Abstract
Background:
Control-IQ technology version 1.5 allows for a wider range of weight and total daily insulin (TDI) entry, in addition to other changes to enhance performance for users with high basal rates. This study evaluated the safety and performance of the updated Control-IQ system for users with basal rates >3 units/h and high TDI in a multicenter, single arm, prospective study.
Methods:
Adults with type 1 diabetes (T1D) using continuous subcutaneous insulin infusion (CSII) and at least one basal rate over 3 units/h (N = 34, mean age = 39.9 years, 41.2% female, diabetes duration = 21.8 years) used the t:slim X2 insulin pump with Control-IQ technology version 1.5 for 13 weeks. Primary outcome was safety events (severe hypoglycemia and diabetic ketoacidosis (DKA)). Central laboratory hemoglobin A1c (HbA1c) was measured at system initiation and 13 weeks. Participants continued using glucagon-like peptide-1 (GLP-1) receptor agonists, sodium-glucose transport protein 2 (SGLT-2) inhibitors, or other medications for glycemic control and/or weight loss if on a stable dose.
Results:
All 34 participants completed the study. Fifteen participants used a basal rate >3 units/h for all 24 hours of the day. Nine participants used >300 units TDI on at least one day during the study. There were no severe hypoglycemia or DKA events. Time in range 70-180 mg/dL was 64.8% over the 13 weeks, with 1.0% time <70 mg/dL. Hemoglobin A1c decreased from 7.69% at baseline to 6.87% at 13 weeks (−0.82%, P < .001).
Conclusions:
Control-IQ technology version 1.5, with wider range of weight and TDI input and enhancements for users with high insulin requirements, was safe in individuals with T1D in this study.
Introduction
The Tandem t:slim X2 insulin pump with Control-IQ technology has been evaluated in multiple randomized controlled trials, showing a mean adjusted group difference of 11.5% improvement in time in range (TIR) across adults, children, and preschoolers with type 1 diabetes (T1D) compared with the control group.1-3 No subgroups were identified that did not benefit from Control-IQ use. 4
The previous version of Control-IQ technology was limited to entry of weight of 25 to 140 kg (55-308 lbs) and entry of total daily insulin (TDI) of 10 to 100 units/day. In the pivotal trial of adolescents and adults with Control-IQ technology, average TDI was 55 units/day with use of the system over six months. 3 Similar insulin use was seen in the pivotal trial of the OmniPod 5, with average TDI use of 46.4 units/day in adolescents and adults. 5 However, with obesity and insulin resistance increasingly emerging in individuals with T1D, and associated higher insulin requirements, 6 there is a need to optimize automated insulin delivery (AID) systems to reflect greater insulin use.
In November 2023, Food and Drug Administration (FDA) cleared version 1.5 of the Control-IQ algorithm, which allows for a wider range of weight and TDI entry. The updated system also removes the 3 unit/h basal rate clipping present in previous versions of Control-IQ technology, potentially enhancing performance and outcomes for users with high programmed basal rates. This study evaluated the safety and performance of the updated system in individuals with T1D with basal rates greater than 3 units/h and high insulin requirements in a multicenter, single arm, prospective study.
Methods
The multicenter study was approved by the Advarra Institutional Review Board, and informed consent was obtained from each participant. The FDA approved an investigational device exemption, and the study was registered at ClinicalTrials.gov (NCT05422053).
Adults with T1D using continuous subcutaneous insulin infusion (CSII) and at least one basal rate over 3 units/h were recruited from four clinical centers across the United States. Key inclusion criteria were as follows: (1) age ≥18 years old and diagnosed with T1D for at least one year, (2) currently using CSII with any brand of insulin pump for at least three months, and planned to use at least one basal rate above 3 units/h with the study pump, and (3) hemoglobin A1c (HbA1c) <10.5%. Key exclusion criteria were as follows: (1) more than one episode of severe hypoglycemia (needing assistance) or diabetic ketoacidosis (DKA) in the past six months, (2) pregnancy, (3) unstable dose of glucagon-like peptide-1 (GLP-1) receptor agonist, sodium-glucose transport protein 2 (SGLT-2) inhibitor, or other medication for glycemic control/weight loss in the last three months, (4) starting a new non-insulin glucose lowering or weight loss agent during the trial, (5) adrenal insufficiency, and (6) other chronic condition determined by investigator to interfere with participation in the study. The use of any insulin other than U-100 Lispro or Aspart was not allowed with the pump. Prior continuous glucose monitoring (CGM) experience was not required. Full inclusion/exclusion criteria are included in the Supplemental Material.
