Abstract
Background:
Removal of diabetes devices, including insulin pumps and continuous glucose monitoring (CGM), is a common practice due to hospital policies, interference with imaging studies, medications, and surgical interventions. Furthermore, these devices are inherently prone to malfunction, adhesive failure, and issues with insertion that can lead to a reduction in wear time. Prescription and dispensing practices provide an exact number of sensors per month without redundancy to account for the realities of daily CGM use.
Methods:
A RedCap survey was completed by adult patients with type 1 or type 2 diabetes (T1D or T2D) who utilize CGM followed in the Diabetes Center at Washington University in St Louis.
Results:
Of 384 surveys sent, 99 were completed. Participants had a mean age of 54 years, T1D 69%, female 70%, White 96%, non-Hispanic 96%, and a mean duration of diabetes mellitus (DM) 28 years. Of the cohort, 100% used CGM (80.2% Dexcom, 13.5% Freestyle Libre, 6.3% Medtronic), 61% insulin pump, and 41% Hybrid closed-loop (HCL) systems. CGM-related disruption events included device malfunction (in 85.4% of participants), insertion problems (63.5%), and falling off (61.4%). Medical care–related disruption occurred most frequently in the setting of imaging (41.7%), followed by surgery/procedures (11.7%) and hospitalization (4.4%). Adverse glycemic events attributed to CGM disruption, including hyperglycemia and hypoglycemia, occurred ≥4 times in 36.5% and 12.4% of the cohort, respectively.
Conclusions:
Disruption in CGM use is common. Lack of redundancy of CGM supplies contributes to care disruption and adverse glycemic events.
Introduction
Home glucose monitoring for individuals with diabetes mellitus (DM) has evolved over the 20th and 21st centuries. Initially, the only monitoring strategy was cumbersome urine glucose testing. In the 1980s, home blood glucose meters were utilized, and in the early 2000s, continuous glucose monitoring (CGM) was developed and has revolutionized diabetes care in the past few years.
CGM is standard of care for individuals on both intensive insulin therapy and basal insulin.1,2 Its efficacy in improving time spent in a desired glucose range and in reducing hypoglycemia has been extensively demonstrated in individuals with type 1 diabetes (T1D) and type 2 diabetes (T2D).3 -5 Further data suggest that discontinuation of CGM leads to loss of glycemic control and other adverse events. 6 The development of Hybrid closed-loop (HCL) insulin delivery systems that integrate CGM with insulin pump therapy further underscores the importance of these devices in the care of individuals with diabetes.7 -9
There are different CGM technologies, those that are intermittently scanned versus others with real-time monitoring, depending on the manufacturer. However, both options have been shown to improve glycemic control and reduce hypoglycemia in a variety of populations. As with any technology, and is the case with human error, it is not infrequent that these devices can malfunction for reasons such as adhesive difficulties, insertion problems, and displacement.
We aimed to assess the frequency of CGM disruption in our clinic population with diabetes and to understand the adverse consequences of interruptions in CGM device use.
Methods
Design
We conducted a retrospective Web-based multiple-choice survey of individuals with diabetes who utilize CGM devices. Our goals were to investigate the prevalence of device disruptions, their etiologies, and the rate of adverse consequences attributed to reduced wear time (hypoglycemia, hyperglycemia, emergency room [ER] visit, hospital admission). We collected demographic data (age, sex, race, and ethnicity), type of diabetes and its duration, and type of CGM and insulin pump. We also included questions about capillary blood glucose monitoring with a glucose meter. When evaluating the number of disruptions or adverse events that occurred in the past year, we asked participants to select from a list of numerical responses with the following options: 0, 1 to 3, 4 to 6, and 7 or more. Adverse event data were self-reported. We defined a disruption event as device removal for the purpose of medical care and/or any truncation of wear time secondary to CGM malfunction, displacement, or insertion problems. In addition, we examined patient satisfaction with the CGM replacement process, and the frequency of out-of-pocket cost incurred using the same frequency rating scale.
Patients
Patients were recruited from the adult population followed in the Washington University/Barnes-Jewish Hospital Diabetes Center in St. Louis, Missouri. Participants were selected based on active CGM or insulin pump use, regardless of make or manufacturer. Individuals without active device use in the past 30 days or age < 18 years were excluded. Participation was voluntary and respondents were not compensated.
Survey Distribution, Data Collection, and Statistical Analysis
The REDCap platform was used for survey creation, electronic consent, response collection, and generation of descriptive statistics.
