Abstract
Background:
Diabetes management and treatment requires the use of many devices that frequently must puncture the skin, creating a risk of unintentional retention in the body as a retained diabetes device. In this article, we reviewed case studies about retained diabetes devices and presented analyses of the success rate of current imaging techniques in identifying retained devices and the success rate of device removal.
Methods:
PubMed and Google Scholar were searched for articles about retained diabetes devices. Relevant articles that included sufficient details about discovery and removal of the device were included. The success rate of identification and the success rate of removal of retained devices were both calculated as percentages.
Results:
Sixteen case studies of retained diabetes devices were identified. These devices included parts of continuous glucose monitors and infusion sets, a lancet, and various types of needles for insulin injection. Each case is presented with details about the year of publication, the retained diabetes device, the company that produced the device, the age and gender of the patient, the type of diabetes that the patient had, the location of the device, the reason for initial discovery of the retained device, the process of locating the device, the success rate for removal of the device, and the removal procedure of the device. Analysis revealed a 100% success rate for the use of imaging technology including X-rays and computed tomography to identify a retained diabetes device. The patients with retained diabetes devices had a 62.5% success rate for eventual removal of the device.
Conclusions:
With the increasing use of injected, inserted, and implanted diabetes wearables for digital health, it is likely that some of the devices will detach, break apart, or otherwise become retained in the body. It is important to be aware of available technologies to identify retained diabetes devices so that it will be possible in most cases to surgically remove these devices if they detach or become retained.
Introduction
Many devices are used for diabetes that must be injected, inserted, or implanted to monitor and treat this disease. These include devices that puncture the skin to measure capillary blood glucose and devices that are inserted into the skin to measure glucose in interstitial fluid. Other devices deliver injections of peptides like insulin and glucagon into the skin. All of these measurements and treatments require frequent, often as much as multiple times per day, skin punctures with needles or implantation of sensors into the subcutaneous space. While these devices allow for proper diabetes management, they come with the risk of unintentional detachment or retention. In this article, we define a retained diabetes device as any fragment or piece of a medical device used for monitoring or treating diabetes retained in the body after its intended usage period. Given the increasing number of injected, implanted, and inserted devices used for diabetes and the associated increasing risk of a retained or detached device, we decided to ask the question: does current imaging technology sufficiently identify and support successful removal of retained diabetes devices? We investigated cases of retained diabetes devices that exist in the medical literature and reviewed the causes as well as methods for detection, removal, and prevention of retained diabetes devices. We then assessed the number of retained devices that could be successfully identified through current imaging techniques and when the device was successfully identified, we assessed the number of cases where the retained device was successfully removed.
Methods
We conducted a database review on May 2, 2022 of the past 26 years, dating back to January 1, 1996, to search for reported cases of retained diabetes devices on PubMed and Google Scholar using the search terms of “retained,” “diabetes,” “device,” and “foreign body.” Searches were conducted with the following two sets of three key terms in quotation marks: (1) “retained,” “diabetes,” and “device” and (2) “retained,” “diabetes,” and “foreign body.” We then reviewed the abstracts of every article from each search that appeared on PubMed and the first 100 results of each search on Google Scholar to determine whether these listings were relevant to the topic. We limited each search on Google Scholar to the first 100 results because our experience with prior searches on this database was that relevant links will almost always appear within the first 100 listings. Inclusion criteria included a thorough discussion of the case with details about discovery, investigation, and removal of the device. We then reviewed the reference section of each included article to find additional case studies, and we also searched each included article on Google Scholar to search for subsequent cases that referenced any of our index cases. In total, we found fourteen articles describing sixteen cases. Figure 1 presents a PRISMA diagram of how we performed our database review to identify these articles.
