Abstract
The development of painful diabetic neuropathy (PDN) is a common complication of chronic diabetes that can be associated with significant disability and healthcare costs. Prompt symptom identification and aggressive glycemic control is essential in controlling the development of neuropathic complications; however, adequate pain relief remains challenging and there are considerable unmet needs in this patient population. Although guidelines have been established regarding the pharmacological management of PDN, pain control is inadequate or refractory in a high proportion of patients. Pharmacotherapy with anticonvulsants (pregabalin, gabapentin) and antidepressants (duloxetine) are common first-line agents. The use of oral opioids is associated with considerable morbidity and mortality and can also lead to opioid-induced hyperalgesia. Their use is therefore discouraged. There is an emerging role for neuromodulation treatment modalities including intrathecal drug delivery, spinal cord stimulation, and dorsal root ganglion stimulation. Furthermore, consideration of holistic alternative therapies such as yoga and acupuncture may augment a multidisciplinary treatment approach. This aim of this review is to focus on the current management strategies for the treatment of PDN, with a discussion of treatment rationale and practical considerations for their implementation.
Introduction
Approximately half of the estimated 425 million patients with diabetes worldwide will be affected by diabetic neuropathy.1-4 Neuropathic complications reportedly comprise up to 27% of the cost of diabetes. 5 Less easily quantified are the burdens of depression, lost productivity, and impaired quality-of-life suffered by patients.6,7 Up to a third of patients with diabetes will also develop painful diabetic neuropathy (PDN), further exacerbating disability.8,9 Thus, the treatment of PDN is focused on strict glycemic control to deter disease progression and pain management. In one study, patients with PDN spent US$7066 annually more than patients without pain, highlighting significant economic costs. 10
Both the American Academy of Neurology 11 and European Federation of Neurological Societies 12 have published guidelines regarding the clinical management of PDN, although the randomized clinical trials (RCTs) forming the basis of these guidelines were generally based on short-term data. 13 In the clinical setting, PDN often involves polypharmacy, which highlights the difficulty in obtaining pain relief with single medications.14,15 This review focuses on the current management strategies for the treatment of PDN including pharmacological, neuromodulation, and alternative therapies.
Clinical Presentation and General Management
PDN is a clinical diagnosis, and there is considerable variability in its presentation.1,4 Burning pain is often the initial presenting complaint, and concurrent paresthesias are common.16,17 A distal, symmetric “stocking” distribution is the most common manifestation. 17 Rarer atypical forms include diabetic radiculoplexopathies (often unilateral/asymmetric), chronic inflammatory demyelinating polyradiculoneuropathy, and autonomic neuropathies. 17 These subtypes are less studied and can be particularly difficult to manage. Onset may be insidious, and therefore a careful history probing for less overt symptoms may be essential for diagnosis. 1
PDN is a diagnosis of exclusion; as such, other metabolic derangements and causes of peripheral neuropathy must be ruled out. This may be particularly difficult given the common associated comorbidities, including metabolic syndrome. 16 To further complicate diagnosis, up to half of patients may be asymptomatic. 1 Therefore, the American Diabetes Association recommends temperature and monofilament testing to screen for neuropathy in patients initially diagnosed with type 2 diabetes, and within five years of a type 1 diabetes diagnosis.1,18 Both subtypes should be tested annually after their initial evaluation, and some research has shown certain prediabetic patients should also be screened.1,18
There are no proven treatments able to reverse the nerve damage; therefore, preventative measures including proper foot care and good glycemic control are the cornerstones of treatment.1,2,4 Measures to slow or halt symptom progression include lifestyle modifications, such as an improved diet, exercise, and weight loss, as well as medications to optimize glycemic control.17,19 Maintaining glycemic control has been associated with significantly lower incidence and slower progression of neuropathy, with particular benefit (up to 81% relative risk reduction) demonstrated in patients with type 1 diabetes.19,20
Pain management is a particular challenge posed by PDN and an important treatment consideration.14,21 For the remainder of this review, we will focus on PDN management and discuss accepted pharmacotherapies, neuromodulatory techniques, and alternative therapies used for pain relief. Figure 1 illustrates a flowchart of recommended management strategies.
