Abstract
Background:
Painful diabetic neuropathy (PDN) can result in the loss of protective sensation, in which people are at twice the likelihood of foot ulceration and three times the risk of lower extremity amputation. Here, we evaluated the long-term effects of high-frequency (10 kHz) paresthesia-independent spinal cord stimulation (SCS) on protective sensation in the feet and the associated risk of foot ulceration for individuals with PDN.
Methods:
The SENZA-PDN clinical study was a randomized, controlled trial in which 216 participants with PDN were randomized to receive either conventional medical management (CMM) alone or 10 kHz SCS plus CMM, with optional treatment crossover after 6 months. At study visits (baseline through 24 months), 10-g monofilament sensory assessments were conducted at 10 locations per foot. Two published methods were used to evaluate protective sensation via classifying risk of foot ulceration.
Results:
Participants in the 10 kHz SCS group reported increased numbers of sensate locations as compared to CMM alone (
Conclusions:
Significant improvements were observed in protective sensation from preimplantation to 24 months postimplantation for the 10 kHz SCS group. With this unique, disease-modifying improvement in sensory function, 10 kHz SCS provides the potential to reduce ulceration, amputation, and other severe sequelae of PDN.
Trial Registration:
The SENZA-PDN study is registered on ClinicalTrials.gov with identifier NCT03228420.
Background
Worldwide there are 537 million adults with diabetes, approximately 25% of whom will experience painful diabetic neuropathy (PDN).1-3 While pain is often the primary motivation for patients to seek treatment for PDN, these patients often also suffer from reduced sensory function. 4 Intact sensory function of the foot, particularly of the plantar surface, is critically important in preventing loss of protective sensation (LOPS) in people with PDN.5,6 Notably, LOPS is associated with double the likelihood of foot ulceration and triple the likelihood of an extremity amputation, which ostensibly contributes to the 15-fold risk of amputation in people with diabetes compared to people without diabetes.7,8 The 5-year mortality rate after amputation related to diabetes is more than 50%, suggesting that preventing diabetes-related amputation would reduce diabetes-related mortality.9,10 Given the high risk of diabetes-related foot ulceration, amputation, and mortality, there is a clear need to evaluate clinical measures, such as sensory function, that might predict loss of function, deteriorating quality of life, and future health risks for patients with diabetic neuropathy.
Owing to the importance of protective sensation, significant work has been done to stratify foot ulceration risk for diabetic neuropathy patients. For example, the American Diabetes Association (ADA) released a report detailing a comprehensive foot examination and the resulting foot ulceration risk categories. 11 Per this report, LOPS and the associated transition to a higher risk category may be diagnosed using a well-established 10-g monofilament test, an objective assessment in which the patient reports whether or not they can feel the monofilament contact at a number of locations on the plantar surfaces of their feet.12-14
Sensory function has been shown to decline during the progression of diabetes and subsequent damage to the nervous system and, though several interventions exist to manage a PDN patient’s pain, there exist limited data on functional sensory changes as a result of these interventions.15-17 Some interventions, such as improved diet and exercise or insulin treatments, may slow the progression and severity of diabetic neuropathy. 18 However, the data available on treatments to restore function of damaged nerves and the resulting sensory function are limited. For example, in a case series, two patients with PDN had leg numbness reversed after 1 month of gabapentin as assessed by 10-g monofilament, whereas a separate randomized controlled trial (RCT) suggested the efficacy of gabapentin for improving sensory function was similar to that of a placebo.19,20
Beyond pharmacological treatments, several studies have investigated spinal cord stimulation (SCS) as an alternative to treat PDN. Broadly, SCS studies investigating PDN treatment have not collected data for or reported changes in functional sensory outcomes. In one study, the Michigan Diabetic Neuropathy Score, which includes sensory function, was collected for all patients, but only at baseline.21,22 Currently, high-frequency (10 kHz) paresthesia-independent SCS is U.S. Food and Drug Administration (FDA) approved for treating PDN and exclusively recommended for SCS treatment of refractory PDN by the American Association of Clinical Endocrinology 23 and by experts in a recent Clinical Compendia Series from the ADA. 24 In the SENZA-PDN study, a recent RCT with 216 PDN participants, 90% of patients who received 10 kHz SCS had at least 50% pain relief after 24 months of therapy, and 65% of participants had overall sensory function improvement in the lower limbs as determined by clinician assessment. 25 Here, we present a more detailed analysis of the improvement in sensory function observed in the SENZA-PDN study via specific evaluation of the objective monofilament test results and associated classification of foot ulceration risk.
