Abstract
Presented is a series of narrative reviews that summarize published information regarding the effect or potential effect of interfering substances on the accuracy of continuous glucose monitoring (CGM) devices. While drawing together what is currently known regarding this topic, the future direction in this field and clinical implications posed by polypharmacy on CGM performance are considered. This third in a series of articles lists substances that have been found to influence readings from specific continuous glucose monitor (CGM) models. Points covered include: (1) A review of non-labeled substances that have been found, through in vivo or in vitro assessment, to influence CGM readings; (2) A consideration of substances found to influence CGM readings to an extent that warrants further investigations to determine if a clinically significant in vivo interfering effect occurs at therapeutically or nutritionally relevant levels of a substance and, as such, would require labeling by the manufacturer; and (3) the possibility of predicting if a new medication or formulation is predisposed to influencing CGM readings, based on an assessment of chemical structure or chemical classification. Evidence suggests that a more systematic approach to interference testing would be of value in determining if additional labeling is required for currently marketed CGMs. It is possible that many of the substances reported have previously been assessed by manufacturers and are within acceptance criteria such that product labeling is not required according to currently recognized definitions of interference. Nevertheless, it would be beneficial for this information to be publicly disclosed.
Introduction
ISO 15197:2013, aimed at blood glucose monitoring (BGM) systems, describes an interfering effect requiring product labeling as a difference in glucose bias between a test and a control sample that exceeds ±0.55 mmol/L (±10 mg/dL) at glucose concentrations of <5.55 mmol/L (<100 mg/dL), or exceeds ±10% at glucose concentrations of ≥5.55 mmol/L (≥100 mg/dL). Applying this definition to CGM systems, a distinction may be made between substances that elicit a measurable influence on a CGM at a given concentration or dosage, but at a level below the designated interference threshold, and substances with interfering effects of a magnitude to require CGM labeling. The former situation, while not requiring labeling, should not be neglected because many CGM users, like BGM users, take concurrent medications and supplements that may cumulatively lead to stacking of interfering effects that could result in clinically significant device inaccuracy. The diversity of compounds implicated in influencing CGM readings, as described in this third article of the review series, indicates that polypharmacy may be a significant factor in CGM accuracy. The issues raised by polypharmacy are described in more detail in part 4 of this review series which also addresses topics related to interfering substance effects during in vivo CGM in vivo performance, clinical implications, and the need for standardization. Parts 1 and 2 of the series review CGM designs, known substance interference mechanisms and a consideration of the behaviors of labeled interfering substances.
In the context of the discussion to follow, it is also relevant to note that CGM glucose measurement algorithms are optimized and parameterized using representative in vivo data. Correspondingly, these algorithms are targeted to nominal levels of endogenous species. Therefore, CGM values will be influenced by deviations from the nominal condition as opposed to a zero-analyte condition as would be the case for exogenous substances, such as medications.
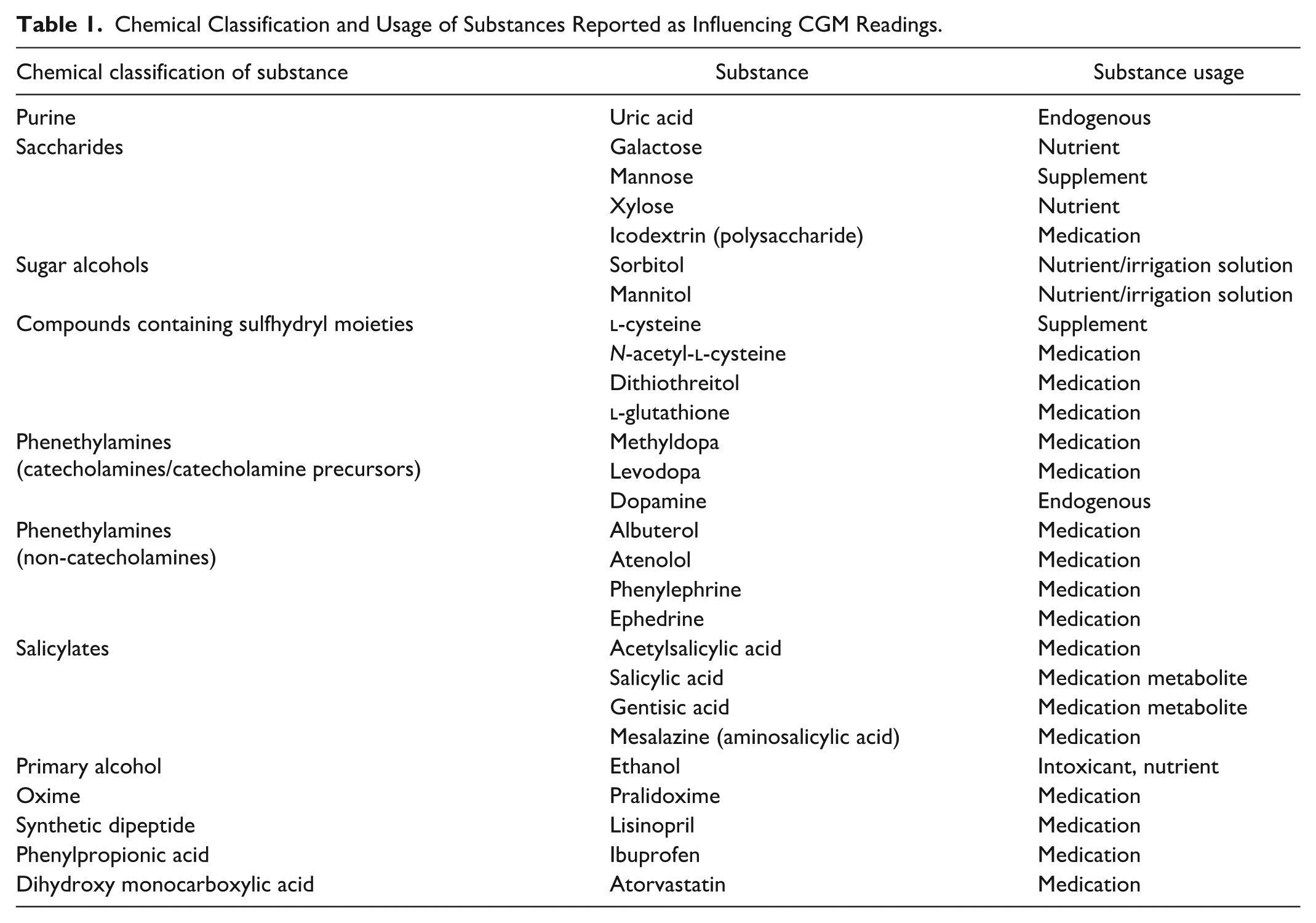
Table 1 lists the chemical classification and usage of substances reported as influencing CGM readings. This may be contrasted with Table 1 (Part 2 of this review series) which summarizes interfering substance labeling for currently marketed CGMs. Except for the phenols, methyldopa and gentisic acid, which are labeled as interferents for the Accu-Chek SmartGuide CGM, none of the substances in Table 1 appear in the labeling of currently marketed CGMs (methyldopa and gentisic acid are addressed in this part of the review series because they may also potentially influence other CGM models). Table 1 groups substances according to their chemical class. Each of these substances will be described in the following sections.
Chemical Classification and Usage of Substances Reported as Influencing CGM Readings.
Uric Acid
Uric acid (Figure 1) is a product of the metabolic breakdown of purine nucleotides, with its endogenous nature and high electroactivity making it a ubiquitous interferent within electrochemical sensors intended for medical applications. In electroanalytical terms, it can be challenging to determine the contribution of interference from uric acid from other biologically important and ubiquitous compounds, such as ascorbic acid and dopamine, due to their overlapping oxidation potentials. 1 Uric acid oxidation is also known to result in progressive fouling of electrode surfaces during electroanalytical applications. 2 Raj and Ohsaka report the electrooxidation of uric acid at a polycrystalline working electrode at ~0.45 V versus a silver/silver chloride (Ag/AgCl) electrode at close to neutral pH (7.2), with the broadness of the voltametric peak being indicative of slow electron transfer kinetics, which they presumed to be due to surface fouling of the electrode by oxidation products. 3
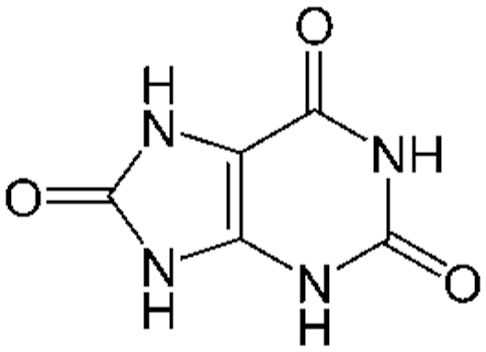
Structure of uric acid.
Uric acid has been shown to influence readings from Dexcom CGM devices when tested at CLSI recommended concentrations.4,5 As discussed in Part 2, Dexcom patent US9572523 describes a CGM system using an enzyme-electrode with a modified permselective membrane design 6 which, when challenged with uric acid, generated a reduced signal compared with the response from a control electrode of unmodified membrane design. While a reduction in signal interference was observed, the test solution containing ~6 mg/dL uric acid, close to the upper physiological level typically found in humans, nevertheless yielded a still clinically meaningful equivalent glucose response of ~25 mg/dL. Given the ionized nature of uric acid under physiological pH conditions, it is reasonable to assume that uric acid permeation may be reduced within Dexcom and similarly designed CGM systems incorporating improved interference membranes with appropriate charge repulsion properties.
A study by Fei and co-workers using a redox polymer containing an osmium bipyridyl complex electrodeposited onto electrode structures, found the material to be an efficient catalyst for the electrooxidation of uric acid, leading to an observation that practical usage of the device could be compromised by the co-oxidation of biological samples containing ascorbic acid and dopamine. 7 While this design of redox polymer is consistent with that used within the FreeStyle Libre (FSL) CGM family, an interfering effect was not observed for uric acid in the FSL2 or FSL3 studies of Pfützner et al.8,9 Fei’s design incorporated a relatively high operating potential of +300 mV (vs a saturated calomel reference electrode) compared with the +40 mV (vs Ag/AgCl) potential employed in FSL CGMs.