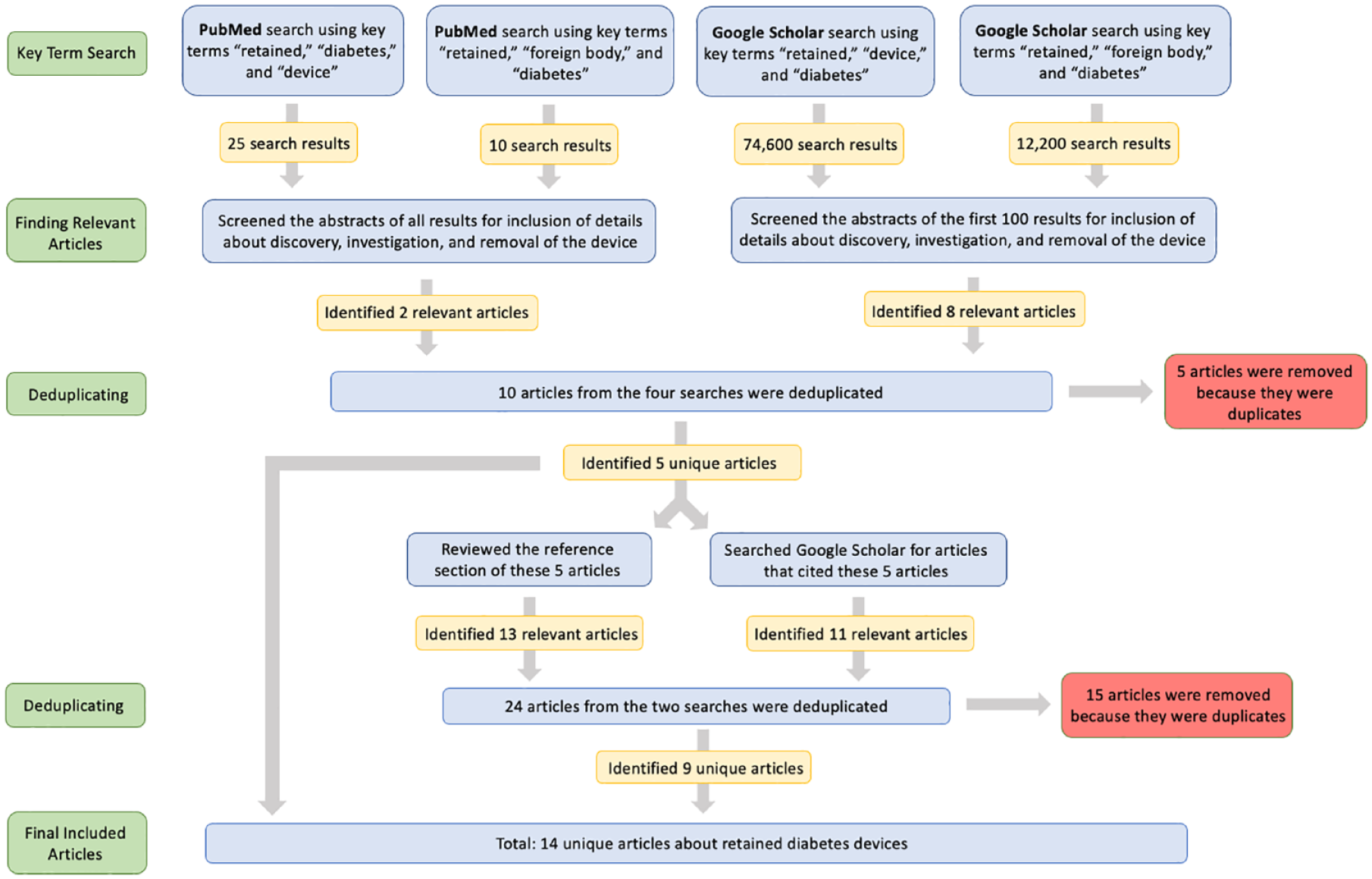
A PRISMA diagram of how we performed our database review to identify articles about retained diabetes devices.
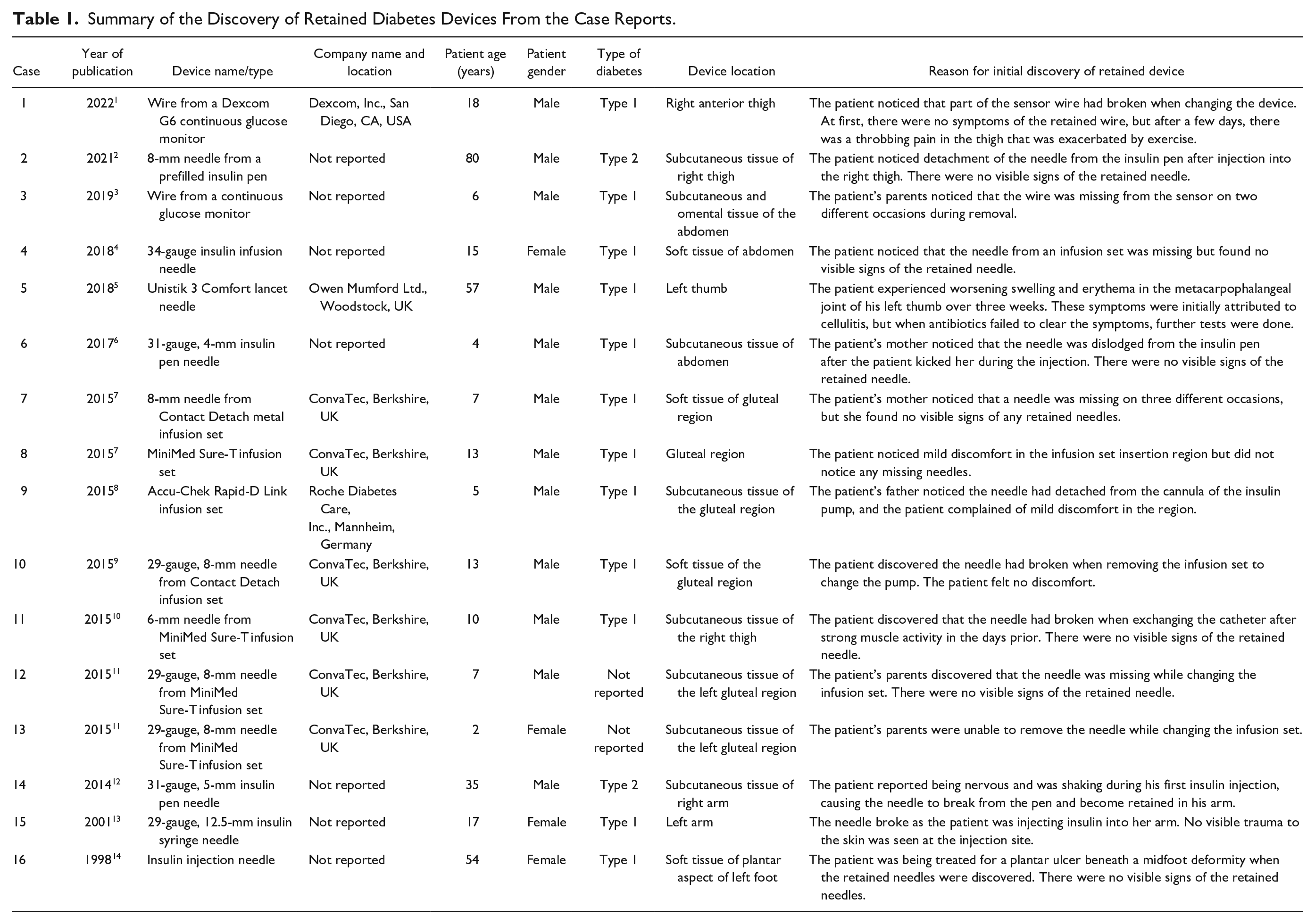
For each article and case study that we included, we noted (1) the year of publication, (2) the name or type of the retained diabetes device, (3) the name and location of the company that produced the device if it was reported in the original case report, (4) the age of the patient, (5) the gender of the patient, (6) the type of diabetes that the patient had, (7) the location of the device, (8) the reason for initial discovery of the retained device, (9) the method for locating the device, and (10) the removal procedure. An analysis of the case studies was then conducted to determine whether imaging techniques could successfully identify the retained device and whether the device was later removed. Success rates were calculated and presented as a percentage.
Results
We found fourteen articles reporting sixteen unique cases of retained diabetes devices in the medical literature, with twelve cases involving people with type 1 diabetes, two cases involving people with type 2 diabetes, and two cases with no type specified. Cases are referenced by their assigned case number, as presented in Tables 1 and 2. Case 1 was a patient with a retained sensor wire from a continuous glucose monitor (CGM) in his right thigh. Case 2 involved a retained needle from a pre-filled insulin pen in the right thigh of a patient. Case 3 involved a wire from a CGM detaching and being retained in the patient on two separate occasions. Case 4 was a patient with a retained needle from an infusion set. The patient noticed that the needle was missing, but there were no visible signs over where the needle was located. Case 5 explores the severe symptoms observed by a patient who was unaware of a retained device in his thumb. His disease history first led physicians to believe that these symptoms were caused by cellulitis, causing him to receive treatment that failed to resolve his symptoms. Later investigation with an X-ray of his thumb revealed the retained lancet needle fragment. Cases 6 and 14 both involve insulin pen needles that became retained in the patient’s body. Both needles were successfully removed surgically. Cases 7 through 13 explore retained needles from various infusion sets located either in the gluteal region or the thigh. Some were able to be removed smoothly whereas others were left in the body where they were found. Case 15 involved an insulin injection needle retained in the left arm that was later surgically removed. Finally, case 16 involved a patient who had two retained needle fragments in her foot that were discovered during an appointment to treat another problem with her foot.
Summary of the Discovery of Retained Diabetes Devices From the Case Reports.
Summary of Identification and Removal of Retained Diabetes Devices From the Case Reports.
