Abstract
Background:
Painful diabetic neuropathy (PDN) is a progressive condition that deprives many patients of quality of life. With limited treatment options available, successful pain management can be difficult to achieve.
Methods:
We reviewed results of recent data evaluating high frequency spinal cord stimulation (SCS).
Results
from the SENZA-PDN randomized clinical trial (NCT03228420), the largest such trial to date, demonstrated 10-kHz spinal cord stimulation substantially reduced PDN refractory to conventional medical management along with improvements in health-related quality-of-life measures that were sustained over 12 months. These data supported the recent U.S. Food & Drug Administration (FDA) approval for 10-kHz SCS in PDN patients and contributed to the body of evidence on SCS available to health care professionals managing the effects of PDN.
Conclusion:
High frequency spinal cord simulation appears to hold promise in treatment of painful diabetic neuropathy. We look forward to future works in the literature that will further elucidate these promising findings.
Keywords
Introduction
At least half of people with diabetes will develop clinically significant peripheral neuropathy.1-4 Associated neuropathic pain and other dysesthesias, including burning, tingling, and numbness, present in the extremities and may progress to a disabling intensity that is difficult to manage with conventional treatments.1,5,6 When pain is present, nonopioid treatments may offer inadequate symptom relief due to intolerable side effects associated with cocktails of neuropathic pain medications and antidepressants—as monotherapy is rarely sufficient.
7
The use of established agents per current clinical guidelines, including amitriptyline, duloxetine, gabapentin, pregabalin, tramadol, tapentadol, and topical agents such as capsaicin for neuropathic pain in diabetic neuropathy is limited by poor efficacy, contraindications, and adverse effects.5,8 Additional agents not currently listed in clinical treatment guidelines but routinely used in practice that have modest benefits and are easily tolerated include alpha-lipoic acid and combination therapy with
Neuromodulation for Patients With Refractory Pain
Spinal cord stimulation (SCS) has been shown to treat an array of chronic neuropathic pain conditions, including chronic peripheral neuropathy. 11 The first randomized controlled studies investigating the efficacy of SCS to specifically treat painful diabetic neuropathy (PDN) were reported by de Vos et al 12 and Slangen et al 13 using low-frequency SCS. These studies showed significant treatment benefit over conventional medical management (CMM), though they were limited by small sample size. In 2020, feasibility results of 10-kHz SCS in patients with chronic peripheral polyneuropathy suggested a significant treatment benefit through 12 months. 14 Petersen et al15,16 have examined the application of neuromodulation for pain relief in PDN. Published reports of SCS for treatment of PDN discussed earlier are summarized in Table 1.
Summary of PDN Studies.
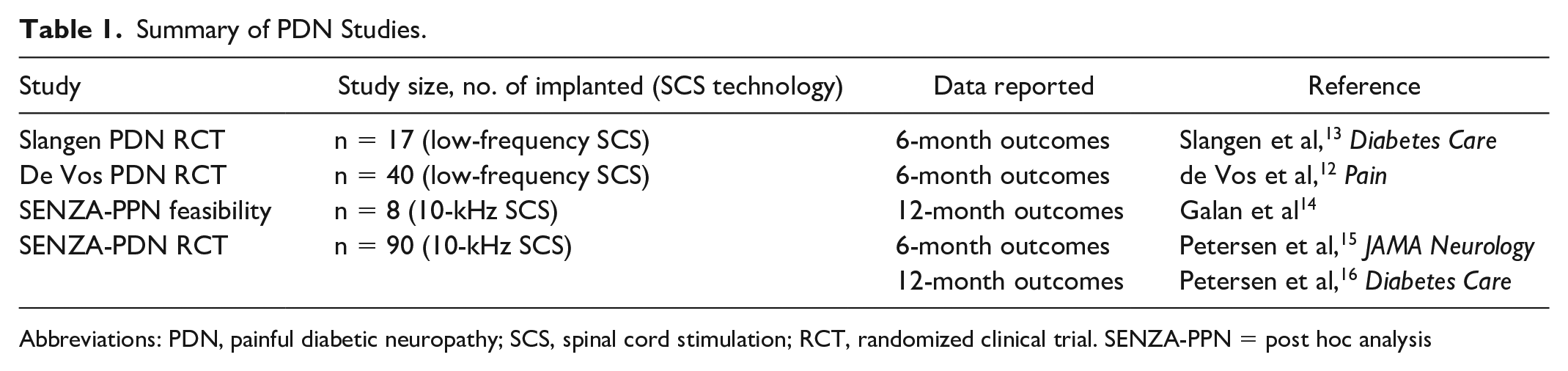
Abbreviations: PDN, painful diabetic neuropathy; SCS, spinal cord stimulation; RCT, randomized clinical trial. SENZA-PPN = post hoc analysis
The 6- and 12-month results of this trial have been reported by Petersen et al, and these data supported the recent U.S. Food & Drug Administration (FDA) 17 approval for 10-kHz SCS as a treatment for lower limb pain due to diabetic neuropathy. The 10-kHz SCS used in the SENZA-PDN RCT involves high-frequency, low-amplitude, short-duration pulses that deliver paresthesia-independent treatment.15,18,19 In contrast, conventional SCS uses frequencies ranging 50 to 1000 Hz that stimulate nerves and mask pain with paresthesia, or a tingling sensation.20,21 High-frequency SCS using a 10-kHz waveform has previously demonstrated efficacy across chronic pain in the back, leg, upper limb, and neck20,22-29 and was shown to be superior to conventional SCS.22,29 This approach to SCS does not produce paresthesias for the patient, an important advantage for PDN patients who often have underlying paresthesias due to their disease.14,30 In addition, some patients treated with 10-kHz SCS reduce or cease opioid treatment altogether.22,24,28,29,31-34
