Abstract
Background:
Neuropathies, the most common complication of diabetes, manifest in various forms, including entrapments, mononeuropathies or, most frequently, a distal symmetric polyneuropathy. Painful diabetic neuropathy (PDN) in the classic “stocking” distribution is a disease of increasing prevalence worldwide and a condition for which standard medical treatment only provides modest relief. Neuromodulation offers a potential alternative to pharmacotherapies given its demonstrated efficacy in other refractory chronic neuropathic pain syndromes. High-quality evidence from randomized controlled trials (RCTs) is available in these other settings for two approaches to spinal cord stimulation (SCS): (1) conventional low-frequency SCS (LF-SCS), which modulates axonal activity in the dorsal column and is paresthesia-dependent, and (2) high-frequency SCS delivered at 10 kilohertz (10 kHz SCS), which targets neurons in the superficial dorsal horn and is paresthesia-independent.
Method:
This review examines the evidence for SCS from published RCTs as well as prospective studies exploring the safety and effectiveness of treating PDN with neuromodulation.
Results:
Two RCTs enrolling 60 and 36 participants with PDN showed treatment with LF-SCS reduced daytime pain by 45% to 55% for up to two years. An RCT testing 10 kHz SCS versus conventional medical management (CMM) in 216 participants with PDN revealed 76% mean pain relief after six months of stimulation. None of the studies revealed unexpected safety issues in the use of neuromodulation in this patient population.
Conclusion:
These well-designed RCTs address the unmet need for improved PDN therapies and provide data on the safety, effectiveness, and durability of SCS therapy.
Keywords
Introduction
Diabetes mellitus is an illness that may cause enormous detriment to a person’s health-related quality of life and is rapidly increasing in prevalence worldwide. 1 In 2018, it was estimated that 34.1 million US adults (13%) had diabetes, 2 and diabetic peripheral neuropathy with both painful and non-painful symptoms as the most common complication.3,4 Painful diabetic neuropathy (PDN) occurs in one-third to one-half of all persons with diabetes, and is a distal, symmetric polyneuropathy that commonly presents as tingling/shooting, burning pain in a stocking distribution that may or may not be accompanied by numbness. 5
Pain due to PDN is challenging to manage, similar to other chronic neuropathic pain conditions. 6 Clinical treatment guidelines recommend anticonvulsant medications such as pregabalin and gabapentin, antidepressants including serotonin and norepinephrine reuptake inhibitors (SNRIs) and tricyclic antidepressants (TCAs), the atypical opioid tapentadol, and topical capsaicin7-13; however, their efficacy is limited and side effects can be intolerable.14,15 In a recent review, Feldman and colleagues 1 issued a clarion call for diabetic neuropathy research: Effective therapies are needed to treat, and perhaps even prevent, diabetic neuropathy to address the growing burden of the disease.
Neuromodulation, including spinal cord stimulation (SCS), dorsal root ganglion (DRG) stimulation, and transcutaneous electrical nerve stimulation (TENS), offer non-pharmacological alternatives for managing the painful symptoms of PDN, particularly in cases that are not responsive to conventional medical management (CMM). High-frequency 10 kHz SCS recently received approval from the Food and Drug Administration for the treatment of PDN. Randomized controlled trials (RCTs) for both conventional, low-frequency SCS (LF-SCS) and high-frequency SCS delivered at 10 kilohertz (10 kHz SCS) have been conducted, providing high-quality evidence for the performance of these therapies and clarifying their role in the PDN treatment pathway. The following narrative review provides a detailed overview of the current state of evidence.
Low-Frequency SCS
Conventional LF-SCS is delivered at frequencies typically ranging from 40 to 60 Hz and has been used for decades to treat chronic pain. Conventional LF-SCS produces paresthesia, a sensation of “tingling” or “pins and needles,” and stimulation parameters are adjusted to overlap paresthesias with the painful area as concordant sensory mapping is necessary for pain relief. 16 The LF-SCS has demonstrated efficacy in neuropathic pain 17 and non-reconstructable peripheral ischemia of the lower extremities.18,19 In the past decade, several studies have explored the application of LF-SCS to various neuropathic pain etiologies, with a specific focus on pain from diabetic neuropathy.