Given that participants with high insulin requirements and high basal rates might already be using additional medications beyond insulin for glycemic control and/or weight loss, participants were allowed to continue use of GLP-1 receptor agonists, SGLT-2 inhibitors, or other medications for glycemic control and/or weight loss if on a stable dose for the last three months. Participants on GLP-1 receptor agonist or SGLT-2 inhibitor therapy were given additional ketone monitoring guidelines.
After screening, participants were trained on the Tandem t:slim X2 insulin pump with Control-IQ technology version 1.5 and the Dexcom G6 CGM and used the system for 13 weeks. Participants were contacted by phone on day 3, day 7, and day 21. There was a six week in-clinic visit to download pump data, a phone contact at nine weeks, and final visit at week 13. Central laboratory HbA1c was measured at system initiation and at 13 weeks.
The primary outcome was safety events for severe hypoglycemia (defined as hypoglycemia requiring assistance due to an altered cognitive state) and DKA (defined by the criteria established in the Diabetes Control and Complications Trial). 7 Continuous glucose monitoring outcomes included time in closed-loop, TIR 70 to 180 mg/dL, time >180 mg/dL, time >250 mg/dL, time <70 mg/dL, time <54 mg/dL, time in the tight glycemic range 70 to 140 mg/dL, coefficient of variation (CV), standard deviation (SD), and mean glucose, for the overall 13 week period, daytime and overnight. Total daily insulin was also calculated at baseline and over the 13 week period.
Results
Thirty-four participants started study device use, and all 34 completed the trial. Mean age was 39.9 ± 11.9 years, 41.2% were female, and diabetes duration was 21.8 ± 11.2 years. Mean TDI at enrollment was 1.2 units/kg per day with a range of 0.5 to 2.0 units/kg per day. At enrollment, 12 participants were using a TDI between 101 and 150 units/day, 11 participants reported a TDI between 151 and 199 units/day, and three participants were using TDI ≥200 units/day, reflecting high insulin use. Body mass index (BMI) ranged from 30 to 52.1 kg/m2. Although the majority of participants (53%) were prior Tandem t:slim X2 pump users, 38% were using a MiniMed 600/700 series pump and 9% were using an Insulet OmniPod at the time of enrollment. Almost all (97.1%) were current CGM users. Central laboratory HbA1c was 7.69 ± 1.08% at Control-IQ start. Full demographics are listed in Table 1.
Participant Demographics (N = 34).

Ten participants were taking metformin, three were taking a GLP-1 receptor agonist (N = 2 semaglutide; N = 1 liraglutide), one participant was taking an SGLT-2 inhibitor (dapagliflozin), and one participant was on phentermine for weight loss throughout the study.
During the 13 weeks of Control-IQ use, there were no severe hypoglycemia or DKA events.
Central laboratory measured HbA1c decreased from 7.69 ± 1.08% at Control-IQ start to 6.87 ± 0.57% at 13 weeks, a mean change of −0.82 ± 0.73% (95% confidence interval [CI] = −1.08, −0.57; P < .001). Hemoglobin A1c improved for all ranges of TDI use with Control-IQ technology (TDI ≤100 units/day, 100-149 units/day, 150-199 units/day, and ≥200 units/day) with the greatest improvement of −1.21 ± 0.67% for mean TDI use ≥200 units/day (Figure 1 and Supplemental Table 1).