Methods
SENZA-PDN Study Overview
The SENZA-PDN study was a multicenter, prospective, randomized, open-label clinical trial conducted in the United States at 18 centers. Study methods have been described previously. 26 The Western Institutional Review Board and local Institutional Review Boards approved the protocol, consent form, and study documents prior to study commencement at each site. Participants were enrolled after providing written informed consent. The study was conducted in accordance with the Declaration of Helsinki as well as good clinical practices and reported in accordance with the Consolidated Standards of Reporting Trials (CONSORT) reporting guideline. 27 Participants had symptoms of PDN for at least 12 months that were refractory to pharmacological treatment, lower limb pain visual analog scale scores of at least 5 out of 10 cm, hemoglobin A1c ≤ 10%, and body mass index ≤ 45 kg/m2.
In total, 216 participants were randomized to receive either conventional medical management alone (CMM, n = 103) or 10 kHz SCS in addition to CMM (10 kHz, n = 113). Of these 113 participants, 90 received a permanent implant. Participants with <50% pain relief after 6 months in either arm were eligible to cross over into the other study arm. At 6 months, none of the 10 kHz participants crossed over to the CMM arm. In contrast, 93% (n = 64) of the eligible CMM participants crossed over to the 10 kHz treatment arm, 154 participants (90 original plus 64 CMM-to-10 kHz crossover participants) received 10 kHz therapy. Participants who received 10 kHz SCS were followed for 24 months postimplantation.
Sensory Assessments
One goal of the SENZA-PDN study was to evaluate neurological function of participants throughout the study. To address this, two neurologists involved with the study developed a comprehensive neurological examination protocol, including 10-g monofilament sensory testing of the feet. Investigators received training from the two neurologists to perform these examinations. These data were collected from participants at baseline and 3, 6, 12, and 24 months. For participants who crossed over from CMM to 10 kHz, neurological examinations were also conducted at 3, 6, 12, and 24 months postimplantation. As a large majority of participants originally assigned to the CMM group crossed over to the 10 kHz treatment after 6 months, we also analyzed the postimplantation results for this crossover group through the 24-month follow-up visit. To compare responses most similarly at different time points between the original and crossover 10 kHz groups after initiating 10 kHz SCS therapy, the most recent visit prior to implantation (preimplantation) was used as the reference time point for evaluating the effects of 10 kHz SCS.
To assess sensory function via 10-g monofilament testing (Neuropen, Owen Mumford, Woodstock, UK), participants were seated or supine with their feet exposed and asked to close their eyes for each of the 10 test sites on each foot (Figure 1), the monofilament was applied for 1 to 2 s and the participant was asked to describe the sensation as Normal, Hypersensitive, Diminished, or Absent. Normal, Hypersensitive, and Diminished responses were categorized as Sensate, whereas an Absent response was Insensate.