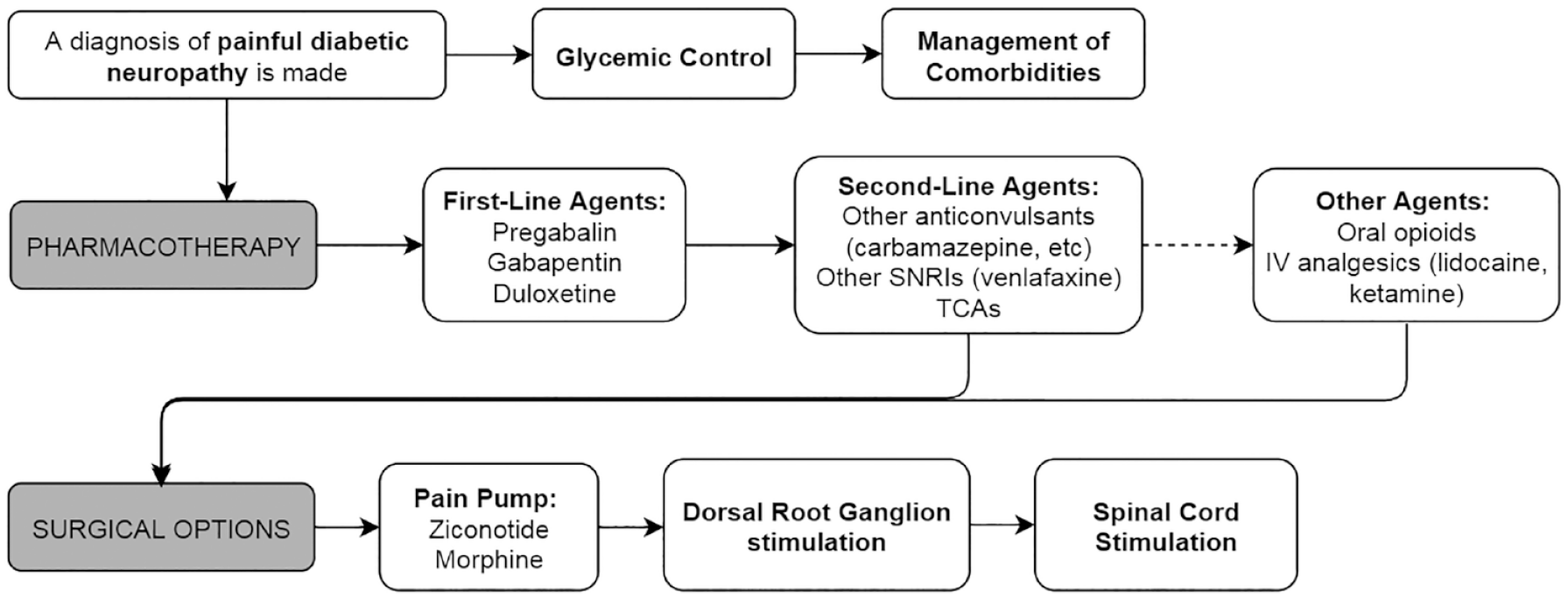
Flowchart of painful diabetic neuropathy management options.
Pharmacotherapy
It is a common misconception that all pain medications are to be taken “as needed.” While this may hold true for musculoskeletal pain, the management of neuropathic pain requires routine and regimented administration regardless of pain severity in order to provide sustained relief. Therefore, it is crucial that specialists in diabetes management educate patients on the need to adhere to the appropriate dosage regimen.
Current pharmacotherapeutic strategies include antidepressants, anticonvulsants, and opioids, which have been demonstrated to be superior to placebo for pain control in PDN. 13 Other therapies such as topical analgesics and intravenous (IV) medications warrant further study. Published guidelines from the American Academy of Neurology recommend pregabalin as a first-line treatment. 11 In addition to pregabalin, first-line treatment recommendations from the European Federation of Neurological Societies include serotonin-norepinephrine reuptake inhibitors (SNRI), tricyclic antidepressants (TCA), and gabapentin. 12 Table 1 outlines pharmacotherapeutic agents, including dose considerations, mechanisms, and side effects.22,23
Pharmacotherapy for Painful Diabetic Neuropathy.