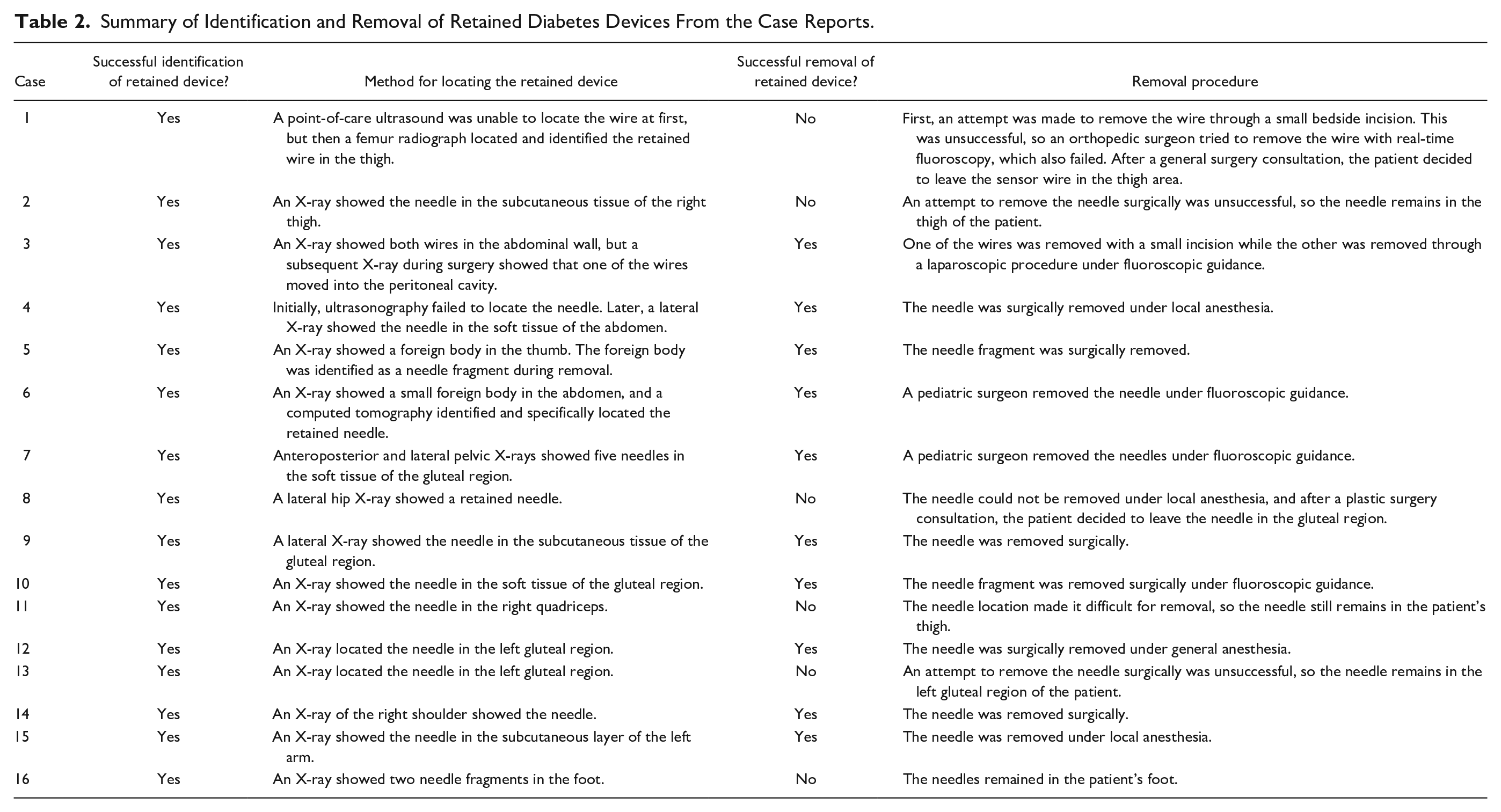
X-ray imaging was used to successfully identify and locate the retained diabetes device in all sixteen cases, resulting in a 100% success rate of current imaging technology for identifying retained devices. In case 1, a point-of-care ultrasound failed to locate the retained CGM sensor wire, but a femur radiograph was later able to locate and identify the retained wire. Ultrasonography was used in case 4 but failed to identify the retained device, so X-ray imaging was later done to identify the insulin infusion needle. In case 6, computed tomography (CT) was used to more specifically pinpoint the retained device after X-ray imaging identified the presence of a foreign body. Out of these sixteen cases with successfully located retained devices, only ten of the cases were resolved through surgical removal of the device from the patient’s body, resulting in a 62.5% successful removal rate. In cases 1, 2, 8, and 13, an attempt was made initially to remove the retained diabetes devices but ultimately failed, leading to the retained device remaining in the body of the patient. In cases 11 and 16, the decision was made to leave the retained devices where they were without a removal attempt because they were located in a difficult area and there was no benefit to removal.
Discussion
Causes of Retained Devices
The increased deployment of minimally invasive injected, inserted, and implanted diabetes devices has led to an increased risk for procedure-related complications, including retention of all or part of the implanted devices. Additionally, needle injection of insulin is a long-established practice for diabetes therapy and recently, other injectable drugs have been introduced, resulting in more needle injections and potentially more risk for needle-related complications. 15 Analysis of the phenomenon of retained diabetes devices requires determination of causes of retention as well as methods for detection, removal, and prevention of this problem.
