Abstract
Spinal cord stimulation (SCS) technology has been recently approved by the US Food and Drug Administration (FDA) for painful diabetic neuropathy (PDN). The treatment involves surgical implantation of electrodes and a power source that delivers electrical current to the spinal cord. This treatment decreases the perception of pain in many chronic pain conditions, such as PDN. The number of patients with PDN treated with SCS and the amount of data describing their outcomes is expected to increase given four factors: (1) the large number of patients with this diagnosis, (2) the poor results that have been obtained for pain relief with pharmacotherapy and noninvasive non-pharmacotherapy, (3) the results to date with investigational SCS technology, and (4) the recent FDA approval of systems that deliver this treatment. Whereas traditional SCS replaces pain with paresthesias, a new form of SCS, called high-frequency 10-kHz SCS, first used for pain in 2015, can relieve PDN pain without causing paresthesias, although not all patients experience pain relief by SCS. This article describes (1) an overview of SCS technology, (2) the use of SCS for diseases other than diabetes, (3) the use of SCS for PDN, (4) a comparison of high-frequency 10-kHz and traditional SCS for PDN, (5) other SCS technology for PDN, (6) deployment of SCS systems, (7) barriers to the use of SCS for PDN, (8) risks of SCS technology, (9) current recommendations for using SCS for PDN, and (10) future developments in SCS.
Introduction
At least 50% of people with diabetes will develop neuropathy, 1 and approximately one-third of them will develop painful diabetic neuropathy (PDN).2-4 Pharmacotherapy for PDN is often not successful and may be associated with intolerable side effects. 5 Noninvasive lifestyle modifications are rarely adequate. 6 In the past year, two devices that deliver spinal cord stimulation (SCS) have been explicitly cleared by the US Food and Drug Administration (FDA) to treat PDN. These two devices are also cleared in the European Union under a general indication for various types of chronic pain, which include PDN. These two devices are a high-frequency (HF) 10-kHz stimulation system that was cleared by the FDA in 2021 7 and a traditional (also known as low-frequency, tonic, or paresthesia-based [PB]) system that was cleared by the FDA in 2022. 8 Both traditional and HF methods have only been tested in a small number of studies to date. A search on PubMed on July 20, 2022 of the terms “spinal,” “cord,” “stimulation,” “diabetes,” and “neuropathy” produced 170 articles (Figure 1). A similar search was performed on Google Scholar on the same day. This article presents the current status of SCS as treatment for refractory PDN.
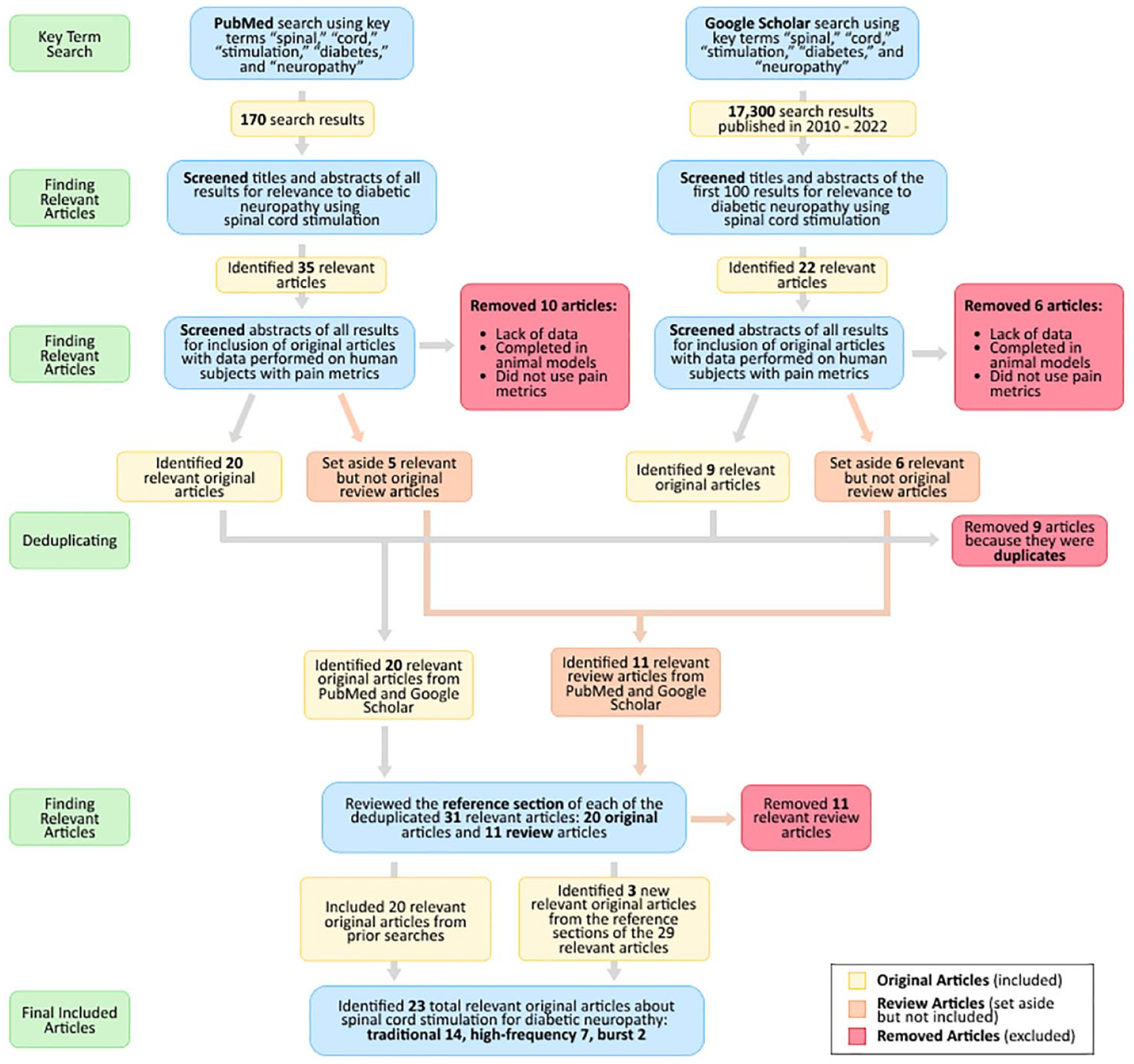
PRISMA diagram for identifying relevant original articles on spinal cord stimulation to treat painful diabetic neuropathy. Searches were completed on July 20, 2022. Studies were included if they used spinal cord stimulation, treated diabetic neuropathy, completed the study on human subjects, reported original data, and presented pain scores as a measurement of pain.
