Abstract
Background:
Optimization of automated insulin delivery (AID) settings is required to achieve desirable glycemic outcomes. We evaluated safety and efficacy of a computerized system to initialize and adjust insulin delivery settings for the t:slim X2 insulin pump with Control-IQ technology in adults with type 1 diabetes (T1D).
Methods:
After a 2-week continuous glucose monitoring (CGM) run-in period, adults with T1D using multiple daily injections (MDI) (N = 33, mean age 36.1 years, 57.6% female, diabetes duration 19.7 years) were transitioned to 13 weeks of Control-IQ technology usage. A computerized algorithm generated recommendations for initial pump settings (basal rate, insulin-to-carbohydrate ratio, and correction factor) and weekly follow-up settings to optimize glycemic outcomes. Physicians could override the automated settings changes for safety concerns.
Results:
Time in range 70 to 180 mg/dL improved from 45.7% during run-in to 69.1% during the last 30 days of Control-IQ use, a median improvement of 18.8% (95% confidence interval [CI]: 13.6-23.9, P < .001). This improvement was evident early in the study and was sustained over 13 weeks. Time <70 mg/dL showed a gradual decreasing trend over time. Percentage of participants achieving HbA1c <7% went from zero at baseline to 55% at study end (P < .001). Only six of the 318 automated settings adaptations (1.9%) were overridden by study investigators.
Conclusions:
Computerized initiation and adaptation of Control-IQ technology settings from baseline MDI therapy was safe in adults with T1D. The use of this simplified system for onboarding and optimizing Control-IQ technology may be useful to increase uptake of AID and reduce staff and patient burden in clinical care.
Introduction
Automated insulin delivery (AID) has been shown to improve outcomes in people with type 1 diabetes (T1D). 1 A recent international consensus recommended AID therapy for most individuals with T1D to improve diabetes care and outcomes. 2
Automated insulin delivery onboarding is often performed at specialized diabetes centers, as it requires trained staff to onboard a person with T1D on an AID system, and depending on the system, requires programming safe insulin delivery settings into the pump (e.g. basal rate [BR], insulin to carbohydrate ratio [ICR], and correction factor [CF]). In addition, glycemic outcomes with AID use depend on optimizing settings for each individual over time. This remains a significant challenge for both patients and providers, especially in primary-care practices.
To reduce this burden, some AID systems now include automated correction boluses to help users improve time in the target range. 3 Other systems have opted to adapt BRs or other internal model parameters over time.4-6 Many reports have been published looking at the effects of adjusting user profile settings for commercially available AID systems to achieve optimal clinical outcomes, but the specific settings to change, as well as what settings are user adjustable, are different for every system.7,8 Given that all of these devices work slightly differently, this creates an ever-growing burden for patients and clinicians to understand how each system functions. 9
The t:slim X2 insulin pump with Control-IQ technology (Tandem Diabetes Care, San Diego, CA) is an AID system that has been shown to improve hemoglobin A1c (HbA1c), time in target range (TIR) 70 to 180 mg/dL and reduce time below 70 mg/dL (time below range; TBR) in youth and adults with T1D from age 2 to 72. 10 Tandem diabetes care developed an automated decision support system that provides recommendations for initial and ongoing changes in pump settings to improve glycemic outcomes. We evaluated the system in a safety and feasibility trial of this AID onboarding and optimization tool among adults with T1D using multiple daily injections (MDI).
Methods
This study tested a settings initialization and adaptation tool developed at Tandem Diabetes Care, which consists of an algorithm that analyzes data from the Control-IQ system and suggests optimal settings. The calculations were derived from analyzing data across the large customer base of Control-IQ technology users who have consented for Tandem to use their data. For the settings initialization, the tool primarily used total daily basal insulin dose to select starting values for BR, ICR, and CF per a preset algorithm. For the ongoing adaptation, current settings and glycemic data from fasting and postprandial periods were used to generate a recommendation for settings adjustments up to 5% change at one time to improve time in range while maintaining nominal hypoglycemia.