Prospective Studies
A single-arm prospective study in 15 participant by Pluijms et al reported 11 (73%) with successful trial stimulations as assessed by ≥50% pain reduction or Patient Global Impression of Change (PGIC) ≥6 on a 7-point Likert scale. 20 This study showed a significant decrease in daytime and nighttime pain scores compared with baseline levels after 12 months of treatment. Of 11 implanted participant, 7 (64%) were responders (≥50% pain relief) during the day, and 3 (27%) were responders at night. No significant change was observed in EuroQol Five Dimensions Questionnaire (EQ-5D-5L) scores, which assesses health-related quality of life. 21 Quality of sleep improved significantly (P = .04) and 74% of participant reported improved sleep. The only adverse event (AE) reported was a lead displacement during long-term follow-up that required stimulation parameter adjustment (Table 1).
Complications Reported in Prospective Studies of Spinal Cord Stimulation in PDN Patients.
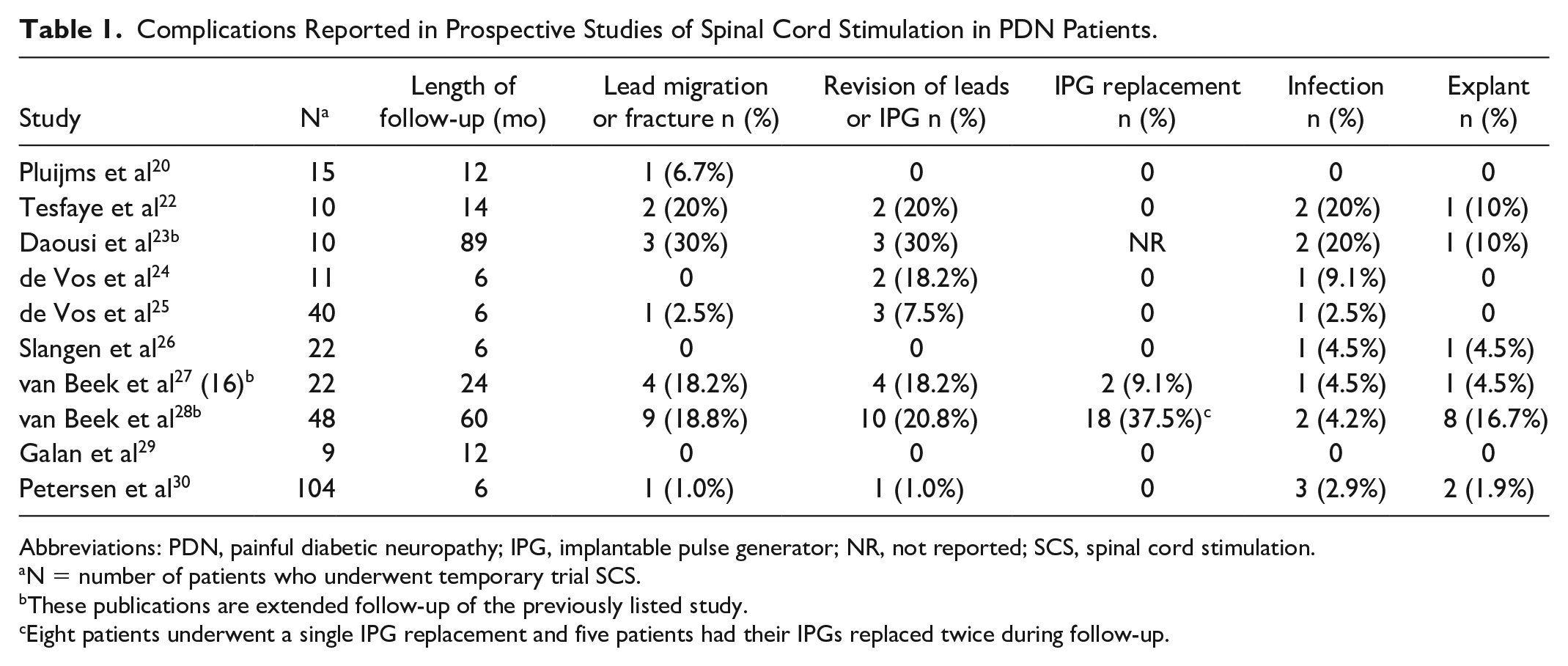
Abbreviations: PDN, painful diabetic neuropathy; IPG, implantable pulse generator; NR, not reported; SCS, spinal cord stimulation.