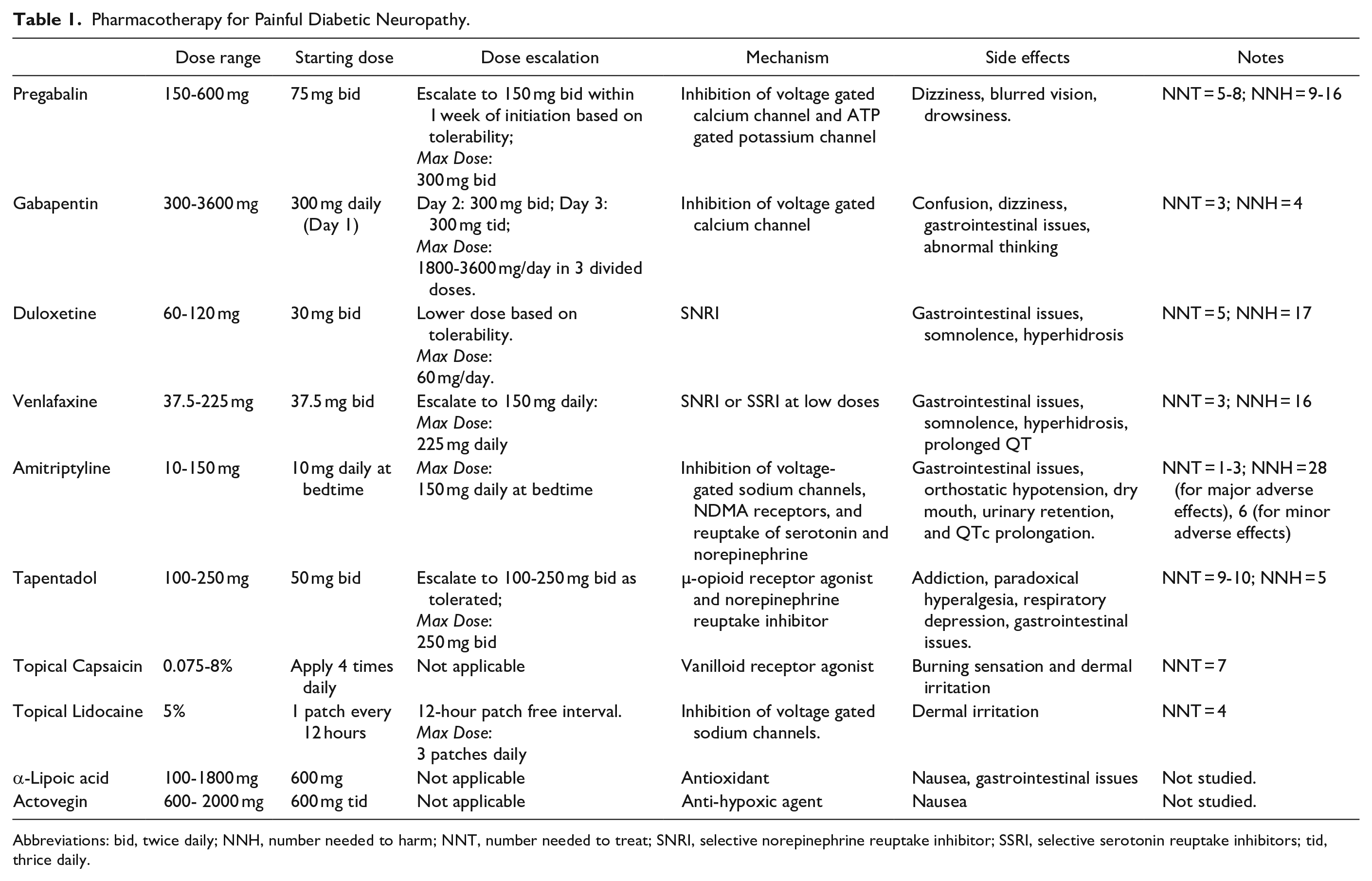
Abbreviations: bid, twice daily; NNH, number needed to harm; NNT, number needed to treat; SNRI, selective norepinephrine reuptake inhibitor; SSRI, selective serotonin reuptake inhibitors; tid, thrice daily.