This algorithm was tested in a single-center, prospective, single-arm study. The study was approved by the Colorado Multiple Institutional Review Board, and informed consent was obtained from each participant. The U.S. Food and Drug Administration (FDA) approved an investigational device exemption, and the study was registered at ClinicalTrials.gov (NCT05204134).
Adults with T1D who were using MDI therapy and who met eligibility criteria were enrolled at a single clinical site (Barbara Davis Center for Diabetes, Aurora, CO). Key inclusion criteria were (1) adults age ≥18 years old with T1D for at least 1 year using MDI therapy, (2) total daily insulin (TDI) dose ≥ 10 units/day, (3) HbA1c between 7.5 and 11% at screening, (4) not pregnant or planning pregnancy, and (5) willingness to follow all study procedures. Key exclusion criteria were (1) two or more episodes of severe hypoglycemia needing assistance in the past 6 months, (2) two or more episodes of diabetic ketoacidosis (DKA) in the past 6 months, (3) significant chronic kidney disease or hemodialysis, (4) significant liver disease, (5) adrenal insufficiency, or (6) other long-term conditions determined by investigator to interfere with participation in the study. Use of any insulin other than U-100 Lispro or Aspart was not allowed with the pump. Use of any noninsulin glucose-lowering agents other than metformin was also not allowed. A listing of all eligibility criteria is included in the Supplemental Material.
Participants completed a continuous glucose monitoring (CGM) run-in period for 2 weeks if they had no prior Dexcom G6 CGM data. Otherwise, their prior home CGM data for the last 2 weeks was used. Participants were then trained on the t:slim X2 insulin pump with Control-IQ technology and used the system for 13 weeks. The settings initialization calculations used in the study was based on population data and used total daily basal insulin dose to select starting values for BR, ICR, and CF per a preset algorithm.
Participants uploaded their pump on day 3, day 7, and every 7 days, thereafter. After each upload, the adaptation algorithm generated a BR, ICR, and CF for each participant (one rate/setting to be used for the entire 24-hour day), limited to a maximum 5% change at a time. These recommended changes were sent to providers at the clinical site for their review, which they then relayed to the participants to manually change on the pump, and verified with a pump upload. Providers could override the automated settings changes for safety concerns. If the user or study provider manually changed settings to more than one timepoint per day, the settings adaptation system continued to adapt back to one setting per day for BR, ICR, and CF.
Central laboratory HbA1c was measured at the pump training visit and again at the 13-week final study visit. A semistructured one-on-one interview was offered concurrent with the 13-week visit or within a 28-day period following that visit to all participants. Participants also completed the Diabetes Impact and Device Satisfaction (DIDS) scale at the 13-week visit, an 11-question validated survey that measures diabetes impact and device satisfaction. 11
Safety outcomes included severe hypoglycemia (defined as hypoglycemia requiring assistance because of an altered cognitive state), DKA; defined by the criteria established in the Diabetes Control and Complications Trial), 12 and other adverse events. Continuous glucose monitoring outcomes included TIR 70 to 180 mg/dL, time >180 mg/dL, time >250 mg/dL, time <70 mg/dL, time <54 mg/dL, time in the tight glycemic range 70 to 140 mg/dL, and median glucose, for the overall period, daytime and overnight, compared from run-in to the last 30 days of Control-IQ technology use. The number of provider overrides/manual adjustments to pump settings were also tracked. Total daily, basal and bolus insulin doses were also calculated. Patient-reported outcomes are reported for the DIDS scale.
Clinical outcomes were computed separately for each participant at multiple frequencies. For each outcome, the CGM run-in baseline period was compared to the overall 13-week study and to each 30-day interval (labeled “Day 30,” “Day 60,” and “Day 90”). First, normality of each measure was tested via a D’Agostino-Pearson Test, then normal groups were compared via a Paired Samples T-Test, and nonnormal groups (most in this study) were compared via a Wilcoxon Signed-Rank Test. A full Bonferroni correction was applied to account for repeated measures across these time intervals.