Results
Participants
Of 384 survey requests sent, 99 were fully or partially completed. Participants had a mean age of 54 years. About 69% had T1D, 70% were female, 96% White, 96% non-Hispanic, and a mean duration of DM was 28 years (Table 1). Of the cohort, 99% used a CGM with the following brand distribution: 80.2% Dexcom, 13.5% Freestyle Libre, and 6.3% Medtronic. About 61% of participants were using an insulin pump and 41% used HCL systems. Only 35% of respondents reported ever using capillary blood glucose monitoring with a glucose meter and the majority of those (61.3%) only checked once daily.
Patient Baseline Characteristics.
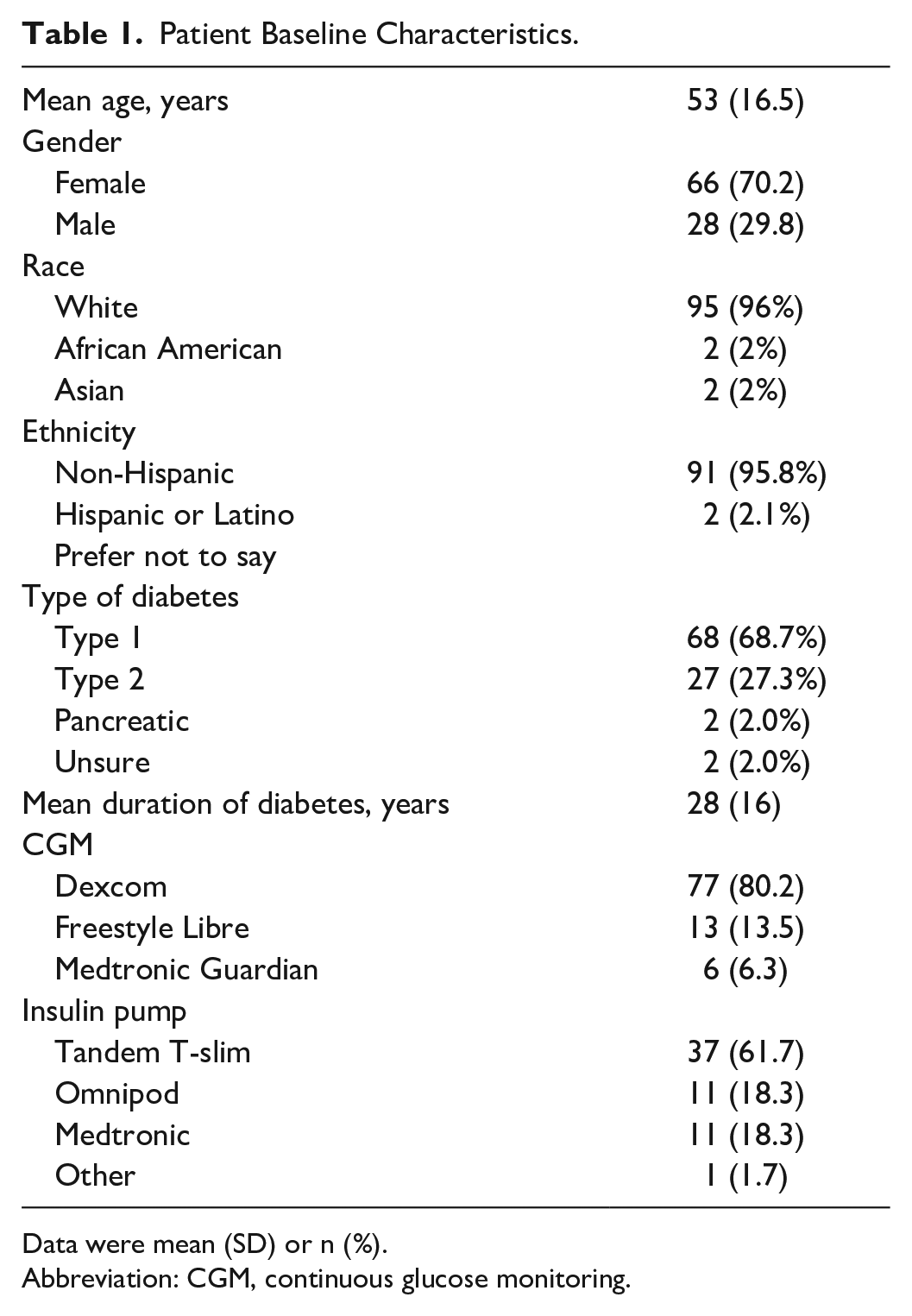
Data were mean (SD) or n (%).
Abbreviation: CGM, continuous glucose monitoring.
Device-Related Disruption Events
CGM device–related disruption events included device malfunction, insertion problems, and issues with device attachment. Only 14.6% of participants reported no disruption to their full CGM wear time. A summary of device distribution event frequency and etiology is shown in Figure 1. The average number of all-cause CGM disruptions in the last year was 8.1 among participants; this number represents the average of the composite of all disruption etiologies queried in our study. The disruption rates were consistent among age groups.