SENZA-PDN (NCT03228420) is currently the largest, randomized, controlled, open-label clinical trial assessing SCS for PDN. The study assigned patients with pain refractory to current treatments to 10-kHz SCS combined with CMM compared with CMM alone. The trial screened 430 candidates and enrolled 216 patients with PDN for 12 months or longer and symptoms refractory to treatment with gabapentin or pregabalin and at least one other analgesic, rating their lower limb pain as at least 5 out of 10 on a 10-cm visual analogue scale (VAS). 35
In PDN, a common challenge has been in predicting which patients will respond to which treatments. 36 During screening in SENZA-PDN, 94% of patients who underwent a temporary trial of 10-kHz SCS device achieved at least 50% pain relief or more, qualifying them for permanent device implantation. The ability to trial 10-kHz SCS prior to having a permanent implantation procedure affords reassurance to patients as well as implanting physicians of the potential benefits from the therapy. SCS trials are conducted for clinical studies as well as in real-world practice.15,22,32,34 Temporary trial stimulation occurred for 5 to 7 days with percutaneous leads placed epidurally along T8 to T11 vertebral bodies. 15 The results reported are equal to, if not better than, pain relief with 10-kHz SCS recorded in other pain types like back pain and upper limb and neck pain.28,37-40
At 3 months after implantation, 79% of patients in the intention-to-treat population achieved ≥50% pain relief plus no worsening of baseline neurological deficits, compared with only 5% of CMM patients (P < .001; study primary end point). 15 In terms of pain relief alone at 3 months, 78% of patients reported pain scores of 3 cm or less on the VAS (P < .001 vs 5% with CMM). 15 At 6 months, pain scores remained stable in the CMM arm, while those on 10-kHz SCS reported a mean pain decrease of 76.3%, with 85% of patients reporting ≥50% pain relief from baseline. 15
Aside from pain, sensory, reflex, and motor deficits are common findings among patients with PDN. 41 Investigators conducted thorough neurological examinations at baseline, 3, 6, and 12 months to evaluate motor strength, reflex, and sensory function, including a diabetic foot examination with pinprick and 10 g monofilament testing that is predictive of protective sensation. 42 In SENZA-PDN, 62% of patients in the 10-kHz SCS plus CMM arm showed a clinically meaningful improvement in neurological function at 6 months compared with only 3% in the medical management arm. 11 Furthermore, by 6 months, patients in the 10-kHz SCS plus CMM arm showed improvements in health-related quality-of-life measures, including sleep quality. Overall, the improvements offered with 10-kHz SCS were also reflected in patient satisfaction with treatment. 11
The SENZA-PDN investigators recently published 12-month results from their study showing durable treatment effects. 16 Patients in the 10-kHz SCS arm averaged 77.1% pain relief with 86% of patients reporting ≥50% pain relief from baseline. The observed neurological improvements reported at 6 months remained present at 12 months with 68% of patients noted to have improved on examination from baseline. The study design included an option to crossover to the alternative treatment arm at 6 months. Although no patients crossed from 10-kHz SCS to CMM, 81% of CMM patients crossed over to SCS. The results postimplant were similar to those for patients originally randomized to 10-kHz SCS: 70.3% mean pain relief, 84% of patients with ≥50% pain relief from baseline, and 62% of patients observed to have meaningful neurological improvement.
Significance of the Findings From SENZA-PDN
Aside from the substantial pain relief achieved in patients with symptoms refractory to conventional pain management options, the potential disease-modifying findings of this study in terms of neurological function may be of significant value. With pain relief and improved sensory perception in the feet, as was observed in SENZA-PDN, patients will be able to meaningfully participate in activities of daily living and potentially avoid injuries. Additional studies are needed to elucidate the potential impact on important quality-of-life measures, including balance and fall risk, in RCTs. The durability of pain relief in this patient cohort continues to be monitored with data collection planned for 24 months postimplantation. Results from real-world experience with 10-kHz SCS for PDN were similar to the randomized trial results. 43
Who Should Be Considered for SCS Referral?