Saccharides and Sugar Alcohols
Introduction
A list of saccharides and sugar alcohols tested within CGM systems are given in Table 2 and Figure 2. Certain of these substances have been identified as influencing CGM readings, including 5- and 6-carbon-containing monosaccharides and sugar alcohols. No reports have been identified regarding sucrose testing in commercial CGM systems.
Saccharide and Sugar Alcohol Compounds Tested Within CGM Systems. Certain CGM Designs Are Influenced by the Monosaccharides Galactose, Mannose and Xylose, and the Sugar Alcohols Sorbitol and Mannitol as Described in the Text.
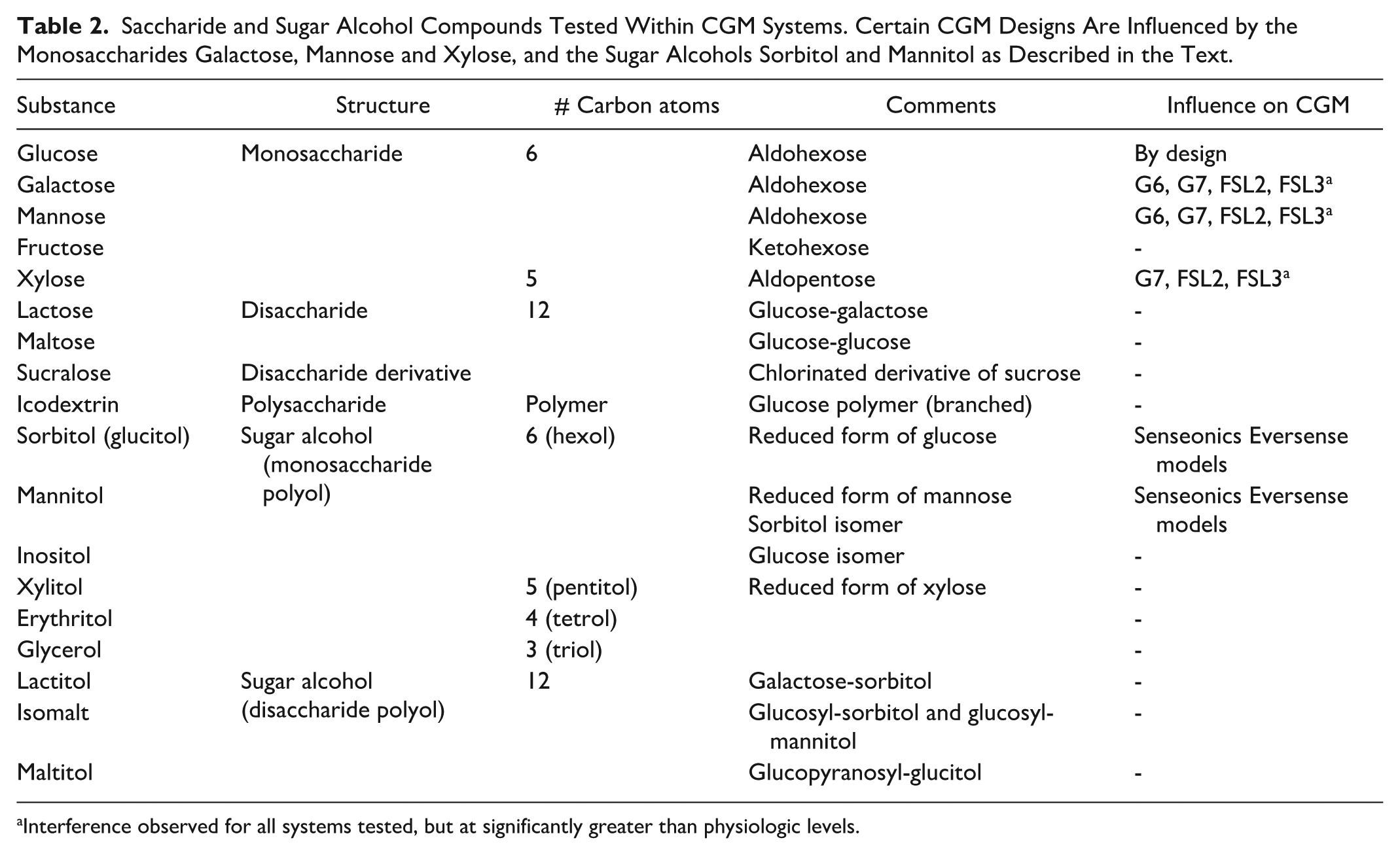
Interference observed for all systems tested, but at significantly greater than physiologic levels.
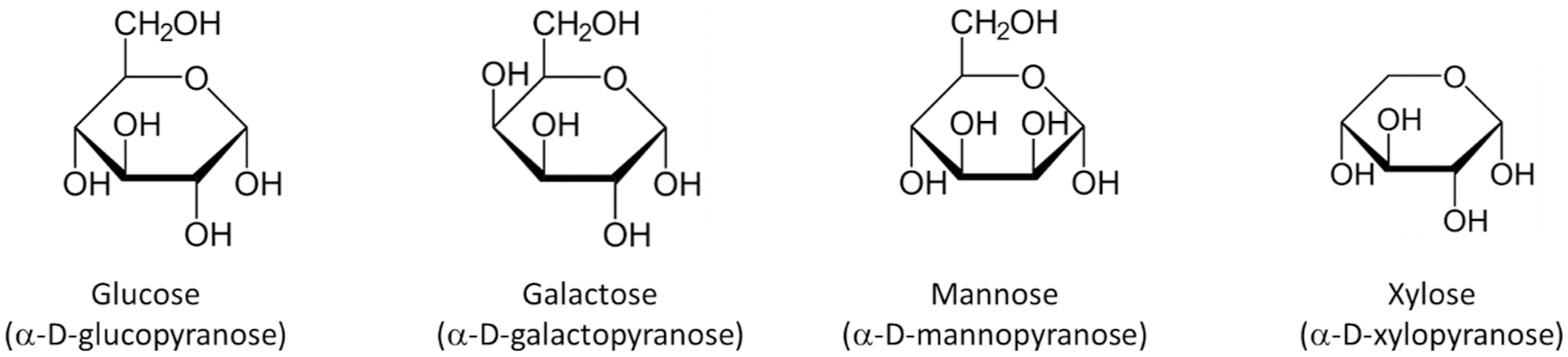
The monosaccharides galactose, mannose, and xylose implicated in influencing enzyme-based CGM readings. Glucose is shown for comparison purposes.
Galactose, Mannose, and Xylose
Pfützner et al 10 constructed a flow-cell design to study in vitro interfering substance effects on Dexcom G6 (G6), Dexcom G7 (G7), FSL2, and FSL3 CGMs. Glucose concentration was maintained at a constant level while analyte concentrations were independently and dynamically changed during passage through the flow-cells. The extent of signal bias from baseline was used to quantify interfering effects. Biases of > +10% from baseline were observed for the aldohexoses galactose and mannose for all models tested, but at concentrations greater than physiologic (eg, 300 mg/dL vs the 60 mg/dL CLSI recommended test concentration for galactose).8-11 These findings agree with the 2018 study of Boehm et al on three CGM systems commercially available at that time (identities not revealed), with devices being similarly allowed to equilibrate in a buffered solution in a flow-cell prior to introduction of test analyte. The observed interfering effect from galactose obtained at high concentrations was further assessed within the clinically relevant range where a maximum mean relative difference of +6.9% was recorded at 10 mg/dL galactose in 70 mg/dL glucose, considered by the authors to be non-substantially interfering.
Boehm et al 11 also reported an interfering effect from the aldopentose xylose, as did Pfützner on the G7, FSL2, and FSL3 systems, but again at higher than physiologic levels.5,8,9 While xylose is more widely implicated as an interferent in glucose dehydrogenase (GDH)-based glucose biosensors, the extent of these interfering effects may be attributable to the higher than physiological concentrations (200-300 mg/dL) with which the CGM systems were challenged. Xylose was also assessed by Lucarelli et al 12 on an early GlucoMen Day CGM design but found not to exceed the ISO bias limit when tested at non-supratherapeutic levels. Boehm et al similarly observed high mean bias values (37%-48%) in CGM systems challenged with 200 mg/dL mannose.
Lactose
Lactose is a disaccharide that does not influence CGM readings when tested in vitro (mean biases were within ±1% of the control 200 mg/dL glucose condition when FSL2 and G6 CGMs were challenged with 20 mg/dL lactose by Pfutzner et al5,9,11). However, in vivo CGM responses to lactose will be influenced by physiological breakdown into its constituent saccharides: glucose and galactose, the latter monosaccharide having both an interfering effect and an effect from being metabolized to glucose. As noted by Hauss, specific interference concerns arise within populations exhibiting galactosemia, when lactose ingestion results in the toxic accumulation of galactose in the body. 13 In addition to increasing the risk for developmental impairment, especially in the young, the interference effect of elevated galactose may mask hypoglycaemia or prompt unnecessary treatment for a falsely elevated glucose reading.
Maltose and Icodextrin
Maltose, a glucose disaccharide, has been tested in vitro with minimally invasive CGMs, that, while having an influence on CGM readings does not require product labeling according to the ISO 15197 definition of an interfering effect (Pfützner et al5,9,11 reported a +4% and +9% mean bias respectively from the 200 mg/dL glucose control condition for the FSL2 and G6 systems). Lucarelli et al 12 found maltose to be within the ISO bias limit when tested at non-supratherapeutic levels on an earlier Glucomen Day CGM model. Icodextrin is a glucose polymer formed from corn starch. It is highly polydisperse, with mean molar masses reported in the range of ~16 to 18 kg/mol.14,15 Pfützner et al 9 recorded a +10% (ie, at threshold level for ascribing an interfering effect to a substance) bias from baseline for the FSL2 system when testing icodextrin in vitro in their dynamic flow-cell set-up. This interference observation was unexpected with the cause not established, although the source material, a natural saccharide product with a widely distributed molecular weight, may include the presence of low molecular weight saccharides.
This potential icodextrin interfering effect is distinct to that reported in those BGM systems that used the GDH enzyme variant incorporating the coenzyme pyrroloquinolinequinone (PQQ-GDH) as the glucose recognition agent. Native PQQ-GDH exhibits cross-reactivity to maltose, a metabolic breakdown product of icodextrin, thus raising the risk of falsely elevated glucose readings in icodextrin-using patient populations, such as those on peritoneal dialysis.16-18 Native PQQ-GDH is no longer used in the manufacture of BGMs due to this issue and so would be unlikely to feature in CGM designs.