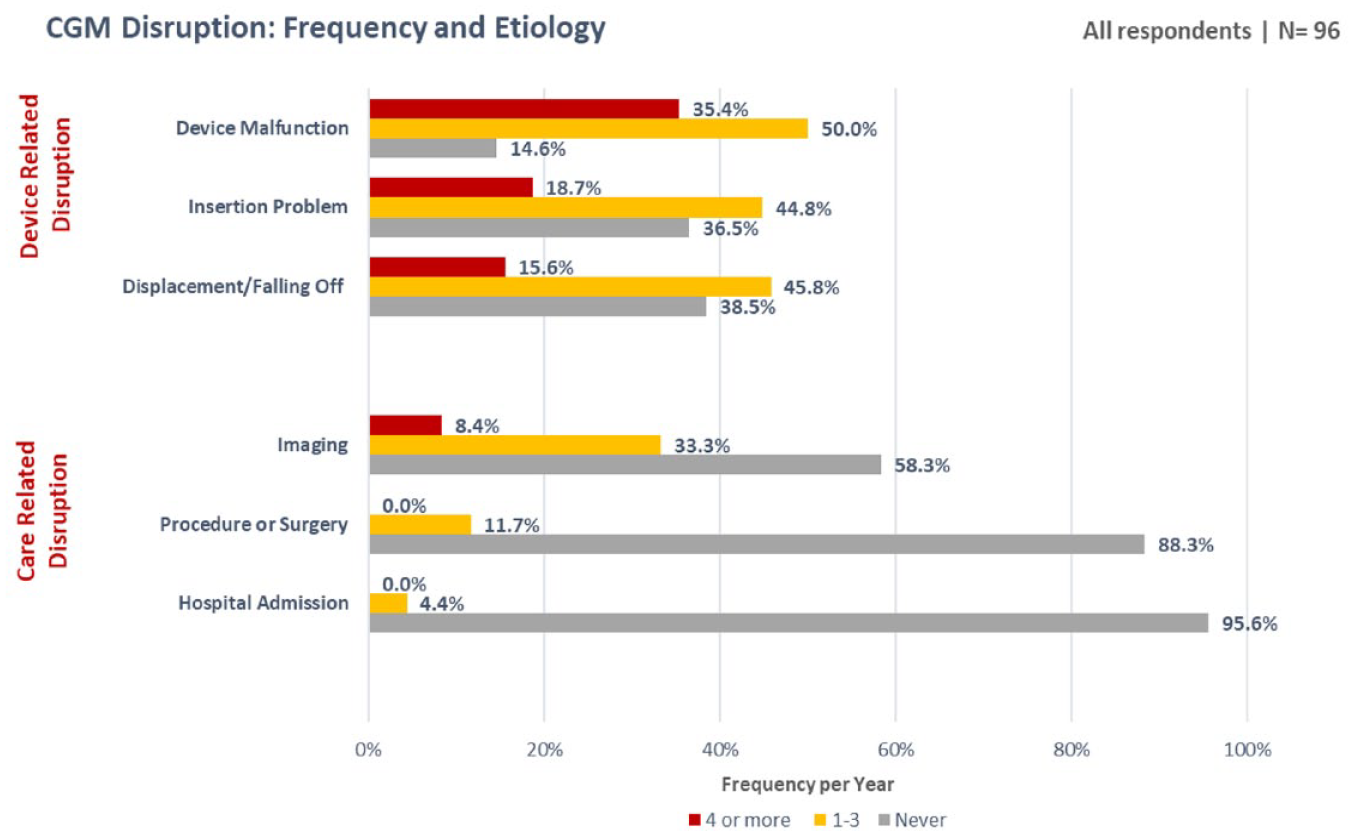
Summary of device distribution event frequency and etiology.
Device malfunction occurred at least once in the past year for 85.4% of respondents with 35.4% reporting four or more malfunctions. Insertion problems were the second most common cause of device-related disruption with 18.7% of respondents reporting more than four events. Sensor attachment issues resulting in dislodgment occurred one to three times for 45.8% of our cohort with 15.6% noting more than four displacement events.
Medical care–related disruptions occurred most frequently in the setting of imaging, followed by surgery/procedures, and hospitalization. Imaging studies lead to device removal at least once for 41.7% of individuals. Radiology studies prompted device removal four or more times in the last year for 8.4% of respondents. Most participants were able to keep their devices on for surgeries/procedures and inpatient hospitalization, 88.3% and 96%, respectively, and none experienced more than three disruptions due to these events.
