Abstract
Painful diabetic neuropathy is a common vexing problem for people with diabetes and a costly problem for society. The pathophysiology is not well understood, and no safe and effective mechanistically-based treatment has been identified. Poor glycemic control is a risk factor for painful diabetic neuropathy. Excessive intraneuronal glucose in people with diabetes can be shunted away from physiological glycolysis into multiple pathological pathways associated with neuropathy and pain. The first three treatments that are traditionally offered consist of risk factor reduction, lifestyle modifications, and pharmacological therapy, which includes only three drugs that are approved for this indication by the United States Food and Drug Administration. All of these traditional treatments are often inadequate for relieving neuropathic pain, and thus, new approaches are needed. Modern devices based on neuromodulation technology, which act directly on the nervous system, have been recently cleared by the United States Food and Drug Administration for painful diabetic neuropathy and offer promise as next-in-line therapy when traditional therapies fail.
Keywords
Introduction
Diabetic neuropathy (DN) affects more than half of individuals with diabetes. 1 Diabetic neuropathy can present clinically as either diabetic peripheral neuropathy (DPN) or autonomic neuropathy. 2 Painful diabetic neuropathy (PDN), contrasting with painless peripheral DN, is a form of DPN that manifests in sensations such as burning, tingling, numbness, and severe pain, especially in the lower extremities.1,3,4 PDN has been reported to affect approximately one-third of individuals with diabetes1,3,5 -8 and can also affect people with prediabetes. 9 PDN affects approximately one-fifth of individuals with type 1 diabetes (T1D) 10 and approximately one-fourth to one-half of individuals with type 2 diabetes (T2D).4,11 The incidence of PDN is often associated with worsening of painless DN symptoms, 12 progressing from sensory loss to dysesthesia, including frank pain. 13 As with many complications of chronic conditions, PDN may generally affect quality of life, resulting in mood changes, lower quality of sleep, restricted mobility, reduced activity, and social withdrawal.3,4 Psychological factors, like emotional distress and depression, can further exacerbate the symptoms of PDN.1,9
Socioeconomic factors, such as income, ethnic minority status, and education, are associated with diabetes prevalence and can also predict the prevalence of DPN. 9 In one study, individuals with T1D who lived in lower-resourced and socially deprived areas, compared with individuals with T1D who did not, were 2.17 times more likely to develop DPN. 9 Lower education levels and reduced access to health insurance also strongly predict prevalence of DPN, even after adjusting for traditional DPN risk factors, such as glycemic control and diabetes duration. 9