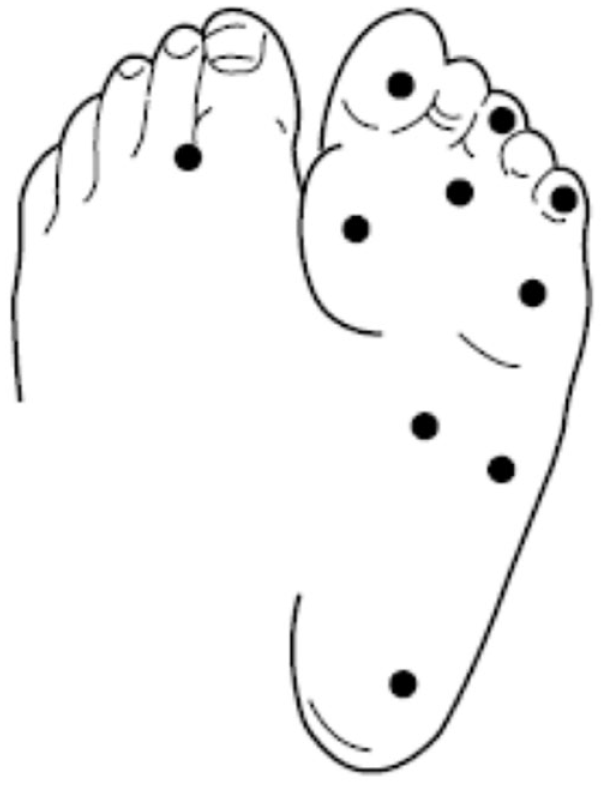
Illustration of 10 test sites used for monofilament assessments. The test sites were (1) distal hallux, (2) third distal phalanx, (3) fifth distal phalanx, (4) first metatarsal head, (5) third metatarsal head, (6) fifth metatarsal head, (7) medial hindfoot, (8) lateral hindfoot, (9) plantar surface of the calcaneus, and (10) one site on the dorsal surface where the first and second toe meet.
Risk Classification Methods Using Monofilament Data
One objective of this analysis was to determine the effect of 10 kHz SCS treatment on the risk of foot ulceration in people with PDN. To this end, two risk classification methods were used. The first was the classification system presented by the ADA Task Force (hereafter referred to as ADA Method), in which four sites per foot were assessed. The four sites assessed were the plantar surfaces of the distal hallux and first, third, and fifth metatarsal heads. If all eight sites were sensate, the participant was in Risk Category 0 (Low Risk), whereas if at least one site was insensate, the participant was in Risk Category 1 (High Risk). 11
The second classification system is based on work investigating the sensitivity and specificity of 10-g monofilament assessments on foot ulceration occurrence when evaluating 10 sites per foot. In that study by Armstrong et al., test specificity increased with little change to sensitivity as the number of insensate responses increased up to four per foot out of 10 sites, but there was a significant decrease in sensitivity and specificity associated with four or more insensate responses. 13 Thus, for the second classification system (hereafter referred to as Armstrong Method), the participant was Low Risk if there were three or fewer insensate responses on both feet and High Risk if there were four or more insensate responses on either foot.
Statistical Methodology
The current statistical analysis evaluated the results during (1) the randomized phase, that is, the 6-month duration after the study baseline visit and (2) the postimplantation phase, that is, the 24-month duration after implantation with a permanent 10 kHz SCS system (this includes the original 10 kHz group and the CMM-to-10 kHz crossover cohort). This analysis was conducted as an exploratory analysis per the study statistical analysis plan. For the ADA and Armstrong risk category analyses, each missing data point was first imputed using multiple imputation at the individual measurement level (Insensate vs. Sensate) for 10 points on each foot using all available response data for each participant. To evaluate time and group effects, estimated odds ratios and test statistics were obtained from a repeated-measures model and summarized using the MIANALYZE procedure in SAS (SAS Institute, Inc., Cary, NC). In the randomized phase, the repeated-measures model included time, group and a time-by-group interaction as fixed effects, and participant as a random effect. In the postimplantation phase, the repeated-measures model included time as a fixed effect and participant as a random effect. An autoregressive correlation structure of order 1 was specified. For the evaluation of 10 kHz versus CMM treatment in the 6-month randomized phase, odds ratios are the odds of a participant being at low risk of foot ulceration, comparing the 10 kHz to CMM groups. For the evaluation of 10 kHz SCS treatment through the 24-month postimplantation phase, odds ratios are the odds of a participant being at low risk of foot ulceration, comparing subsequent time points to preimplantation.