While not an interfering effect, in vivo CGM responses to maltose will be influenced by metabolism of the glucose disaccharide parent compound to its glucose monosaccharide constituents.
Other Saccharides
No interference has been found with inulin, a fructo-oligosaccharide, 11 sucralose, an artificial sweetener and chlorinated sucrose derivative, 11 or heparin, a high-molecular-weight sulfated polysaccharide of the glycosaminoglycan family. Other sugar-related compounds tested with CGM systems include the expectorant, guaifenesin (an ether of guaiacol and glycerine), the SGLT-2 inhibitor empagliflozin (a glycosyl compound) and triglyceride, which contains a glycerol backbone, with none found to have significant interfering effects.5,9 Ribose was assessed by Lorenz et al, on the Senseonics Eversense system, but was found not to exceed the ISO bias limit when tested at physiologic levels. 19
Sugar Alcohols
Several CGM in vitro interference studies have variously assessed a range of common sugar alcohols, including glycerol, erythritol, xylitol, sorbitol (glucitol), inositol, lactitol, isomalt (isomaltitol), maltitol, and mannitol. The interfering effects of mannitol and sorbitol on the Senseonics Eversense systems are described in Part 2 of this review series.
Thiolated Compounds
Four sulfhydryl (-SH) compounds have been identified as influencing electrochemical CGM readings when tested in vitro:
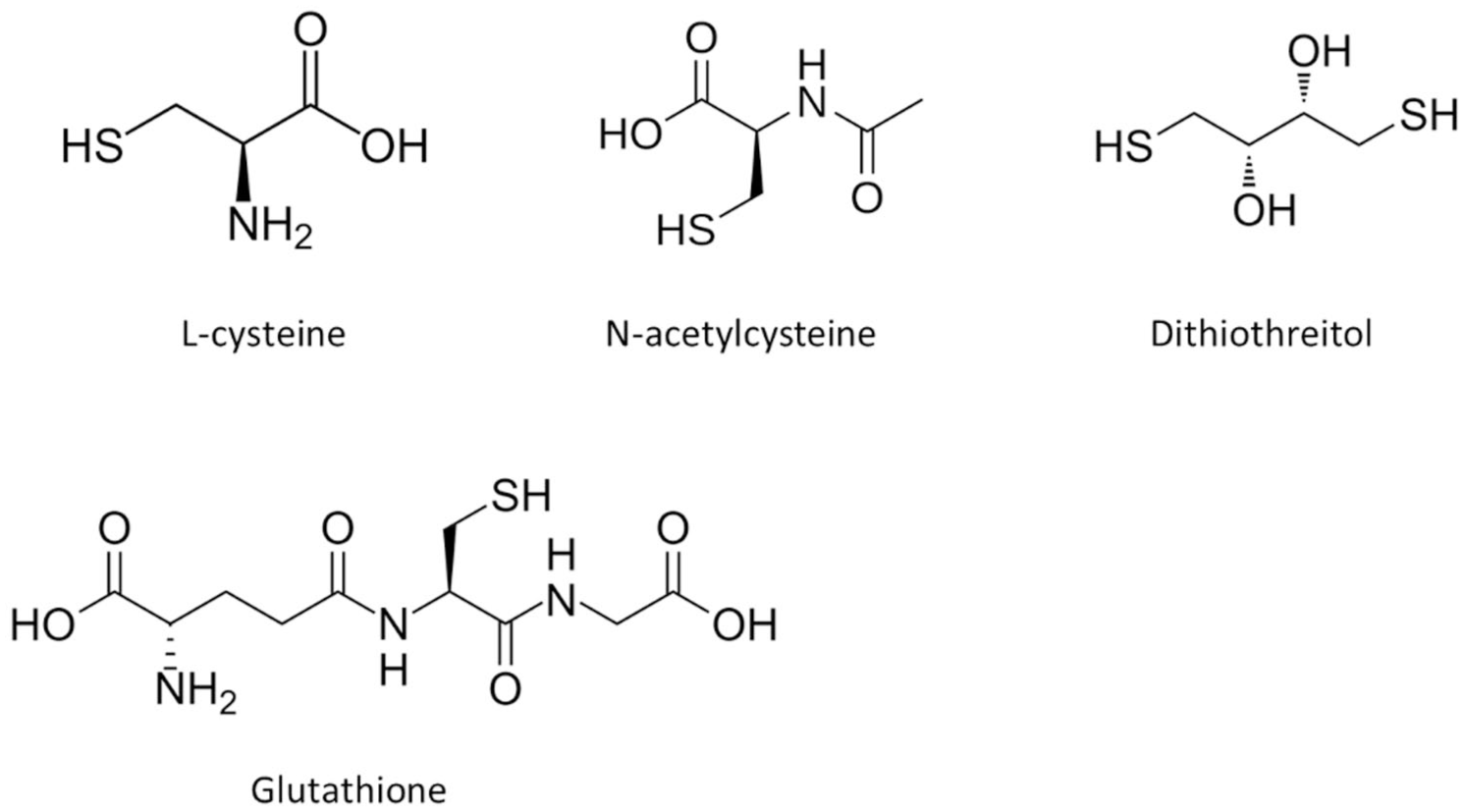
Compounds having sulfhydryl (-SH) moieties found to influence CGM readings.
Pfützner et al,4,5 using a dynamic experimental design, recorded a signal deterioration on introduction of
While Hauss et al also reference certain BGM systems that are influenced by NAC, no such effect was observed by Basu et al
27
during an in vivo assessment of earlier versions of the Dexcom (G4) and Medtronic (Guardian Sof-sensor) systems. These differing observations indicate that using in vitro observations to predict in vivo glucose biosensor behavior of potential interfering substances requires detailed consideration of the test design and applied measurement conditions. Pfützner also recorded an interfering effect with NAC and DTT on FSL2 and FLS 3 devices, manifested as a signal increase, but with no evidence of subsequent sensor passivation which may be due to the working electrode being formed from carbon paste rather than a precious metal.8,9 Lucarelli et al,
12
reporting on an earlier generation GlucoMen Day CGM, a second-generation biosensor containing a carbon working electrode and Prussian Blue mediator, identified interference from the thiolated tripeptide
Piroxicam is also a sulfur-containing compound but carries a sulfonyl as opposed to a sulfhydryl structure. It has not been tested with electrochemical CGMs but was observed by Lorenz et al 19 as having a minor interfering effect in the Senseonics Eversense design. Piroxicam was selected as one of four test compounds known to exhibit either absorption or fluorescence in the wavelength region corresponding to anthracene absorption/fluorescence. 28 While a bias of −47% was recorded at a 6 mg/dL piroxicam concentration at high glucose, subsequent regression analysis showed this to be non-clinically significant at therapeutic levels.
Dexcom patent US7651596 identifies a small interfering effect from the sulfonyl compound tolbutamide when tested with an earlier iteration of a system based on the Dexcom CGM design. 29 No records have been found as to whether this interfering effect persists within currently marketed designs. FSL 14-day labeling, although identifying tolbutamide as a non-interfering species, also states that potential interferents in interstitial fluid (ISF) are unknown compared to circulating blood.
Salicylic Acid and Derivatives
Salicylic acid and gentisic acid are both metabolic breakdown products of acetylsalicylic acid (aspirin, Figure 4). Both are phenols, a class of organic compound having one or more hydroxyl (-OH) groups attached to an aromatic hydrocarbon ring, a feature common to other substances associated with CGM interference, for example acetaminophen (as discussed in Part 2). Many phenols, when investigated within classical electrochemical studies, cause electrode passivation, with the electrochemical oxidation process being described as notoriously complex. 30
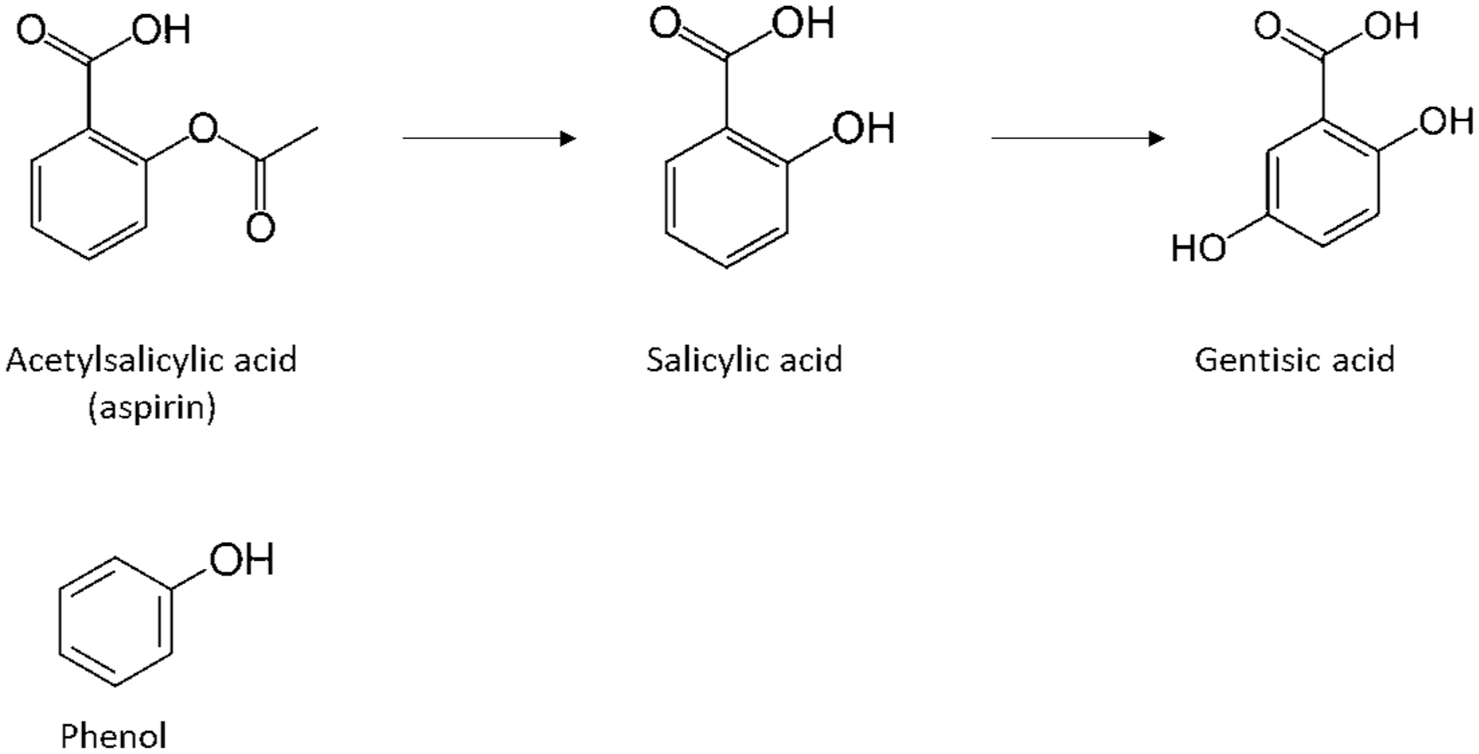
Acetylsalicylic acid (aspirin) metabolism to salicylic acid and gentisic acid. Phenol structure shown below.
