Abstract
Continuous glucose monitoring (CGM) has been shown to improve glycemic control and self-monitoring, as well as to reduce the risk of hypoglycemia. Integrated CGM (iCGM) FDA-cleared systems with published performance data are established nonadjunctive and accurate CGM tools that can directly inform decision-making in the treatment of diabetes (i.e., insulin dosing). Studies have assessed accuracy and safety data of CGMs that were eventually cleared for iCGM by the FDA and that informed the recommendation for their nonadjunctive use. Subsequent robust clinical trials and real-world studies demonstrated clinical effectiveness with improvements in a range of patient outcomes. In recent years, a number of non-iCGM-approved CGM devices have entered the market outside the United States worldwide. Some of these non-iCGM-approved CGM devices require additional user verification of blood glucose levels to be performed for making treatment decisions, termed adjunctive. Moreover, in many non-iCGM-approved CGM devices, accuracy studies published in peer-reviewed journals are scarce or have many limitations. Consequently, non-iCGM-approved CGM devices cannot be automatically perceived as having the same performance or quality standards than those approved for iCGM by the FDA. As a result, although these devices tend to cost less than iCGMs that carry FDA clearance and could therefore be attractive from the point of view of a health care payer, it must be emphasized that evaluation of costs should not be limited to the device (such as the usability preference that patients have for nonadjunctive sensors compared to adjunctive sensors) but to the wider value of the total benefit that the product provides to the patient.
Keywords
Introduction
Diabetes mellitus (DM) is a public health care problem that is now epidemic in scale worldwide. 1 Glucose monitoring is an important part of self-management of diabetes. Of note, although self-monitoring of blood glucose is associated with an improvement in control of HbA1c, the information it provides is limited, and people living with diabetes may be unaware of their glucose levels or trends, leading to an increased risk of hypoglycemic events.2-4 In contrast, continuous glucose monitoring (CGM) has been shown to improve glycemic control, as well as to reduce the risk of both severe and nonsevere hypoglycemia.5-11 In fact, CGM supports people with DM to optimize their therapy, diet, and activity, facilitating improved HbA1c, increased time in target range of 70 to 180 mg/dL, reduced time below range (<70 mg/dL), and improved Glycemia Risk Index scores.5-11 A CGM measures interstitial glucose, which correlates well with plasma glucose; trend arrows provide useful information about whether glucose levels are rising or falling. CGM devices can be divided into two groups, “adjunctive” devices, which require measurement of glucose levels through concomitant use of blood glucose meters for adjusting DM therapy (i.e., insulin dose or treatment of hypoglycemia), and “non-adjunctive” devices, which are not subject to these requirements.12-14
Many but not all CGM systems are nonadjunctive. Nonadjunctive systems do not require confirmation by stand-alone blood glucose monitoring (BGM) before making treatment decisions. The benefits of nonadjunctive CGM systems are adequate accuracy deemed to be sufficient by a regulatory agencies (the United States Food and Drug Administration [US FDA], the European Union, and other comparable regulatory agencies in other countries) to not require the inconvenience of blood glucose confirmation. A nonadjunctive CGM (compared to an adjunctive CGM) offers greater convenience, less pain, less blood waste, and an opportunity to see glucose readings without having to prick the finger to obtain a blood sample.6,14,15
An integrated continuous glucose monitoring system (iCGM) is intended to automatically measure glucose in bodily fluids continuously or frequently for a specified period of time. Integrated CGM systems are designed to reliably and securely transmit glucose measurement data to digitally connected devices, including automated insulin dosing systems and are intended to be used alone or in conjunction with these digitally connected medical devices for the purpose of managing a disease or condition related to glycemic control. This category of CGM performance specified by the U.S. FDA is the strictest accuracy standard defined by any regulatory agency in the world. 16
In this review, quality standards of CGM systems meeting iCGM criteria required by the FDA to be approved for clinical use in the United States were compared to those of other systems, particularly non-iCGM-approved CGM devices that have recently come onto the market outside the United States.15,17-23
Importance of an iCGM
Table 1 contains the iCGM special control performance requirements. It is a worthy accomplishment for a CGM to achieve this designation. Currently, the only CGMs which do meet those requirements are Abbott FreeStyle Libre 2, Abbott FreeStyle Libre 2 Plus, Abbott FreeStyle Libre 3, Abbott FreeStyle Libre 3 Plus, Dexcom G6, and Dexcom G7.15,17-19 Achieving clearance from FDA as non-iCGM puts a device into a level-3 category, whereas clearance as an iCGM puts it into a level 2 category. Food and Drug Administration defines a class 2 device as moderate to high risk and class 3 as high risk. We believe that the lesser amount of risk from using an FDA-cleared iCGM compared to using an FDA-cleared non-iCGM defines the important advantage of using an iCGM product rather than a FDA-cleared non-iCGM. 16
iCGM Special Control Performance Requirements.