Results
Thirty-five participants signed the consent form to enroll in the study. Two participants failed screening. Demographics for all participants who passed screening criteria and began study device use (N = 33) are listed in Table 1. Of these, there were 14 participants with a screening visit HbA1c in the 8% to 8.9% range, and nine participants with a screening HbA1c >9%. Participants had a wide range of prior CGM experience, ranging from none (N = 9) to >5 years of use (N = 8).
Participant Demographics Who Started Study Device Use (N = 33).

Abbreviations: SD, standard deviation; BMI, body mass index; CGM, continuous glucose monitor.
During the study, three participants dropped out shortly after pump start (on day of pump start, 4 days after pump start, and 10 days after pump start, respectively). There were no DKA events. There was one participant who, early in the study, was admitted to the hospital for pancreatitis and then later had two episodes of severe hypoglycemia needing assistance. The first event was a low after a breakfast meal bolus followed by increased physical activity. The second event was also after breakfast on a different day. This participant was withdrawn from the study by the investigator and medical monitor on Day 71. There were no other severe hypoglycemic events. In all, 29 participants finished the full study.
Time in closed loop averaged 95.3%. Overall glycemic results are listed in Table 2. Glycemic outcomes improved from baseline to the end of 13 weeks, and at 30-day intervals (Day 30, 60, and 90). TIR 70-180 mg/dL improved from 45.7% during run-in to 69.1% during the last 30 days of Control-IQ use, with most of the improvement in TIR evident early on, and then sustained through 13 weeks (Figure 1). This represents a median improvement of 18.8% TIR 70 to 180 mg/dL (95% confidence interval [CI]: 13.6-23.9, P < .001). There was also an increase in percentage of time in the tight target range 70 to 140 mg/dL. There were decreases in percentage of time >180 mg/dL, percentage of time >250 mg/dL, and median glucose. Time below range showed a gradual decreasing trend over time (without a decrease in time in range), although the differences were not statistically significant after adjusting for multiple comparisons.
Median (IQR) CGM Outcomes Overall and by 30-Day Interval, Compared to the Run-in Phase.
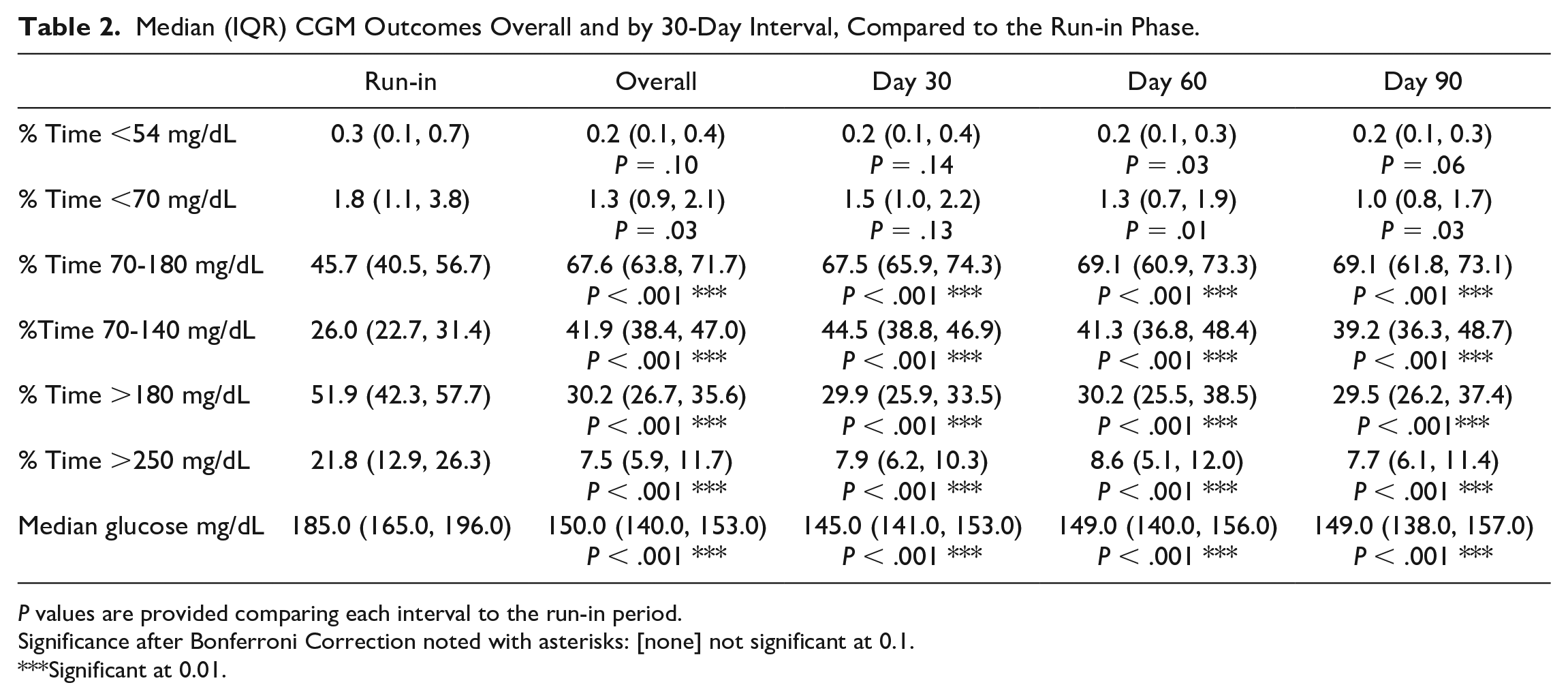
P values are provided comparing each interval to the run-in period.
Significance after Bonferroni Correction noted with asterisks: [none] not significant at 0.1.
Significant at 0.01.
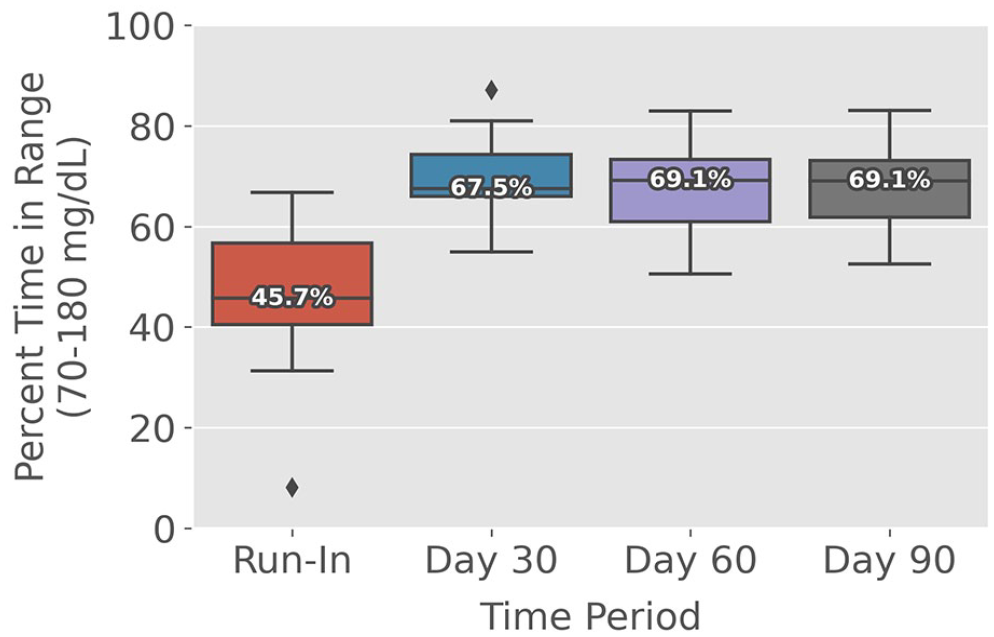
Median time in range 70 to 180 mg/dL compared from run-in through each 30-day period.
Similar improvements were seen for daytime and nighttime (Supplemental Table 1), with nighttime outcomes showing the highest time in range, consistent with prior studies of Control-IQ technology.10,13-15 Insulin use showed no differences between run-in and the last 30 days of Control-IQ use (Table 3).
Median (IQR) Insulin Use Overall and by 30-Day Interval, Compared to the Run-in Phase.