Overview of SCS Technology
Neuromodulation related to SCS is a technology that acts directly upon nerves through alteration or modulation of neuronal activity by delivering titratable electrical stimulation to a target area. This process interferes with the transmission of pain signals from the periphery to the central nervous system.2,9 The goal of neuromodulation is to decrease a patient’s pain and increase their functionality and quality of life. 2 The mechanism of spinal neuromodulation appears to be that electrical stimulation of the spinal cord impacts the processing of painful inputs from peripheral nerve fibers in the symptomatic body area. The columns of the spinal cord contain white matter, which consist of myelinated axon bundles, and the horns of the spinal cord contain gray matter, which consist of unmyelinated cell bodies of neurons (Figure 2). Traditional SCS stimulates neurons in the dorsal columns, resulting in (1) antidromic propagation to inhibit painful signals which enter the spinal cord from the periphery and (2) orthodromic propagation to activate supraspinal pain-processing centers in the brain.10-12 High-frequency 10-kHz SCS stimulates the dorsal horns.13,14 Thus, neuromodulation techniques reduce pain by altering the function of the structures that elicit the sensation of pain. Five factors affect the amount of energy delivery during SCS (Table 1). 15
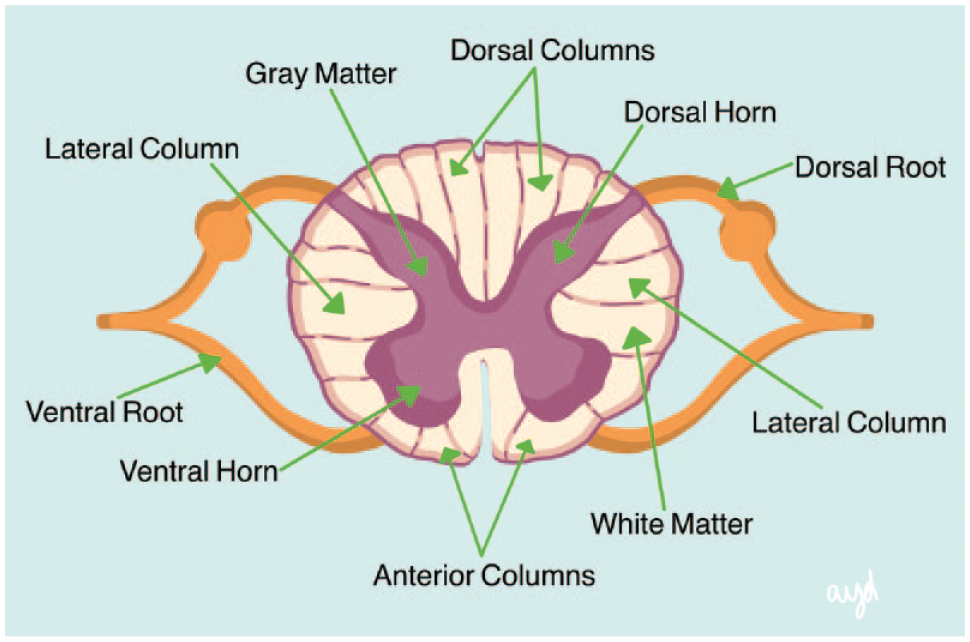
Spinal cord anatomy.Source: Figure provided by Ashley Y. DuBord.
Five Factors Affecting the Amount of Energy Delivery During SCS.

An SCS device is a neuromodulator consisting of three components: (1) an implantable pulse generator; (2) two lead wires, each with electrodes that are placed in the epidural space to deliver electrical impulses to the spinal cord; and (3) a hand-held remote controller that turns the device on and off and adjusts the settings (Figure 3). Usually two leads are implanted (one for each side of the spinal cord), but for some patients, a single lead is adequate; very rarely will a patient require more than two leads. 16 The pulse generator contains a battery and sends electrical signals to the leads that stimulate the spinal cord. The battery can either be nonrechargeable or rechargeable via induction at regular intervals by an external wearable power source 17 placed over the implanted pulse generator. 18 An implanted stimulator with a nonrechargeable battery must be replaced every two to five years, and a rechargeable implanted stimulator with an external battery may last eight to ten years. 19 Some patients prefer to avoid battery recharging and instead select a nonrechargeable SCS generator, which requires more frequent replacement. Battery size affects recharge interval rather than the time-to-replacement. 16 In some cases, patients may be directed to a pulse generator containing a rechargeable battery, which requires less frequent recharging and surgical replacement. A rechargeable battery as opposed to a nonrechargeable battery could save up to $100,000 from fewer procedural expenses over a patient’s lifetime. Fewer replacement procedures can also result in fewer procedural complications. 20 However, patients with diabetes have an increased risk of surgical site infections 21 for various types of procedures but not necessarily for SCS implantation. 22
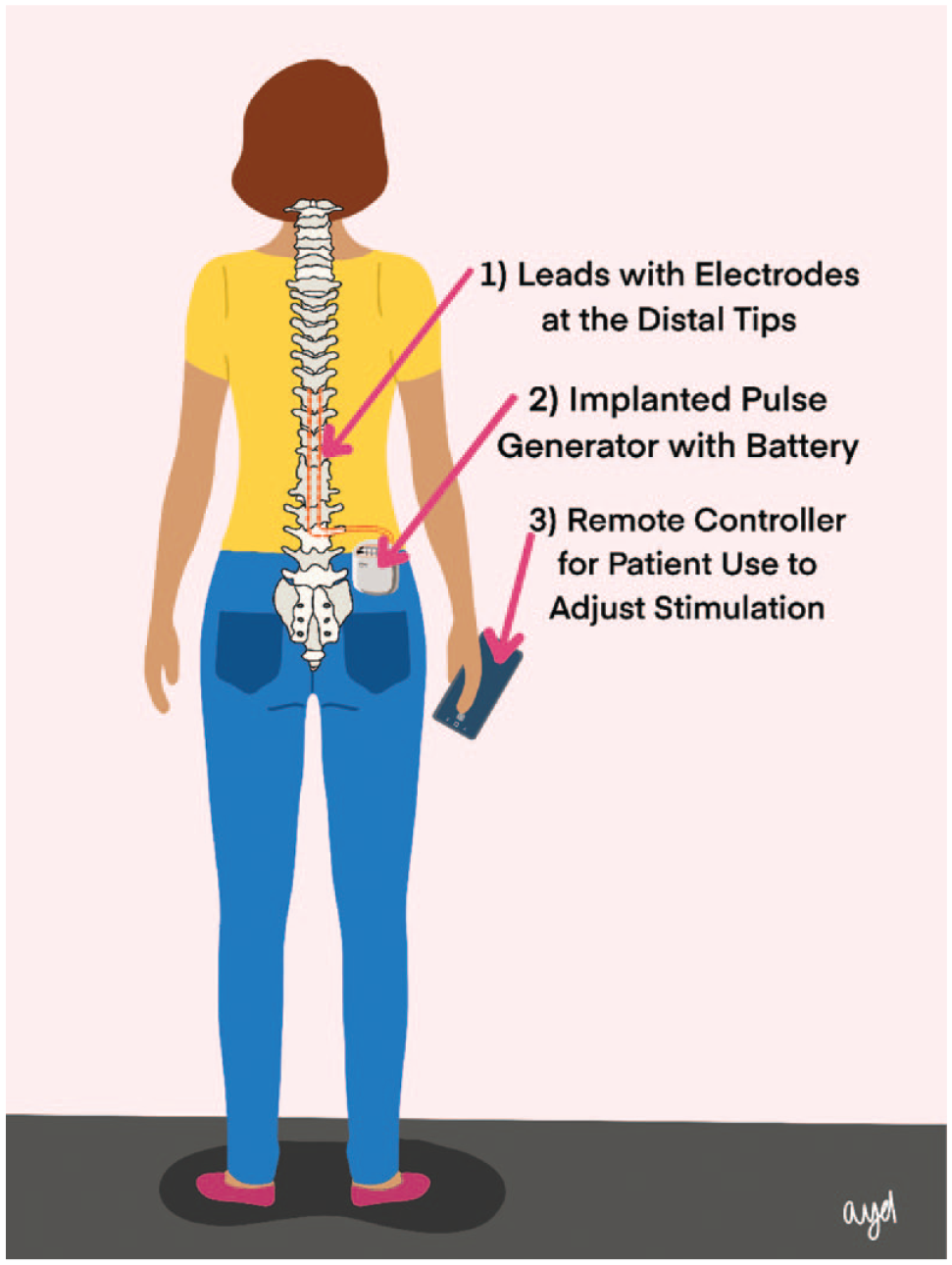
Image of a person with a spinal cord stimulator with two leads implanted adjacent to the thoracic spine.