Mean data (±95% confidence intervals) are presented for all data (available data for Sensate/Insensate point analysis and available data plus imputed data for the ADA and Armstrong risk category analysis) at each time point, with SCS data only for participants who received a permanent implant.
Results
Average Responses
To assess broad trends in sensory outcomes, counts of Sensate and Insensate responses were averaged at each time point (Figure 2) during the randomized phase (CMM and 10 kHz groups, Figure 2a) and the postimplantation phase (all implanted participants, Figure 2b). For the randomized phase, baseline evaluations showed no statistical differences between the two treatment groups for the odds of a Sensate response at a given test site with monofilament testing (
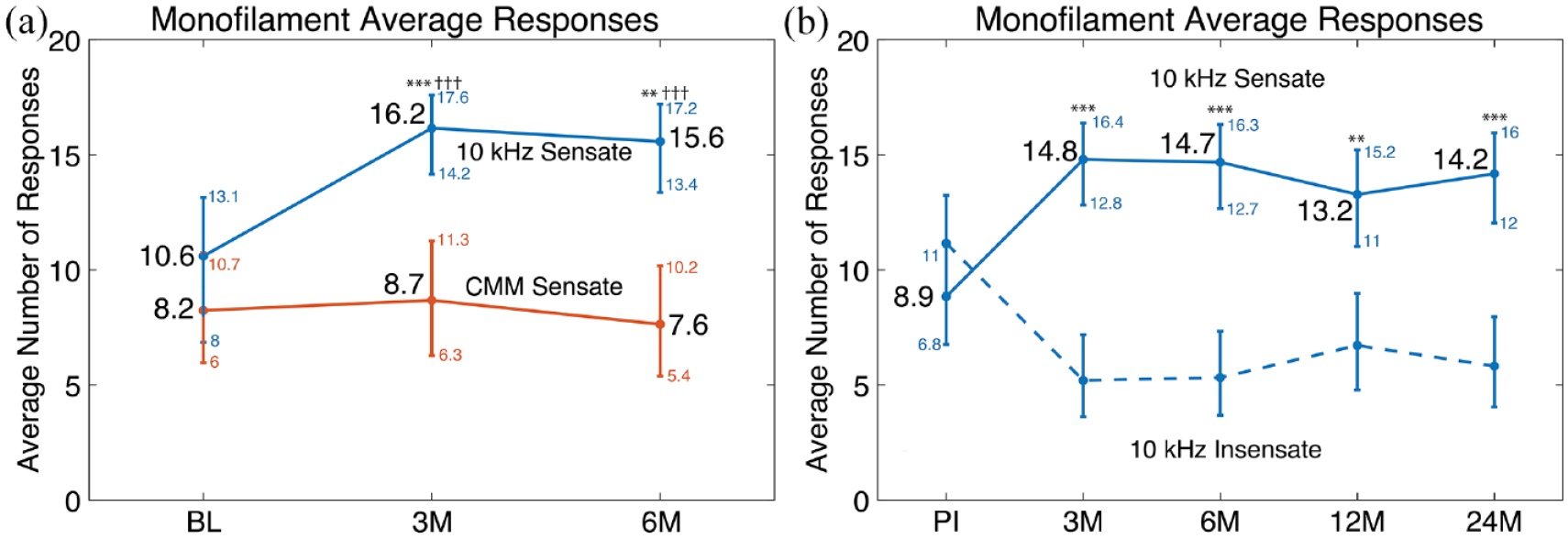
Average number of responses for monofilament assessments. Comparison of average number of Sensate monofilament responses per participant in the 10 kHz and CMM groups from Baseline through 6 months (a). Comparison of average number of Insensate and Sensate monofilament responses per participant with the 10 kHz SCS treatment (including both original 10 kHz SCS arm participants and participants that crossed over from CMM to 10 kHz SCS) from Preimplantation through 24 months (b). For (a), n = 101 (CMM group) and n = 90 (10 kHz group). For (b), n = 154, including both the original 10 kHz arm and CMM-to-SCS crossover participants.
