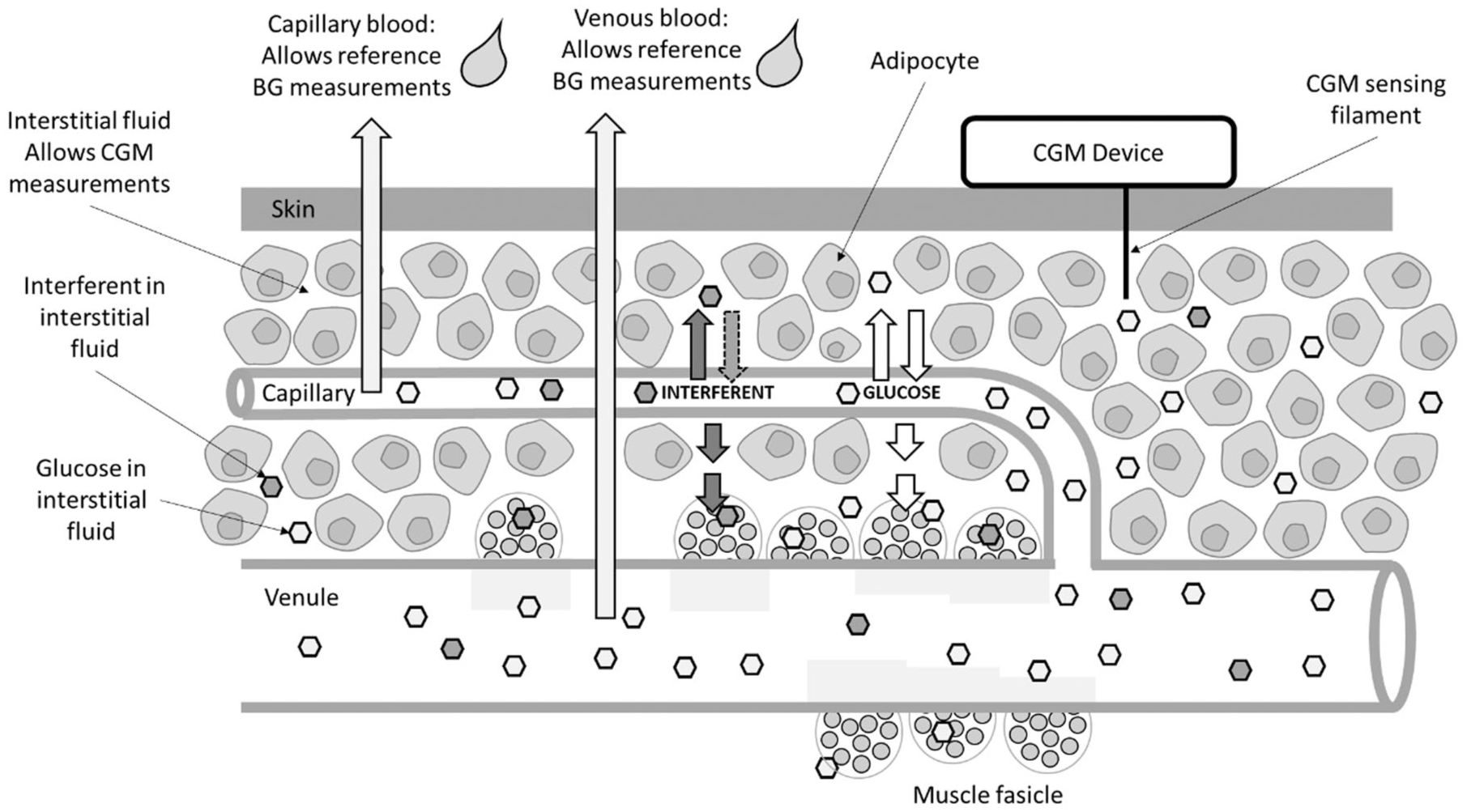
CGM systems perform readings in interstitial fluid (ISF), whilst reference methods measure blood glucose (BG), due to challenges in extracting sufficient ISF. Depending on relative concentrations, glucose may diffuse into or out of the interstitial space and capillaries and may also transition to adipocytes and muscle. Interfering substances may similarly distribute between the different compartments, with absolute concentrations depending on the pharmacokinetic properties, site of action and elimination pathway of the substance. Thus, glucose levels as measured by a reference method in blood may differ from ISF measurements performed by CGM devices.