Three types of electrical waveform patterns with SCS for PDN have been reported most frequently, including traditional, HF, and burst.23-25 Traditional SCS therapy provides steady delivery of electric current with a low frequency of 40 to 60 Hz, with pulse amplitudes ranging from 2 to 8 mA and typical pulse widths of 200 to 500 µs. 26 This type of current elicits paresthesias, and programming is targeted to maximize overlap of the paresthesias with painful regions. Newer paresthesia-free systems deliver HF current with low-amplitude current of up to 5 mA. Burst SCS delivers intermittent bursts of electric pulses at HF (five pulses at 500 Hz delivered 40 times/s). 27 These latter two modes do not generate paresthesias. 27
Traditional SCS stimulates the dorsal column large fibers that carry paresthesia sensations, but HF 10-kHz SCS does not affect these pathways (resulting in an absence of paresthesias associated with pain relief). 28 HF 10-kHz SCS, but not burst SCS, stimulates dorsal horn inhibitory neurons, resulting in decreased spinal pain processing. 29 The exact mechanism of burst SCS is not known, but as burst and traditional stimulation in the same location of the thalamus can activate distinct, nonoverlapping perceptive pathways, 30 a unique mechanism for this form of SCS will likely be eventually discovered. Traditional SCS (50 Hz) was originally approved by the FDA as a treatment for intractable back and leg pain in 1989, 31 and in 2015, the FDA also approved HF SCS for these indications. 32 A burst SCS system was approved by the FDA in 2016. 33
The Use of SCS for Diseases Other Than Diabetes
The first trial of HF SCS for painful neuropathies of any type was the SENZA-PPN trial, which assessed 26 patients, nine of whom had PDN. 34 A post hoc analysis of the outcomes in the nine patients with PDN by the same investigator team demonstrated improvements in pain score and responses to pain by 87.5% of patients. 35
The next major trial to build evidence of treatment efficacy (which, like most of the trials of SCS technology, was sponsored by the product manufacturer) was a large randomized clinical trial (RCT) of HF SCS, the SENZA-RCT that compared HF SCS at 10 kHz with traditional SCS for back and leg pain. In this trial, HF SCS therapy, compared with traditional SCS, resulted in significantly greater improvement in all pain at 3 months 36 and 24 months. 37
Contrary to the results of the SENZA-RCT, four studies have found no significant difference between traditional and HF SCS for treating different kinds of pain. In 2017, De Andres and colleagues reported results of a prospective comparison of traditional and HF SCS for back pain. They found that outcomes did not differ between patients receiving these two modalities of SCS. 38 In 2018, Thomson and colleagues conducted a prospective trial of multiple frequencies of SCS for multiple causes of chronic pain (called PROCO RCT) and concluded that all frequencies tested, from 1 to 10 kHz, provided equivalent pain relief and improvement in quality of life. They defined a successful outcome as pain relief at least 30% instead of the more widely used end point of at least 50% pain relief. 39 In 2019, Bolash compared traditional and HF SCS for pain due to failed back surgery in a prospective study and found no significant difference in pain relief between the two modalities. 17 In 2021, Hagedorn and colleagues reported a retrospective comparison of 163 persons receiving either traditional SCS or HF SCS for a variety of diagnoses and found no statistically significant difference in outcomes between the two groups. 40 To the extent that explantation rates reflect device failure, these rates have been similar around 1% to 4% per year for both traditional and HF SCS systems in the literature in studies conducted with widely divergent protocols.23,41-44
The Use of SCS for PDN
Current pharmacotherapy for PDN is frequently disappointing because of poor results or side effects. Therefore, many patients have resorted to new treatment methods, such as SCS. For PDN, both traditional SCS (Table 2) and HF SCS (Table 3), compared with pharmacotherapy, have been shown to improve pain scores significantly more effectively. Burst SCS has also been effective for PDN in a very limited number of trials (Table 4). However, the use of SCS for PDN has been limited by (1) a dearth of RCTs with long-term follow-up showing benefits and (2) a circuitous path for symptomatic patients to obtain the treatment.
Traditional SCS Studies.
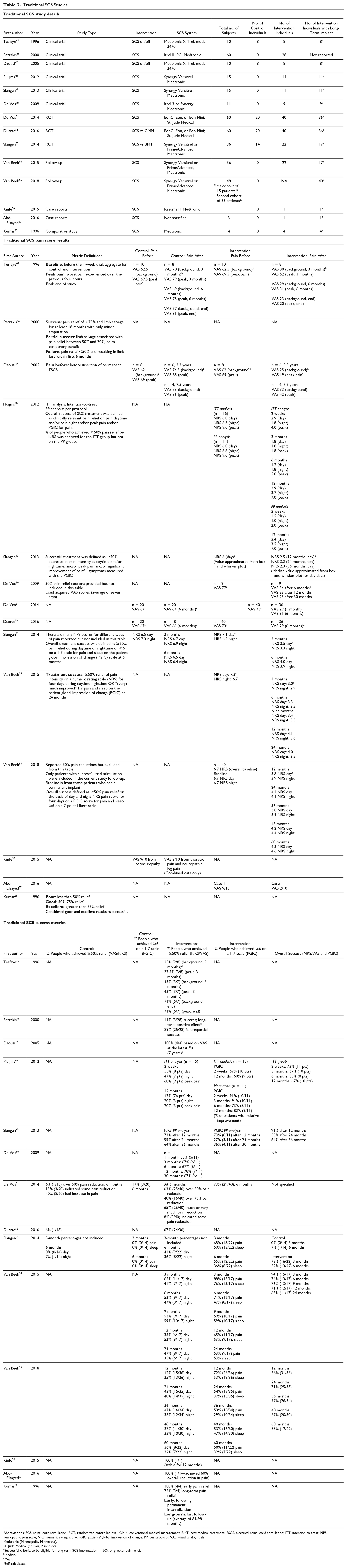
Abbreviations: SCS, spinal cord stimulation; RCT, randomized controlled trial; CMM, conventional medical management; BMT, best medical treatment; ESCS, electrical spinal cord stimulation; ITT, intention-to-treat; NPS, neuropathic pain scale; NRS, numeric rating score; PGIC, patients’ global impression of change; PP, per protocol; VAS, visual analog scale.
Medtronic (Minneapolis, Minnesota).
St. Jude Medical (St. Paul, Minnesota).
Successful criteria to be eligible for long-term SCS implantation = 50% or greater pain relief.
Median.
Mean.
Self-calculated.
High-Frequency SCS Studies.