People with diabetes often require a regimen of insulin delivered via subcutaneous injections or infusion sets. Since injections are usually needed multiple times per day and infusion sets must usually be replaced every few days, manufacturers try to design these devices to minimize patient discomfort. As a result, these devices are now equipped with increasingly thin needles that are less painful but more prone to break. However, these needle breaks often leave no scarring or visible signs of breakage, making it difficult to locate a needle fragment. In the sixteen retained diabetes device cases we identified, there were five cases involving CGM wires (cases 1 and 3), a lancet (case 5), and a piece of an infusion set (cases 8 and 9), as well as eleven cases involving a retained needle. The eleven patients with retained needles used either an 8-mm needle from a prefilled insulin pen (case 2), a 34-gauge insulin infusion needle (case 4), a 31-gauge and 4-mm insulin pen needle (case 6), an 8-mm needle from a ConvaTec Contact Detach metal infusion set (case 7), a 29-gauge and 8-mm needle from a ConvaTec Contact Detach infusion set (case 10), a 6-mm needle from a Medtronic MiniMed Sure-T infusion set (case 11), a 29-gauge and 8-mm needle from a Medtronic MiniMed Sure-T infusion set (cases 12 and 13), a 31-gauge and 5-mm insulin pen needle (case 14), a 29-gauge and 12.5-mm insulin syringe needle (case 15), or a pair of insulin injection needles whose sizes were not specified (case 16). They may have noticed that the needles were missing from their devices upon removal, but they observed no significant symptoms of the broken needles being retained in their bodies. The retained devices were essentially asymptomatic, although patients noted swelling and discomfort with a retained Unistik 3 Comfort lancet needle (case 5), mild discomfort with a retained Medtronic MiniMed Sure-T infusion set (case 8), and mild discomfort with a retained Accu-Chek Rapid-D Link infusion set (case 9) in the general area where the devices were inserted. These patients did not, however, attribute these local symptoms to the retained needles. These retained needles could only be located through imaging techniques.
Regarding causes of diabetes device retention, continuous glucose monitoring is becoming an important component of diabetes management and is now regarded by many as the standard-of-care for monitoring diabetes. 3 Components of CGMs have also been found to be retained in patients. In case 1, the patient noticed that a CGM sensor wire fragment broke off and was retained in his thigh during the process of changing the CGM. The patient in case 3 found detached wires from a CGM retained in their body during the removal process on two separate occasions. Similar to the needles used for insulin delivery, CGM wires for subcutaneous insertion to monitor glucose levels are very thin. These thin wires are predisposed to migration within the body if retained. In this particular case, one of the retained wires migrated from the area of initial insertion at the abdominal wall into the peritoneal cavity, where it was eventually removed. Figure 2 shows this CGM wire in the peritoneal cavity during surgery when removed under fluoroscopic guidance.
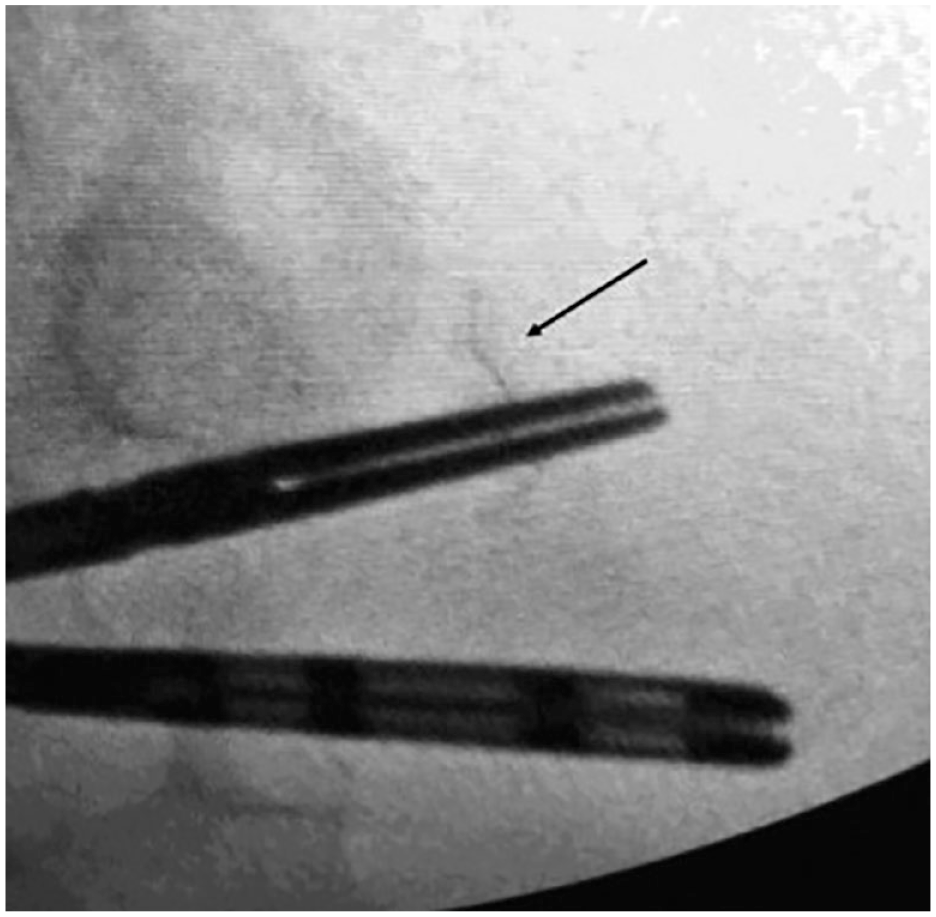
Under fluoroscopic guidance, an intra-abdominal curved CGM wire (indicated by the arrow) was surgically removed using blunt graspers.