Different reference methodologies, whilst being traceable to the primary higher order method, can exhibit different mean relative biases that may influence the reported accuracy of a glucose measurement device. A comprehensive evaluation by Freckmann et al 44 compared the clinical accuracy of BGM systems against a glucose oxidase-based and hexokinase-based comparator and showed significant systematic differences between mean bias, linear regression and mean absolute relative difference (MARD) values, prompting a call for measurement procedure harmonization. The same researchers have also proposed a methodology by which a recalibration approach can be retrospectively applied to CGM datasets to reduce the impact of choice of reference method. 45 Furthermore, should a CGM device have a similar mechanism of glucose measurement to that of the selected reference method, it may be supposed that a lesser interfering effect will be observed compared to a reference methodology that responds differently in the presence of a given interfering substance. This may be of benefit in that, whilst reference method standardization is key to allow more comparative assessments of CGM performance, observed differences between CGM readings and those obtained from different reference instruments may aid in identifying and quantifying interference effects.
Standardization of In Vitro CGM Interferent Assessments
Recommended test concentrations for the evaluation of many clinically relevant interfering compounds in blood have been established by CLSI46,47 and form a basis for in vitro BGM interferent testing. Similar guidelines do not exist for CGM systems, in large part due of the difficulties in extracting ISF in sufficient quantities to establish clinically relevant test concentrations in this matrix. In the absence of actual clinical data, many researchers work on the basis that the level of a substances in blood will serves as a surrogate indicator of the corresponding level of that substance in ISF. As described earlier, pharmacokinetics, and the mode of interferent delivery (oral, topical, injection et cetera) will mean that these assumptions may not be valid. Whilst practically challenging, efforts to better understand therapeutically relevant levels of substances in ISF will aid improved understanding of the effect of interfering substances on CGM performance. The microdialysis investigations of Basu et al 4 are pioneering in this respect. In the meantime, an agreed standardization of the test levels of potentially interfering substances, either based on, or inspired by those established by CLSI would be beneficial. Additionally, a common methodology for the practical evaluation of in vitro CGM performance in the presence of interfering substances should be considered, whether flow cell based, or other readily adoptable design.
Postmarket Surveillance
Both the Diabetes Technology Society and regulators advocate programs in which the manufacturer plays a more active role in assessing product performance on intended user populations once a product has been cleared for sale and has entered the market.48-51 Such initiatives offer the potential for identification of currently unsuspected or newly marketed interfering substances and possible medication and/or supplement combinations that may impact on BGM or CGM device performance. The implementation of robust postmarket surveillance programs should help manufacturers identify potential interfering substances. An example is hydroxyurea, which was identified as an interferent in CGMs of the first generational electrochemical biosensor design due to clinicians observations of higher-than-expected glucose readings in a hospitalized patient. 52
Summary
The effect of potentially interfering substances on commercial CGM devices is an evolving area of research. Most systems are minimally invasive, with the glucose-sensing element sited within the ISF compartment under the skin. These systems require a sensing element to identify glucose within the complex ISF sample medium, coupled to a transducer to convert the glucose recognition process into a measurable, quantifiable signal. As such, CGM accuracy is compromised by substances that interact with the sensor and/or transducing mechanism. This issue has long been recognized in the BGM field, where approved international standards and guidelines exist to quantify such interfering effects, and comprehensive requirements exist regarding system testing within an in vitro setting, albeit only with single agents, not combinations or so-called cocktails of agents. The issue is more complicated in CGM systems in that such standards and guidelines do not yet fully exist and that CGM systems, by their nature, generate high frequency glucose readings, over an extended time. Additionally, the challenges of collecting sufficient ISF for analyte determination by an independent comparator method mean that CGM readings obtained during clinical studies are paired to readings from the designated comparator device, which are generally obtained via venous draw. This gap is being addressed by the IFCC, who have created the CGM working group to propose appropriate performance metrics, study designs and assessment methodologies, and to consider the metrological traceability issue.
A combined summary of the chemical substances identified as influencing the readings of current, and some previously commercially available CGM systems as identified within this review series, is presented in Table 2.
Chemical Substances That Have Been Identified as Possible Interferents Within Currently or Previously Commercially Available CGM Systems.