Anticonvulsants
Pregabalin is a Food and Drug Administration (FDA)-approved anticonvulsant used for the management of PDN, administered daily at 150-600 mg. Evidence shows significant pain reduction with comparable efficacy to duloxetine and gabapentin,24-27 as well as improvements in sleep and global impression scales without affecting nerve conduction.26,28 Gabapentin is characterized as second-line therapy, typically administered at a total daily dose of 300-3600 mg. 29 It is well-tolerated, demonstrating up to 50% pain relief compared to placebo, and noninferior pain relief compared to amitriptyline and pregabalin.30,31 At higher doses, both pregabalin and gabapentin are administered in divided doses.
Secondary to their pharmacokinetics, pain relief is observed within one to four weeks from initiation of either pregabalin or gabapentin. 23 During this time, however, side effects may be experienced including confusion, dizziness, drowsiness, and gastrointestinal issues.26,32 The use of anticonvulsants is also associated with the development of tachyphylaxis. It is important to emphasize to patients to continue taking their medication in order for pain control to be realized, and dose adjustments can be made to balance pain relief and side effects. Furthermore, pregabalin is often cost-prohibitive or not commonly covered by insurance.
Other anticonvulsants for PDN management include carbamazepine, valproic acid, and lamotrigine. While these medications have demonstrated improvement in PDN symptoms, they are not first-line and have shown inconsistent results.33-35
Antidepressants
Antidepressants, such as duloxetine, are considered first-line pharmacotherapy for PDN. While initially prescribed for depression, duloxetine is an SNRI that is widely studied in PDN management, and is prescribed as a daily dose of 60-120 mg. Duloxetine improves long-term pain outcomes, depressive symptoms, and overall quality-of-life in PDN patients.36,37 Effects are typically observed as early as three to five days from drug initiation. It is associated with mild elevations of fasting plasma glucose and weight gain, but insignificant changes in hemoglobin A1c. 38 Adverse effects include gastrointestinal issues, somnolence, and hyperhidrosis. 36
Venlafaxine is an SNRI that is administered at a daily dose of 37.5-225 mg, 29 and functions as a selective serotonin reuptake inhibitor at low doses. While not FDA-approved for PDN, studies show significant pain improvement compared to placebo; however, there is comparatively better literature on the use of duloxetine in PDN, and thus venlafaxine should be considered only if duloxetine fails or the side effects are intolerable. Venlafaxine is noninferior to TCAs and gabapentin, 39 but inferior to pregabalin. 40 Adverse effects are similar to those of duloxetine; however, patients should also be routinely monitored for QTc prolongation. 39
TCAs, specifically amitriptyline, are also used in PDN management; however their use requires careful monitoring and tends to be avoided in elderly patients and those with significant comorbidities. Amitriptyline is administered daily at 25-150 mg. 29 Compared to duloxetine,41,42 gabapentin, 43 and pregabalin, 44 amitriptyline confers noninferior pain relief. TCAs concurrently target histaminergic, adrenergic, and cholinergic receptors leading to a robust side effect profile, including gastrointestinal issues, orthostatic hypotension, dry mouth, urinary retention, and QTc prolongation. 42 The use of this drug is recommended only as a last resort given its high risk of side effects.
Opioids
Chronic opioid therapy for PDN can be helpful for a subset of people; however, their use is associated with certain unique concerns. For example, now well-recognized is the concern of misuse and abuse with long-term opioid treatment: misuse is the use of a drug in a nonindicated manner, whereas abuse is when use becomes detrimental or unlawful. 45 Furthermore, it is important to understand the distinction between tolerance (physiological adaptation resulting in reduced drug efficacy) and physical dependence (physiological adaptation in which withdrawal can be induced with drug cessation or rapid dose reduction) with chronic opioid use. 45 Evaluating a person with PDN for the potential use of chronic opioid therapy should include obtaining a history of past/current substance abuse and addiction so that the prescriber can address all relevant issues before prescribing opioids.
Since the development of tolerance may lead to dose escalation, the prescriber needs to be aware of dose-dependent risks of opioid therapy. Opioids should always be used at the lowest effective dose. Side effects and aberrant behaviors must be monitored consistently by the prescriber and opioid-sparing approaches including interventional treatments need to be considered and routinely incorporated. Additional concerns of chronic therapy include the development of opioid-induced hyperalgesia—although not well understood, some patients will actually experience increased pain. 46 If identified, dose reduction can be very effective.