Source: Table constructed with data from FDA. 16
Abbreviations: iCGM, integrated continuous glucose monitoring system; LCL, lower confidence limits; ROC, glucose rate of change.
(a)
(b)
(1) Design verification and validation must include the following:
(i) Robust clinical data demonstrating the accuracy of the device in the intended use population.
(ii) The clinical data must include a comparison between iCGM values and blood glucose values in specimens collected in parallel that are measured on an FDA-accepted laboratory-based glucose measurement method that is precise and accurate and that is traceable to a higher order (eg, an internationally recognized reference material and/or method).
(iii) The clinical data must be obtained from a clinical study designed to fully represent the performance of the device throughout the intended use population and throughout the measuring range of the device.
(iv) Clinical study results must demonstrate consistent analytical and clinical performance throughout the sensor wear period.
(v) Clinical study results in the adult population must meet the following performance requirements:
(A) For all iCGM measurements less than 70 mg/dL, the percentage of iCGM measurements within ±15 mg/dL of the corresponding blood glucose value must be calculated, and the lower one-sided 95% confidence bound must exceed 85%.
(B) For all iCGM measurements from 70 to 180 mg/dL, the percentage of iCGM measurements within ±15% of the corresponding blood glucose value must be calculated, and the lower one-sided 95% confidence bound must exceed 70%.
(C) For all iCGM measurements greater than 180 mg/dL, the percentage of iCGM measurements within ±15% of the corresponding blood glucose value must be calculated, and the lower one-sided 95% confidence bound must exceed 80%.
(D) For all iCGM measurements less than 70 mg/dL, the percentage of iCGM measurements within ±40 mg/dL of the corresponding blood glucose value must be calculated, and the lower one-sided 95% confidence bound must exceed 98%.
(E) For all iCGM measurements from 70 to 180 mg/dL, the percentage of iCGM measurements within ±40% of the corresponding blood glucose value must be calculated, and the lower one-sided 95% confidence bound must exceed 99%.
(F) For all iCGM measurements greater than 180 mg/dL, the percentage of iCGM measurements within ±40% of the corresponding blood glucose value must be calculated, and the lower one-sided 95% confidence bound must exceed 99%.
(G) Throughout the device measuring range, the percentage of iCGM measurements within ±20% of the corresponding blood glucose value must be calculated, and the lower one-sided 95% confidence bound must exceed 87%.
(H) When iCGM values are less than 70 mg/dL, no corresponding blood glucose value shall read above 180 mg/dL.
(I) When iCGM values are greater than 180 mg/dL, no corresponding blood glucose value shall read less than 70 mg/dL.
(J) There shall be no more than 1% of iCGM measurements that indicate a positive glucose rate of change greater than 1 mg/dL per minute (/min) when the corresponding true negative glucose rate of change is less than −2 mg/dL/min as determined by the corresponding blood glucose measurements.
(K) There shall be no more than 1% of iCGM measurements that indicate a negative glucose rate of change less than −1 mg/dL/min when the corresponding true positive glucose rate of change is greater than 2 mg/dL/min as determined by the corresponding blood glucose measurements.
(vi) Data demonstrating similar accuracy and rate of change performance of the iCGM in the pediatric population as compared to that in the adult population, or alternatively a clinical and/or technical justification for why pediatric data are not needed, must be provided and determined by FDA to be acceptable and appropriate.