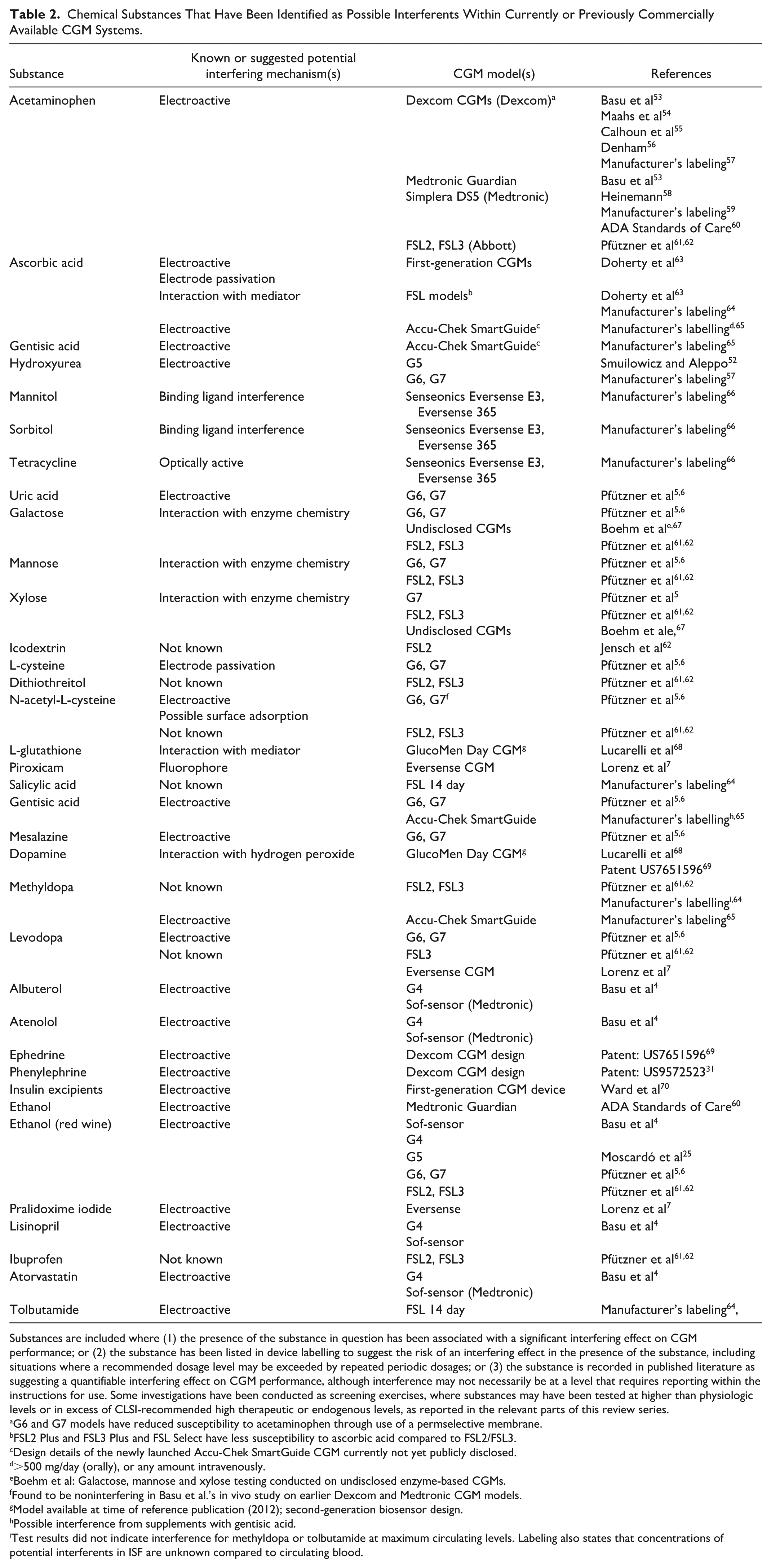
Substances are included where (1) the presence of the substance in question has been associated with a significant interfering effect on CGM performance; or (2) the substance has been listed in device labelling to suggest the risk of an interfering effect in the presence of the substance, including situations where a recommended dosage level may be exceeded by repeated periodic dosages; or (3) the substance is recorded in published literature as suggesting a quantifiable interfering effect on CGM performance, although interference may not necessarily be at a level that requires reporting within the instructions for use. Some investigations have been conducted as screening exercises, where substances may have been tested at higher than physiologic levels or in excess of CLSI-recommended high therapeutic or endogenous levels, as reported in the relevant parts of this review series.