N = number of patients who underwent temporary trial SCS.
These publications are extended follow-up of the previously listed study.
Eight patients underwent a single IPG replacement and five patients had their IPGs replaced twice during follow-up.
Another prospective study of LF-SCS by Tesfaye et al included ten men with median baseline pain score of 6.3 cm on a 10-cm visual analogue scale (VAS). 22 All participant were trialed with both SCS and sham stimulation, and eight (80%) proceeded to permanent implant. At the end of the study (median stimulation time 14 months), LF-SCS improved peak pain from 8.1 to 2.0 cm on the VAS (P = .03), and six (75%) participant continued to benefit at the last follow-up. Complications included loss of effect after four months in one participant, wound infections, and lead migrations (Table 1). Continued pain relief was documented in the remaining responding participant at follow-up intervals of 3.3 (n = 6) and 7.5 years (n = 4). 23
Finally, a small prospective study of LF-SCS in 11 participant with refractory PDN reported that 9 (82%) experienced significant pain relief and were converted to permanent systems, and 6 (67%) remained responders after six months of treatment. 24 Surgical revisions successfully resolved two cases of inadequate connection between the lead and extension cables, and an infection was also resolved.
A small prospective study tested another stimulation paradigm, Burst SCS, for two weeks in 12 participant with PDN who were already successfully using LF-SCS. 31 Burst SCS delivers clusters of pulses separated by quiescent intervals. 32 Patients had used LF-SCS for an average of 1.8 years which reduced pain from 7.0 to 2.8 cm. Burst stimulation for two weeks further decreased pain to 1.6 cm on average. Adverse events reported by participant who received burst stimulation included headaches, dizziness, and the sensation of “heavy legs.” Several participant reported feeling paresthesia with burst stimulation when lying on their backs despite attempts to set the amplitude below the level of perception.
De Vos et al
The previous single-arm studies established preliminary efficacy data for LF-SCS and warranted further evaluation for Level I RCT evidence. This multicenter, open-label study by de Vos and colleagues was one of the first RCTs of LF-SCS in PDN patients including 60 participant with treatment-refractory symptoms for at least one year and average pain score of 5 cm on a VAS. 25 participant were randomized 2:1 to best medical treatment (BMT) with or without LF-SCS, and 37 of 40 (92%) participant had successful trial stimulation. Outcomes at six months for 36 participant receiving SCS and 18 control participant showed that pain scores decreased by 55% with LF-SCS, while there was no decrease in mean pain among control participant (Table 2). The responder rate was 69% (25 of 36) among participant treated with SCS, while 6% (1 of 18) of controls were responders after six months of treatment.
Summary of Randomized Controlled Trials of Spinal Cord Stimulation Therapy for PDN.