(vii) Data must demonstrate that throughout the claimed sensor life, the device does not allow clinically significant gaps in sensor data availability that would prevent any digitally connected devices from achieving their intended use.
(2) Design verification and validation must include a detailed strategy to ensure secure and reliable means of iCGM data transmission to provide real-time glucose readings at clinically meaningful time intervals to devices intended to receive the iCGM glucose data.
(3) Design verification and validation must include adequate controls established during manufacturing and at product release to ensure the released product meets the performance specifications as defined in paragraphs (b)(1) and (b)(2) of this section.
(4) The device must demonstrate clinically acceptable performance in the presence of clinically relevant levels of potential interfering substances that are reasonably present in the intended use population, including but not limited to endogenous substances and metabolites, foods, dietary supplements, and medications.
(5) The device must include appropriate measures to ensure that disposable sensors cannot be used beyond its claimed sensor wear period.
(6) Design verification and validation must include results obtained through a usability study that demonstrates that the intended user can use the device safely and obtain the expected glucose measurement accuracy.
(7) The labeling required under § 809.10(b) of this chapter must include a separate description of the following sensor performance data observed in the clinical study performed in conformance with paragraph (b)(1) of this section for each intended use population, in addition to separate sensor performance data for each different iCGM insertion or use sites (eg, abdomen, arm, buttock):
(i) A description of the accuracy in the following blood glucose concentration ranges: less than 54 mg/dL, 54 to less than 70 mg/dL, 70 to 180 mg/dL, greater than 180 to 250 mg/dL, and greater than 250 mg/dL.
(ii) A description of the accuracy of positive and negative rate of change data.
(iii) A description of the frequency and duration of gaps in sensor data.
(iv) A description of the true, false, missed, and correct alert rates and a description of the available glucose concentration alert settings, if applicable.
(v) A description of the observed duration of iCGM life for the device.
We recommend that the accuracy, reliability, and safety of CGM systems should be evaluated with objective and transparent studies before approval, as this may translate into better glycemic outcomes. It is possible that the specific requirements for iCGM designation will evolve in the future based on either new research or external input to FDA, but at this time, the requirements for iCGM are the most rigorous of any major regulatory body. 16,24-27 In this context, critical understanding of accuracy is key to interpreting data. However, as assessment of the accuracy of CGM has not been standardized globally, accuracy data should be considered in the context of the study design and data reporting. Of note, Pemberton et al 28 and also Freckmann et al 29 clearly defined the issues to be addressed when evaluating the accuracy of study design and reporting. They stated that studies presenting accuracy of CGMs should ideally be based on whether the study was published in a peer-reviewed journal, whether the population was clearly defined by DM type and demographics, and whether it represented the target population. Other areas that should be specified according to their articles include the testing