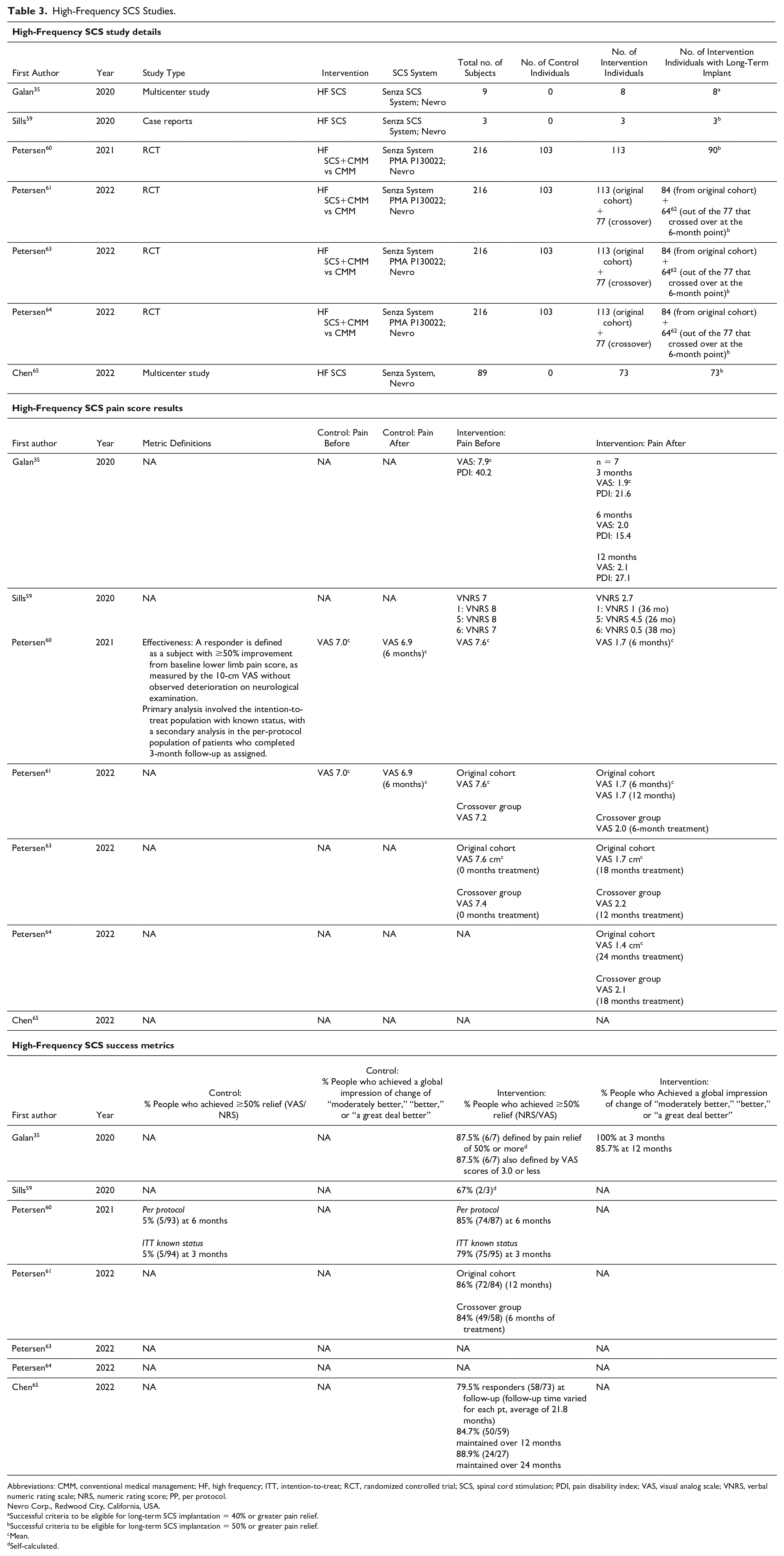
Abbreviations: CMM, conventional medical management; HF, high frequency; ITT, intention-to-treat; RCT, randomized controlled trial; SCS, spinal cord stimulation; PDI, pain disability index; VAS, visual analog scale; VNRS, verbal numeric rating scale; NRS, numeric rating score; PP, per protocol.
Nevro Corp., Redwood City, California, USA.
Successful criteria to be eligible for long-term SCS implantation = 40% or greater pain relief.
Successful criteria to be eligible for long-term SCS implantation = 50% or greater pain relief.
Mean.
Self-calculated.
Burst SCS Studies.
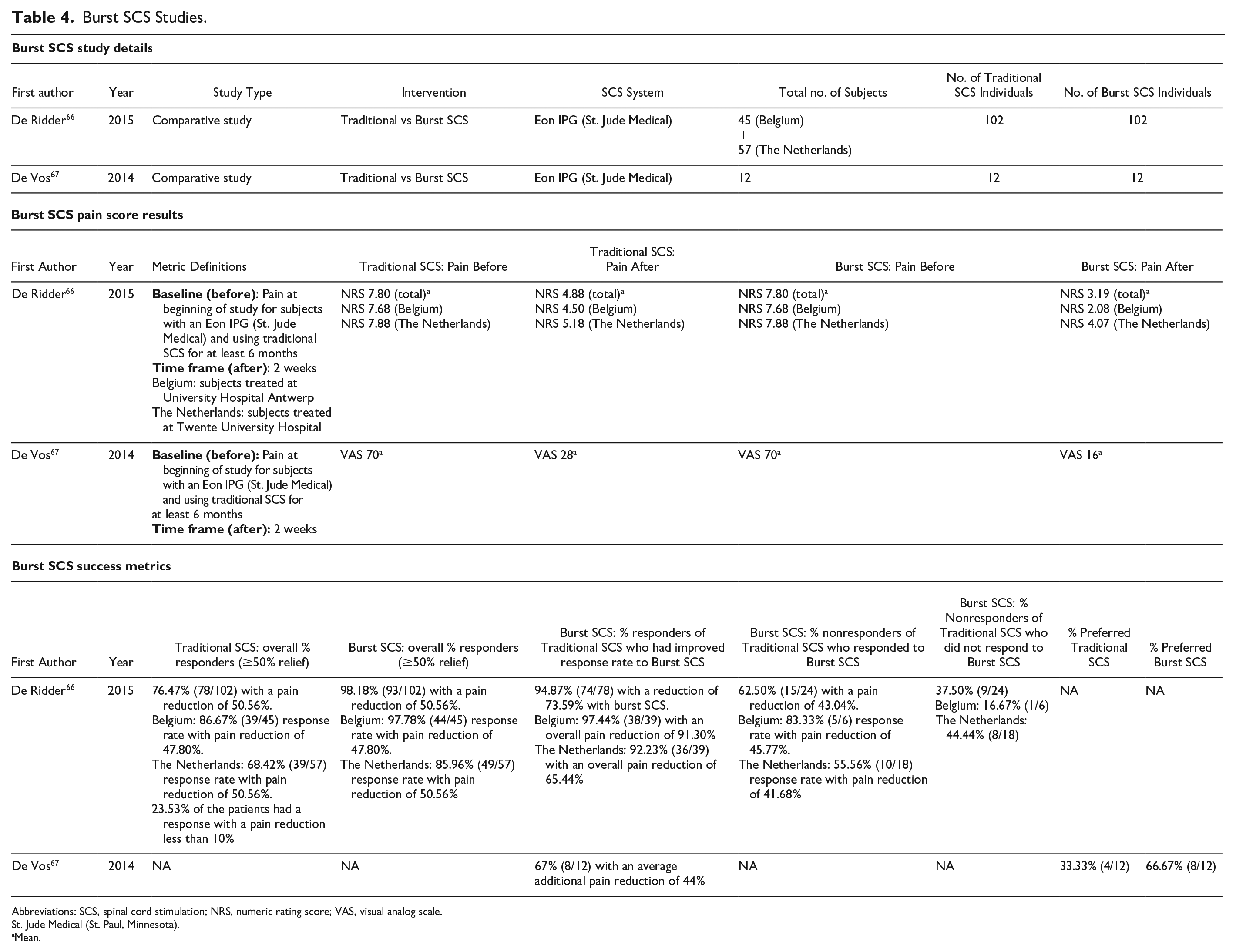
Abbreviations: SCS, spinal cord stimulation; NRS, numeric rating score; VAS, visual analog scale.