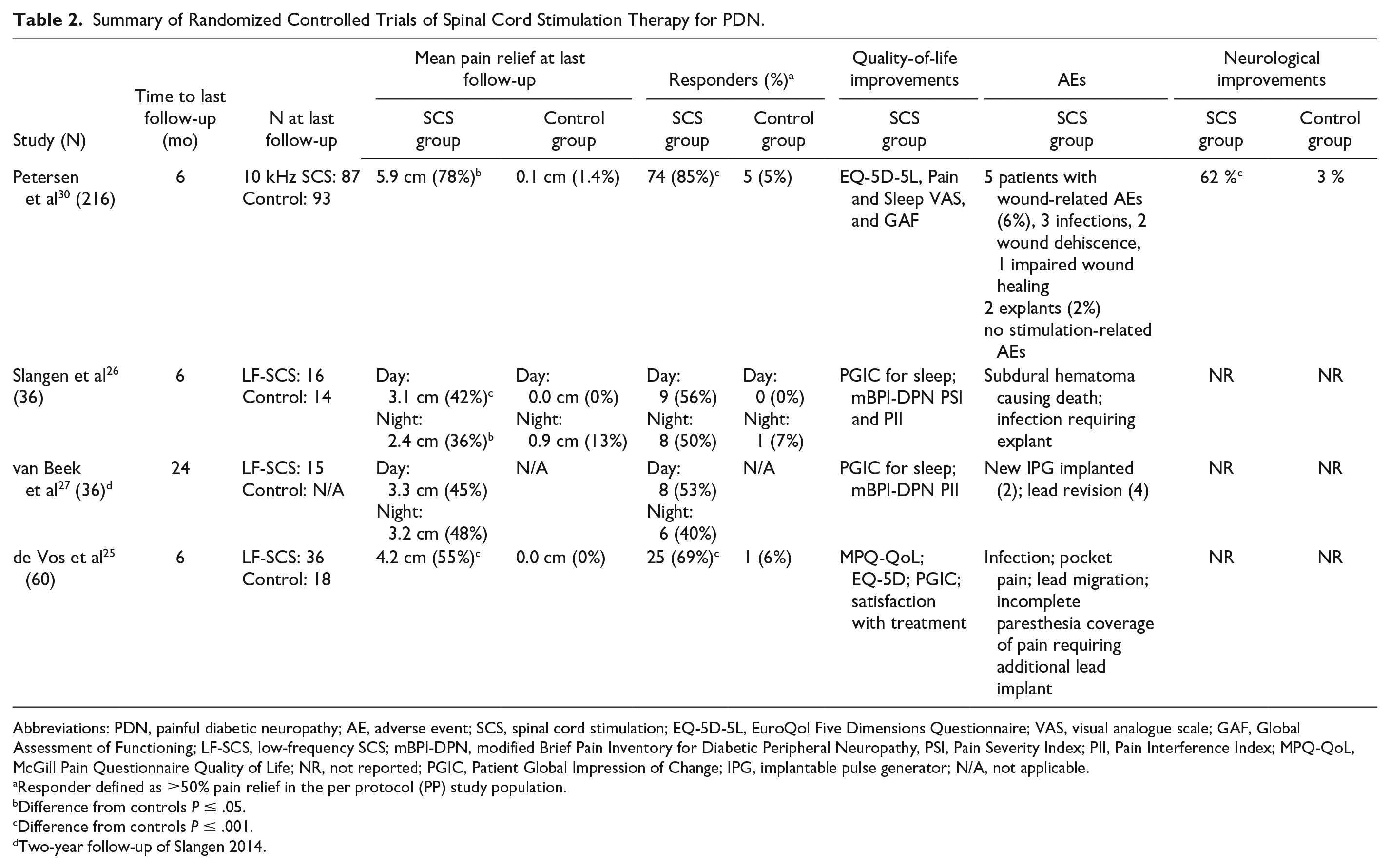
Abbreviations: PDN, painful diabetic neuropathy; AE, adverse event; SCS, spinal cord stimulation; EQ-5D-5L, EuroQol Five Dimensions Questionnaire; VAS, visual analogue scale; GAF, Global Assessment of Functioning; LF-SCS, low-frequency SCS; mBPI-DPN, modified Brief Pain Inventory for Diabetic Peripheral Neuropathy, PSI, Pain Severity Index; PII, Pain Interference Index; MPQ-QoL, McGill Pain Questionnaire Quality of Life; NR, not reported; PGIC, Patient Global Impression of Change; IPG, implantable pulse generator; N/A, not applicable.
Responder defined as ≥50% pain relief in the per protocol (PP) study population.
Difference from controls P ≤ .05.
Difference from controls P ≤ .001.
Two-year follow-up of Slangen 2014.
Quality-of-life assessments showed that EQ-5D-5L scores improved in the SCS group and worsened in the control group, and mean McGill Pain Questionnaire Quality of Life (MPQ-QoL) scores decreased from 16 to 8 after six months of SCS treatment, indicating less pain interference with daily activities and sleep, and this was significantly different from control participant’ mean score, which decreased from 15 to 14 (P < .001). About 70% of both groups reported problems with sleep at baseline, but after six months, 30% of the SCS group and 65% of the control group still had disturbed sleep. Multiple AEs were reported related to device implant and stimulation and are listed in Table 1. Adverse events related to the implantation procedure included pain due to the implantable pulse generator (IPG) in two participant, lead migration, incomplete paresthesia overlap with painful area during trial stimulation leading to placement of another electrode in two participant, and one infection during trial stimulation, which was successfully resolved and followed by a permanent implant. There were no reports of neurological improvement among SCS patients.