In addition to the mechanical issues leading to device retention, there can also be clinical factors that increase the likelihood of retained devices. Diabetic peripheral neuropathy decreases the sensitivity of a person’s sensory mechanisms, especially in the feet. 16 Therefore, patients with diabetic peripheral neuropathy are more susceptible to sustaining a skin puncture in the feet, including those from retained devices, without noticing them. We identified two cases of retained devices where people with diabetic peripheral neuropathy were unaware of the skin punctures. In case 5, the patient developed swelling in his metacarpophalangeal joint near where the retained device was located. Since the patient did not recall any injury to his thumb that may have caused the swelling, the physician first attributed his symptoms to cellulitis, given his medical history of poorly managed type 1 diabetes. The physician prescribed antibiotics. It was only when the patient’s condition failed to improve after eight days of antibiotic therapy that the physician ordered an X-ray of the thumb that revealed the retained Unistik 3 Comfort (Figure 3). In case 16, a patient’s foot was being evaluated for a plantar ulcer when X-rays revealed four retained insulin injection needles in various locations on her foot. The patient was unaware of any of these punctures but reported disposing of insulin injection needles onto her bedroom floor, where it was likely she stepped on several of them. Diabetic arthropathy is another complication that can decrease the manual dexterity of people with diabetes, affecting their ability to self-inject. 2 In case 2, the patient was observed to have poor dexterity when using his insulin pen during retraining. This incorrect self-injection technique is believed to have caused the needle to detach and become retained in the right thigh.
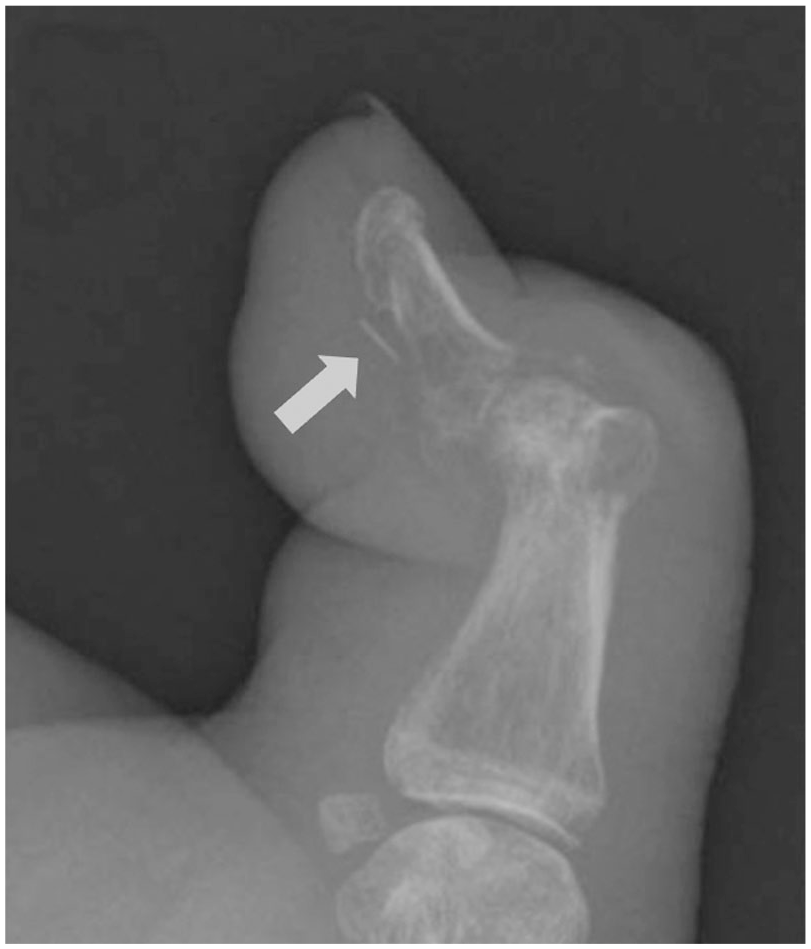
A retained Unistik 3 Comfort lancet, indicated by the arrow, in the thumb of a patient with diabetic peripheral neuropathy.