Gentisic acid is a labeled interferent for the Accu-Chek SmartGuide CGM and was found to have an observable interfering effect on the G6 and G7 CGMs in the study of Pfützner et al.4,5 Labeling for the earlier FSL 14-day system references an interfering effect from salicylic acid, manifested as a slight lowering of glucose readings depending on the amount of active substance present. The Senseonics study of Lorenz et al 19 also indicated an interfering effect from salicylic acid, but this was not considered clinically relevant at normal therapeutic doses. Exclusion criteria for the Accu-Chek SmartGuide clinical study NCT06704672 include current or recent use of acetylsalicylic acid. 31
The structurally related compound mesalazine (5-aminosalicylic acid, mesalamine, Figure 5) is a medication used to treat inflammatory bowel disease, with in vitro electrochemical investigations indicating electrooxidation at relatively modest operating potentials. 32 Pfützner et al4,5 found this compound had an apparent fouling effect when tested in vitro on the G7 system, but was considered non-interfering in Basu et al’s 27 in vivo assessment.
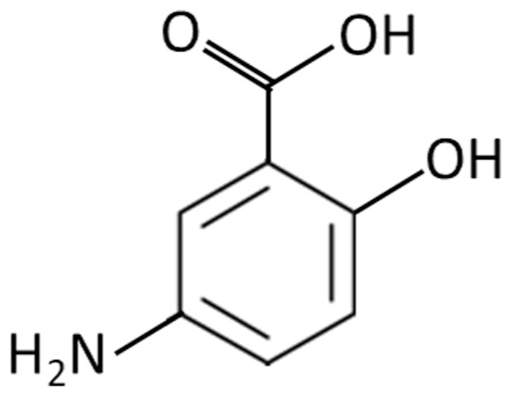
Mesalazine (aminosalicylic acid); structurally related to the salicylates.
Interfering Effects From Phenols Used as Excipients in Insulin Formulations
The combined in vivo/in vitro study conducted by Ward et al 33 to investigate the effects of integrating laboratory-fabricated first-generation CGM devices into insulin delivery cannulas, identified a significant artifact within the in vitro arm of the experiment. The introduction of insulin aspart resulted in a rapid increase, then decrease, in CGM current response, followed by a subsequent current decline to a level lower than the baseline condition, indicating irreversible electrode fouling. This effect was not seen in the study arm featuring a second-generation CGM, this observed behavior being attributed to the lower operating potential applied. The interfering effect was subsequently traced to the use of phenol and m-cresol excipients in the insulin formulation (other phenolic excipients are also used in insulin formulations). 34 Conversely, Hermanides et al 35 found no deterioration in device performance when injecting fast-acting insulin in the locality of a GlucoDay S microdialysis CGM (Menarini Diagnostics), although details regarding the particular insulin formulation used, excipient content and CGM sensing mechanism were not reported.
It is open to question if Ward et al’s observation is a consideration in routine CGM usage, particularly those CGMs of the first-generation design, since recommended practice is for CGMs to be spatially distanced from insulin delivery locations on the body. Furthermore, the CGMs used in the study were investigational, fabricated by the investigators and potentially lacking the more sophisticated designs and algorithmic refinements of the current leading CGMs. Responses will also be dependent upon specific insulin formulations from different manufacturers, who may not necessarily consider potential glucose measurement interfering effects when improving insulin formulation stability and absorption kinetics. Nonetheless, the wider question of excipient addition to medical formulations warrants consideration because, as the claimed wear duration of newer generation of CGM increases, the potential risk is raised for cumulative sensor fouling over time caused by the repeated administration of medication formulations containing low levels of interferent.
Phenethylamines
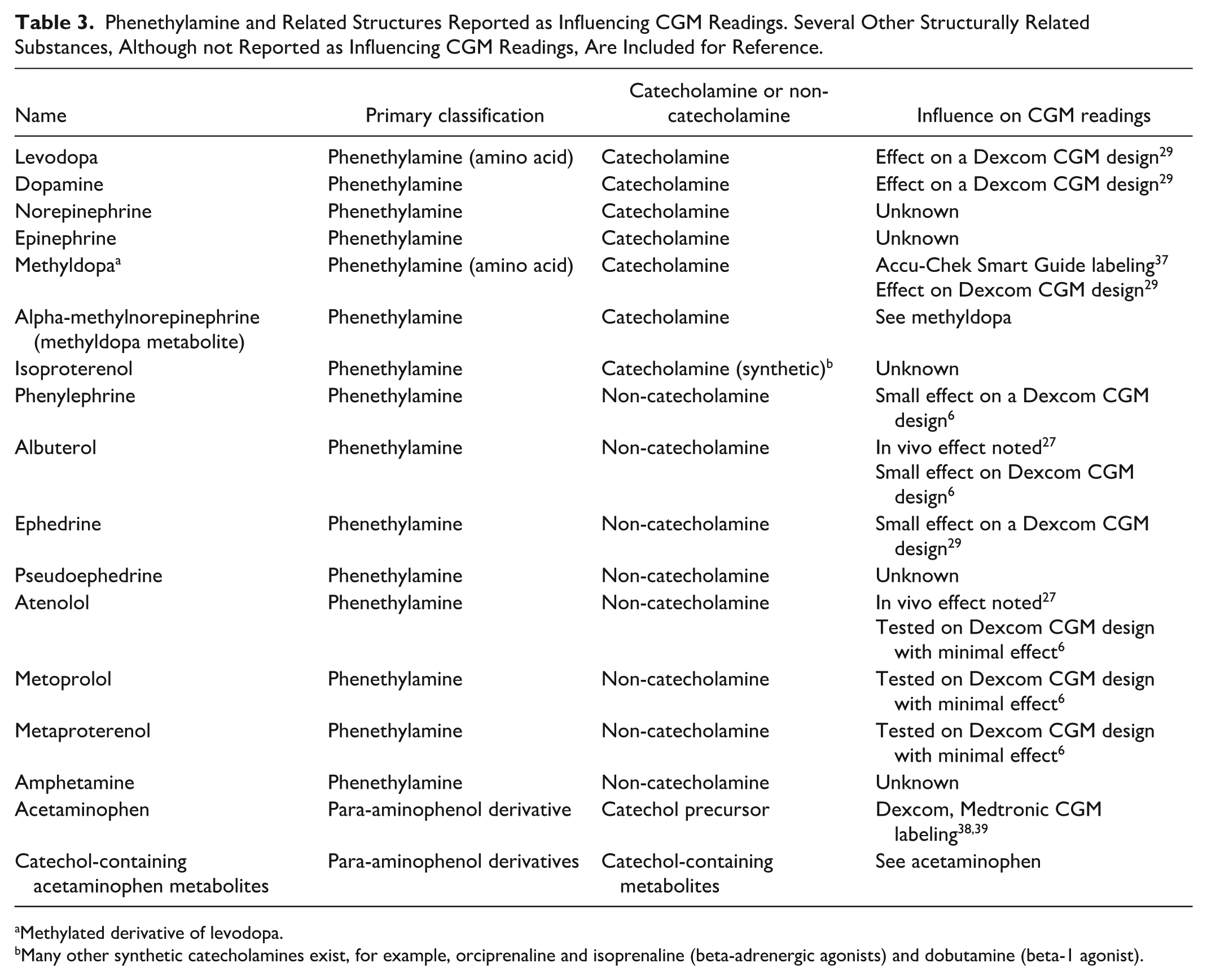
The phenethylamines are group of compounds composed of a phenyl ring linked to an ethylamine side chain (Figure 6). An important subclass of the phenethylamines are the catecholamines, direct-acting adrenergic compounds characterized by an ethylamine side chain attached to a catechol structure, comprising two adjacent hydroxyl (-OH) groups attached to a phenyl ring (Figure 6). The non-catecholamines constitute a further subclass of the phenethylamines, exhibiting an ethylamine side chain, but with no catechol moiety. Many non-catecholamines act as adrenergic agonists, capable of mimicking actions of the sympathetic nervous system. Some common phenethylamine compounds and associated structures are listed in Table 3. Acetaminophen (as reviewed in Part 2) has been included as, while an aminophenol derivative, is metabolized to multiple compounds including some containing catechol structures. 36
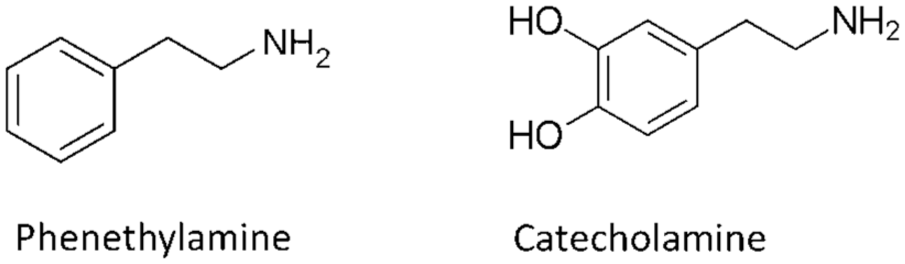
Structures of the phenethylamines and the catecholamines. Dopamine has the simplest catecholamine structure, corresponding to the skeletal catecholamine structure shown here.