In addition to physical challenges, individuals with PDN experience socioeconomic obstacles and financial strain because of the combination of higher health care costs and a reduced capacity to work. 9 It is estimated that individuals with PDN spend US$7066 more per year than individuals without PDN. 1 Overall, symptom management for DPN, including treatment for diabetic foot ulcers and lower-limb amputations, 14 accounts for up to 27% of the cost of diabetes care and 9% of total health care costs for people with diabetes. 1 This excludes the cost of reduced productivity, decreased quality of life, and increased risk of other complications, such as burns and falls.3,4,15 Using the MarketScan database 16 from 2010 through 2015, 23% of 360,559 patients had a diagnosis of PDN. Of these patients, individuals with PDN, compared with individuals without PDN, had annual health care costs that were two times greater over a five-year time frame and a risk of lower-limb amputation that was 16 times higher during the first year after diagnosis. 16
Characteristics of Painful Diabetic Neuropathy
Painful diabetic neuropathy is a complication of both T1D and T2D and presents as a painful symmetrical disease of the sensorimotor nervous system, first affecting distal nerves and then progressing proximally. The first nerves affected are the longest peripheral nerve fibers in the lower extremities. PDN initially involves the small unmyelinated sensory nerves of the toes and feet, and then the hands in a stocking-glove distribution. The pain of PDN is frequently described as burning, sharp, tingling, aching, or electrical sensations.17,18 PDN is associated with adverse systemic symptoms, such as poor quality of sleep, 3 mood disorders, social withdrawal,19,20 and decreased productivity at work due to absenteeism 21 and presenteeism. 19
In clinical trials of pain-relieving interventions, pain intensity is often assessed with a visual analogue scale (VAS), verbal categorical rating scale (VRS), or numerical rating scale (NRS) where 0 is no pain and 10 is the worst pain imaginable22 -24 (Figure 1). Furthermore, several instruments for measuring the effects of neuropathic pain on quality of life have been used. 25
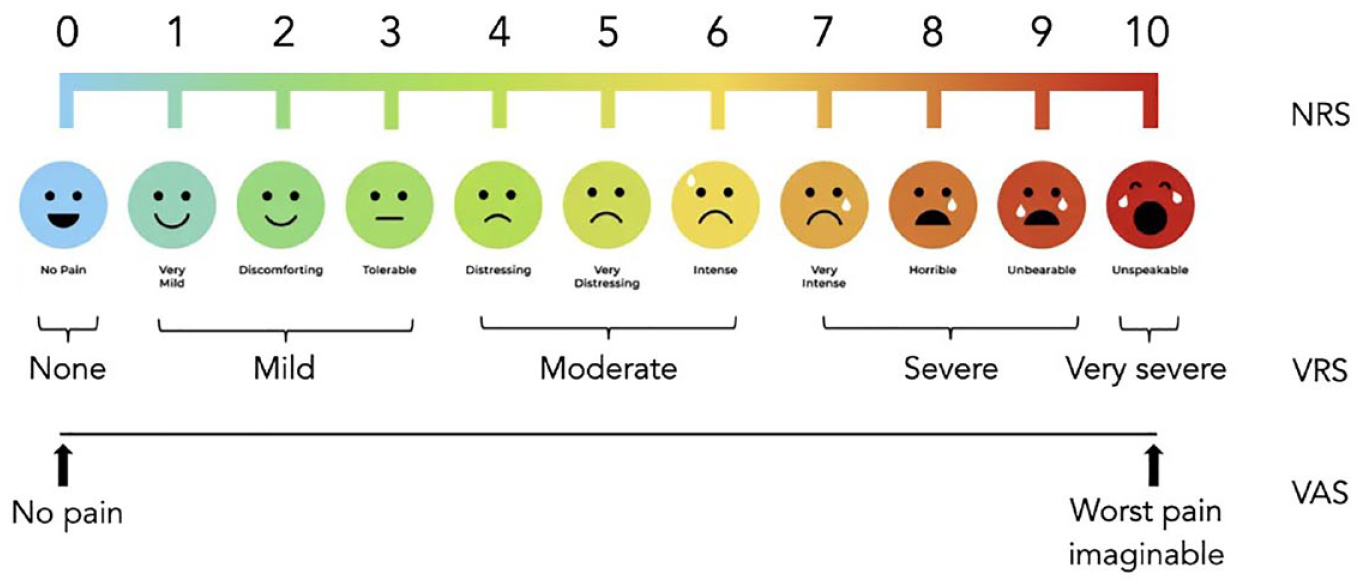
Three types of pain rating scales to measure the severity of painful diabetic neuropathy symptoms.
Pathology