protocol, the selection of comparator samples and methods, and the statistical analysis. Moreover, they stated that an ideal assessment of published CGM performance should evaluate whether at least three unique sensor lots were used, whether sufficient readings were taken at each anatomical insertion site and in extreme glucose ranges, and whether readings were taken across days of sensor use. Freckmann and Pemberton also stated that for reporting of accuracy, additional evaluation should include the following: whether data were provided according to DM type and age; agreement rates across the full glucose range or outside the range; time in range; time above range; time below range, coefficient of variation, days of sensor use; and time between sensor activation and first glucose reading recorded.28,29 Other authors have emphasized the need to assess accuracy from 3 viewpoints: the analytical viewpoint (i.e., evaluation of deviations between CGM values and blood glucose concentrations measured with a suitable comparator), the clinical accuracy, and the accuracy of the trend (i.e., the characterization of deviations between the glucose concentration rate of change and blood glucose concentrations measured with a suitable comparator).30,31 Other important issues to be analyzed for a comprehensive evaluation of CGM systems include sensor stability, calibration stability, threshold alert reliability for hypo- and hyperglycemia, sensor survival, and data availability. 25 In summary, a comprehensive characterization of the different approaches for design and presentation of study results is warranted, and a standard method for designing CGM performance studies to improve the clarity of their results should be applied.28,29
The accuracy studies of non-iCGM-approved CGM entrants may be subject to a series of limitations. Thus, whereas some products are supported by an extensive and growing body of clinical evidence across patient subgroups and in real-world settings, non-iCGM-approved entrants that are marketed outside the United States may be supported by little or no published evidence of outcomes in the public domain. 28 For instance, accuracy studies published in peer-reviewed journals are not available for some devices, and the result is that their pivotal accuracy studies for regulatory clearance might not be replicated in independent studies.32,33 A published article might not be relevant to performance in situations specified by the product label because it might include testing in a study population that exceeds the age range of the population for whom the product is approved, or the range of glucose concentrations tested might be mainly in a range where most CGMs tend to perform at their best. Furthermore, a description of the characteristics of patients is lacking in some studies, thus limiting the assessment of data validity. Unfortunately, critical appraisal of publicly available accuracy studies reveals that many indications for several non-iCGM-approved CGM devices outside the United States may not be necessarily based on publicly available accuracy data, but only on non-public data sets submitted to the regulatory agencies.28,29
All CGM devices must be calibrated to maintain accuracy, and this task can be performed by either the patient or the factory. Factory-calibrated systems are considered preferable because they reduce patient burden and eliminate patient-calibration errors. However, given that they are not required to be verified by BGM testing, it is critical that factory-calibrated devices are manufactured in a controlled process to a consistently high quality. 34 However, it is also essential that factory-calibrated devices be supported by robust accuracy data.