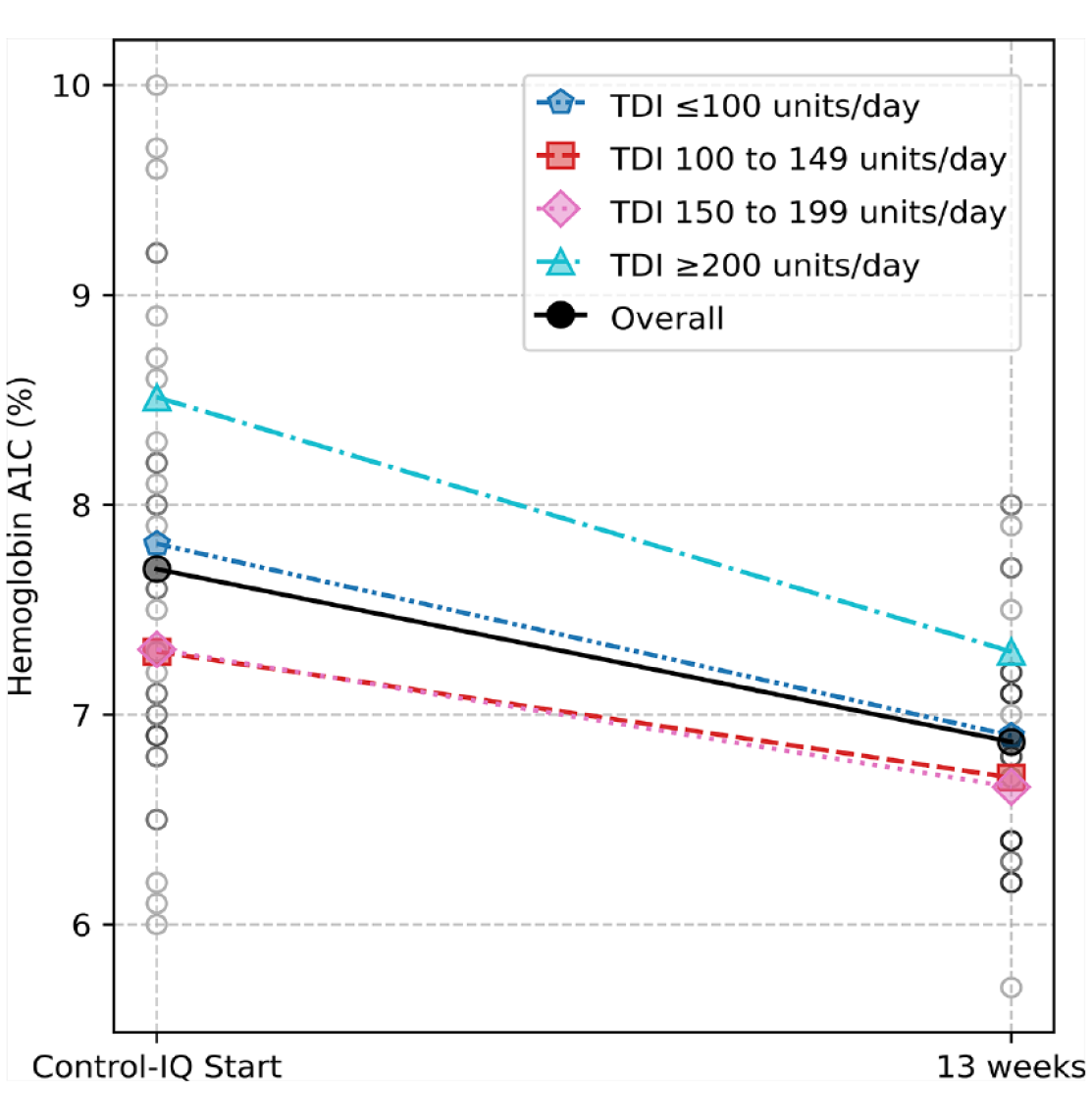
Change in hemoglobin A1c for different ranges of TDI use with Control-IQ technology version 1.5. Central laboratory measured hemoglobin A1c decreased from 7.69 ± 1.08% at Control-IQ start to 6.87 ± 0.57% at 13 weeks overall, a mean change of −0.82 ± 0.73% (95% CI = −1.08, −0.57; P < .001). Similarly, HbA1c improved for all ranges of TDI use with Control-IQ technology, with the greatest improvement shown for users averaging ≥200 units/day (mean change −1.21 ± 0.67%, P < .001).
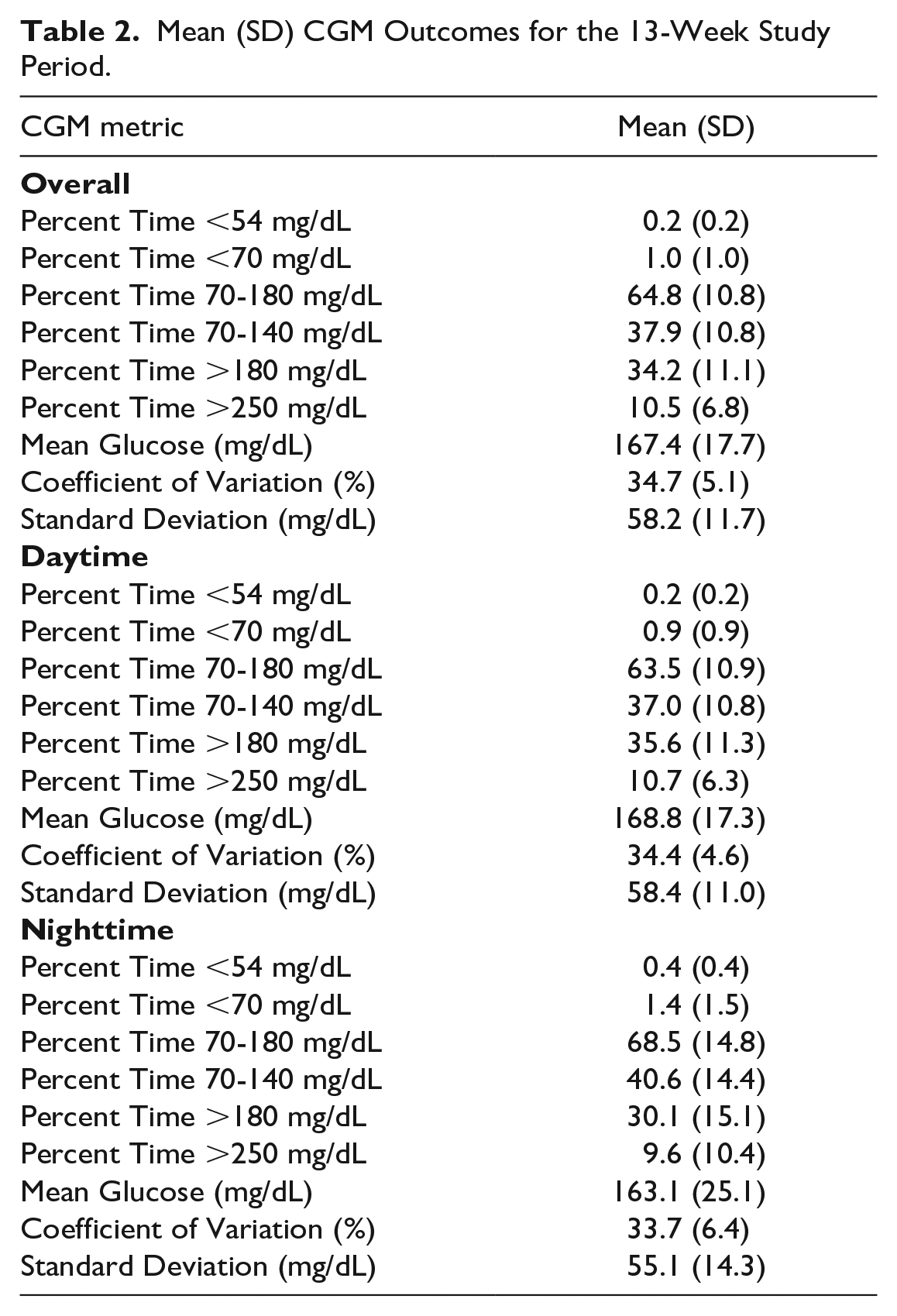
Glycemic outcomes for the 13 week period are shown in Table 2. Sensor use was 97.9 ± 1.3%, with 93.1 ± 3.5% time in closed loop. Time in range 70-180 mg/dL was 64.8 ± 10.8% overall, with only 1.0 ± 1.0% time below 70 mg/dL. This TIR was achieved in the first week of Control-IQ use and was sustained throughout the study.
Mean (SD) CGM Outcomes for the 13-Week Study Period.
A wide range of TDI and insulin delivery profile settings were used with the study device. All participants met the inclusion criteria of using at least one programmed basal rate >3 units/h. Fifteen participants (44%) used a basal rate >3 units/h all 24 hours of the day throughout the 13 weeks. Twenty-three participants entered a TDI value into the pump of >100 units/day, 15 participants entered a value >150 units/day, and four participants entered a value of 200 units/day (the max enterable TDI) at pump start. The range of TDI administered by the study device was also very high, where 32 participants used >100 units/day on at least one day, 20 participants used >200 units/day on at least one day, and nine participants used >300 units/day on at least one day. Mean TDI increased from 138.6 ± 51.4 units/day at baseline to 155.5 ± 59.7 units/day at 13 weeks, a change of 17.0 ± 32.5 units/day (Table 3). The majority of this change was in bolus insulin, with Control-IQ autoboluses representing 48.2% of all boluses given.
Mean (SD) Insulin Use for the 13-Week Study Period Compared With Baseline.
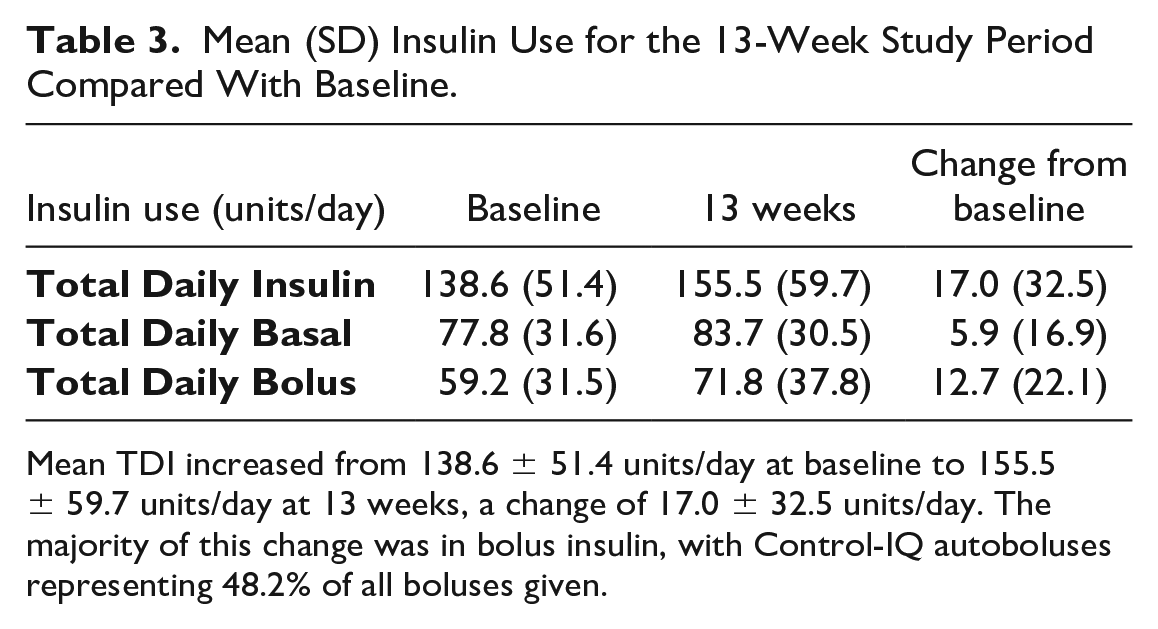
Mean TDI increased from 138.6 ± 51.4 units/day at baseline to 155.5 ± 59.7 units/day at 13 weeks, a change of 17.0 ± 32.5 units/day. The majority of this change was in bolus insulin, with Control-IQ autoboluses representing 48.2% of all boluses given.
There was one severe adverse event, new onset atrial fibrillation, unrelated to the study device. The participant was able to continue device use and completed the study.
Conclusions
With a wider range of allowable weight entry of 9 to 200 kg (20-440 lbs), compared with 25 to 140 kg (55-308 lbs) previously, and TDI entry of 5 to 200 units/day, compared with 10 to 100 units/day previously, Control-IQ technology version 1.5 now supports an expanded range of TDI use. Control-IQ technology version 1.5 also adds support for temporary basal rates and extended boluses up to 8 hours with Control-IQ active.
The updated system also removes the 3 units/h basal rate clipping present in previous versions of Control-IQ technology, potentially enhancing performance and outcomes for users with high programmed basal rates. Although previous versions of Control-IQ could always raise basal rate and/or give autoboluses to address predicted hyperglycemia, Control-IQ technology version 1.5 can now deliver programmed basal rates up to 15 units/h when the 30 minute future glucose prediction is in range. Although Control-IQ technology version 1.5 has been used in prior studies, 8 it had not been specifically evaluated in this population of individuals with T1D and higher insulin requirements. In this study, the system was used to evaluate high insulin use in individuals with T1D, in particular for those using basal rates above 3 units/h. The use of the system was safe in this study, with no DKA or severe hypoglycemia events.
Perhaps the most interesting aspect of this study was the population studied, as previous AID studies have not focused on this population of users with T1D and high insulin requirements. Yet there is clearly a need to evaluate performance of these systems in users with high insulin requirements and to enhance AID systems to improve performance for these users. In this study, at baseline mean HbA1c was 7.7% with use of a variety of different insulin delivery systems, suggesting despite significant insulin use (TDI average of 138.6 units/day at screening) participants were not achieving target A1c goals. After three months of Control-IQ technology version 1.5 use, average HbA1c was below 7%, meeting target.
All participants in the study were at one point in time diagnosed with T1D, but now were taking larger doses of insulin due to insulin resistance. Many of these participants likely had what is now termed “double diabetes,” 9 with a BMI ranging from 30.0 to 52.1 kg/m2, putting them at higher risk for cardiovascular complications and other adverse events. 6 In a recent analysis from the T1D Exchange Registry, 49% of participants were overweight or obese, 10 a major cause of insulin resistance, indicating that the ability to safely accommodate high insulin requirements among individuals with T1D will continue to be important in developing flexible and effective AID systems.
The study additionally afforded the opportunity to examine use of the Control-IQ system by these individuals in a likely real-world setting, where a GLP-1 receptor agonists, SGLT-2 inhibitors, and other medications for glycemic control and/or weight loss are used as adjunct treatments for individuals with T1D. Although only a small number of participants used GLP-1 receptor agonists or SGLT-2 inhibitors, there were no device-related serious adverse events among these participants or in the study overall.
Strengths of the study include a use of the updated Control-IQ version 1.5 system and a prospective study design. Participants had a high mean TDI at baseline yet were not meeting HbA1c goals. By the end of the study, mean HbA1c was below 7%. Limitations of the study include lack of a control arm, and as a safety study, the sample size was relatively small, making it difficult to examine any subgroups. There was no comparison to baseline CGM values, as participants were using a number of different therapy modalities (different pumps and/or sensors) at baseline, preventing accurate comparison with baseline CGM data.
In conclusion, Control-IQ technology version 1.5, with support for a wider range of weight and TDI inputs, and enhancements for users with high insulin requirements, was safe in individuals with T1D in this study.