Slangen et al and van Beek et al
Another RCT tested LF-SCS in 36 participant with moderate to severe PDN for at least one year that was refractory to conservative treatments with mean intensity during either the day or night of ≥5 out of 10 on a numeric rating scale (NRS). 26 Of 22 participant assigned to SCS treatment, 17 (77%) had successful trial stimulation and received permanently implanted devices. Daytime mean pain NRS scores decreased by 3.1 points after six months of stimulation in the SCS group but did not change in the BMT group (P < .001), and corresponding nighttime pain scores declined 2.4 points in the SCS treatment arm and 0.9 points in the BMT arm (P < .003). The six-month daytime responder rate (≥50% pain relief) was 9 out of 16 (56%) in the per protocol (PP) population of the SCS arm and 0 out of 14 (0%) in the BMT arm (P < .001; Table 2), while nighttime responder rates were 8 out of 16 (50%) in the SCS group and 1 out of 14 (7%) in the BMT group (P < .05). At the last follow-up, 12 of 16 participant (75%) treated with SCS and followed for six months reported a PGIC for pain score ≥6, demonstrating “much” or “very much” improvement, while none of the participant receiving BMT did (P < .001).
Investigators found significant differences in the PGIC for sleep (P < .05); however, there were no improvements in health-related quality-of-life outcomes with SCS treatment as the Medical Outcomes Study SF-36, the Medical Outcomes Study Sleep Scale (MOS-SS), 33 and ED-5D-5L did not reveal any differences between the groups. Serious adverse events (SAEs) were reported in two participant. One participant developed a subdural hematoma following the attempted implantation of the lead for trial stimulation and later died, while another experienced an infection of the SCS system six weeks after it was implanted, which required an explant (Table 1).
After 24 months of stimulation with LF-SCS, mean NRS pain scores were significantly lower than baseline both during the day (–3.3; P < .001) and night (–3.2; P < .001). 27 The responder rates were 53% during the day and 40% at night (Table 2), comparable with six-month rates. Two participant required IPG replacement by 24 months and four participant had lead revisions. The authors did not report any changes in neurological function in the study.
Recently, five-year results have been published for participant who had completed the Slangen et al RCT (n = 33) combined with those from the single-arm pilot study by Pluijms et al (n = 15). 20 Forty of the 48 participant received permanent device implants and five-year follow-up data were available for 22 participant. 28 Mean pain scores (NRS) were significantly lower than baseline scores (–2.4 points/day; –2.1 points/night) and did not differ significantly from the 12-month results. The daytime responder rate at five years was 36% (8 of 22), down from 42% (15 of 36) at one year of follow-up, while the responder rate was 32% at night, down from 36% at one year. The five-year follow-up also revealed 6 participant had device explants due to sustained lack of effectiveness, 7 required lead revisions or replacements, and 13 had at least one battery replacement, as listed in Table 1. These long-term data suggest that LF-SCS can continue to be effective for at least five years for some patients, but surgical interventions required for explant, IPG replacement, and lead revision affect a substantial proportion of participant.
High-Frequency 10 kHz SCS
High-quality RCT data have recently been published for the use of 10 kHz SCS in participant with chronic pain due to PDN. These results are of interest as 10 kHz SCS is the only neuromodulation modality shown to be paresthesia-independent, which obviates concerns about discomfort associated with paresthesias. 34 In addition, pain relief from 10 kHz SCS has been shown to be superior to that produced by LF-SCS in participant with back and leg pain in a head-to-head study comparing the two approaches. 35 Long-term effectiveness has been documented for two years or more in prospective studies of 10 kHz SCS,36-38 in addition to decreased opioid intake, a particularly important advantage in the context of the opioid crisis. 39 Finally, the effectiveness of 10 kHz SCS has been demonstrated for multiple types of pain, including those with a neuropathic component,35,40-44 making it a promising approach to PDN treatment.