Tapentadol is an opioid that is FDA-approved specifically in PDN management as third-line therapy, 47 administered at an optimal dose of 100-250 mg twice daily. Several studies demonstrate that extended-release tapentadol significantly reduces pain intensity compared to placebo.48-50 Levorphanol, a synthetic opioid, has also been reported to significantly reduce neuropathic pain when used in high-strength formulations (around 9 mg/day), with efficacy comparable to gabapentin and TCAs. 51 Methadone, another synthetic opioid, has demonstrated efficacy in treating neuropathic pain, including patients who have previously failed trials of conventional opioids. 52 Opioid-naïve patients are typically started at doses of 2.5-5 mg every 8-12 hours, and those patients currently taking other opioids require careful dose conversion. 53 Although effective for general neuropathic pain, both levorphanol and methadone require further studies to evaluate their use in PDN.
Topical Analgesics
Capsaicin is a topical analgesic utilized in low (0.075%) and high (8%) dose formulations for PDN management. Significant reductions in neuropathic pain intensity have been reported,54,55 although results can be inconsistent.56,57 Additional studies have reported 8% capsaicin as noninferior to pregabalin, duloxetine, and gabapentin with a comparatively more tolerable side effect profile. 55 Adverse effects include dermal irritation. 58 Qutenza is an 8% capsaicin patch approved for the treatment of PDN in Europe, and is currently pending FDA-approval.
Topical lidocaine (5%) can be utilized for the treatment of localized neuropathic pain, 59 with several studies demonstrating significant pain reduction, improvement in quality-of-life, and patient satisfaction compared to placebo.60,61 Noninferiority testing with pregabalin, gabapentin, capsaicin and amitriptyline has demonstrated sustained efficacy with comparatively improved tolerability.62,63 Side effects similarly include dermal irritation. 64
Other topical analgesics include ketamine cream and clonidine gel. 65 One study demonstrated that topical 5% ketamine did not improve PDN symptoms compared to placebo. 66 There is currently limited and inconsistent evidence on the clinical utility of clonidine for PDN.67,68
Novel Agents
α-Lipoic acid is a potent antioxidant used in multivitamin formulas. 69 Its properties, including reduced oxidative stress, sustained microvascular blood flow, and improved nerve conduction velocity,70,71 prompted investigation into its effects on PDN. Various placebo-controlled RCTs administered α-Lipoic acid with an oral or IV infusion of 600 mg once to thrice daily. Overall, improved neuropathic sensory symptoms were reported, without significant improvement in neuropathy impairment scores, levels of inflammatory or oxidative stress markers, or nerve conduction.72-76 In the United States, it is available as a nutritional supplement and does not require a prescription. This has raised concerns regarding drug purity and safety.
Actovegin is a deproteinized ultrafiltrate of calf serum with potent anti-hypoxic effects. It has been investigated in several trials assessing a potential neuroprotective role in PDN, with regimens consisting of daily infusions (2000 mg) for 20 days followed by three times a day oral formulations (200 mg) for 140 days. These studies demonstrated improvement in neuropathic symptoms and vibration thresholds compared to placebo with a tolerable safety profile.77,78 These novel agents require further investigation to further define their role in PDN management.
Intravenous Therapies
IV Lidocaine
Lidocaine is a local anesthetic with antiarrhythmic properties that also exerts analgesic effects. A recent systematic review evaluating IV lidocaine as a treatment for neuropathic pain reported superiority to placebo in the early post-infusion period. 79 However, infusions over four weeks had no further significant effect. While IV lidocaine was associated with an increased risk of minor adverse effects, no serious adverse effects were reported. Studies specific to the use of IV lidocaine for PDN were included in this review;80-82 common infusion dosages were 5 and 7 mg/kg, providing varying degrees of pain relief from 10 to 28 days following infusion.