Unmyelinated sensory neurons called C-fibers, or small fibers, are responsible for pain and heat sensation whereas thinly myelinated sensory neurons called A-delta fibers carry information about touch, pressure, and cold temperature. Fully myelinated nerves called A-beta and A-alpha fibers carry sensations of vibration and position sense. PDN, represented by tingling, pain, and burning in the feet, co-exists with numbness and loss of proprioception. PDN is accompanied by degeneration and loss of C-fibers as well as demyelination and loss of A-beta and A-alpha nerves of the feet. PDN symptoms in the lower extremities eventually travel proximally and later can also involve the upper extremities.9,17,26
Pathophysiology
Poor glycemic control is the risk factor most frequently linked to DN and DPN. Good glycemic control can prevent neuropathy, reduced nerve conduction, and vibration threshold abnormalities in T1D. However, in T2D, while good glycemic control can delay reduced nerve conduction and vibration threshold abnormalities, it does not necessarily prevent onset of neuropathy. 27
Excessive intraneuronal glucose in people with diabetes can be shunted away from physiological glycolysis to produce the intermediate metabolite pyruvate and into one of four pathological pathways associated with neuropathy and pain. This alternate series of biochemical reactions includes (1) activation of the polyol pathway, (2) increased flux through the hexosamine pathway, (3) increased activation of protein kinase C (PKC), and (4) increased production of advanced glycation end products (AGEs)9,28-30 (Figure 2). Alternatively, excessive glucose shunted into the pentose phosphate pathway can act as a protective mechanism. 31
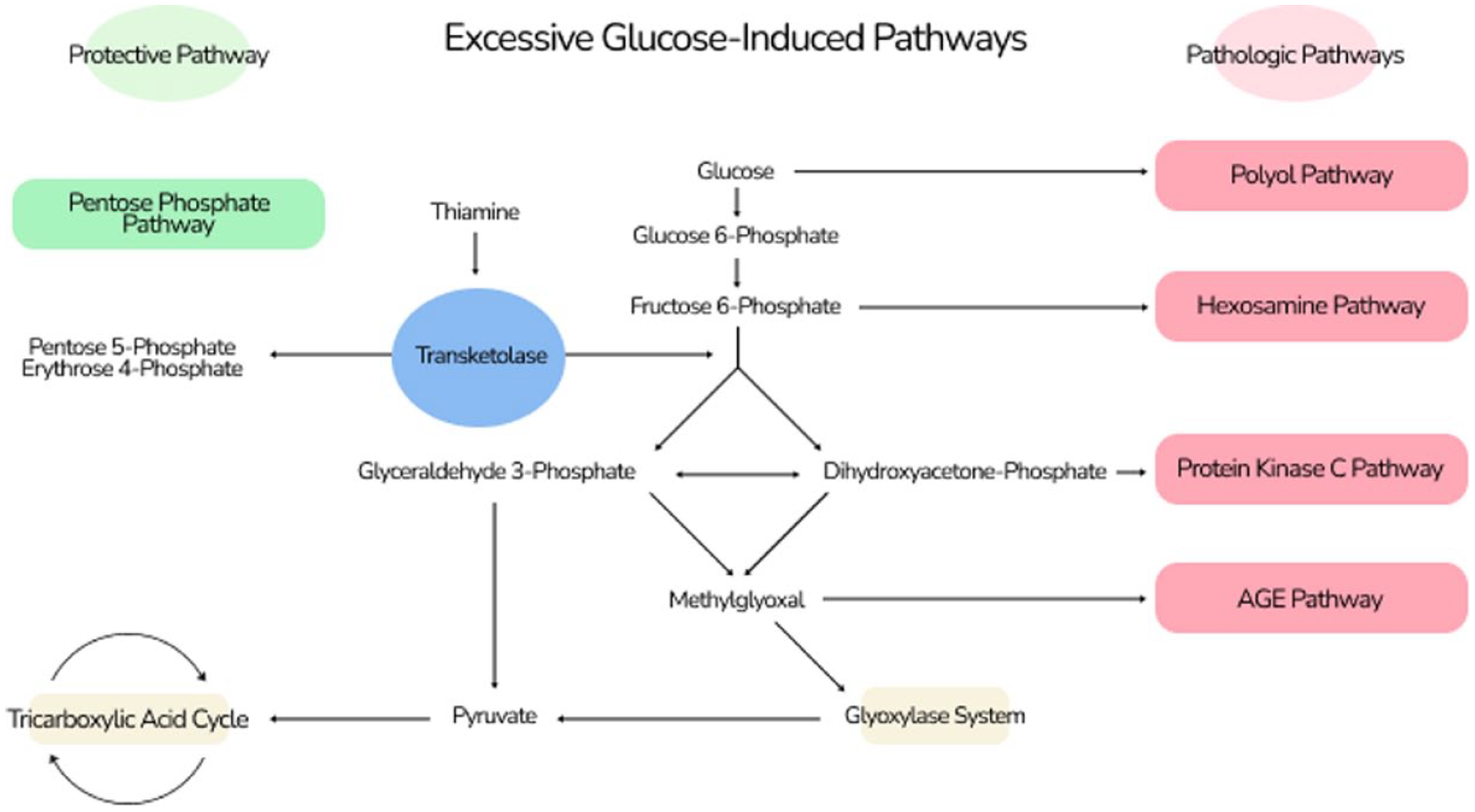
Excessive glucose-induced biochemical pathways. Excessive glucose can be shunted into one of four pathological pathways associated with neuropathy and pain: (1) the polyol pathway, (2) the hexosamine pathway, (3) the protein kinase C pathway, and (4) the AGE pathway. Transketolase permits entry into the pentose phosphate pathway, which acts as a protective mechanism. Four pathologic pathways are in pink. One protective pathway is in green.
The polyol pathway refers to the conversion of excess glucose to sorbitol by the enzyme aldose reductase. The downstream effects of increased sorbitol within neurons are thought to include oxidative stress, intraneuronal ischemia, and increased intracellular osmotic stress, all of which contribute to neuronal damage. The aldose reductase inhibitor, epalrestat, has shown reduction of symptoms 32 and blood sorbitol concentrations, 33 but the effectiveness of this class of drugs has generally been disappointing. 34
Elevated glucose entering the hexosamine pathway is converted into glucosamine molecules that can bind various intracellular enzymes. This process results in endoplasmic reticulum stress and β-cell dysfunction, leading to neuronal damage. No drug has been found to be useful for blocking the effects of increased flux through the hexosamine pathway.