Galan et al
A small, prospective, observational study evaluating 10 kHz SCS for the treatment of peripheral polyneuropathy included participant with various etiologies, including nine with PDN. 45 A post hoc analysis for participant with PDN showed that 10 kHz SCS was not only effective at relieving pain due to PDN in this sample, but was also associated with improved neurological functioning, which has not been reported with any other neuromodulation therapies to date. 29 Among the nine participant with PDN in this study, there was one SAE related to the study treatment, implant site dehiscence, as well as two study-related AEs, pain in extremity and implant site seroma, all of which were resolved.
Petersen et al
While initial results were promising for 10 kHz SCS, the small sample makes it difficult to draw major conclusions. A current study underway will establish the effectiveness of this treatment in a large, well-designed RCT. The SENZA-PDN study (NCT03228420) is a prospective, multicenter, open-label RCT with 24-month follow-up comparing the safety and effectiveness of 10 kHz SCS with CMM. 30 The study randomized 216 participant evenly between the two treatment groups, the largest such RCT to date. participant had PDN for at least 1 year with lower limb pain intensity of ≥5 cm on a 10-cm VAS that was refractory to treatment with gabapentin or pregabalin and at least one other class of analgesic.
The mean VAS score decreased in the 10 kHz SCS group from 7.6 cm at baseline to 1.7 cm at six months, corresponding to 78% pain relief, as shown in Table 2. 30 In contrast, mean pain scores for the CMM group decreased from 7.0 cm at baseline to 6.9 cm at six months, and this change was not statistically significant. In addition, pain worsened in 48 CMM participant (52%) and 2 SCS participant (2%) after six months. The responder rate (≥50% pain relief) was significantly higher in the 10 kHz SCS arm (85%) than the CMM treatment arm (5%; P < .001), and the pain remission rate, which has been defined as a VAS of 3 cm or less for at least six consecutive months, 46 was also significantly different between treatment groups, at 60% in the 10 kHz SCS group and 1% in CMM group (P < .001).
The baseline mean score on the Douleur Neuropathique 4 (DN4) questionnaire was used to assess the neuropathic nature of pain in the study participant, and scores above the threshold value of three are consistent with clinically confirmed PDN. 47 The mean DN4 decreased from 6.5 at baseline to 3.5 at six-month follow-up in the 10 kHz SCS group, and the proportion of participant with DN4 scores of three or above decreased by 34.5% during that time. In contrast, there was a non-significant increase from 6.4 at baseline to 6.6 at six months in the mean DN4 score of the control group, and the proportion of control participant with DN4 ≥3 declined by 1.1%.
The SENZA-PDN RCT evaluated patients’ neurological function, including sensory, motor, and reflex testing at baseline and each follow-up. 48 At six months, three participant in the CMM group (3%) and 52 in the 10 kHz SCS group (62%) demonstrated neurological improvements over baseline in at least one category of function without deficits in any category (P < .001). 30 Improvements were primarily observed in assessments of sensory function. In contrast, 17 participant in the CMM arm (19%) and 5 in the 10 kHz SCS arm (6%) had clinically meaningful deficits in at least one category compared with baseline, regardless of any concurrent improvements in other categories.
The SENZA-PDN study included several assessments of health-related quality of life, including Diabetes Quality of Life (DQOL), 49 EQ-5D-5L, and Pain and Sleep Questionnaire (PSQ-3). 50 Mean scores on the EQ-5D-5L, which assesses general health-related quality of life, did not change significantly in the CMM group, but improved by 0.129 in the 10 kHz SCS group, which was both statistically significant and was two- to four-fold the minimally important clinical difference of .03 to .05. 51 Similarly, sleep disturbances due to pain increased by 5.3% in the CMM group while decreasing 61.9% in the 10 kHz SCS group. No stimulation-related AEs were reported for the 10 kHz SCS group. Explants were required for 2 of 90 participant in the 10 kHz SCS group (2%), and 5 participant (5.6%) experienced AEs related to wound healing, including wound dehiscence (2), impaired healing (1), and infections (3).