Importance of Nonadjunctive Use
Nonadjunctive CGM provides valuable information on glucose levels and is indicated to replace information obtained from standard BGM, without requiring verification, and to aid daily and long-term DM-related treatment decisions, such as insulin dosing (Table 2).7,15,17-19,24,29,35 By contrast, adjunctive CGM devices require confirmation by BGM prior to making any relevant treatment decisions.
US FDA Definition of Non-Adjunctive CGM and US FDA Standards for CGM Accuracy Studies.
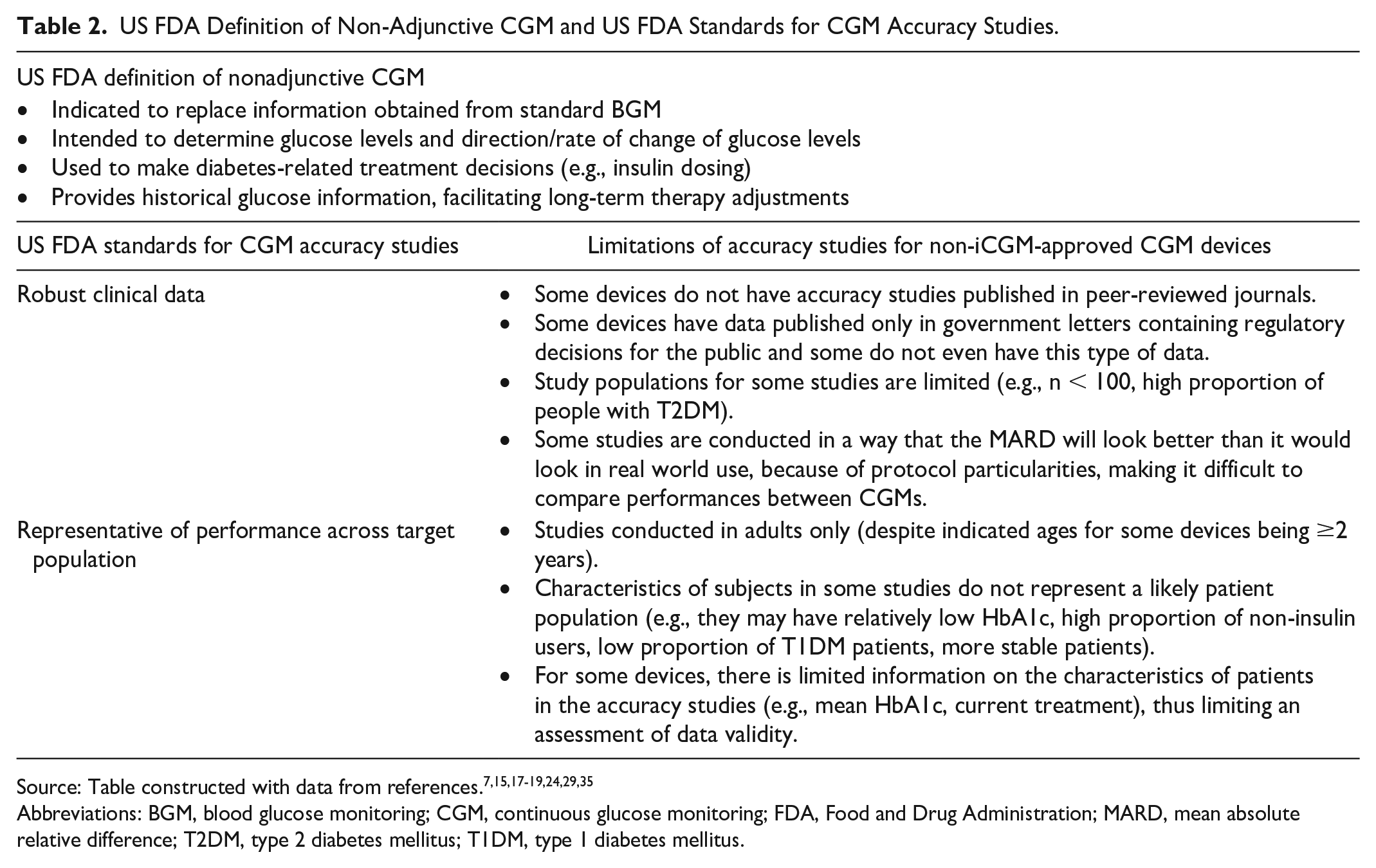
Source: Table constructed with data from references.7,15,17-19,24,29,35
Abbreviations: BGM, blood glucose monitoring; CGM, continuous glucose monitoring; FDA, Food and Drug Administration; MARD, mean absolute relative difference; T2DM, type 2 diabetes mellitus; T1DM, type 1 diabetes mellitus.
Importance of Robust Evidence
To understand the accuracy of CGM devices, it is necessary to conduct studies that include validation of accuracy at critical low- and high-level glucose readings and under conditions of rapidly changing glucose concentrations. In contrast, the manufacturers of non-iCGM devices have not published accuracy data in these important settings or have not shown a documented probability of adequate sensor survival until the end of the claimed sensor life.20-23,36 Furthermore, clinical outcomes and user experience should be demonstrated across patient groups in randomized clinical trials and real-world settings.
Importance of Other Benefits Beyond the Device
When comparing CGMs, it is important to consider not only the sale price, but the overall net cost, including the additional cost of transmitters and/or readers and/or any additional finger prick tests needed, as well as other support services and digital data security provided by the company.
For example, tailored support and education to equip people living with diabetes and health care providers to help make informed diabetes management decisions may or may not be provided by the manufacturer. In addition, there are specific customer service programs to support the resolution of issues, including sensor replacement in some countries and system troubleshooting if needed. In addition, training programs for patients and health care professionals have been developed.
On the contrary, the increasing digitalization of health care creates a complex regulatory environment – health care systems and patients must be confident that their data are secure and not at risk from malicious actors. As a result, digital data security should be considered a priority.14,24,37
Discussion
In the last few years, a number of non-iCGM-approved CGM devices have entered the market worldwide and an assessment of the quality of these devices should be considered the key to understand the role they may play in clinical practice. Those CGM devices are entering markets outside the United States, having often been cleared by regulatory agencies in the countries where they are produced by the EU CE marking system. Often these non-iCGM-approved, which may have been performing at a lower level than FDA-cleared iCGMs, are supported by little or no published evidence of outcomes in the public domain. Without publicly available performance data, or standardized performance data patients and clinicians might lack confidence in their performance. Many CGMs that do not meet the iCGM level of performance have been sold, but the highest level of performance is achievement of iCGM status.