Excessive intraneuronal glucose can lead to increased PKC activation. Elevated glucose levels stimulate diacylglycerol, which in turn activates PKC. This enzyme in turn activates numerous intracellular pathways that result in neuronal damage. Unfortunately, PKC inhibitors have not been successful at treating DN. 35
Excessive intraneuronal glucose can react with amino groups on proteins to form glycation compounds known as AGEs. When AGEs bind receptors for these molecules (RAGEs), then a downstream cascade of inflammation, vasoconstriction, and inflammation occurs, resulting in neuronal cell damage. The enzyme glyoxalase can detoxify some AGEs and alleviate metabolic stress associated with PDN. 36 Drug treatment to block AGE formation has been successful in preclinical trials, 37 but compounds tested in human trials have been unsuccessful because of off-target toxicity. 9 Antioxidants have been shown to decrease AGE formation. 38
Excess glucose can also be shunted into the pentose phosphate pathway, a protective mechanism. 31 In the presence of active transketolase enzyme, excess glucose can be diverted into a protective pathway, rather than entering the four pathological pathways. Genetic variability in transketolase activity might contribute to susceptibility to PDN in patients with diabetes. 28 Benfotiamine, a synthetic form of thiamine, increases the levels of intracellular transketolase and, in animals, has been shown to improve neuropathy. However, there is little data for the use of this compound in humans. 39
Furthermore, bioenergetic failure can occur from a combination of excess glucose and high fatty acid flux, donating excessive electrons, which results in alterations of intermediate metabolism, neuronal reactive oxygen species production, inflammatory pathway activation, and eventually decreased adenosine triphosphate (ATP) production. This cascade causes neuronal mitochondrial damage and cell death.29,40 Treatments to lower glucose and lipid levels can help reverse this process. 41
Diagnosis of Painful Diabetic Neuropathy
Currently, no single established gold-standard diagnostic test exists for PDN.42,43 The most common diagnostic approach is to evaluate the patient for symptoms, signs, and confirmatory tests. The earliest changes indicative of DN occur at the level of the small unmyelinated C-fibers.44,45 Later, large myelinated nerves are involved. Small fiber testing assesses pain, temperature, and crude touch, whereas large fiber testing assesses light touch, vibration, and proprioception. Thermal sensation is frequently the first nerve function to be compromised in PDN. 46 In addition, loss of ankle reflexes and weakness of small foot muscles and dorsiflexors occur early in the course of PDN. 29 Diagnostic tests of small fibers and large fibers are presented below. 47
For Small Fiber Function
Quantitative sensory testing (QST), point-of-care sweat testing, and skin biopsy may be utilized to test small fiber function. QST is a psychophysical measure of the perception of different external stimuli of controlled intensity to assess a range of sensory modalities. QST reveals loss of function from damage to both small fibers and large fibers with PDN, using thermal pain thresholds to test small fiber function and vibration thresholds to test large fiber function. In PDN, compared with painless DN, thermal hyposensitivity is more severe, whereas mechanical stimuli produce fewer differences in response. 48 This indicates a unique association of somatosensory phenotype with PDN, characterized by more severe small fiber dysfunction resulting in thermal hyposensitivity. 49
For Large Fiber Function
Electrodiagnostic studies, such as nerve conduction studies (NCS) and electromyography (EMG), are used for quantitative assessment of nerve damage or to confirm a diagnosis in equivocal clinical presentation. NCS are performed in the lower extremities, including at the sural nerve, peroneal nerve, or tibial nerve, to measure the shape, amplitude, latency, and conduction velocity of a nerve after stimulation. Axonal injury is indicated by loss of amplitude whereas demyelination is indicated by prolonged latency and conduction velocity. Generally, large fiber tests alone without small fiber tests cannot distinguish painful and painless DN. EMG measures ectopic muscle activity and can diagnose axonal damage to nerves innervating the muscles being tested.