Risk Classification of Monofilament Assessments
Baseline evaluations of the randomized phase showed no statistical differences between the two treatment groups for the odds of being at low risk of foot ulceration (Figure 3) when using either the ADA (
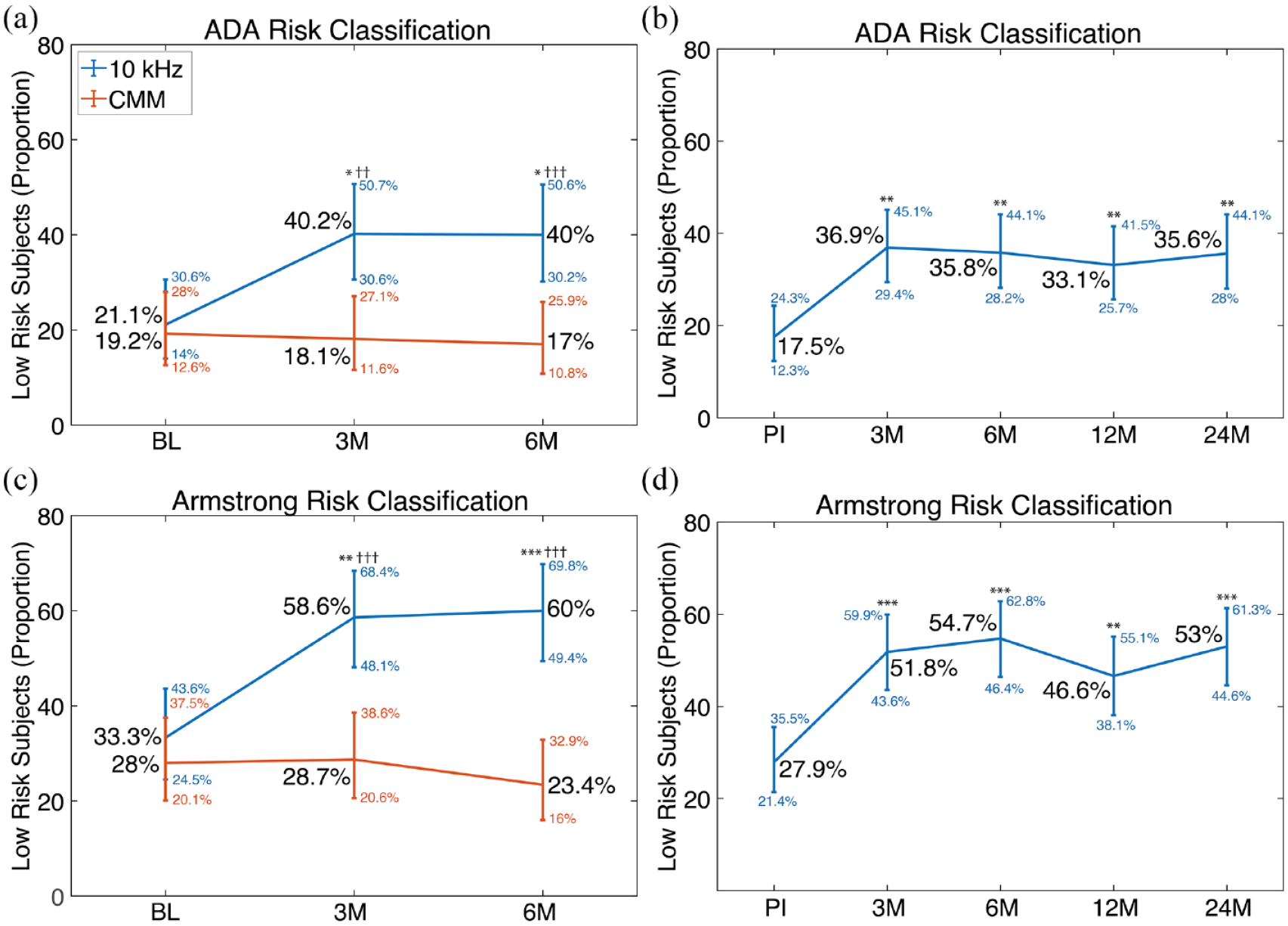
Risk of foot ulceration based on analysis of monofilament assessments. The 10-kHz SCS provides a significant improvement in foot ulceration risk using both the ADA (a and b) and Armstrong (c and d) methods. The 10 kHz group had significantly higher odds of being at low risk of foot ulceration compared to the CMM group at 3 and 6 months (a and c). The entire 10-kHz group, which includes both the original 10-kHz arm participants and participants that crossed over from CMM to 10 kHz SCS, showed a significant increase in the odds of being at low risk of foot ulceration. For (a) and (c), n = 101 (CMM group) and n = 90 (10 kHz group). For (b) and (d), n = 154, including both the original 10 kHz arm and CMM-to-SCS crossover participants.
Discussion
Loss of protective sensation is a critical threshold in the progression of diabetic neuropathy and overall limb health. Accordingly, clinical assessments, such as 10-g monofilament testing, have been developed to evaluate and monitor LOPS. 28 Our study results demonstrated that participants who received 10-kHz SCS therapy for their PDN experienced a significant increase in the average number of sensate locations on their feet as evaluated via 10-g monofilament assessments, whereas participants receiving CMM alone did not.
In comparing the 10 kHz SCS and CMM groups, 10 kHz SCS therapy is clearly effective for providing pain relief, as demonstrated previously 25 and for providing sensory improvement, as demonstrated here. While the 10-kHz group experienced significant improvements in the odds of being in the low risk of foot ulceration category from preimplantation to 3 months postimplantation (ADA: 21.1 to 40.2%, Armstrong: 33.3 to 58.6%), the CMM group’s