G6 and G7 models have reduced susceptibility to acetaminophen through use of a permselective membrane.
FSL2 Plus and FSL3 Plus and FSL Select have less susceptibility to ascorbic acid compared to FSL2/FSL3.
Design details of the newly launched Accu-Chek SmartGuide CGM currently not yet publicly disclosed.
>500 mg/day (orally), or any amount intravenously.
Boehm et al: Galactose, mannose and xylose testing conducted on undisclosed enzyme-based CGMs.
Found to be noninterfering in Basu et al.’s in vivo study on earlier Dexcom and Medtronic CGM models.
Model available at time of reference publication (2012); second-generation biosensor design.
Possible interference from supplements with gentisic acid.
Test results did not indicate interference for methyldopa or tolbutamide at maximum circulating levels. Labeling also states that concentrations of potential interferents in ISF are unknown compared to circulating blood.
Whilst not a substitute for in vitro or in vivo testing, performing chemical-structure assessments to identify potentially interfering substances can be performed at minimal cost. This approach should form part of any strategy to risk-assess the effect of medications, especially those new to market, and medications within a particular compound class where it has been shown other family members have elicited an interfering effect. The catecholamine and noncatecholamine classes of compound are a particular case in point.
This review series further demonstrates that the issue of interfering substances on CGM performance is widely recognized, but that there are significant gaps in knowledge, especially with respect to the impact of combinations of multiple prescribed or OTC medicines and/or food supplements that people take daily. The ever-increasing wear time of CGMs (typically now 10 to 15 days) may also compound interfering effects, whereas the very few in vivo studies to date only replicated effects over a few hours or a single day. A more systematic approach, mimicking CGM model wear times, is required to assess the effect of medications and other substances on performance, from both an individual substance and polypharmacy perspectives. The onus must be on the manufacturer to lead in this area, such as proactively running risk assessments and evaluating their specific CGM design with respect to the (bio)chemical nature of potentially interfering substances. There is some evidence that manufacturers are accepting this challenge, as evidenced by the de novo G6 and G7 CGMs, as well as the FSL2 Plus and FSL3 Plus CGMs, which have included design modifications that have partially addressed performance issues arising from certain interfering compounds (although CGM manufacturers are not currently mandated by regulators to disclose the results of interference testing for nonlabeled substances). This is especially important when viewed from the perspective of automated insulin delivery systems, in which the output of a CGM device is used as an input in calculating the delivery of insulin to people with diabetes. Given the ethical challenges of administering complex pharmaceutical combinations in clinical trials, it is evident that a combined approach of in vitro bench testing of risk-assessed compounds, coupled with manufacturer-led postmarket surveillance activities on patient populations with diabetes, is needed to identify and quantify potentially interfering substance effects before harm is caused by an interfering substance to a person using a CGM device.
Footnotes
Acknowledgements
The author in indebted to Dr Elizabeth Holt MD, FACE, Head of Global Clinical, Medical and Safety; Dr Mike Grady, Director of Clinical Affairs; and Mr Brian Heald, Senior Vice President, Product, for their invaluable suggestions and in-depth review of this manuscript.
Abbreviations
APS, analytical performance specification; BG, blood glucose; BGM, blood glucose monitor; CGM, continuous glucose monitor; CLSI, Clinical and Laboratory Standards Institute; DKA, diabetic ketoacidosis; FSL, FreeStyle Libre; G4, Dexcom G4 CGM; G6, Dexcom G6 CGM; G7, Dexcom G7 CGM; IFCC, International Federation of Clinical Chemists; ISF, interstitial fluid; MARD, mean absolute relative difference; OTC, over-the-counter; RMP, reference measurement procedure; WG-CGM, working group on continuous glucose monitoring.
Declaration of Conflicting Interests
The author declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: SJS is an employee of LifeScan Scotland Ltd.
Funding
The author disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: SJS is a salaried employee of LifeScan Scotland Ltd.