Skin biopsy with intraepidermal nerve fiber density (IENFD) quantification is a laborious but useful test for diagnosing PDN because in the earliest stages of PDN, one can see intraepidermal small nerve fibers with decreased length and density. 45 This reduction of small nerve fibers is progressive and can also be associated with demyelination and degeneration of large myelinated fibers 42 (Figure 3). However, small fiber pathology does not automatically mean that DPN will evolve into PDN. 50
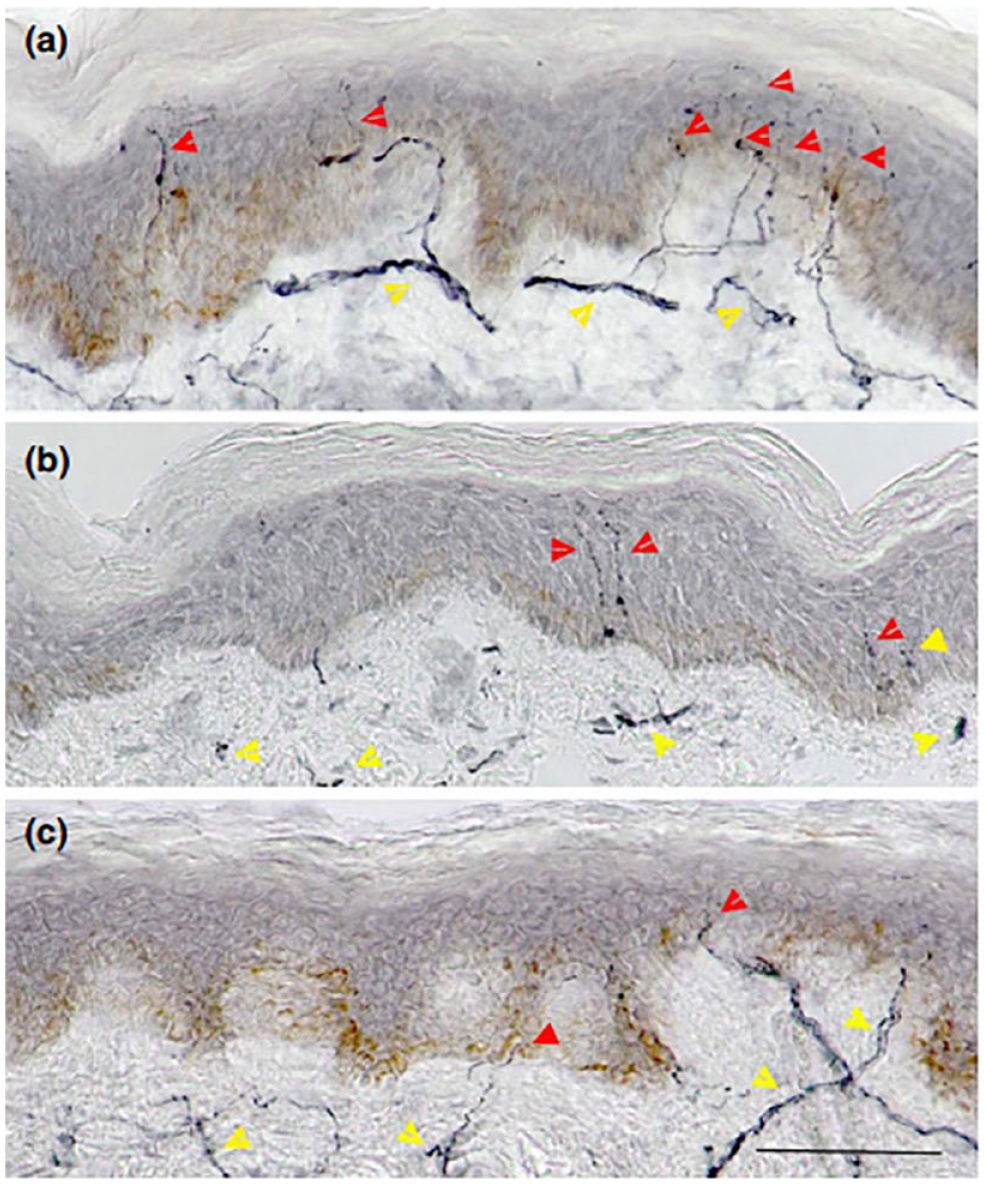
Pathophysiology of nerves in patients with different forms of neuropathy: (a) healthy control, (b) patient with painless diabetic neuropathy, and (c) patient with PDN. A biopsy from a healthy control (a) shows numerous long branching intraepidermal nerve fibers reaching upper levels of the epidermis (red arrowheads) and well-developed subepidermal nerve plexus (yellow arrowheads). A biopsy from a patient with painless diabetic neuropathy (b) shows scant but well-developed intraepidermal nerve fibers. A biopsy from a patient with PDN (c) shows scant short intraepidermal nerve fibers. Figure reproduced from Ferdousi et al 45 under the terms of the Creative Commons Attribution-NonCommercial License: https://creativecommons.org/licenses/by-nc/4.0/.