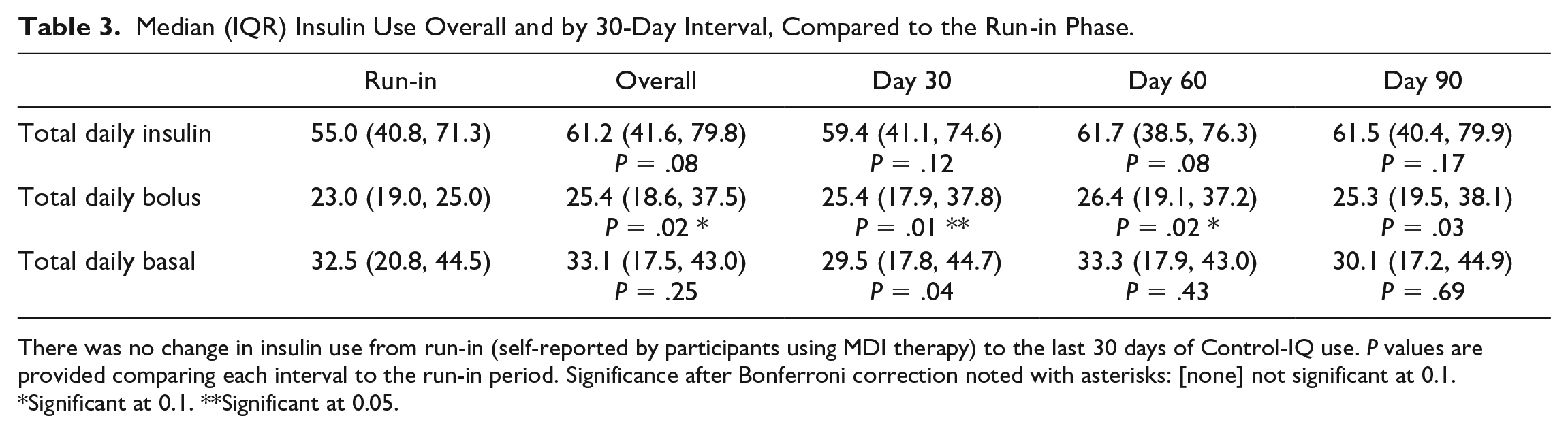
There was no change in insulin use from run-in (self-reported by participants using MDI therapy) to the last 30 days of Control-IQ use. P values are provided comparing each interval to the run-in period. Significance after Bonferroni correction noted with asterisks: [none] not significant at 0.1.
Significant at 0.1. **Significant at 0.05.
The percentage of participants achieving an HbA1c <7% went from zero at baseline to 55% at study end (P < .001).
Of the 33 settings initializations performed with the settings initialization algorithm, there was one manual override. Out of 318 automated settings changes recommended by the system during the course of the study, there were only 6 (1.9%) manual overrides of the adaptation system by study physicians (Supplemental Table 2). There were no overrides by the users alone. An analysis of the automated settings changes for BR and ICR showed many participants were recommended less insulin earlier in the study, with BR decreasing and ICR increasing during the first week. After that, there was no consistent pattern to the recommendations, as settings changes were personalized for each participant. Correction factor was adapted in line with ICR and BR.
Patient-reported outcomes were collected at the end of the clinical trial via the DIDS scale. Results were comparable with other recent studies using the Tandem t:slim X2 insulin pump, 16 with a high Device Satisfaction score of 8.9 (1.1) out of 10, and a low Diabetes Impact score of 3.5 (2.2) out of 10. Six participants agreed to perform the semistructed interviews at the end of the study. Each interview lasted ~30 minutes using a script of open-ended questions to gather feedback and reactions to the use of Control-IQ technology as well as trust and confidence in the automated therapy settings recommendations. All participants reported a positive experience, particularly due to improved glucose control, relief from the burden of managing glucose, improved sleep, and overall better quality of life. Participants were generally confident with the settings that were recommended to them, and while two of the six participants questioned some therapy suggestions, they were comfortable enough with the recommended changes to implement them and felt in the end they had well-tuned therapy settings.