The effects of IV lidocaine are mainly limited to the immediate post-infusion period.53,83,84 As such, its use may be better suited for acute adjunctive care while allowing for up-titration of oral analgesics to therapeutic dosages, as well as incorporation of nonpharmacologic modalities. Results are mixed, with other research showing no significant short- or long-term analgesic benefit up to four weeks post-infusion. 85 Overall, IV lidocaine is a safe alternative to opioid medications for acute management of PDN. Additional well-designed studies are needed to provide clear guidelines on clinical use.
IV Ketamine
Ketamine is a dissociative anesthetic that, when given in sub-anesthetic doses, has analgesic and anti-hyperalgesic effects. The duration of analgesic effect with a single dose of IV ketamine is approximately 60 minutes, and up to six hours when administered orally. 53 Although most studies have evaluated IV ketamine, oral administration has been reported to significantly improve neuropathic pain compared to methadone in one study. 86 A study evaluating patients with peripheral neuropathy treated with ketamine bolus and infusion reported a significant reduction in allodynia and spontaneous pain. 87 A recent systematic review showed that patients treated with IV ketamine demonstrated some type of pain relief for a variety of neuropathic pain syndromes, with comparatively poorer results for oral and topical administration. 88
In one study, the most common side effect was dizziness (44% for infusion, 22% for oral administration). Other side effects included sedation, loss of appetite, nausea, and vomiting. 89 Importantly, the use of IV ketamine has addictive potential similar to that of opioids. As it can be considered a drug of abuse, its use should be considered only in carefully selected patients, and under the strict supervision of a pain physician. Patient evaluation and screening should be similar to patients taking opioid therapy, including delineating a history of substance abuse and addiction. Overall, the use of ketamine in all forms for the treatment of PDN requires further study.
Neuromodulation
Neuromodulation therapies are nonablative pain treatments that are both titratable and reversible; these include intrathecal (IT) pain therapy and spinal cord stimulation (SCS). Both modalities have been demonstrated to be effective treatments option for neuropathic pain, including failed back surgery syndrome and complex regional pain syndrome (CRPS). However, few studies have assessed the use of IT therapy or SCS specifically in the treatment of PDN. Currently, the use of neuromodulation in the treatment of PDN is underutilized, which can be attributed to the lack of high-quality studies supporting its use, as well as limited referral to neuromodulation specialists. Accordingly, these are rare interventions that require careful consideration of a patient’s comorbidities and goals of therapy. Potential patients should be referred to a pain management specialist to determine their candidacy, and ideally their care should be discussed at a multidisciplinary pain conference. The use of neuromodulation therapies should be considered only following the failure of conservative management with evidence-based medications such as anticonvulsants and antidepressants.
Intrathecal Pain Therapy
The use of IT pain therapy is indicated for the treatment of chronic neuropathic pain, including peripheral neuropathy related to diabetes. Morphine (a µ-receptor agonist) and ziconotide (a nonopioid calcium channel blocker) are the only FDA-approved agents for the IT treatment of pain, and are both recommended by the Polyanalgesic Consensus Conference as first-line monotherapy in the treatment of localized or diffuse nonmalignant neuropathic pain. 90 IT therapies are recommended for the treatment of refractory pain, defined as a failure of conservative management with multiple evidence-based treatments. 91 Importantly, IT therapy should not be used as salvage therapy following the failure of high-dose systemic opioids. 90 IT therapy can be used concurrently with oral therapies, and doses should be carefully managed by a pain specialist.