Confocal corneal microscopy can detect small nerve fiber loss in early PDN reliably and with high sensitivity and specificity, 51 even in the absence of large fiber signs, symptoms, or electrodiagnostic findings. 52 This fiber loss manifests as decreased corneal nerve fiber density, corneal nerve branch density, and corneal nerve fiber length. 45 This noninvasive reproducible test has been proposed as a clinical trial surrogate end point for recognizing early PDN prior to the onset of symptoms. 53 Investigational imaging studies have demonstrated that PDN involves not only the peripheral nervous system but also the central nervous system and is associated with reduced spinal cord and primary somatosensory cortical gray matter volume. 50
Treatment of Painful Diabetic Neuropathy
Strategies
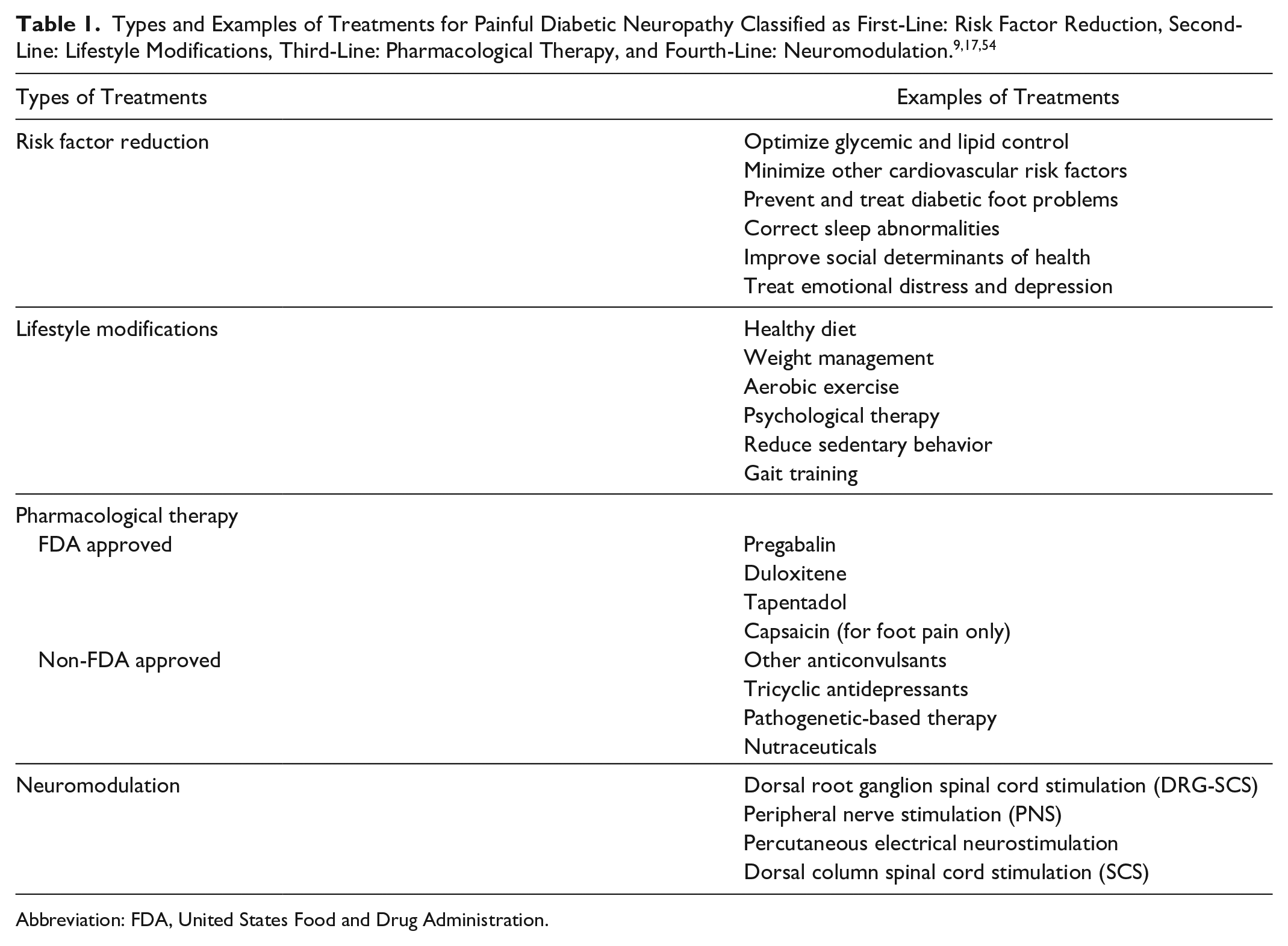
Treatment of PDN includes preventing progression of PDN and managing pain symptoms. The former is achieved through risk factor reduction and the latter is achieved through both lifestyle modifications and pharmacological treatments. Table 1 presents treatments for PDN classified as first-line: risk factor reduction, second-line: lifestyle modifications, third-line: pharmacological therapy, and fourth-line: neuromodulation.