Discussion
Painful diabetic neuropathy, like other neuropathic pain conditions, can be difficult to treat, resulting in a large patient population whose pain does not adequately respond to recommended pharmacotherapies.14,15 Over the last decade, the recommended treatments have remained largely unchanged. Patients continue to suffer not only from pain, but also from depression, anxiety, decreased quality of life, employment disruption, and sleep deprivation.52-54 There is a large unmet need for treatment for these patients, and neuromodulation may help meet this unmet need. The highest quality evidence comes from RCTs enrolling participant with PDN, and several of these have been published for conventional LF-SCS and high-frequency 10 kHz SCS.
The LF-SCS has been used for various chronic pain conditions for decades and has high-quality evidence supporting its use in the treatment of PDN. First shown to be effective in patients with peripheral neuropathies in the 1990s, 55 the evidence now includes two RCTs, one of which has five-year follow-up data on a subset of patients. The LF-SCS is associated with a loss of therapeutic effect, which is the most common reason for device explant, 56 and it is known that tolerance is one of the primary limitations of conventional SCS resulting in a loss of effect over time. 57 The studies available in PDN have, likewise, shown effectiveness of conventional SCS trended down over time.25-28
The SENZA-PDN study now underway has shown improved results over six months compared with LF-SCS, including a greater proportion of pain responders with at least 50% pain relief and a larger decrease in pain scores (Figure 1). This well-designed study includes a large sample size, long-term follow-up, and monitoring of neurological function.45,29,48 In addition, an independent panel of physician medical monitors reviewed each participant to provide oversight and maximize appropriate patient selection. 48
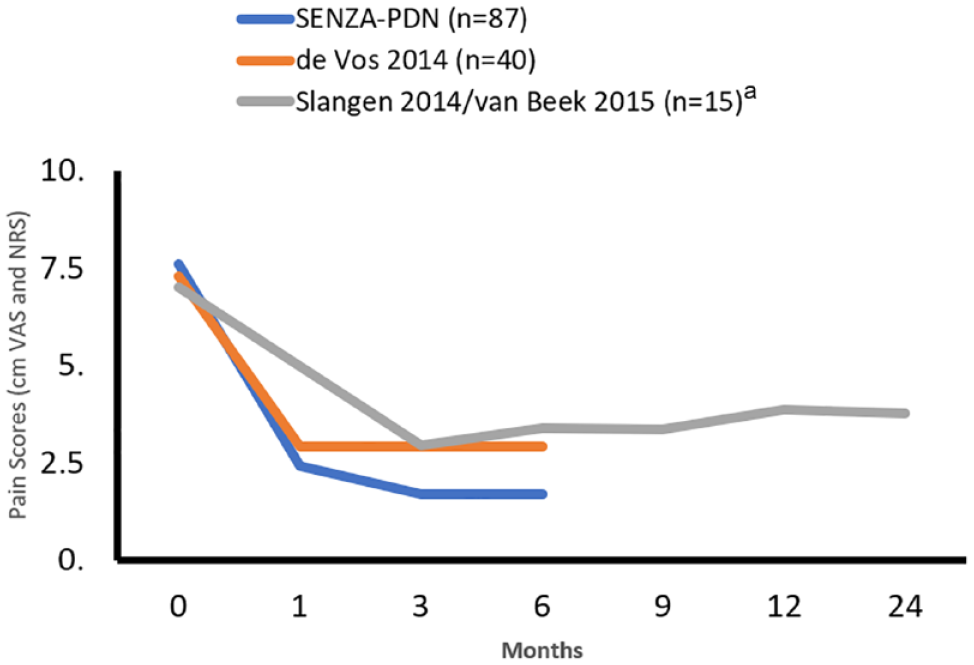
Change in mean pain scores over time in PDN RCTs. Abbreviations: PDN, painful diabetic neuropathy; RCT, randomized controlled trial.