Ziconotide has certain advantages due to its nonopioid properties that make it a preferable first-line monotherapy. In particular, its use is not associated with an elevated risk of respiratory depression, and there is no withdrawal with abrupt discontinuation. However, a history of psychosis or concurrent use of anti-epileptics or sedatives is a contraindication. 92 Initial IT dosing starts at 1.2 mcg/day, with titration of 1.1-2.8 mcg/day at two to four week intervals. 93
The use of IT morphine is also considered first-line; however, the risk of respiratory depression can be deadly, especially with the concurrent use of hypnotics or sedatives. Additional concerns include the development of tolerance, withdrawal with drug discontinuation or pump malfunction, and the rare development of a catheter tip granuloma which can cause neurological injury. 94 The recommended starting dosage for IT morphine is 0.1-0.5 mg/day, which should be titrated conservatively with consideration for the aforementioned side effects. 90
Tonic SCS
Conventional, tonic SCS utilizes low-frequency stimulation of 40-100 Hz to create paresthesias that overlaps painful anatomic regions. 95 In practice, lower extremity stimulation can typically be achieved with lead placement between T9-T11, although sometimes retrograde lead placement is required to achieve foot coverage. In the first clinical study of SCS in medically-refractory PDN, permanent SCS leads were implanted between T9-T11, with patients achieving significant pain relief at a median of 14 months, and with reduced or eliminated oral analgesic intake. 96 Significant long-term pain relief was demonstrated with follow-up at 3.3 and 7.5 years. 97 Similar small, uncontrolled studies demonstrated promising results with long-term SCS treatment.98-100
The success of these pilot studies lead to the 2014 publication of two prospective, multi-center RCTs comparing the efficacy of conventional medical management with and without SCS.101,102 In the study reported by de Vos et al, 36 PDN patients had SCS electrodes implanted between T9-T12. 101 At six months follow-up, mean pain intensity was significantly decreased in the SCS group, with improved quality-of-life metrics and decreased analgesic intake. In the concurrent study by Slangen et al, 17 PDN patients underwent SCS implantation. Treatment success (≥50% improvement in pain severity) at six months follow-up was reported in 59% of SCS patients versus 7% of those receiving medical management alone. 102 The patients in this latter study were reassessed at 24 months and continued to have sustained pain relief. 103
Sub-Paresthesia SCS
Although high-quality evidence has been reported for the use of tonic SCS in neuropathic pain, a significant proportion of patients in these studies did not achieve adequate pain relief. Subsequently, sub-paresthesia waveforms have been developed, including high-frequency and burst stimulation, in which minimal to no paresthesia is perceived. 95 Compared to tonic SCS, both high-frequency and burst SCS have been demonstrated to provide superior relief of back and leg pain in prospective RCTs.104,105
There has been recent enthusiasm in applying these novel waveforms to improve the clinical outcomes of patients with PDN. The SENZA-PDN study is an ongoing prospective, multicenter RCT, with patients assigned to high-frequency SCS at 10 000 Hz plus medical management versus medical management alone (NCT03228420). 106 The primary endpoint is a composite measure of both safety and effectiveness at three months, with follow-up to continue for 24 months. Enrollment was recently completed in 2019, with preliminary results presented at the 2020 North American Neuromodulation Society Annual Meeting; 107 patients with PDN demonstrated improved pain severity, sleep, and walking tolerance at three months, and without higher rates of infection.
Dorsal Root Ganglion Stimulation
Conventional SCS targets the dorsal columns and thus has certain coverage limitations, and dorsal root ganglion (DRG) stimulation has emerged as a therapeutic modality to improve coverage of specific dermatomal distributions. Compared to tonic SCS, DRG stimulation in patients with complex regional pain syndrome has been reported to provide higher rates of treatment success. 108
As patients with PDN predominantly have lower extremity and foot pain, the specificity of coverage provided by DRG stimulation is attractive, although the current evidence is limited. In one retrospective study, seven PDN patients were implanted with a permanent system after a successful trial, and four patients with 12-month follow-up data reported a 64.16% mean relative reduction in perceived pain; 109 the majority of patients were implanted at L5. Based on dermatomal coverage, it is recommended to target L5, possibly with the addition of S1. A staged trial may consist of unilateral electrode placement, with the addition of bilateral electrode(s) for permanent implantation should the trial be successful. In clinical practice, the use of DRG stimulation may be preferable to dorsal column SCS due to its dermatomal specificity.
Other DRG studies have been preclinical. Using conventional and burst DRG stimulation in rat models of PDN, Franken et al observed attenuation of mechanical hypersensitivity using both waveforms compared to sham stimulation.110,111 Although preliminary, these data suggest a potential role for DRG stimulation in the management of refractory PDN.