proportions of low-risk participants did not improve over time or decreased slightly. In evaluating the durability of the improvements in foot ulceration risk, the proportion of low-risk participants approximately doubled from preimplantation to 24 months postimplantation as classified by the ADA (17.5%-35.6%) and Armstrong (27.9%-53%). Taken together, these results support that 10-kHz SCS can improve protective sensation for many PDN patients, and may even reverse LOPS in some cases, and that this benefit is durable through 24 months.
For patients with diabetic neuropathy, LOPS is associated with foot ulceration and poor outcomes in ulcer healing, critical elements of diabetic foot health. As such, an accurate assessment of LOPS is clinically important. 3 Given that overall sensory function is a largely subjective experience, current analysis from the monofilament tests, which objectively classify protective sensation, lends confidence to observations that 10-kHz SCS meaningfully slows the progression of LOPS in patients with PDN. Furthermore, it is notable that this improvement occurs in the context of a normally progressive deterioration of sensation in advanced cases of PDN.
While the results presented here provide information about the changes in participants’ sensory function, the physiologic mechanism responsible for the sensory improvement is not clear. One marker of disease progression that has demonstrated a physiological change in response to treatment is intraepidermal nerve fiber density (IENFD). In a pilot study, 10-kHz SCS was found to significantly increase lower limb IENFD after 6 and 12 months of therapy. 29 A recent preclinical study has shown significant improvements in measures of pain and sensory function in rodent models of PDN as a result of 10-kHz SCS. 30 When taken together with the outcomes presented here, these findings suggest that 10-kHz SCS improves sensory function in patients with PDN while the underlying mechanism of action is still being investigated.