St. Jude Medical (St. Paul, Minnesota).
Mean.
There is often a disconnect between clinicians and patients with chronic PDN. Primary care physicians must refer them to an endocrinologist for diabetes management, who must then refer them to a neurologist for pharmacotherapy, who must finally refer them to a pain specialist who must recommend and then perform SCS electrode insertion. This lengthy process deflects many patients from this treatment. At a tertiary care center in 2019, only 1% of patients who saw endocrinology, neurology, or neurosurgery specialists received SCS treatment. 68
In the past year, two SCS systems have been cleared by the FDA for the specific purpose of treating PDN. On July 19, 2021, the Nevro Corporation (Redwood City, California) announced approval by the FDA of their Senza System to treat chronic pain associated with PDN (Figure 4). 69 On January 24, 2022, Medtronic (Dublin, Ireland) announced approval by the FDA of their traditional Intellis rechargeable neurostimulator and Vanta recharge-free neurostimulator for the treatment of chronic pain associated with diabetic peripheral neuropathy. 8 Two other SCS systems are cleared by the FDA but not for PDN. They are the Proclaim XR System by Abbott (Alameda, California) and the WaveWriter Alpha portfolio of Spinal Cord Stimulator Systems by Boston Scientific (Marlborough, Massachusetts). The four FDA-cleared SCS systems are presented in Figure 4. 8
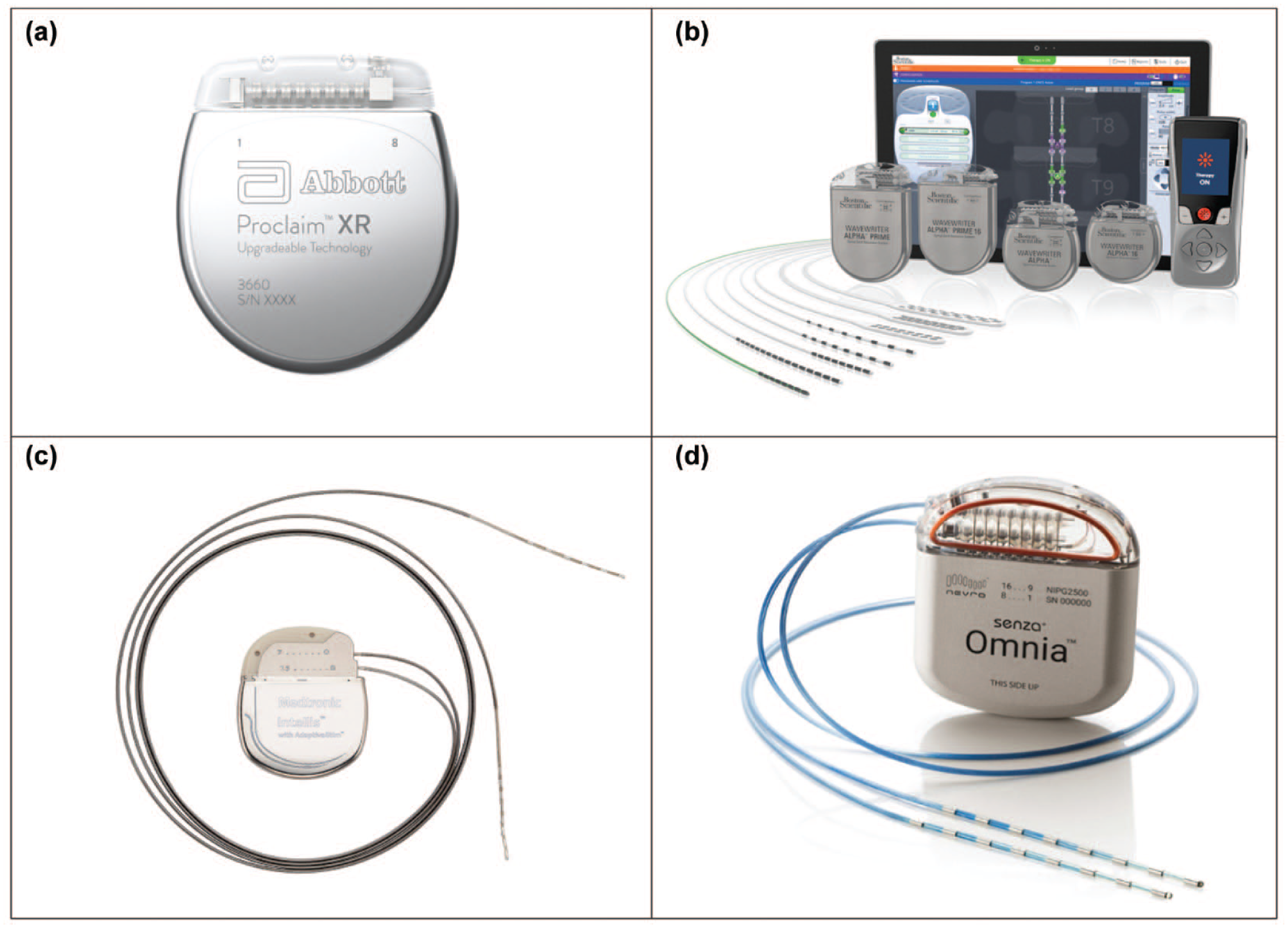
FDA-approved Spinal Cord Stimulation Systems (in alphabetical order according to the manufacturer). (a) Proclaim XR System (Abbott Neuromodulation, Austin, Texas), (b) WaveWriter Alpha portfolio of Spinal Cord Stimulator Systems (Boston Scientific, Marlborough, Massachusetts), (c) The Intellis with AdaptiveStim rechargeable neurostimulator (Medtronic, Minneapolis, Minnesota), (d) Senza Omnia (Nevro, Redwood City, California). Abbreviation: FDA, US Food and Drug Administration.
HF 10-kHz Versus Traditional SCS for PDN
Two RCTs of traditional SCS for PDN have been reported.51,53 In both studies, a significantly higher percentage of patients treated with SCS in addition to the best practice of medical management, compared with the best practice of medical management alone, had a successful response, which was defined differently in each RCT. A successful response was defined by De Vos et al 51 as a 50% or greater reduction in pain based on a numeric pain score, such as either a numerical rating score (NRS) or visual analog scale (VAS). A successful response was defined by Slangen et al 53 as a 50% or greater reduction in pain on an NRS during daytime or nighttime or an improvement in pain and sleep of six or more on a one-to-seven subjective scale of overall improvement, called the patient’s global impression of change (PGIC). Successful responses with SCS versus control therapy occurred in 63% versus 6% of participants, respectively, in the Netherlands 51 and in 59% versus 7%, respectively, of participants in four European country cohorts. 53 When their results were pooled together in a meta-analysis, the conclusion was that SCS is an effective therapeutic in addition to the best medical therapy in reducing pain intensity and improving health-related quality of life in patients with PDN. 70 Favorable results of 14 trials of traditional SCS, including these two trials, plus 12 other trials that were either non-RCTs or else a combination of other primary trials, are all presented in Table 2.
The most important study of HF 10-kHz SCS is the ongoing multicenter open-label randomized controlled 24-month SENZA-PDN study of patients with lower limb PDN that was (1) present for at least 12 months, (2) refractory to treatment with gabapentanoids and at least one other class of analgesics, and (3) characterized by an intensity of 5-cm or more on a 10-cm VAS. This study at 6, 60 12, 61 and 18 63 months has demonstrated that HF SCS (n = 113), compared with conventional medical management (CMM) (n = 103), has provided superior pain relief36,37 and health-related quality of life 71 measures.