Treatment of chronic, treatment-refractory pain with 10 kHz SCS has been shown to be effective and durable in a variety of contexts.36-38,41,43,58-60 The SENZA-PDN will have 24 months of follow-up data to evaluate long-term pain relief and the durability of neurological improvements that have been reported at six months. Improvement in neurological functioning, including reflex and sensory function, is a compelling finding that has not been reported for other treatments of PDN, either pharmacological or neuromodulation. Quality-of-life outcomes, including EQ-5D-5L and sleep disturbances, have also been improved through six months of treatment with 10 kHz SCS, and will allow direct comparison with past LF-SCS studies. Finally, medication use will be assessed in SENZA-PDN to determine whether this patient population will benefit from the opioid-sparing effects reported for 10 kHz SCS in multiple other studies including the SENZA-RCT, prospective, and real-world studies.35-37,40,44,61-64
Despite a high burden of disease from PDN, pharmaceutical companies have decreased research and clinical trials in this area due to incomplete understanding of the condition. 1 Targeting the dorsal horn of the spinal cord represents a more efficient site for the treatment of neuropathic pain than systemic medications whose effect on forebrain networks involved in pain processing can lead to undesirable side effects on cognition, mood, and other limbic functions. 65 Recent studies have demonstrated that SCS procedures can be performed safely for persons with diabetes without elevated risk of wound healing complications.30,66,67
Alternate neuromodulation strategies have been proposed as potential avenues of study, including peripheral nerve and DRG stimulation. Identifying optimal targets for peripheral nerve stimulation is a challenge for PDN given the diffuse nature of disease. Several small retrospective case series with DRG neuromodulation have suggested effectiveness in patients with PDN, although rigorous studies are lacking.68,69 There are potential drawbacks to consider for DRG stimulation as a treatment for PDN, including using a paresthesia-dependent approach and possible lengthier procedures for device implantation. 70 There is high-quality evidence for DRG stimulation to treat complex regional pain syndrome (CRPS), another type of neuropathic pain condition 71 ; however, the pathology of CRPS differs from that of PDN, and it is unclear whether similar outcomes could be achieved in this patient population. Dorsal root ganglion provides targeted stimulation more appropriate for mononeuropathies instead of diffuse polyneuropathies and would require placement of several leads bilaterally to address PDN, further increasing the length of the implant procedure and concomitant risk to the patient. 72
Conclusion
There is currently a substantial unmet need for safe and effective treatments for PDN. Many patients with PDN do not benefit from pharmacotherapies in current use and are candidates for treatment with neuromodulation. Conventional LF-SCS and high-frequency 10 kHz SCS are supported by high-quality evidence from RCTs and prospective studies. High-frequency 10 kHz SCS offers several advantages over LF-SCS, including greater pain relief, a higher proportion of patients achieving treatment success, paresthesia-independence, and evidence of improved neurological function. Neuromodulation with SCS, especially with 10 kHz SCS, offers a pathway forward for improving the lives of PDN patients.
Footnotes
Acknowledgements
The authors thank Erik MacLaren, from Galen Medical Writing, LLC for writing assistance during preparation of this manuscript.
Abbreviations
10 kHz SCS, high-frequency spinal cord stimulation delivered at 10 kilohertz; AE, adverse event; BMT, best medical treatment; CGIC, Clinician Global Impression of Change; CMM, conventional medical management; DN4, Douleur Neuropathique 4; DQOL, Diabetes Quality of Life; EQ-5D-5L, EuroQol Five Dimensions Questionnaire; GAF, Global Assessment of Functioning; IPG, implantable pulse generator; MOS-SS, Medical Outcomes Study Sleep Scale; NRS, numeric rating scale; PDN, painful diabetic neuropathy; PGIC, Patient Global Impression of Change; PII, Pain Interference Index; PSI, Pain Severity Index; PSQ-3, Pain and Sleep Questionnaire; RCT, randomized controlled trial; SAE, serious adverse event; SCS, spinal cord stimulation; SNRIs, serotonin and norepinephrine reuptake inhibitors; TCAs, tricyclic antidepressants; DRG, dorsal root ganglion; TENS, transcutaneous electrical nerve stimulation; VAS, visual analogue scale; CRPS, complex regional pain syndrome
Declaration of Conflicting Interest
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr Strand has received consulting fees from Abbott and Nevro, and has stock ownership in Abbott, Dexcom, Nevro, and Tandem Diabetes. Dr Burkey has received consulting fees from Nevro.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Nevro Corp. provided funding for a medical writer to help draft this manuscript.