It is important to acknowledge some limitations of this analysis. First, while the SENZA-PDN study included a neurological exam, the analysis of risk of foot ulceration was not a prespecified study endpoint, so this analysis was performed as exploratory per the study statistical analysis plan. Second, the ADA foot exam for LOPS allows for any of five tests to be used but recommends 10-g monofilament testing and at least one other test. The assessment presented here is based solely on 10-g monofilament testing. Third, both ADA and Armstrong methods are designed to stratify risk and not necessarily assess changes in risk classification over time.31,32 To follow this intent, risk classification was presented at each study visit via proportions of participants in the low-risk category, and these proportions were then compared between treatment groups and between study visits within a treatment group. Fourth, the SENZA-PDN study was conducted during the COVID-19 pandemic, leading to missed study visits. To address this, missing data were imputed using all available data, but this does not preclude the results from being different had all participant data been collected. Specifically, there were collectively 16 missing data collections (of 270 opportunities) from the preimplantation, 3-month, and 6-month follow-up visits of the 90 participants randomized to CMM. There were 73 missing data collections (of 770 opportunities) from the preimplantation, 3-month, 6-month, 12-month, and 24-month follow-up visits of the 154 participants who received 10-kHz SCS. Fifth, the ADA risk classification specifically indicates “No LOPS, no PAD [peripheral arterial disease], no deformity” for the low-risk category. 11 As PAD and deformity were not assessed, we cannot be certain that all patients classified to the low-risk category had no PAD or deformity. Nonetheless, we can be confident in the assessment of LOPS based on 10-g monofilament testing alone, which showed a clear benefit of 10 kHz SCS in the reversal of LOPS. The final limitation of this study is that, despite the calculated improvements to risk of ulceration presented here, there is no demonstration that this translates into clinical outcomes.
Conclusions
In conclusion, we used a standardized neurological examination and associated foot ulceration risk assessment consistent with the ADA Task Force recommendations to evaluate LOPS in PDN patients treated with 10-kHz SCS versus CMM alone. Compared to CMM alone, 10-kHz SCS significantly increased the odds of a participant being at low risk of foot ulceration. Furthermore, with 10-kHz SCS therapy, the proportion of participants at low risk of foot ulceration approximately doubled from preimplantation to 24 months postimplantation. Improving protective sensation may reduce the likelihood of ulceration, amputation, or other severe sequelae, but further studies must be conducted to test this hypothesis.
Footnotes
Acknowledgements
The authors would like to thank Colleen Kelly, PhD, a paid, independent statistician, for conducting the statistical analyses and reviewing the presentation of the statistical results. The authors would also like to thank the entire SENZA-PDN investigator team for executing the study.
List of Abbreviations
AACE, American Association of Clinical Endocrinology; ADA, American Diabetes Association; CMM, conventional medical management; LOPS, loss of protective sensation; PDN, painful diabetic neuropathy; RCT, randomized controlled trial; SCS, spinal cord stimulation.
Author Contributions
CEA has received consulting fees from AbbVie, Amgen, Biohaven, Clexio Biosciences, Collegium, Eli Lilly, Elsevier, Flowonix, Gene Pharma, Lundbeck, Nevro Corp, Novartis, Pfizer, SK Life Science, Teva Pharmaceutical, and Vertex as well as research support from AbbVie, Allergan, Amgen, Daiichi Sankyo, Eli Lilly, Novartis, Teva Pharmaceutical, and Vertex Pharmaceuticals. DGA has received consulting fees from Nevro Corp. as a member of the company’s Scientific Advisory Board. ZBK, MJJ, MB, KB, and DLC are employees of Nevro Corp. EAP has received consulting fees from Abbott Laboratories, Biotronik, Boston Scientific, Medtronic Neuromodulation, Nalu Medical, Neuros Medical, Nevro Corp, Presidio Medical, Saluda, and Vertos Medical, research support from Mainstay, Medtronic Neuromodulation, Nalu Medical, Neuros Medical, Nevro Corp, ReNeuron, Saluda, and SPR, and stock options from neuro42 and SynerFuse.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: All funding was provided by Nevro Corp.