Hagedorn and colleagues stated in 2022 that, for PDN, if lifestyle management and improved glycemic control followed by pharmacotherapy are not successful, then the next step should be neuromodulation therapy with HF 10-kHz SCS. They also stated that for PDN, the recent SENZA-PDN study has demonstrated clear improvement in pain and quality of life compared with lifestyle management, pharmacotherapy, and other SCS programming options. 72
Advantages of HF 10-kHz SCS over traditional SCS include (1) no unpleasant or persistent paresthesias, which means that activities (including driving and sleeping) are less impacted by uncomfortable paresthesias; (2) superiority during the implant procedure because there is no need to map painful areas with HF SCS, which means that the procedure time for HF 10-kHz SCS is faster and less dependent on exposure to fluoroscopic radiation 73 ; and (3) evidence that HF 10-kHz SCS, but not traditional SCS, excites pain-inhibitory neurons in the dorsal horns of the spinal column and spares ascending pathways that carry sensations of paresthesias26,74,75 (Table 3). On the contrary, traditional SCS, compared with HF 10-kHz SCS, has been used longer, so its properties are better known, and there is a more robust data set supporting the use of traditional SCS (Table 2) for diseases other than PDN. As the total number of RCTs is low for both forms of SCS, more research must be done and more RCTs with larger populations must be performed to definitively compare the two most widely used types of SCS. Nonetheless, both traditional SCS and HF 10-kHz SCS appear to be safe and effective.
Other SCS Technology for PDN
In addition to traditional, HF, and burst SCS, various alternative waveforms and stimulation patterns have been used to a limited extent for SCS but have not been reported for use with PDN. 76 Combinations of various frequencies have been used with SCS, including (1) differential target multiplexed SCS, which uses multiplexed electrical pulses that differ in frequency, pulse width, and amplitude 77 ; (2) closed-loop SCS, which records spinal cord-evoked compound action potentials in patients during daily use and adjusts the stimulation current to maintain constant activation within patients’ therapeutic windows 78 ; and sweet spot selection, which first identifies the optimal location of the spinal cord for pain relief (known as the sweet spot) using 10-kHz stimulation and then titrates down the frequency to the lowest rate that provides pain relief without paresthesias. 39
Deployment of SCS Systems
Patients considered for SCS technology are selected when more conservative treatments, including psychological treatment, physical therapy, and pharmacotherapy, have failed to provide sufficient symptom reduction.79,80 SCS candidates undergo formal psychological evaluation to assess whether there are psychological barriers that may inhibit a response to SCS therapy. 81
Prior to undergoing surgical implantation of permanent SCS hardware, patients undergo a trial (or “test drive”) for approximately 7 to 14 days to allow them to experience the expected reduction in pain and/or associated symptoms of permanent SCS implantation without the risk of an infection from an implanted system. The SCS trial is an outpatient procedure requiring a field block by the surgeon using local anesthetic with minimal or no sedation. The trial is performed with full barrier surgical precautions to minimize the risk of infection. 82
During the trial, one or more SCS leads (wires with electrodes at the distal tips) are inserted percutaneously via needles (usually 14-gauge) into the posterior epidural space using intermittent fluoroscopic guidance, such that the electrodes are able to modulate the pain pathways of the dorsal spinal cord at the appropriate level. The SCS leads extend from the patient’s epidural space through the skin and connect to an external pulse generator to mimic long-term treatment, which identifies the subjects who are likely to benefit from permanent implantation.
The physician and the patient can select from electrical stimulation “programs” designed to reduce pain at various points within the trial. 83 Multiple variables for stimulation can be altered during programming of SCS generators, including intensity, frequency, pulse width, and even patterns of stimulation. With traditional SCS, the patient perceives this electrical stimulation as paresthesias substituted for pain. However, modern SCS systems can provide HF stimulation that the patient does not consciously sense. Staffing for the SCS trial requires an interventional pain physician or neurosurgeon to perform the procedure, a radiologic technologist to operate the fluoroscopy machine, and a representative employed by the manufacturer of the SCS equipment. A surgical technologist and a circulating nurse may also be present.
A reduction in pain by at least 50% is considered a successful trial. Such success during a trial of SCS is achieved by approximately 80% of patients with PDN. 84 Other outcomes, including reduced opioid consumption or improved functional status, also indicate a positive response.
Permanent surgical implantation of a spinal cord stimulator, which consists of the SCS leads and pulse generator, is an outpatient procedure that requires either sedation or general anesthesia and is performed with similar staff as a trial implantation. This operation typically requires two surgical incisions. A vertical incision is made over the lumbar spine and a separate horizontal incision is made in the lateral mid-lumbar region. The SCS leads are inserted through the vertical incision into the posterior epidural space using intermittent fluoroscopic guidance to stimulate the spine at the appropriate level. Once the leads are in position, they are generally sutured into position (often to the supraspinous ligament) with nonabsorbable sutures. The leads are tunneled to connect with the SCS pulse generator, which is generally surgically implanted at an approximate depth of 1 to 2 cm in the lateral mid-lumbar region. An X-ray of an implanted SCS is presented in Figure 5.
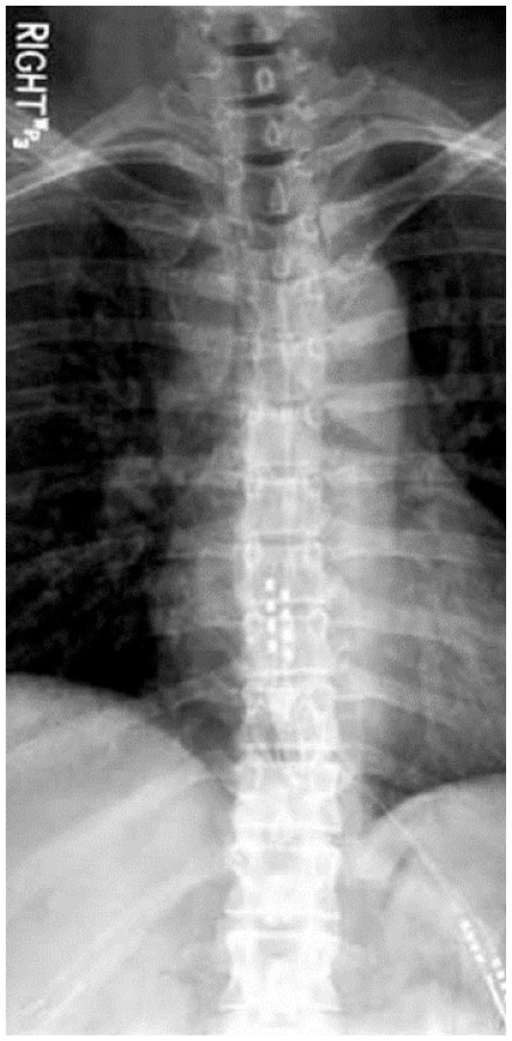
Anteroposterior radiograph showing a pair of spinal cord stimulation electrodes implanted in the posterior epidural space.