Adverse Disruption Outcomes
Adverse glycemic outcomes due to the lack of CGM use, such as hyperglycemia and hypoglycemia, occurred four or more times per year in 36.5% and 12.4% of the cohort, respectively. An ER visit occurred only in two patients, and no hospital admissions were reported. A breakdown of adverse glycemic outcome frequency due to CGM disruption can be found in Figure 2.
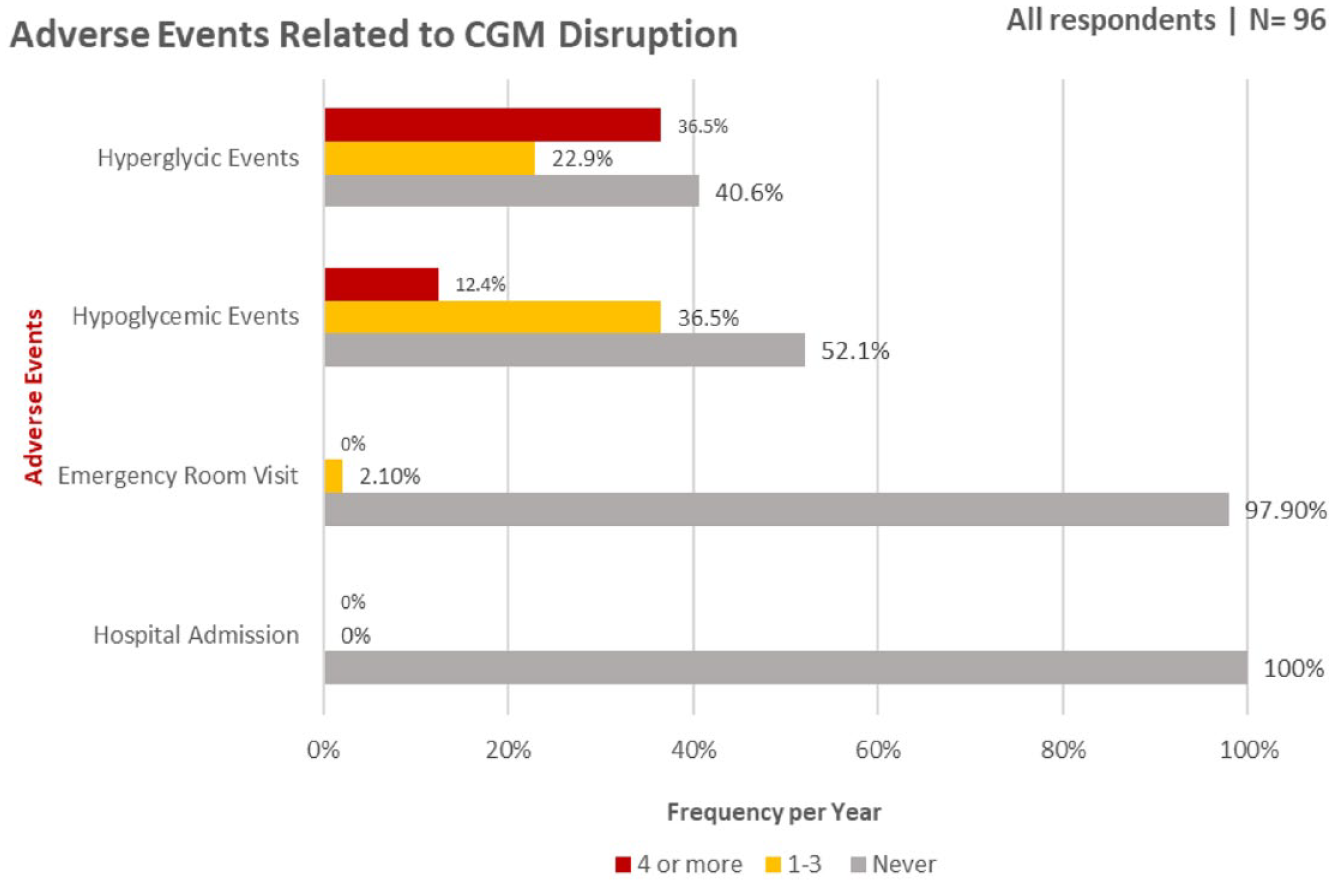
Summary of adverse glycemic outcomes secondary to CGM disruption by frequency and etiology.
The Replacement Experience
As a result of CGM device disruptions, 19.6% of those surveyed were unable to use CGM for seven or more days. A substantial proportion of individuals (47.9%) needed a CGM replacement one to three times, with 9.4% needing seven or more replacement devices. CGM replacement requests were made by 74% of those surveyed with 72.9% receiving one or more replacement devices (Figure 3). The cost of replacement was covered predominantly by the manufacturers (67.4% of cases) and insurance companies (15.2%). Out-of-pocket cost was incurred by 6.6% of participants (Figure 4). