Abstract

Keywords
Tandem Mobi is a new automated insulin delivery (AID) system that consists of the smallest durable insulin pump in the United States to date, the Control-IQ technology AID algorithm, and supports both Dexcom G6 and G7 continuous glucose monitoring (CGM) sensors. The Control-IQ algorithm is programmed into the pump hardware, the CGM pairs directly to the pump, and the pump and CGM communicate by Bluetooth. The system offers multiple wear options, including an on-body sleeve which allows the user to adhere the pump to their body, similar to a patch pump (Figure 1).
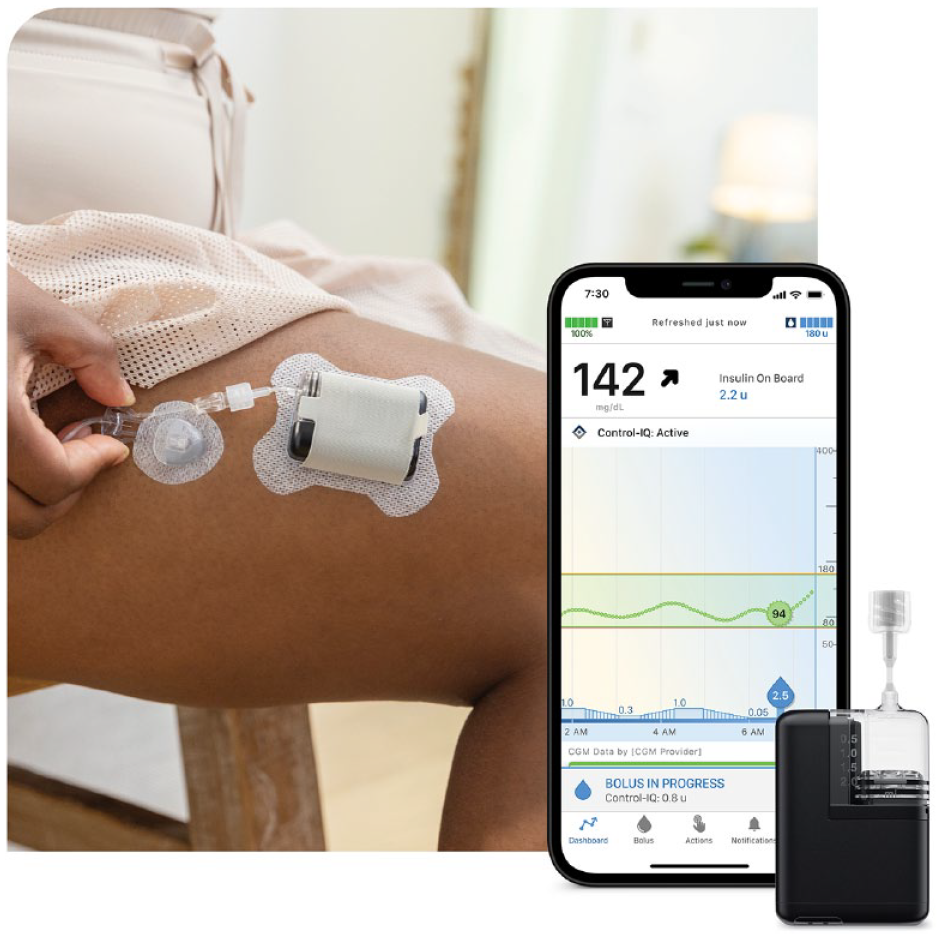
The new, compact AID systems consist of the pump, the Control-IQ technology AID algorithm embedded in the pump, and a CGM that pairs directly to the pump via Bluetooth. As there is no screen on the pump, the user interacts with the system via a smartphone app on their personal phone. The small size of the pump allows for unique wear options, such as using 5-inch short tubing with an adhesive sleeve for an on-body wear experience, similar to a patch pump.
Similar to prior early use evaluations of the Tandem t:slim X2 insulin pump, 1 we evaluated glycemic outcomes and patient satisfaction after six weeks use of this new AID system to determine the early user experience. Results were stratified by previous insulin therapy: multiple daily injections (MDIs), non-Tandem insulin pump users, and Tandem insulin pump users.
A total of 1280 users of the system were included. These users had at least 14 days of CGM data with 70% data availability. Mean age was 35 ± 16.7 years and 62.7% were female. Ninety-seven percent reported as having type 1 diabetes.
Glycemic outcomes for all groups met international consensus guideline goals at six weeks of use. 2 Prior MDI users (n = 235) achieved a median (interquartile range) glycemic management indicator (GMI) of 7.0% (6.6%, 7.4%). During system use, time in range (TIR) was 72.1% (62.5%, 83.8%) and time below range (TBR) was 1.0% (0.5%, 2.2%). Prior non-Tandem users (n = 208) achieved a GMI of 7.0% (6.6%, 7.3%), with TIR of 72.5% (65.0%, 81.3%) and TBR of 1.6% (0.8%, 2.9%).
For prior Tandem pump users (n = 837), all of whom had prior CGM values available, there was a significant reduction in GMI from 7.0% (6.7%, 7.4%) to 6.9% (6.6%, 7.3%; P < .001). For these users, TIR increased significantly from 70.5% (60.5%, 79.0%) to 73.0% (63.8%, 81.3%; P < .001), with a slight but statistically significant increase in TBR from 1.4% (0.6%, 2.4%) to 1.6% (0.8%, 3.0%; P < .001).
A total of 433 users responded to the feedback survey at six weeks. Among them, 114 were previous MDI users and 307 were previous pump users. Seventy-four were children (<18 years old), and 359 were adults. Overall, 84% reported being satisfied or very satisfied with the system, with strong agreement on usefulness and ease of use. Eighty-eight percent reported that the system was consistent with expectations or better than expected. A total of 394 users answered the questions on psychosocial effects of the system. Most of the group agreed or strongly agreed that the system was easy to use (87%), reduced the burden of diabetes management (79%), improved quality of life (85%), helped people to manage their diabetes effectively (86%), helped people to feel more freedom (77%), and people could wear it the way they wanted (79%).
In conclusion, early users of this new AID system met glycemic goals2,3 and reported high satisfaction. Future research should clarify ongoing satisfaction with the system, as indicated by continued use.
Footnotes
Abbreviations
AID, automated insulin delivery; CGM, continuous glucose monitor; GMI, glycemic management indicator; HbA1c, hemoglobin A1c; MDI, multiple daily injection; TBR, time below range; TIR, time in range.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: HKA reports receiving research support and consultation fees from Tandem Diabetes Care, Medtronic, and Dexcom. KEK does not have any conflict of interest. ED, KA, JP, and LM are full-time employees and shareholders of Tandem Diabetes Care.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by Tandem Diabetes Care.