Risk Factor Reduction
Attention to risk factor reduction, including aiming for good glycemic 55 and lipid control 56 and reducing cardiovascular risk, 57 can prevent progression of PDN. Patients with neuropathy are at increased risk of diabetic foot ulcers because they cannot necessarily feel and react in a timely manner to tight shoes or a foot injury, so they should check their feet regularly and avoid trauma. 58 Correction of any other physical, social, or psychological problems will increase resilience against the symptoms of PDN. 9
Lifestyle Modifications
Lifestyle modifications, including healthy diet and weight management, have been shown to improve small fiber neuropathy. 59 Exercise and psychological therapy have been shown to decrease pain and improve quality of life.60,61 A reduction in sedentary behavior has been postulated to improve neuropathy pain, but the benefit of this strategy is not proven. 9 Aerobic exercise and gait training have been shown to decrease the risk of falling in people with PDN. 62
Pharmacological Therapy
Pharmacological therapy for neuropathy pain differs from pharmacological therapy for musculoskeletal pain. Traditional analgesics and narcotics can successfully treat the latter condition when used on an as-needed basis, but these types of drugs are not usually effective when used as needed for neuropathy pain, and they frequently are not tolerated. Drugs for the symptoms of PDN are not intended to treat the pathophysiological mechanisms of PDN. 63 To be effective, drugs for neuropathy pain must be taken consistently regardless of the level of pain. 1
Four drugs are approved by the United States Food and Drug Administration (FDA) for PDN: (1) pregabalin (a gabapentanoid anticonvulsant), (2) duloxetine (a serotonin and norepinephrine reuptake inhibitor), (3) tapentadol (a weak to moderate opioid), and (4) capsaicin (a topical alkaloid) for foot pain only.17,54 Other drugs in the anticonvulsant, antidepressant, and analgesic families have also been used, usually without great success. In some cases, combinations of FDA-approved drugs have been used. 17 There is often little or no response to these nonspecific symptomatic drugs, and fewer than one-third of patients with PDN achieve at least a 50% decrease in their pain from such treatments. 42 Generally, pain relief with pharmacotherapy is limited, and side effects can be intolerable. 64 Medications used for neuropathy pain are intended to improve symptoms and are not able to treat the underlying disease mechanism. What is needed is a drug that treats the underlying pathology of PDN, but neither a definite mechanism for PDN nor a drug to clearly improve that mechanism has been identified to date.
Other drugs that are frequently used for PDN pain include the anticonvulsant gabapentin, the tricyclic antidepressant amitriptyline, and the weak opioid tramadol. 1 A variety of nutraceutical agents, which have not been approved by FDA for the purpose of treating PDN, include neuromodulators, antioxidants, and vitamins. One such example of an antioxidant used to treat PDN pain is alpha lipoic acid, which has shown some promise in reducing diabetic micro- and macrovascular complications and pain through the following mechanisms: (1) normalizing AGE formation, (2) reducing flux through the hexosamine pathway, and (3) selectively inhibiting neuronal T-type calcium channels. 65 Many variations of these agents have been deployed without statistically significant benefit.