Other Stimulation Modalities
Despite being considered low-risk procedures, IT and SCS therapies are invasive and require the chronic implantation of hardware. Transcutaneous electrical nerve stimulation (TENS) is a noninvasive neuromodulation modality that delivers electricity via patches placed on painful areas, with efficacy demonstrated for some patients with musculoskeletal pain. The mechanism of action in PDN is unclear, and has been attributed to improved microcirculation and/or the stimulation of cutaneous afferents, leading to nociceptive inhibition. 112
Some of the earliest studies evaluating TENS in the treatment of PDN reported decreased pain scores and improvement in neuropathic symptoms, although with a high placebo effect with sham stimulation.113,114 Additional studied therapies include external muscle stimulation,115,116 pulsed electromagnetic fields, 117 and frequency rhythmic electrical modulation. 118 Although multiple studies have been performed, they are limited by their short duration, small patient numbers, lack of study rigor, and significant placebo effects. 112
Alternative Therapies
The use of nonpharmacological interventions for PDN has a great deal of support, and the most studied modalities include lifestyle modification with diet and exercise, dietary supplements, and holistic therapies such as yoga and acupuncture. 119
Yoga has been reported to improve multiple health parameters in patients with diabetes including reduced BMI, systolic blood pressure, and fasting glucose, as well as a reduction in oxidative stress parameters.120,121 In a study of older female patients with PDN, meditation and progressive relaxation resulted in significant daily pain reduction for at least 24 hours compared to baseline. 122 Acupuncture has also been shown to be an effective treatment for PDN; one study reported significant pain relief when participants were treated once per week for 10 consecutive weeks with individualized acupuncture regimens. 123 Subsequent systematic reviews have reported favorable outcomes with acupuncture, although the practice is quite variable, employing different methodologies and outcome measures.124,125
Low-level laser therapy (aka photobiomodulation) employs the use of red and near infra-red light over painful areas to purportedly improve tissue healing and promote pain relief. Some studies have demonstrated short-term pain relief in patients with PDN;126,127 however, the lasting efficacy is unclear, with some studies reporting no benefit with longer-term follow-up.128,129 As such, the use of alternative therapies should be integrated into a comprehensive management strategy and should not be used as sole interventions.
Conclusion
PDN is a relatively common complication of diabetes associated with significant disability and cost. Although primary prevention of diabetes is required at a societal level, the treatment of diabetic complications, including aggressive glycemic control, is essential in controlling the development of neuropathic complications. Guidelines exist regarding the pharmacological management of PDN, and first-line agents include anticonvulsants (pregabalin, gabapentin) and antidepressants (duloxetine). Oral opioids may be helpful in a subset of patients; however, their use should be carefully monitored, and should always be used at the lowest effective dose. Despite multiple conservative treatment options, there are considerable unmet needs in this patient population, and neuromodulation therapies, including IT drug delivery and SCS, have the potential to improve pain management outcomes. Improving pain outcomes will require an individualized patient approach, with consideration for pharmacotherapy, surgical treatments including neuromodulation, and other holistic alternative therapies.
Footnotes
Abbreviations
DRG, dorsal root ganglion; FDA, Food and Drug Administration; IT, intrathecal; PDN, painful diabetic neuropathy; RCT, randomized control trial; SCS, spinal cord stimulation; SNRI, serotoninnorepinephrine reuptake inhibitor; SSRI, selective serotonin reuptake inhibitor; TCA, tricyclic antidepressant; TEN, transcutaneous electrical nerve stimulation.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr Argoff is a Speakers Bureau member for Allergan, Inc., AstraZeneca Pharmaceuticals LP, Depomed, Inc., Iroko Pharmaceuticals, LLC, Millennium Laboratories, LLC, Teva Pharmaceuticals Industries Ltd., and XenoPort, Inc.; Advisory Board member for AstraZeneca Pharmaceuticals LP, Collegium Pharmaceutical, Inc., Depomed, Inc., Endo Pharmaceuticals Inc., Pfizer Inc., Purdue Pharma L.P., Shionogi & Co. Ltd., Teva Pharmaceuticals Industries Ltd., Vertex, and XenoPort, Inc.; Research support from Eli Lilly and Company, Endo Pharmaceuticals Inc., and Forest Laboratories, Inc. Dr Pilitsis is a consultant for Boston Scientific, Nevro, TerSera, Medtronic, Saluda and Abbott and receives grant support from Medtronic, Boston Scientific, Abbott, Nevro, TerSera, NIH 2R01CA166379-06 and NIH U44NS115111. She is medical advisor for Aim Medical Robotics and Karuna and has stock equity. All other authors declare no conflicts of interest.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