The FDA maintains the Manufacturer and User Facility Device Experience (MAUDE) database, which contains adverse events reported by manufacturers, including retained devices. 17 FDA guidance mandates that if a manufacturer becomes aware of a literature case report, then the manufacturer must submit it as an adverse event. It should be noted that there is no guarantee that all case reports from the medical literature will be reported to MAUDE by manufacturers or that all cases reported to MAUDE by manufacturers will also appear in the medical literature.
Detection of retained devices
Detection methods are important for the discovery and potential removal of retained devices because many patients are unable to self-identify the existence and location of retained devices. X-ray imaging is the main method of detection used in the case studies in this report, which is consistent with a recent review article that found radiography to be the most widely used imaging technique to discover retained surgical sharps. 18 However, X-ray imaging may not be effective for identifying retained items without knowing the general shape, size, and location of the retained item. People with diabetes will usually know the general area of device insertion on their body, so this limitation with respect to surgical retained devices may not be as relevant for diabetes devices. There is a possibility that a retained medical device will migrate within the body between the time of imaging and the time of removal, but other imaging or surgical techniques can be used in these situations. For identifying needles as small as 4-mm, CT scanning may be no more sensitive than X-rays for detection, although CT scanning can be superior for localizing these retained devices. 19 Ultrasound can also be used to locate a retained device although this imaging method was not successfully used for any of the retained diabetes devices in this report. Advantages of ultrasound, compared with X-rays, are a lower cost and no exposure to radiation. 20 Thus, X-rays, in combination with other imaging techniques, when necessary, are sufficient for identifying retained diabetes devices.
Removal strategies for retained devices
Small incisions with local anesthesia or open surgery under general anesthesia are the two typical methods for removal of retained devices. Depending on the location of the retained device, surgery may require the use of fluoroscopic guidance. Sometimes, if the retained device does not cause harm or discomfort to the patient and the risks and discomfort associated with removal are as great or greater than those of leaving the device in place, then a retained device may also be left in the body instead of being removed.
Fluoroscopy can track the motion of bones and other tissues through multiple X-ray images. 21 This approach uses various angles and directions to view images, aiding surgeons in locating retained devices. However, these imaging methods, compared with standard X-rays, expose the patient to more radiation. In case 4 with a retained 34-gauge insulin infusion needle, the surgeons chose to remove the device under local anesthesia because of the location of the needle. It was retained in the abdomen of a 15-year-old patient, and the surgeons did not want to expose the organs in the abdomen, especially the ovaries and uterus, to radiation. They opted to first use an ultrasonography detection because of its minimally invasive nature, but they ultimately failed to detect the needle using this method. The surgeons later located the device using an X-ray image.
Sometimes, the use of fluoroscopy is unavoidable despite the associated health risks. In case 1, after a failed attempt to remove a retained CGM sensor wire with a small bedside incision, orthopedic surgeons used fluoroscopy to try to remove the wire. Unfortunately, this attempt was also unsuccessful. In case 3, however, surgeons were able to locate a retained CGM wire that continued to migrate in the body after imaging and remove it by using fluoroscopic guidance when conducting the laparoscopic procedure.
Prevention of retained devices
To prevent occurrences of detached or retained devices, there needs to be improved insertion, detection, and removal methods for diabetes devices that puncture the skin. This includes assuring production quality of devices, developing effective devices to aid insertion, detection, and removal, and investigating new materials to create sensors.
In case 8 with the retained device from an Accu-Chek Rapid-D Link infusion set, the needle became detached from the infusion set on two separate occasions. This problem was attributed to a manufacturing error on a batch of needles, so ensuring the quality of all components of diabetes devices will help reduce the risk of breakage that can lead to retention.
Regarding insertion, devices intended for use with more shallow insertion can be more readily located than those that are inserted more deeply into the body. The pharmaceutical company Intarcia (Hayward, California) developed ITCA 650, an implantable delivery device requiring replacement two to four times per year to deliver a glucagon-like peptide-1 (GLP-1) receptor agonist for treatment of type 2 diabetes. This device was intended to be shallowly inserted in the subdermal plane. Among the first set of more than 18,000 placements, replacements, and removals, 99% were initially successful. 22 Any implantable device comes with a risk of retention, so Intarcia also designed a depth guide to implant their device at a particular depth beneath the skin and a removal tool to decrease risk.