Supplemental Material
sj-docx-1-dst-10.1177_19322968241234072 – Supplemental material for Control-IQ Technology Use in Individuals With High Insulin Requirements: Results From the Multicenter Higher-IQ Trial
Supplemental material, sj-docx-1-dst-10.1177_19322968241234072 for Control-IQ Technology Use in Individuals With High Insulin Requirements: Results From the Multicenter Higher-IQ Trial by Anders L. Carlson, Timothy E. Graham, Halis K. Akturk, David R. Liljenquist, Richard M. Bergenstal, Becky Sulik, Viral N. Shah, Mark Sulik, Peter Zhao, Peter Briggs, Ravid Sassan-Katchalski and Jordan E. Pinsker in Journal of Diabetes Science and Technology
Supplemental Material
sj-docx-2-dst-10.1177_19322968241234072 – Supplemental material for Control-IQ Technology Use in Individuals With High Insulin Requirements: Results From the Multicenter Higher-IQ Trial
Supplemental material, sj-docx-2-dst-10.1177_19322968241234072 for Control-IQ Technology Use in Individuals With High Insulin Requirements: Results From the Multicenter Higher-IQ Trial by Anders L. Carlson, Timothy E. Graham, Halis K. Akturk, David R. Liljenquist, Richard M. Bergenstal, Becky Sulik, Viral N. Shah, Mark Sulik, Peter Zhao, Peter Briggs, Ravid Sassan-Katchalski and Jordan E. Pinsker in Journal of Diabetes Science and Technology
Footnotes
Acknowledgements
The authors acknowledge Kirstin White, Alex Wheatcroft, Karen Carmelo, Nicole Piquette, and David Berger for their assistance with the study.
Abbreviations
AID, automated insulin delivery; CGM, continuous glucose monitor; CV, coefficient of variation; DKA, diabetic ketoacidosis; GLP-1, glucagon-like peptide-1; SD, standard deviation; SGLT-2, sodium-glucose transport protein 2; HbA1c, hemoglobin A1c; T1D, type 1 diabetes; TDI, total daily insulin.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: A.L.C.’s employer, International Diabetes Center, has received research support and/or consulting fees from Eli Lilly, Novo Nordisk, Sanofi, Medtronic, Tandem, Insulet, Dexcom, Abbott Diabetes, and MannKind. T.E.G.’s employer, Diabetes & Endocrine Treatment Specialists, has received research support/and or consulting fees from Novo Nordisk, Eli Lilly, Sanofi, Provention Bio, Novartis, Tandem, Insulet, Ascendis Pharma, and Progenitor Life Sciences. H.K.A. has received research grants through University of Colorado from Tandem Diabetes Care, Medtronic, Dexcom, Eli Lilly, and MannKind and has received consulting fees through University of Colorado from Tandem Diabetes Care, Medtronic, and Dexcom. D.R.L. has received funding for clinical research from Abbott Diabetes Care, Biolinq, Senseonics, Dexcom, Diasome, Sanvita, Medtronic, Tandem, Provention, Eyenuk, TrialNet, and JAEB Center for Clinical Research. R.M.B. has received research support, has acted as a consultant, or has been on the scientific advisory board for Abbott Diabetes Care, Ascensia, Bigfoot Biomedical, Inc, CeQur, Dexcom, Eli Lilly, Embecta, Hygieia, Insulet, Medtronic, Novo Nordisk, Onduo, Roche Diabetes Care, Tandem Diabetes Care, Sanofi, United Healthcare, Vertex Pharmaceuticals, and Zealand Pharma. R.M.B.’s employer, non-profit HealthPartners Institute, contracts for his services, and he receives no personal income from these activities. B.S. has received payments from Medtronic, Insulet, and Tandem as a certified pump trainer, consulting fees from Eli Lilly, honorariums from Medtronic, the Academy of Nutrition & Dietetics, the Association of Diabetes Care & Education Specialists, and the American Diabetes Association. V.N.S. has received research grants through University of Colorado from Tandem Diabetes Care, Insulet, Novo Nordisk, Alexion, JDRF, and NIH and consulting fees through University of Colorado from Dexcom, Insulet, Tandem Diabetes Care, Embecta, Novo Nordisk, and Medscape. M.S. has received consulting fees from Sanvita. P.Z., P.B., R.S.-K., and J.E.P. are employees of Tandem Diabetes Care, Inc.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by Tandem Diabetes Care, Inc.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