Wireless external power sources offer an alternative to a trial using temporary percutaneous electrodes followed by permanent implantation. With this approach, the trial period can be extended to as long as 30 days because the absence of external wires reduces the risk of infection. 86 Because percutaneous electrodes are no longer needed, an SCS trial can be extended as long as necessary with the use of as many waveforms and settings needed to achieve success or demonstrate failure of the system. A successfully implanted SCS can remain in place permanently after the trial, eliminating the need for its removal and for implantation of a new set of stimulator components: a pulse generator and new leads containing new electrodes. 87 Thus, wireless technology-enabled single-stage direct-to-permanent implantation may be a safer and less costly approach to SCS implantation. 88
Barriers to the Use of SCS for PDN
At this time, the main barriers to using SCS for PDN include (1) lack of recognition by clinicians that two SCS systems are now specifically cleared by the FDA for this indication, (2) slow dissemination of data from RCTs showing benefits of SCS, (3) the absence of professional guidelines based on recent trial evidence to support SCS as a second-line treatment if pharmacotherapy fails, and (4) concern for the risk of complications from the invasive nature of insertion. The first three barriers can be overcome through educational initiatives. The risks of complications from the insertion process can be minimized first through patient selection and second by weighing risks against the benefits of the technology.
Thoughtful patient selection can help mitigate the risks of SCS, beginning with those who have a successful trial. Psychosocial factors considered to predict poor outcomes of SCS therapy include lack of engagement, dysfunctional coping, unrealistic expectations, an inadequate daily activity level, problematic social support, secondary gain, psychological distress, and an unwillingness to reduce high-dose opioids. 89 In addition, it is important to separate a placebo effect from the effect of the actual SCS intervention. 90 Similar short-term improvement in pain due to failed back surgery has been observed with a sham procedure, compared with paresthesia-free SCS at 1200 and 3030 Hz, but in this study, stimulation at 5882 Hz significantly outperformed sham stimulation. 91 A follow-up study is now underway to compare pain relief from 10 kHz HF SCS with sham stimulation for chronic neuropathic low back pain. 92 Clinical factors can also inform patient selection. All reported studies of SCS for patients with PDN have been for lower extremity pain, and it is not known how well SCS works for upper extremity PDN. 84 People with diabetes are at increased risk of poor wound healing after surgical procedures, 93 and HF SCS has not been studied in people with diabetes whose A1C exceeds 10%. 94
Risks of SCS Technology
Placement of an SCS has procedural and technical risks. During and following SCS implantation, patients should be carefully monitored for evidence of a surgical site infection (the most common procedural complication) as well as epidural hematoma, mechanical neural injury, or late spinal compression related to fibrous lead encapsulation, post-dural puncture headache, or in rare cases, paralysis. The most frequent technical complications are lead migration and loss of stimulation.95-97 A meta-analysis published in 2021 of 32 longitudinal studies of complications that occur after SCS placement via either percutaneous or open (laminotomy/laminectomy) placement for multiple types of pain reported an overall average complication rate of 21.1%, with equipment, technical, and medical complications occurring at rates of 12.1%, 1.1%, and 6.3%, respectively. 95
A survey of the FDA Manufacturer and User Facility Device Experience (MAUDE) database for entries related to complications of SCS devices between January 1, 2016 and December 31, 2020 reported the following types of complications: (1) procedural complications, (2) serious adverse events, (3) device-related complications, (4) patient complaints, (5) surgically managed complications, and/or (6) other complications (Figure 6). Most complications were managed surgically with explantation (50.9%) rather than through revision (5.0%) or incision and drainage (6.6%). The types and numbers of surgical procedures described in the database for procedural and device-related complications are presented in Figures 7 and 8, respectively. 98
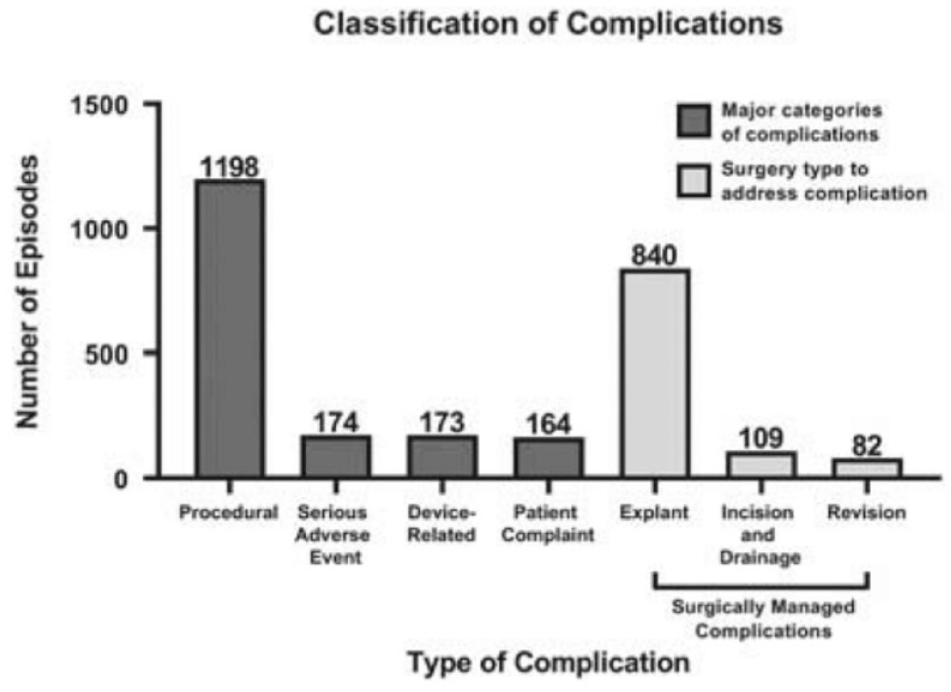
Classification scheme of complications due to surgical insertion of 10-kHz high-frequency spinal cord stimulators from the MAUDE database. Categories include (1) procedural, (2) serious adverse event, (3) device-related, (4) patient complaint, (5) surgically managed complications, (6) other. Abbreviation: MAUDE, Manufacturer and User Facility Device Experience.
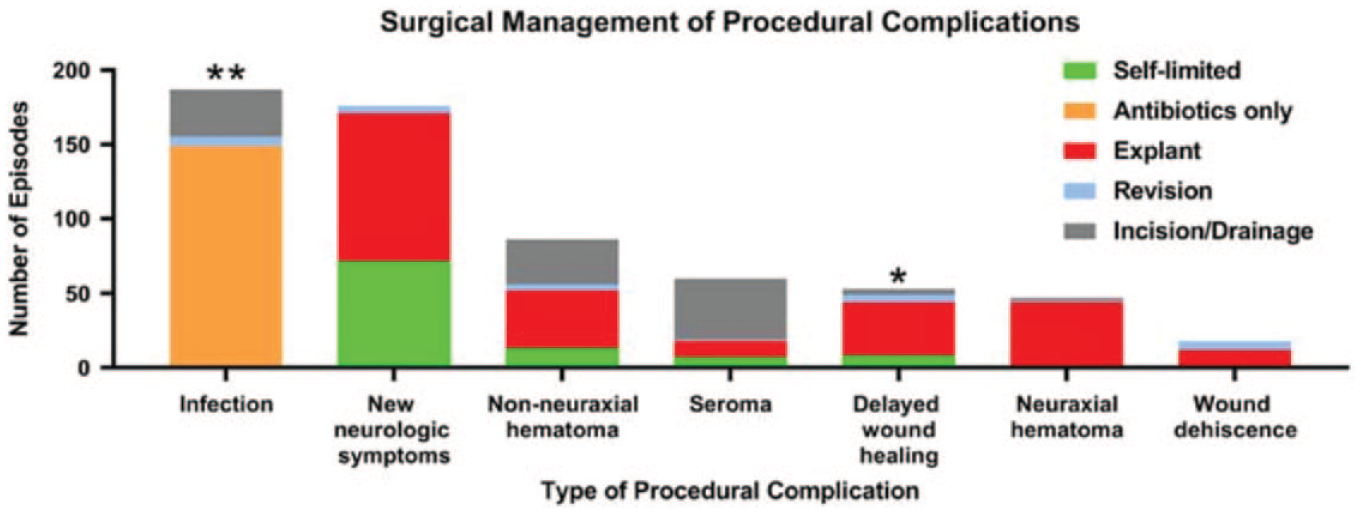
Type and frequency of surgical management espisodes for procedural complications described in the MAUDE database. Abbreviation: MAUDE, Manufacturer and User Facility Device Experience.