Besides X-rays and ultrasound, other novel methods have been developed to detect retained implanted devices, since one of the main barriers to the use of implanted devices is the difficulty in locating and removing them. Tools using near infrared light (NIR), light emitting diodes (LEDs), and ultrasound can be used to guide venipuncture and are already FDA-approved. 23 Utilizing different absorption wavelengths of light, NIR projects a contrast image onto the skin that can be used to help locate diabetes devices under the skin. In a trial involving thirty patients with implanted glucose sensors, NIR was able to locate the sensors in twenty-four patients within five seconds. The patients whose sensors were located using NIR were each able to have their sensors removed surgically in under three minutes. The average removal procedure time for sensors that NIR was unable to locate was twenty minutes. 24 This technique was also shown to be successful in locating hormone-releasing contraceptive implants. 25 In a small case series, NIR was able to locate etonogestrel implants within five seconds. NIR technology is a promising technique but still comes with limitations because NIR is affected by the presence of tattoos, excessive freckles, and scars.
Technologies to better keep track of and detect surgical devices have been developed to try to limit retained devices, especially surgical sponges. 26 Retained surgical sponges often either become infected and cause a severe inflammatory response that leads to formation of an abscess or induce a fibrous reaction that creates a mass. 26 Various solutions to better detect retained surgical sponges can be adapted to detect retained diabetes devices.27,28 One method is a computer-assisted sponge count that uses a handheld device to scan the matrix labels on sponges that are unique for each sponge. These labels cannot be scanned when they are inside of a person, so sponges must be scanned prior to use in the person and then once again after being removed from the person. This technology may be used in combination with NIR to create a new technique that can both detect unique identifying labels of diabetes devices and scan through people in order to locate a device if it is retained in a person. The FDA has established the Unique Device Identification System in order to track medical devices that have been sold. This requires manufacturers to put unique labels on the packaging of medical devices and also the medical device itself in cases where the device is used more than once or will be reprocessed. 29 The system can facilitate detection of some retained diabetes devices through their unique identifiers, although it is not likely that this system will be applied to needles or CGM sensors. Another technology utilized for locating retained surgical sponges is a radiofrequency detection and identification system. This method requires sponges to have small radiofrequency tags sown into them in order to be detected by handheld detection system. 26 If diabetes devices were to also have very small radiofrequency tags embedded within them, then this could allow for easier detection of the devices and potentially reduce the need for X-rays to locate them.
Based on the established principle that in medicine, prevention is better than cure, if sensors and other medical devices could be constructed from biodegradable materials, then they would naturally degrade in the body once the usage period has passed. 30 Dissolvable microneedles have been three-dimensionally printed using polylactic acid, a biodegradable material. 31 Microneedles may be used for insulin injections in people with diabetes, and if they were made of biodegradable materials, then there would be no need for the surgical removal of broken needles since the needles could dissolve in the human body. Glucose sensors can also be made of biodegradable materials. A research group has created a biodegradable sweat glucose sensor that is noninvasive, but there are still many challenges that remain for bioresorbable components of medical devices for inserted or implanted devices. 32 Difficulties include the need to account for differing physiological conditions in people, movement of a device within the body, and a need for developing a power supply from biodegradable materials. 33
Conclusion
In the future, injected, inserted, and implanted diabetes devices will likely become even more widely used for digital health applications. Patients who need to perform self-injections often feel anxious about needles or have an actual fear of needles. 34 Implanted or inserted devices hold the possibility of decreasing anxiety and fear while still serving the same purpose as self-injections. New implantable devices are being developed for diabetes, such as the already FDA-approved Eversense CGM and the non-FDA-approved ITCA 650 implantable drug delivery system. Each time a new implantable or inserted device comes on the market, the global risk to the entire population of device retention increases. We conclude that based on the success rates for identification (100%) and removal (62.5%) of retained diabetes devices in the literature, it is safe for people with diabetes to use inserted or implanted devices and that current imaging technology is extremely capable of identifying retained diabetes devices. Studying cases of retained devices from the medical literature can lead to improved device design, creation of supplementary devices to assist with prevention of retention, and importantly development of new methods for detecting and removing retained devices, preferably without exposing the patient to radiation.
Footnotes
Acknowledgements
The authors thank Annamarie Sucher-Jones for her expert editorial assistance.
Abbreviations
CGM, continuous glucose monitor; CT, computed tomography; FDA, US Food and Drug Administration; GLP-1, glucagon-like peptide-1; LEDs, light-emitting diodes; MAUDE, Manufacturer and User Facility Device Experience; NIR, near infrared light.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Disclosures
NYX is a consultant to Abbott Diabetes Care. KTN is a consultant to Abbott Diabetes Care. HKA received research support through University of Colorado from REMD, Dexcom, Senseonics, Eli Lilly. VNS reports receiving honorarium through University of Colorado from Dexcom, Insulet, Sanofi and Medscape LLC. VNS reports research funding through University of Colorado from Eli-Lilly, NovoNordisk, Sanofi, Dexcom, Insulet, Abbott, JDRF and NIH (NIDDK and NIAMS). DCK is a consultant for Dexcom, Eli Lilly, EOFlow, Integrity, Lifecare, Medtronic, Novo Nordisk, Roche Diagnostics, and Rockley Photonics. JSK has nothing relevant to disclose.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