Conclusions
Automated therapy settings initialization and adaptation was demonstrated to be safe with Control-IQ technology use and contributed to a significant improvement in glycemic control. In prior randomized, controlled pivotal trials of adults, children, and preschoolers with T1D, TIR 70 to 180 mg/dL improved an average 11.5% with use of Control-IQ technology compared to the control arm.10,13-15 Similar results were seen in the pivotal trial of the insulin-only bionic pancreas, with an 11% improvement in TIR 70 to 180 mg/dL. 17
In our study, time in range improved far more than in those prior pivotal trials. Median TIR 70 to 180 mg/dL in this study improved from 45.7% to 69.1%, without physician intervention to initialize and then optimize user profile settings. Median glucose also improved from 185 mg/dL at baseline to 149 mg/dL at the end of the study. While the study design and outcomes were not aimed to differentiate glycemic improvement due to Control-IQ technology compared to settings adaptation, the longitudinal changes are interesting to note. The significant improvement in TIR occurred immediately with the introduction of Control-IQ technology. This was likely due primarily to the addition of Control-IQ technology. Time below range, however, was more incrementally reduced over the 90-day course of trial, from 1.8% at baseline, to 1.5%, 1.3%, and 1.0% at 30, 60, and 90 days, respectively. This gradual reduction in hypoglycemia is best explained by the gradual adaptation of settings over the course of the trial, when Control-IQ use was consistent throughout. Future trials are needed beyond safety and feasibility to understand the additive glycemic effects of the two technologies.
Few resources exist to help health care providers optimize settings for AID systems. As a result, standard insulin pump dosing guidelines are often used.18,19 Previous reports show a wide range of settings in use with AID systems that vary across age groups. 20 While it would be ideal to regularly tune these insulin delivery settings to improve outcomes, most users of diabetes devices do not download or analyze data from these devices at regular intervals. 21 In addition, only 20% of clinicians feel they have time to work with patients with diabetes devices, have positive attitudes around diabetes technology, and feel they can keep up with the advancing technology. 22 An automatic initialization and adaptation system allows for frequent adjustment of these settings without additional workload.
As automation advances, designing for trust is critical, and health care providers may need to accept that a thoroughly, clinically validated algorithm is designed for useful clinical outcomes. 23 Previous research has indicated that when humans are given a choice between their own judgment and an algorithm, they generally choose the former. 24 That there were less than 2% manual overrides of the adaptation system by study physicians indicated high trust in the system. The study physicians’ knowledge of insulin dosing titration with AID systems may have helped contribute to their trust of the adaptation as well. It is possible that future acceptance of automated adjustments may follow a diffusion curve of more knowledgeable health care providers adopting first, followed by those with less comfort in insulin titration. Ultimately, trusting the automation to handle the task of frequent settings changes may reduce time needed for clinic visits, reduce burden on patients and providers, and ultimately lead to cost savings.
In this study, there was no specific pattern to the recommended settings changes across the entire study cohort. This speaks to the importance of individualizing settings optimizations. At the same time, TDI did not change. Although some individuals would be expected to have a lower TDI after starting pump therapy, those with a higher HbA1c, such as this study population with a mean HbA1c of 8.5% at baseline, are often under-insulinized and would be expected to use more insulin with AID therapy.
Strengths of this study include recruitment of participants who were all on MDI therapy, so all participants were able to use both the initialization and the adaptation elements of the system. In addition, at screening, most of the participants had an HbA1c >8%, and none had a HbA1c <7%, indicating this was a population likely to benefit from continued updates to their profile settings. These participants had a wide variety of prior CGM use, ranging from none to many years. By the end of the study over half of the participants achieved their HbA1c goal of <7%. Although the system used in this study required manual updating of the recommended user profile settings, users gave the system high satisfactions scores and noted a low impact on diabetes management. Even with settings changes limited to only 5% maximum change each week, these changes could accumulate over the 13 weeks, allowing settings to double or halve from their initial values.
There are limitations as well. As a safety and feasibility study, the sample size was relatively small. The study was not powered to show specific changes in glycemic outcomes. Furthermore, there was not a control arm of people using Control-IQ technology without the settings adaptation, which makes it difficult to evaluate the effect of the settings adaptation alone.