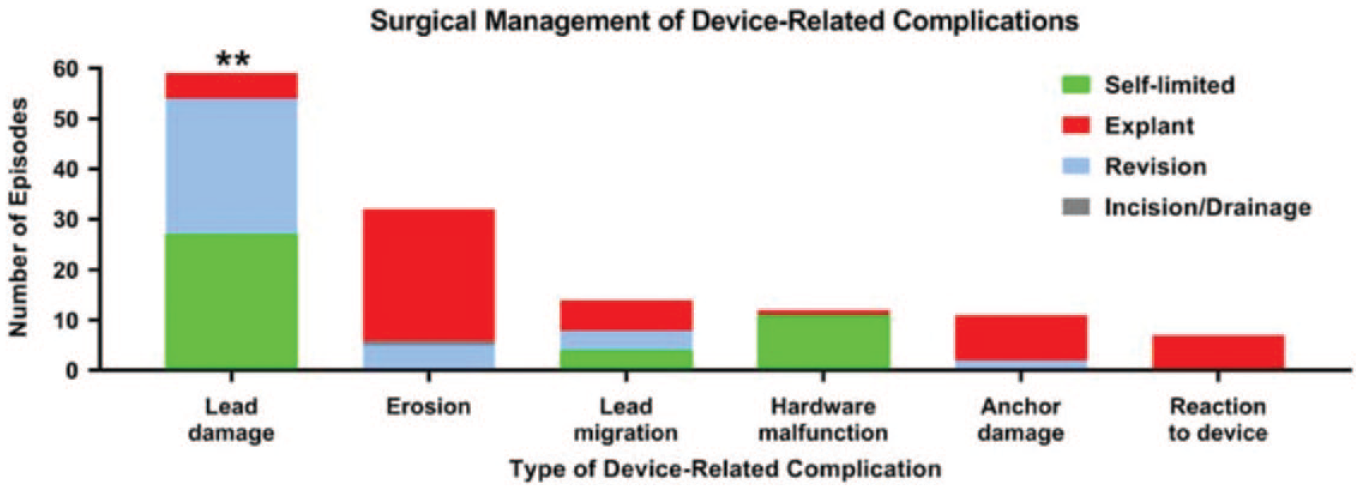
Type and frequency of surgical management episodes for device-related complications described in the MAUDE database. Abbreviation: MAUDE, Manufacturer and User Facility Device Experience.
A potential drawback to implanting an SCS for PDN is that the most stimulation is delivered to a specific level of the spinal cord. If a patient experiences progressive involvement of more dermatomes, then the stimulation might eventually not cover the entire symptomatic area. 2
Current Recommendations for Using SCS for PDN
Neuromodulation is becoming an established treatment for pain when pharmacotherapy fails. According to Xu and colleagues, for patients with refractory PDN in the lower extremities, “SCS should be considered to reduce pain and improve quality of life.” 84 D’Souza and colleagues stated that SCS should be offered to patients who have refractory PDN and fail to benefit from conservative options, such as optimization of glucose control and neuropathic analgesic medications, before strong opioids are prescribed. 2 Meanwhile, current evidence favors a shift away from traditional SCS and toward paresthesia-free HF SCS 99 as first-line neuromodulation treatment72,100 based on an accumulation of evidence showing better outcomes for HF SCS compared with traditional SCS101-103 for many types of pain, including PDN.
Future Developments in SCS
As additional research is conducted on the effects of SCS on PDN pain and quality of life in the future, data will be needed in four areas: (1) measuring outcomes from RCTs neutralizing the placebo effect of SCS placement with sham procedures as controls, especially as pain has a subjective component (although the added risk to a control subject receiving sham therapy must also be considered 104 ); (2) determining who is a good candidate for SCS (including the degree of glycemic control where people with diabetes can be safely treated); (3) matching clinical indications with which type of SCS treatments to administer; and (4) defining statistics, types of analyses, and target outcomes to compare research studies. One term which is not consistently applied in the SCS literature is “intention-to-treat.” In most SCS studies, achievement of the pain reduction outcome is expressed as a fraction of the number of patients achieving success divided by the total number of patients at various time points following device implantation. The denominator of total patients in the study is expected to decrease over time, both early in the study when some patients fail an initial trial of stimulation and again later as subjects drop out. The term “intention-to-treat analysis” is not exactly correct when applied to such multistage studies of SCS with evolving and shrinking denominators. 52 Incorrect use of the term “intention-to-treat” in a per-protocol analysis can create bias, suggesting an exaggerated favorable effect of the intervention. 105
Conclusion
Recent evidence indicates that SCS can provide safe and effective treatment for the pain from PDN (although not necessarily the underlying nerve damage) especially when risk factor modification, nonpharmacological therapy, and pharmacotherapy have all failed. It is difficult to compare outcomes for trials of various modalities because of a scarcity of robust head-to-head comparison data. Also, many of the studies of a single modality have used methodologically flawed protocols. Based on existing limited outcomes data, however, we conclude that for PDN, HF 10-kHz SCS, compared with traditional SCS, offers a greater likelihood of significant pain reduction and improved quality of life by eliminating paresthesias as well as allowing for a faster implant procedure.25,72,106 Nevertheless, more data is needed for both of these forms of FDA-approved technologies for treating PDN.
The use of any type of SCS for treating PDN—both traditional and HF forms—is a relatively new approach to this vexing problem, and the results so far look promising. As long as current noninvasive treatments for PDN are problematic and no effective FDA-approved pathogenetic treatments exist, the appeal of SCS will grow. If current ongoing research continues to show favorable outcomes for SCS in PDN, then this type of neuromodulation therapy will become increasingly adopted and will become a source of hope to many people with a currently hopeless disease.
Footnotes
Acknowledgements
The authors thank Annamarie Sucher-Jones for her expert editorial assistance and Ashley Y. DuBord for her excellent artwork.
Abbreviations
BMT, best medical treatment; CMM, conventional medical management; ESCS, electrical spinal cord stimulation; DTM, differential target multiplexed; FDA, US Food and Drug Administration; HF, high frequency; HF 10 kHz, high-frequency 10 kilohertz; ITT, intention-to-treat; MAUDE, Manufacturer and User Facility Device Experience; NPS, neuropathic pain scale; NRS, numeric rating score; PB, paresthesia-based; PDI, pain disability index; PDN, painful diabetic neuropathy; PGIC, patients’ global impression of change; PP, per protocol; RCT, randomized controlled trial; SCS, spinal cord stimulation; VAS, visual analog scale; VNRS, verbal numeric rating scale.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: KTN is a consultant to Abbott Diabetes Care. NYX s a consultant to Abbott Diabetes Care. DCK is a consultant for EOFlow, Fractyl Health, Integrity, Lifecare, Rockley Photonics, and Thirdwayv. AMY, JH, LTH, BKA, and NE have nothing relevant to disclose.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Diabetes Technology Society, which received no external funding and paid all costs for conducting the study.