Phenethylamine and Related Structures Reported as Influencing CGM Readings. Several Other Structurally Related Substances, Although not Reported as Influencing CGM Readings, Are Included for Reference.
Methylated derivative of levodopa.
Many other synthetic catecholamines exist, for example, orciprenaline and isoprenaline (beta-adrenergic agonists) and dobutamine (beta-1 agonist).
Catecholamines
As a class, catecholamines are known to readily undergo oxidation at electrodes poised at relatively modest potentials, yielding corresponding o-quinone derivatives. 40 The amine group on the catecholamine side chain may also undergo a cyclization reaction, with further oxidation to form aminochrome. As such, this class of compound has the potential to influence electrochemical CGM readings, particularly those of the first-generation biosensor design.
Methyldopa, an antihypertensive agent and levodopa, used in the treatment of Parkinson’s disease (Figure 7) have been implicated in the in vitro interference of electrochemical CGM devices, with methyldopa being labeled as an interferent on the Accu-Chek SmartGuide CGM (the recently posted Accu-Chek SmartGuide clinical study NCT06704672 also includes exclusion criteria regarding current or recent use of levodopa). 31 Dexcom patent US7651596 describes an in vitro test in which a sensor with a cellulosic-based interference was exposed to methyldopa and levodopa in phosphate buffered saline at therapeutically relevant test concentrations (0.1-0.5 and 0.02-0.3 mg/dL, respectively). 29 The resultant sensor signals were used to calculate “equivalent glucose” signals of 52 and 14 mg/dL, respectively. US7651596 provides definitions of interferent effects, with <10 mg/dL (equivalent glucose signal) described as “very minimal,” while a “minimal signal” was described as “less than 20mg/dl equivalent) which is believed to provide sufficient interferent blocking (resistance) to enable functional (useful) sensor data even in the presence of these interferents at the tested concentrations.” This is a less stringent definition than suggested within general guidance documentation on interfering substances. It is likely that modifications to the membrane-based interferent-blocking technology described in this patent have further evolved within Dexcom CGM designs since the time of filing.
The catecholamine precursors levodopa and methyldopa, and associated metabolites, norepinephrine, epinephrine, and alpha-methylnorepinephrine, all catecholamines.
The FSL Pro system summary of safety and effectiveness data (SSED) document issued by the FDA in 2016 stated that levodopa and methyldopa had been tested with no adverse impact on sensor performance. Pfützner reported interfering effects for methyldopa on both the FSL2 and FSL3 systems and for levodopa on FSL3, albeit just above the 10% bias threshold required for identifying a substance to be labeled as interfering.8,9 Levodopa was also found to have an interfering effect on the optically based Senseonics Eversense CGM design, although below therapeutically relevant levels.
While classified as catechols, both methyldopa and levodopa are sometimes referred to as catecholamine precursors, because the metabolism of these compounds yields the methyldopa metabolite alpha-methylnorepinephrine and levodopa metabolite dopamine (Figure 7). Dopamine has been identified as influencing the readings from a CGM of first-generation biosensor design with Dexcom patent US7651596 describing an in vitro test in which the interference-membrane modified sensor was exposed to dopamine at therapeutically relevant test concentrations (0.03-0.10 mg/dL). 29 The resultant sensor signal yielded an “equivalent glucose” signal of 10 mg/dL.
The FSL Pro CGM SSED states that dopamine has been tested with no adverse impact on CGM performance. The historical Glucomen CGM model of 2012 suggests a direct dopamine redox reaction with system-generated hydrogen peroxide, resulting in peroxide consumption and a consequent reduction in electrochemical signal. However, this model has been superseded by later CGM designs carrying the Glucomen name. 12
Dopamine can be further metabolized to yield norepinephrine and epinephrine, also catecholamines (Figure 7). As such, these metabolites maintain their electroactive nature and therefore their presence in ISF has the potential to contribute to signals generated from electrochemical CGMs in vivo.
The Non-catecholamines
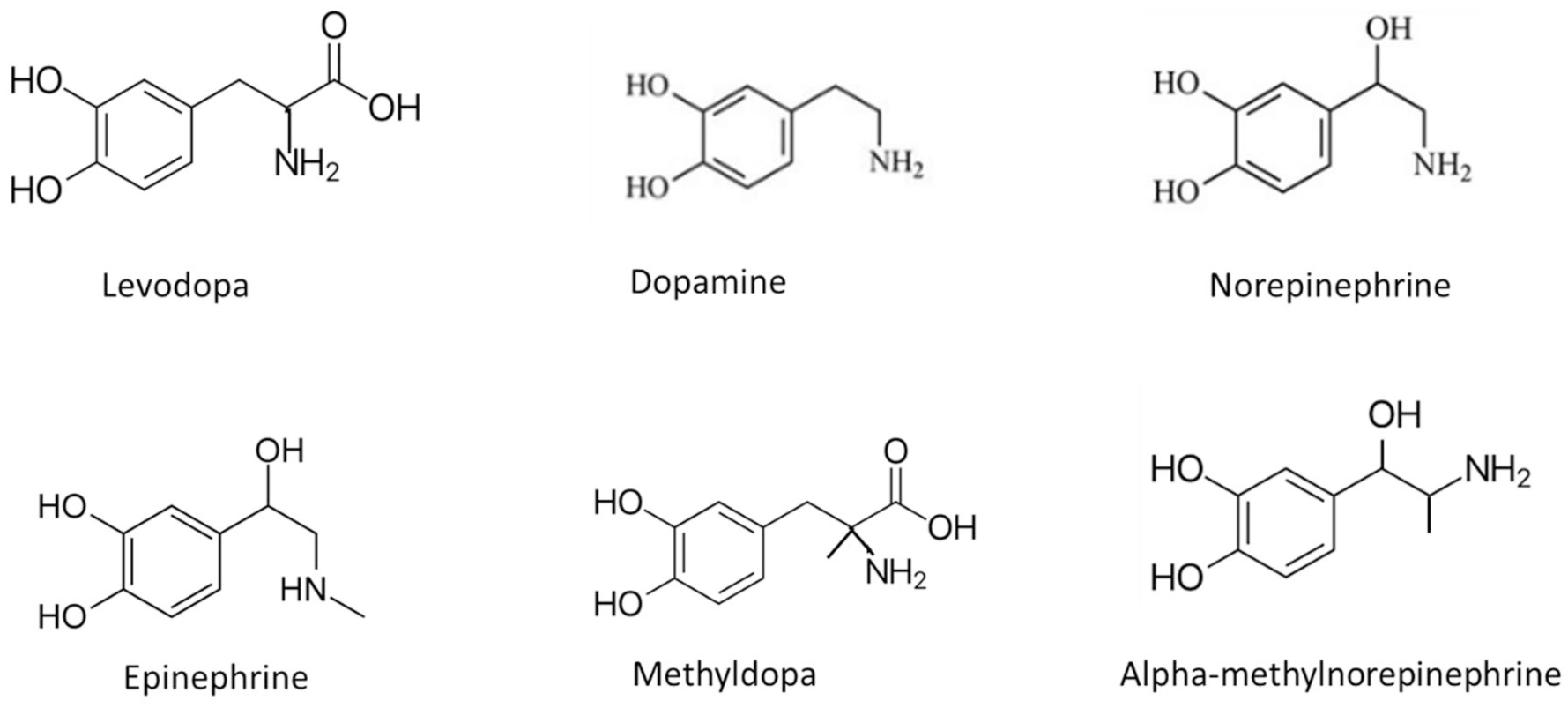
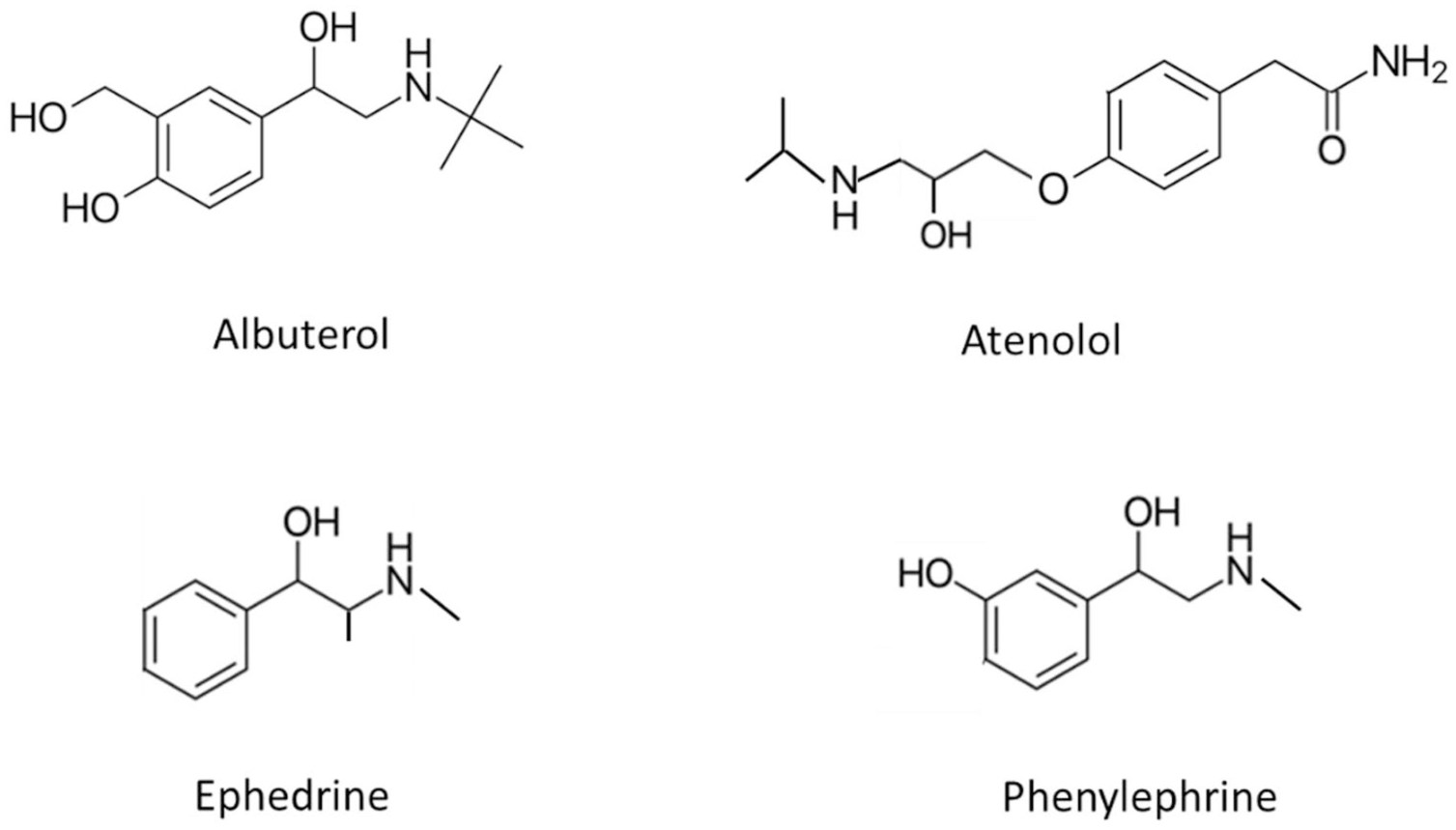
Figure 8 shows structural commonalities between the four non-catecholamines found to influence CGM readings: albuterol (salbutamol), atenolol, ephedrine and phenylephrine. Basu found that in vivo administration of albuterol in subjects wearing G4 and Guardian Sof-Sensor CGMs led to observable influences on CGM values compared with paired plasma glucose reference measurements. 27 Dexcom patent US9572523, filed in 2015, reports an equivalent glucose signal decrease of 3 mg/dL to CGMs exposed to a 0.04 mg/dL albuterol, albeit at a test concentration greater than recommended by CLSI.6,41 This same patent also quantifies the interference due the presence of 4 mg/dL of the decongestant phenylephrine, with an equivalent 10 mg/dL glucose response recorded, again at test concentrations in significant excess of CLSI recommendations.