Multiple classes of drugs that target the various pathogenetic mechanisms of PDN (Figure 2) have been studied in an attempt to improve neuropathy symptoms. 66 These agents have been prescribed to favorably impact the underlying pathophysiological abnormalities that are present in PDN (Figure 2). Unfortunately, trials of these agents have not been successful. 67
Neuromodulation Therapy
Neuromodulation is a technology that acts directly upon nerves to alter or modulate nerve activity by delivering electrical or pharmaceutical agents directly to a target area. 68 Four types of neuromodulation have been tested for PDN: (1) dorsal root ganglion spinal cord stimulation (DRG-SCS), 69 (2) peripheral nerve stimulation (PNS), 70 (3) percutaneous electrical neurostimulation, 71 and (4) dorsal column spinal cord stimulation (SCS). 3 The evidence for improved clinical outcomes for the first two methods is sparse, and they will not be discussed in this article. The third method is used by the First Relief auricular neurostimulator (DyAnsys, San Mateo, California), which received FDA clearance for treatment of PDN on July 14, 2022. It is a wearable device placed on the ear, which administers continuous pulses of a low-level electrical current through tiny needles. 72 The device was tested in a 90-day randomized controlled trial on 63 subjects in India, in which a VAS pain score analysis showed a significant reduction of pain with the device for users. 73 No data has been published from this trial. Dorsal column spinal SCS is administered by two devices cleared by the FDA for PDN that, compared with best medical therapy, have been shown to be effective for patients with PDN. 74
Overall, SCS is a well-established treatment. A search of the PubMed database for the term “spinal cord stimulation” on July 16, 2022, elicited 25,687 references on the topic. Whereas this type of treatment has long been considered as a last resort, 1 recent FDA approval in 2015 75 of a newly developed form of SCS, high frequency SCS, has advanced this treatment to a potential next-in-line treatment if pharmacological therapy fails. 63 Spinal cord stimulation is discussed in detail in the second part of this two-part series, “Spinal Cord Stimulation for Painful Diabetic Neuropathy” by Yeung and colleagues.
Future Developments in Painful Diabetic Neuropathy Research
Treatment of PDN has been limited by an inadequate understanding of the pathophysiology of this disease. More information is needed about the mechanisms by which hyperglycemia, microvascular disease, and other risk factors result in various types of nerve damage. 12 Research in the pathophysiology of PDN will help produce disease-modifying treatments that mechanistically address the disease pathology, rather than just its symptoms.17,76 Treatment algorithms for stratifying subpopulations of patients with PDN, including genetic studies, can be developed using systems biology to create precision medicine tools. These new tools can incorporate recent research into the use of new pharmacological treatments and lifestyle modifications that can be directed at novel therapeutic targets.77,78 Research into nonprescription bio-factors or nutraceuticals might uncover natural substances that can serve as (1) antioxidants to prevent the oxidative stress that accompanies DPN, (2) promoters of carbohydrate and lipid metabolism to improve glycemic patterns in diabetes, or (3) essential nutrients that can provide extra health benefits to provide protection against DPN in addition to their basic nutritional value. 9 These are examples of areas where future research will uncover better tools to understand, treat, and prevent PDN.
Conclusion
Painful diabetic neuropathy is a major source of morbidity in diabetes because of chronic pain and a diminished quality of life. Because the pathophysiology is poorly understood, there are no FDA-approved therapies that mechanistically treat or reverse this condition. Risk factors have been identified and potential mechanisms have been hypothesized, but these mechanisms have not been amenable to treatments for PDN that target the presumed pathogenetic bases of the pain. Current widely used pharmacological therapies are inadequate because of poor effectiveness and side effects. New approaches are needed. Neuromodulation therapy is one such emerging and promising treatment.
Footnotes
Acknowledgements
The authors thank Annamarie Sucher-Jones for her expert editorial assistance.
Abbreviations
AGE, advanced glycation end product; ATP, adenosine triphosphate; DN, diabetic neuropathy; DPN, diabetic peripheral neuropathy; DRG, dorsal root ganglion; EMG, electromyography; FDA, United States Food and Drug Administration; IENFD, intraepidermal nerve fiber density; NCS, nerve conduction studies; NRS, numerical rating scale; PDN, painful diabetic neuropathy; PKC, protein kinase C; PNS, peripheral nerve stimulation; QST, quantitative sensory testing; RAGE, receptors of advanced glycation end products; SCS, spinal cord stimulation; T1D, type 1 diabetes; T2D, type 2 diabetes; VAS, visual analogue scale; VRS, verbal categorical rating scale.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: DCK is a consultant for EOFlow, Fractyl Health, Integrity, Lifecare, Rockley Photonics, and Thirdwayv. AMY, JH, KTN, NYX, LTH, BKA, and NE have nothing relevant to disclose.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Diabetes Technology Society, which received no external funding and paid all costs for conducting the study.