Insulin dosing is an important feature when considering CGM use in DM. However, some non-iCGM-approved CGM devices cannot be used for insulin dosing without BGM testing (i.e., adjunctive use).13,14 Moreover, a lack of information on study design, or limitations in published studies for some devices limits critical evaluation of accuracy data, with the result that the reported accuracy of non-iCGM-approved devices might not be replicated in independent studies.33,38
Furthermore, the body of evidence is scarce, insufficient, or even lacking for many non-iCGM-approved entrants, in terms not only of accuracy, but also of glycemic outcomes (i.e., time in range, time above range, time below range, coefficient of variation).20-23,29 For example, publicly available accuracy data from both observational and randomized clinical trials supporting some non-iCGM-approved entrants include small sample sizes and/or very few data points in the critical range of <70 mg/dL, more stable patients, and no glucose and insulin challenges, and data may not have been published in peer-reviewed journals.8,11,20-24,35,36,39-48
On the contrary, many non-iCGM-approved entrants may have a low cost, and this could be attractive from the viewpoint of a health care system because cost may be a value driver. However, evaluation of costs should not be limited to the device, but also to the wider value of an integrated and nonadjunctive CGM, including support beyond the sensor.11,36 In this context, costs should focus mainly on the quality of the device, as well as the comprehensive service provided. Such service includes whether a product’s manufacturer provides a full digital ecosystem with tailored support and education to equip patients and help clinicians to effectively use the system to make more informed DM management decisions. In fact, there is a growing body of evidence suggesting improved glycemic outcomes (i.e., time in range; time above range; time below range, coefficient of variation) due to these additional benefits.49 -51 Therefore, when considering reimbursement for CGM products, the potential added value of such added services should also be considered.11,15,17-19,41,43,45,52-54
Conclusions
Integrated CGM FDA-cleared systems are an established integrated and nonadjunctive type of product that can directly inform decision-making in treatment of DM. The benefits of these cleared products have typically been established in robust clinical trials and real-world studies, demonstrating state-of-the-art accuracy and improvement in patient outcomes. In addition, support services that provide value beyond the system’s glucose sensing technology have been developed for these systems. By contrast, the manufacturers of many non-iCGM-approved devices sold outside the United States have not published robust accuracy data or randomized controlled trial data to support their use. These devices may not be approved for nonadjunctive use and might require additional patient verification of glucose levels (e.g., finger prick tests) before making treatment decisions. Thus, there is more to consider when selecting a CGM than cost – one should also consider the ease of use for how the product functions, its level of technical performance at its task, and the amount of support services provided by the manufacturer. Many non-iCGM-approved devices come up short in these three areas.
Addendum
After this article was accepted, the FDA granted iCGM designation to the Senseonics Eversense CGM.
Footnotes
Acknowledgements
Writing and editorial assistance was provided by Content Ed Net with funding from Abbott Diabetes Care (Sao Paulo, Brazil).
Correction (October 2024):
Article updated; for further details please see the Article Note at the end of the article.
Abbreviations
BGM, blood glucose monitoring; CE,
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: DCK is a consultant for Afon, Atropos Health, Glucotrack, Lifecare, Nevro, Novo, Samsung, and Thirdwayv. MG is a consultant for Abbott, Medtronic, Novo, Roche. SJM is consultant for Abbott, Curestream, Daewoong, EOFlow, G2e, Huons, iSense, Medtronic, Novonodisk, and Sanofi. EGW has received personal fees from Abbott, AstraZeneca, Dexcom, Eli Lilly, Embecta, Insulet, Medtronic, Novo Nordisk, Roche, Sanofi, Ypsomed and research support from Abbott, Embecta, Insulet, Novo Nordisk, Sanofi.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Editorial assistance and open access license were funded by Abbott Diabetes Care (Sao Paulo, Brazil).
Article Note
The following updates were made to the Table 1 in this article:
iCGM special control performance requirement B and E, “<” has been removed.
iCGM special control performance requirement C and F, “<” has been corrected to “>”.
iCGM special control performance requirement J, “CGM ROC >−1 mg/dL/min & ROC <−2 mg/dL/min” has been corrected to “CGM ROC >1 mg/dL/min & Ref ROC <−2 mg/dL/min”
iCGM special control performance requirement K, “CGM ROC <−1 mg/dL/min & ROC >−2 mg/dL/min” has been corrected to “CGM ROC <−1 mg/dL/min & Ref ROC >2 mg/dL/min”.
Under the column “95% LCL”, % has been added to the values from A to G.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