In conclusion, computerized therapy settings initialization and adaptation, implemented with Control-IQ technology, was safe in this population of MDI users who showed a substantial improvement in time in range. The use of this simplified system for onboarding and optimizing Control-IQ technology may be useful to increase uptake of AID and reduce staff and patient burden in clinical care.
Supplemental Material
sj-docx-1-dst-10.1177_19322968241229074 – Supplemental material for Safety and Feasibility Evaluation of Automated User Profile Settings Initialization and Adaptation With Control-IQ Technology
Supplemental material, sj-docx-1-dst-10.1177_19322968241229074 for Safety and Feasibility Evaluation of Automated User Profile Settings Initialization and Adaptation With Control-IQ Technology by Viral N. Shah, Halis K. Akturk, Alex Trahan, Nicole Piquette, Alex Wheatcroft, Elain Schertz, Karen Carmello, Lars Mueller, Kirstin White, Larry Fu, Ravid Sassan-Katchalski, Laurel H. Messer, Steph Habif, Alex Constantin and Jordan E. Pinsker in Journal of Diabetes Science and Technology
Supplemental Material
sj-docx-2-dst-10.1177_19322968241229074 – Supplemental material for Safety and Feasibility Evaluation of Automated User Profile Settings Initialization and Adaptation With Control-IQ Technology
Supplemental material, sj-docx-2-dst-10.1177_19322968241229074 for Safety and Feasibility Evaluation of Automated User Profile Settings Initialization and Adaptation With Control-IQ Technology by Viral N. Shah, Halis K. Akturk, Alex Trahan, Nicole Piquette, Alex Wheatcroft, Elain Schertz, Karen Carmello, Lars Mueller, Kirstin White, Larry Fu, Ravid Sassan-Katchalski, Laurel H. Messer, Steph Habif, Alex Constantin and Jordan E. Pinsker in Journal of Diabetes Science and Technology
Supplemental Material
sj-docx-3-dst-10.1177_19322968241229074 – Supplemental material for Safety and Feasibility Evaluation of Automated User Profile Settings Initialization and Adaptation With Control-IQ Technology
Supplemental material, sj-docx-3-dst-10.1177_19322968241229074 for Safety and Feasibility Evaluation of Automated User Profile Settings Initialization and Adaptation With Control-IQ Technology by Viral N. Shah, Halis K. Akturk, Alex Trahan, Nicole Piquette, Alex Wheatcroft, Elain Schertz, Karen Carmello, Lars Mueller, Kirstin White, Larry Fu, Ravid Sassan-Katchalski, Laurel H. Messer, Steph Habif, Alex Constantin and Jordan E. Pinsker in Journal of Diabetes Science and Technology
Footnotes
Abbreviations
AID, automated insulin delivery; BR, basal rate; CF, correction factor; CGM, continuous glucose monitor; DIDS, Diabetes Impact and Device Satisfaction; DKA, diabetic ketoacidosis; ICR, insulin-to-carbohydrate ratio; MDI, multiple daily injections; T1D, type 1 diabetes; TBR, time below range; TIR, time in target range.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: VNS has received research grants through University of Colorado from Tandem Diabetes Care, Insulet, NovoNordisk, Alexion, JDRF and NIH. He has received fees through University of Colorado for speaking or consulting work for Dexcom, Insulet, Tandem Diabetes Care, Embecta, NovoNordisk and Medscape. HKA has received research grants through University of Colorado from Tandem Diabetes Care, Medtronic, Dexcom, Eli Lilly, Mannkind. He has received consulting fees through University of Colorado from Tandem Diabetes Care, Medtronic and Dexcom. AT, NP, AW, ES, KC, LM, KW, LF, RSK, LM, SH, AC, and JEP are employees of Tandem Diabetes Care, Inc.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by Tandem Diabetes Care, Inc.
Prior Publication
Data from this study was presented in abstract form at the Advanced Technologies & Treatment for Diabetes (ATTD) meeting, February 2023.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