For health care professionals managing patients with PDN, the SENZA-PDN trial results provide evidence supporting a new potential option to consider for patients with refractory pain. Patients with inadequate pain relief despite best available medical treatment should be referred to a pain medicine physician or functional neurosurgeon who offers 10-kHz SCS to determine if they are appropriate candidates for this therapy. Conditions that may preclude the success of SCS therapy are psychiatric conditions that make managing an active implant challenging, opioid dependence, and HbA1c > 10. Just as screening for diabetic retinopathy has become relatively mainstream, 36 so too should screening for PDN with a robust patient referral pathway. The SENZA-PDN study population was clearly refractory, including patients with moderate-to-severe lower limb pain, who had tried at least two classes of analgesic medications, had diabetes (97% Type 2) for more than 12 years on average, and more than 7 years on average since their diagnosis of peripheral neuropathy. The infection rate was low and similar to population without diabetes at 5.2%, despite a median HbA1c of 7.3 (range 5.3-10), and a median body mass index (BMI) of 34.1 (range 18.8-49.9).
Barriers to Establishing Treatment
The primary barrier for the entry of SCS therapy into the established treatment flow for patients with refractory PDN is lingering questions around cost and safety of the device implant and procedure. Contrary to these concerns, the study results demonstrated relatively low complication rates in this patient group, with a 5.2% infection rate, and only 3.9% of the 154 patients explanted their implants over the first 18 months of follow-up. 44 The rate of SCS system repositioning or replacement procedures was only 3.2% over the same period.
SCS is covered by all major insurance plans including Medicare and Medicare Supplemental plans. Individual patient coverage or costs may vary based on specific plan benefits, and can be answered by a patients pain management provider or insurer before they decide to move forward with the procedure. In terms of cost to health care payers, the upfront cost of the SCS system and procedure is significant; however, because health care use is intensive for patient with refractory PDN, SCS can be cost-effective within 2 to 3 years of implantation, 45 as observed with chronic low back pain patients. 46 The health care utilization data collected in SENZA-PDN shows a 38% reduction in hospital visits in the first 6 months postimplant. 44
Another barrier is that SCS providers are not currently treating the PDN patients who could benefit. Many patients suffering with this condition are refractory to CMM, so they are not being actively treated by a specialist for their PDN. Therefore, strong referral pathways, interdisciplinary care models, and dissemination of the study findings to primary care providers, or even direct to patient, could be important for patient access to the therapy.
The staggering socioeconomic costs for patients with PDN coupled with a paucity of effective treatments and enormity of the patient population suffering requires a collaborative perspective for care delivery. 7 Further research, especially studies targeting potential disease-modifying benefits for improvements in sensation, are required to better understand the comprehensive benefits of 10-kHz SCS for PDN.
The authors wish to break down the barrier to SCS for PDN by informing health care professionals across the disciplines who treat these patients, from endocrinology, primary care, podiatry, neurology, and pain management, along with policymakers, of the promising results of this multicenter RCT. Furthermore, the authors aim to highlight how the study’s inclusion criteria may inform selection of the PDN patients most likely to benefit from this therapy. Given the current opioid crisis, ineffectiveness of current pain relief options, lack of disease-modifying treatments, and financial burden of medical management of PDN, patients may be receptive to alternative treatment options. 36 The results of the SENZA-PDN study led to FDA approval of 10-kHz SCS treatment 17 for patients with PDN that can improve a patient’s quality of life. Widespread, interdisciplinary sharing of these results, and others inevitably forthcoming in the space, will encourage further clinical development and benefit patients with refractory PDN.
Footnotes
Acknowledgements
The authors thank Catherine Panwar of Panwar Health for her assistance in language content and manuscript preparation and Lisa Brooks, PhD, from the Clinical Research Department at Nevro Corp. for further manuscript review.
Abbreviations
PDN, painful diabetic neuropathy; kHz, kilohertz; SCS, spinal cord stimulation; FDA, U.S. Food & Drug Administration; VAS, visual analogue scale; CMM, conventional medical management.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr DGA has received consulting fees as scientific advisor for Nevro Corp. Dr GG has received speaker fees from Eli Lilly, Novo Nordisk, and Abbott Diabetes, and has received consulting fees as a member of the scientific advisory board for Nevro Corp. Assistance in medical writing for this manuscript was funded by Nevro Corp.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This manuscript was partially supported by the National Institutes of Health, National Institute of Diabetes and Digestive and Kidney Diseases Award Number 1R01124789-01A1, and National Science Foundation (NSF) Center to Stream Healthcare in Place (#C2SHiP) CNS Award Number 2052578.