Structures of non-catecholamines found to have influenced CGM readings.
Atenolol, a beta blocker medication was also evaluated within the in vivo study of Basu 27 and found to exhibit an interfering effect on both the G4 and Medtronic Guardian Sof-sensor systems. However, Dexcom patent US9572523 6 indicates a minimal effect when tested in vitro, which may suggest the presence of electroactive atenolol metabolites in vivo. This same patent indicated that the non-catecholamines metoprolol and metaproterenol had a negligible equivalent glucose signal. The antihypotensive agent ephedrine was found to elicit a “minimal signal; namely, less than 20 mg/dL equivalent” from the Dexcom CGM design described in US patent 7651596. 29
The interfering behavior of acetaminophen has been reviewed in Part 2 of this review. This compound, when metabolized in the human body, is known to produce multiple metabolites including some containing catechol structures, for example 3-hydroxyacetaminophen (Figure 9). 36 The presence of such catechol moieties suggests a potential mechanism for the in vivo influencing effect of acetaminophen on electrochemical CGM signals.
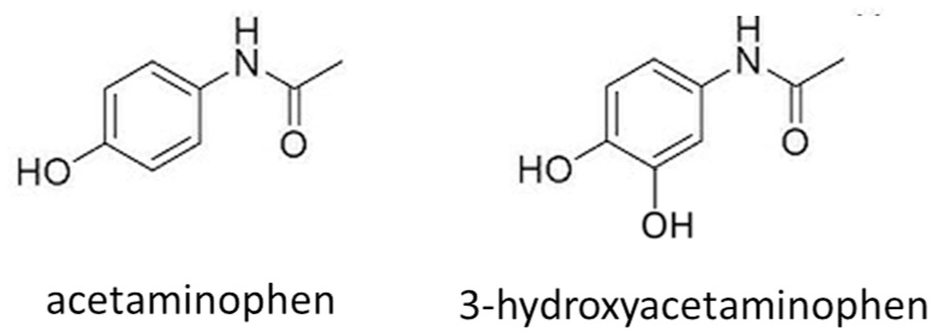
Acetaminophen and a catechol-containing acetaminophen metabolite, 3-hydroxyacetaminophen.
Consideration of Other Catecholamine-Related Structures
The preceding sections indicate that several compounds and metabolites exhibiting catecholamine or catecholamine-related structures have the potential to influence electrochemical CGM readings. Given that many medications are based on compounds formed from such structures (for example the large class of non-catecholamine adrenergic agonists), there may be benefits in a more detailed assessment of this class of medication, including new-to-market products, for potential electrochemical CGM interfering effects.
Ethanol
In vitro studies of ethanol reveal a complex electrooxidation mechanism influenced by electrode structure, adsorption of reaction components to the electrode, sample matrix factors and pH. 42 The American Diabetes Association (ADA) Standards of Care 43 report the “Medtronic Guardian” system to exhibit interference from alcohol (ethanol), while ethanol has been identified as having an in vitro interfering effect by Pfützner et al on the G6 and G7 CGM at a physiologically relevant level. Moscardo et al, 44 studying the effect of ethanol (beer) consumption alongside a mixed meal on the accuracy on a Dexcom G5 system in a population with Type 1 diabetes, found a significant effect on system accuracy, with interference behavior being described as complex and potentially driven by ethanol-induced metabolic effects, including pH shifts. The picture is further complicated by the study of Fazio et al 45 who found that co-administration of ethanol with the known CGM interferent ascorbic acid, alongside a meal, led to significantly lowered levels of plasma ascorbic acid for at least 24 hours in a healthy subject population.
Basu et al, 27 investigating the in vivo effect of the consumption of two glasses of red wine (374 mL) on subjects’ CGM profiles, noted that changes in CGM readings were temporally related to ingestion of the alcohol. Attempts were made to follow changes in ethanol levels within plasma and ISF, the latter matrix accessed by microdialysis, which proved technically challenging due to limited sample volumes and evaporation. Pfützner et al8,9 also found a slight interfering effect from red wine on the FSL2 and FSL3 systems. While red wine contains significant levels of ethanol, it is complex, containing trace amounts of many compounds such as simple saccharides, polysaccharides and antioxidant species, including multiple polyphenolic compounds. 46 Many components of red wine are readily oxidizable, with Kilmartin observing that “quality winemaking is all about redox reactions,” with the extent of oxygen exposure and application of antioxidants contributing enormously to the final wine style. 47 Thus, the imbibing of complex alcoholic drinks may exert different effects on CGM performance depending on beverage composition, co-consumption of food, CGM design, and a complex individual metabolic response, the latter factor not being a consideration within in vitro studies. Other mechanisms may also be involved, with the polyphenol resveratrol for example, being found in red wine and known to improve insulin sensitivity. 48
Pralidoxime
Pralidoxime (PAM, Figure 10), generally formulated as a chloride or iodide salt, is used to treat organophosphate poisoning. It is an interferent associated with electrochemical BGM systems. 49 While no reference to PAM testing on electrochemical CGM systems has so far been identified, Lorenz et al’s study of the fluorescence-based Senseonics design identified PAM iodide as a potential interferent during the screening phase of the study but was assessed above normal therapeutic levels.
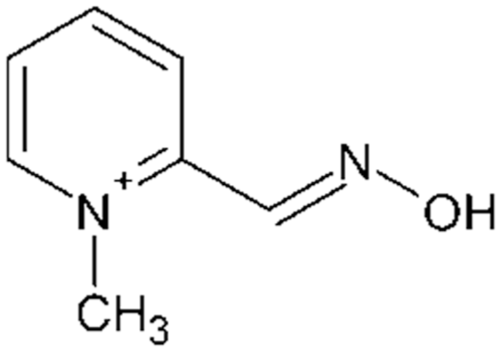
Pralidoxime is an oxime, generally formulated as an iodide or chloride salt.
Amino Acids and Peptides
In addition to the amino acids already reviewed as influencing CGM readings (levodopa, methyldopa,
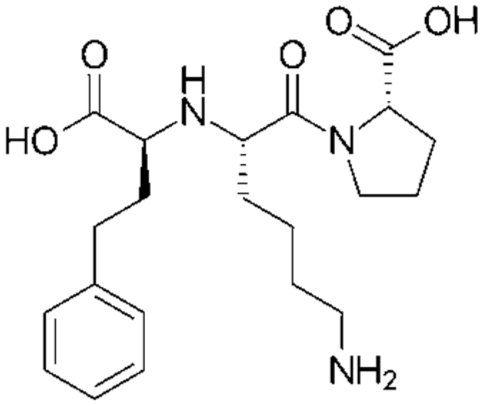
Lisinopril, a synthetic peptide derivative with an in vivo influence on CGM readings.
Ibuprofen
An interfering effect from ibuprofen (Figure 12) on both the FSL2 and FSL3 systems was observed by Pfützner et al8,9 using their in vitro flow-cell based test system, where a +10% deviation from baseline was observed at a test concentration consistent with CLSI recommendations. 41 Abbott documentation states that while ibuprofen is an electroactive compound that can affect sensor performance, the FSL2 system exhibited no clinically significant interfering effect when tested at high therapeutic treatment levels. 50 Dexcom labeling states no reports of ibuprofen interference with their systems. 38
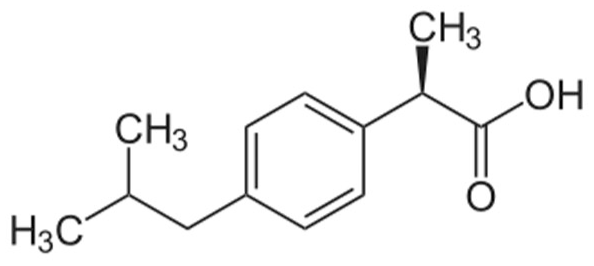
Structure of ibuprofen, a phenylpropionic acid and non-steroidal anti-inflammatory agent.
Atorvastatin
Atorvastatin (Figure 13), a statin medication and dihydroxy monocarboxylic acid, was also evaluated within the in vivo study of Basu et al 27 and found to exhibit an effect on both the G4 and Guardian Sof-sensor systems.
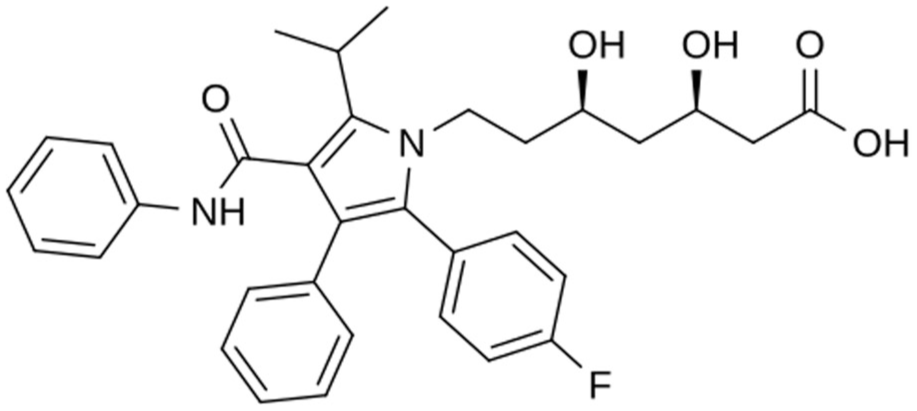
Atorvastatin, a statin medication found to influence CGM readings in vivo.
Oxygen
Oxygen Susceptibility in First-Generation CGM Models
First-generation CGMs rely on oxygen dissolved in the sample matrix to facilitate electron transfer from the enzyme active site to the electrode interface (as described in Part 1 of this review). Given the sparingly soluble nature of oxygen in ISF, membrane designs that limit glucose flux, while promoting ready oxygen access to the GOx enzyme layer, are deployed to ensure that oxygen is not the rate-limiting reactant. For first-generation CGMs to be accurate, they must therefore be capable of operating across a wide oxygen/glucose range, such as conditions of hypoxia coupled to hyperglycemia.
Published literature is sparse regarding the clinical accuracy of first-generation CGM devices under hypoxic conditions, 51 which may prevail in some critically ill patients or maybe due to compression artifacts at the CGM wear site due to body tissue pressurization and subsequent gradual oxygen depletion, most notably when a person is lying on their device at rest. However, first-generation designs have been studied in vivo under hyperoxic conditions by several researchers, on subjects with and without diabetes.52,53 An evaluation by Baines et al, 54 measuring venous and capillary blood by BGM and ISF glucose by CGM (Medtronic Guardian Connect) from 10 subjects with diabetes during hyperbaric oxygen treatment, found no statistically significant difference between the capillary and ISF arms of the study. However, Huang found a significant difference in G6 CGM values within subjects with diabetes as they transitioned from normobaric to hyperbaric air and vice versa (mean 3.76 mg/dL difference). 55 This difference was considered non-clinically relevant, with the investigators unable to attribute this difference to an oxygen interfering effect. The study of Gamarra, 56 who assessed the performance of both G7 and FSL3 in recreational scuba divers with type 1 diabetes, recorded overall mean absolute relative differences (MARDs) of 31% for G7 and 14.2% for FSL3 versus a BGM comparator. While a significant elevation in partial oxygen pressure would be expected during diving at depth, blood oxygen partial pressure measurements were not conducted, with the authors concluding that further investigation is required to understand the factors responsible for the observed impacts on G7 accuracy.
Oxygen Susceptibility in Second-Generation CGM Models Using Glucose Oxidase
The enzyme-mediator design of second generation electrohemical CGM biosensors has been described in Part 1 of this review. Empirical in vivo data suggests that dissolved oxygen does not have a significant effect on the performance of the second-generation GOx enzyme-based FSL CGM models (as will be discussed). However, mechanistic laboratory evaluations suggest a more complex picture, with two potential interference mechanisms being described:
The donation of electrons from the reduced form of the mediator species to dissolved oxygen
The donation of electrons from the reduced form of the glucose oxidase enzyme redox center to dissolved oxygen
In both cases, this can result in the generation and accumulation of hydrogen peroxide (electrochemically inactive at the electrode potentials employed by second-generation systems).
Mediator interference mechanism 1: transfer of electrons from mediator to oxygen
An oxygen sensitivity due to direct reduction of oxygen by polymeric osmium redox centers, has been demonstrated by Prevoteau and Mano in electrochemical systems. In a first study, they examined the reduction of molecular oxygen on osmium-based redox polymers (akin to those used in FSL CGM models) at glassy carbon electrodes in the absence of enzyme. 57 A range of osmium polymers of different redox potentials were assessed under non-physiologic (forced-convection) conditions to maximize oxygen levels at the electrode interface. Catalytic reduction of oxygen to hydrogen peroxide by the osmium complexes were observed (Figure 14) at fixed redox potentials (followed by a further slower kinetic reduction of the peroxide to water, again facilitated by the osmium mediator). The authors suggested that this behavior had potential consequences for systems employing osmium redox centers for biosensing applications.
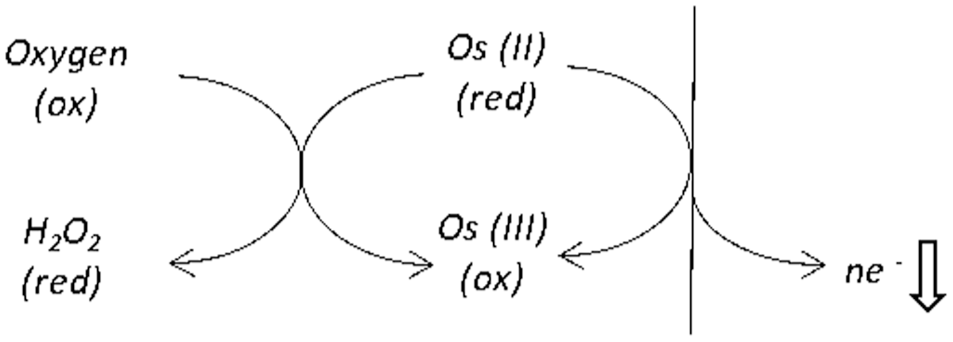
Electron acceptance by oxygen from a reduced form of osmium mediator. In this schematic, the reduced form of the mediator has a charge of +2. A decrease in the amount of reduced mediator due to oxygen consumption will correspondingly lower the CGM signal response.
Mediator interference mechanism 2: transfer of electrons from the glucose oxidase enzyme redox center to oxygen
In the related BGM field, where artificial mediators are routinely employed with GOx enzyme, literature hypothesizes that dissolved oxygen in capillary blood samples compete with artificial mediator for electrons from the reduced cofactor of GOx, giving rise to an oxygen dependent interfering effect (Figure 15). Electron acceptance by the oxygen molecule results in its reduction to hydrogen peroxide, this effect being most evident at low glucose concentrations when the current signal from the device is lowest. Commercial labeling indicates that many BGM reagent formulations include an excess of mediator to minimize the relative effect of this interaction. One may therefore speculate that a similar effect may be evident with second-generation CGM models given dissolved oxygen is present within ISF.
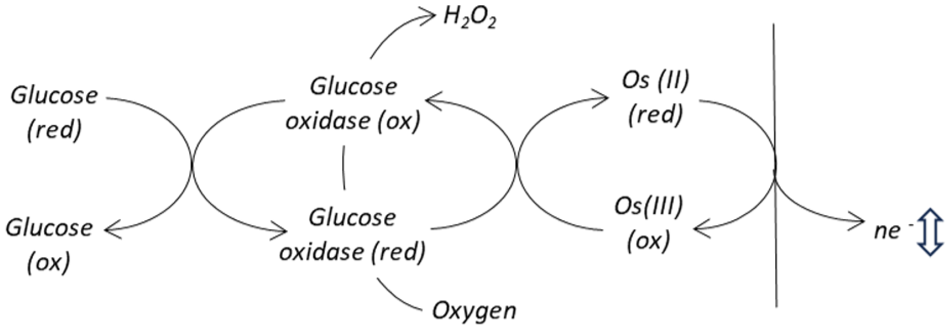
Oxygen can compete with mediator for electrons at the active site of the enzyme, generating hydrogen peroxide which is electrochemically inert under the measurement conditions applied. This effect correspondingly influences the glucose specific signal.
The extent of this interfering oxygen effect appears non-linear for in vivo assessments. Findings from a large-scale clinical database of unmanipulated capillary blood samples paired to an oxygen insensitive comparator method, showed that, while a relatively modest systematic increase in mean bias was evident at low oxygen levels, as oxygen tension increased, no systematic change in bias was observed above the nominal condition. 58 This is contradictory to published benchtop (in vitro) studies in which a significant BGM oxygen dependency has been reported across the range of oxygen partial pressures encountered in manipulated venous blood samples.59-61 The authors suggest that as these laboratory tests require the artificial manipulation of venous blood samples to meet target oxygen partial pressure ranges, there is an inadvertent introduction of artifacts into the test method, resulting in deviations from clinical reality.
Combined oxygen interference in the presence of mediator and glucose oxidase enzyme
Returning to Prevoteau and Mano, these researchers conducted a follow-on study in which GOx enzyme was introduced alongside an oxygen sensitive osmium redox polymer (formal potential of 170 mV vs Ag/AgCl) under conditions of purged and maximal dissolved oxygen. 62 Correspondingly, both oxygen interference mechanisms occurred simultaneously. A pronounced sigmoidal-shaped reduction in glucose signal was obtained in the presence of oxygen versus the oxygen-purged control. The proportionally greater signal reduction at low glucose was attributed to the reduction of oxygen by both the mediator and glucose oxidase enzyme. At higher glucose concentrations, it was concluded that most oxygen was consumed in the outer layers of the reagent film and therefore had a more limited effect on the current signal, as predicted by classical modeling studies.63,64
Empirical Observations of Variable Oxygen Partial Pressure in Second-Generation CGM Models
At a population level, oxygen partial pressures within fingertip capillary blood samples follow a near-normal distribution, with 99% of values within the range 6.40 to 13.68 kPa (48.0-102.6 mm Hg), 65 and it is reasonable to assume that a comparable situation pertains in ISF. As manufacturers calibrate their devices using clinical samples drawn from broad populations within the intended user group, device calibration is centered around the nominal level of dissolved oxygen (and other potentially interfering endogenous substances). In the case of oxygen-sensitive systems, device calibration should be targeted to deliver zero bias between the device reading and that of an oxygen-insensitive comparator method under nominal oxygen conditions. Because most samples encountered will have partial oxygen pressures within a narrow range, systematic bias effects are not obvious. Hypoxic samples may exhibit a positive bias relative to the oxygen-insensitive comparator, because the reduced availability of oxygen will result in a proportionally greater transfer of electrons to the artificial mediator. Theory would predict, therefore, that a systematic negative bias should be seen at higher oxygen partial pressure, which is the case within laboratory studies where samples are artificially manipulated to increase oxygen tension. However, such behavior is not apparent in real-world clinical studies using BGM systems. 58
Support for the relative oxygen insensitivity of the FSL systems under high oxygen conditions comes from the scuba diving study of Gamarra et al. 56 While blood oxygen tensions were not measured, it is reasonable to suppose that ISF partial oxygen pressures in subjects while diving using air (Nitrox) cylinders is relatively elevated (the average 27-m dive depth representing a 2.7-fold pressure increase over atmospheric pressure). In this case, the FSL3 system yielded a MARD similar to clinical studies performed under normobaric conditions (CGM values matched to oxygen-insensitive BGM capillary blood measurement performed pre-dive and immediately post-dive).66-68 Contrary to the mechanistic observations of Prevoteau and Mano, this data suggests no obvious systematic interfering effect due to elevated oxygen. Similarly, a study by Herold and Weh indicates no systematic bias with diving depth for FSL3 systems. 69
The CareSens Air CGM (second-generation biosensor design) is reported to incorporate FAD-GDH within its design. Correspondingly, the enzymatic reaction has no oxygen dependency, with the reaction mechanism being reliant upon the presence of an undisclosed artificial mediator. There are currently no reports regarding the behavior of this system under conditions of physiological extremes of oxygen partial pressure.
Thus, while it is evident that, from a mechanistic perspective, oxygen has the potential to interfere with second-generation CGM devices that use GOx enzyme, this is not borne out by empirical data which suggests little sensitivity to oxygen, even under physiological extremes. Further device-specific studies are warranted to fully understand this issue.
Oxygen Susceptibility in Third-Generation CGM Models
Continuous glucose monitor models of the third-generation biosensor design do not have a dependence on dissolved oxygen. However, as noted by Toghill and Compton, this design does not necessarily resolve the interference effect of dissolved oxygen competing with the electrode to regenerate enzyme (GOx), but the impact can be considerably lower. 18 This again suggests that further device-specific studies are warranted.
Summary
Substances that have been reported as influencing readings from currently or previously available CGM models, but not reflected in product labeling, are listed in Table 4 (substances that are labeled as interferents of commercial CGM models have been reviewed in Part 2). It is not known how many of these substances have been subject to more rigorous assessments by CGM manufacturers as there is no requirement to publicly disclose information relating to substances that do not breach interference assessment criteria. The data suggest that many substances undergo direct electrochemical oxidation at the sensing element of CGMs based on first-generation electrochemical biosensor design. For those CGMs based on the second-generation biosensor design, the mode of interference is less clear, but may be influenced by the presence of the mediating species. Certain monosaccharide sugars have been shown to influence readings from CGMs that use an enzyme as the glucose recognition element. The implantable Senseonics Eversense systems, being of a different sensing design to other marketed CGMs, exhibits its own distinct interference profile.
A Summary of Chemical Substances that Have Been Identified as Influencing Readings From Currently or Previously Commercially Available CGM Systems.
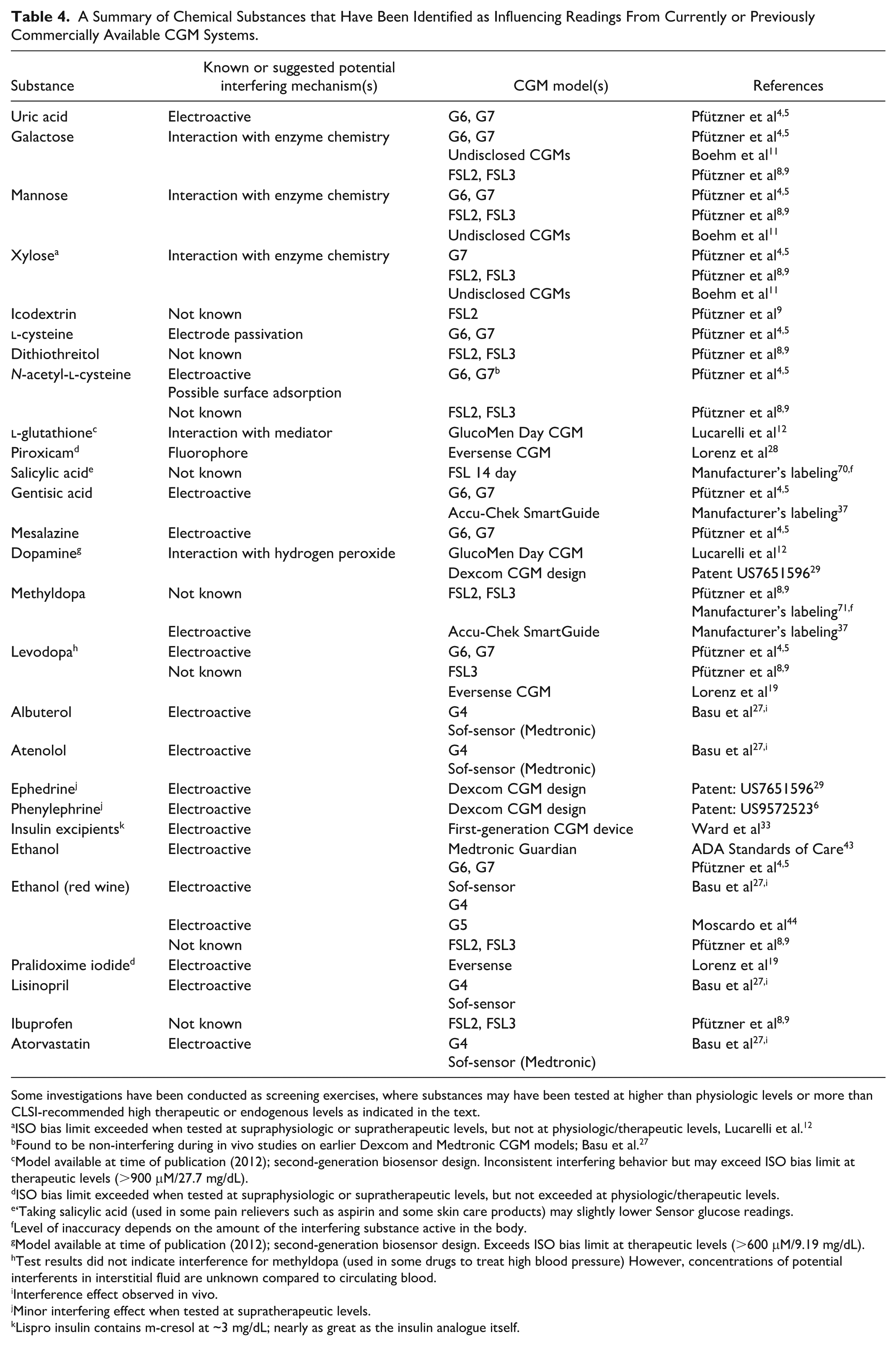
Some investigations have been conducted as screening exercises, where substances may have been tested at higher than physiologic levels or more than CLSI-recommended high therapeutic or endogenous levels as indicated in the text.
ISO bias limit exceeded when tested at supraphysiologic or supratherapeutic levels, but not at physiologic/therapeutic levels, Lucarelli et al. 12
Found to be non-interfering during in vivo studies on earlier Dexcom and Medtronic CGM models; Basu et al. 27
Model available at time of publication (2012); second-generation biosensor design. Inconsistent interfering behavior but may exceed ISO bias limit at therapeutic levels (>900 μM/27.7 mg/dL).
ISO bias limit exceeded when tested at supraphysiologic or supratherapeutic levels, but not exceeded at physiologic/therapeutic levels.
‘Taking salicylic acid (used in some pain relievers such as aspirin and some skin care products) may slightly lower Sensor glucose readings.
Level of inaccuracy depends on the amount of the interfering substance active in the body.
Model available at time of publication (2012); second-generation biosensor design. Exceeds ISO bias limit at therapeutic levels (>600 μM/9.19 mg/dL).
Test results did not indicate interference for methyldopa (used in some drugs to treat high blood pressure) However, concentrations of potential interferents in interstitial fluid are unknown compared to circulating blood.
Interference effect observed in vivo.
Minor interfering effect when tested at supratherapeutic levels.
Lispro insulin contains m-cresol at ~3 mg/dL; nearly as great as the insulin analogue itself.
As observed in Part 2 of this review series, little is known regarding the levels of potentially interfering substances in ISF, with no clear guidelines regarding recommended test concentrations in ISF. Consequently, test levels of substances and hence outcomes may vary between different studies. For example, for those substances listed in Table 4, some have been assessed above therapeutic or physiologic levels. Test methodology and the selection of test compounds may also vary according to manufacturer or researcher, making it difficult to definitively define the true interference profile of different CGM designs. The need for standardization of substance interference behaviors on CGM systems is therefore required and is discussed further in part 4 of this review series.
Footnotes
Acknowledgements
The author is indebted to Dr Elizabeth Holt MD, FACE, Head of Global Clinical, Medical and Safety, Dr Mike Grady, Director of Clinical Affairs and Mr Brian Heald, Senior Vice President, Product, for their invaluable suggestions and in-depth review of this manuscript.
Abbreviations
ADA, American Diabetes Association; Ag/AgCl, silver/silver chloride; BGM, blood glucose monitor; CGM, continuous glucose monitor; CLSI, Clinical and Laboratory Standards Institute; DTT, dithiothreitol; FDA, Food and Drug Administration; FSL, FreeStyle Libre; G4, Dexcom G4 CGM; G6, Dexcom G6 CGM; G7, Dexcom G7 CGM; GDH, glucose dehydrogenase; GOx, glucose oxidase; ISF, interstitial fluid; ISO, International Standards Organization; MARD, mean absolute relative difference; NAC, N-acetyl-
Declaration of Conflicting Interests
The author declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Steven John Setford is an employee of LifeScan Scotland Ltd.
Funding
The author disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Steven John Setford is a salaried employee of LifeScan Scotland Ltd.
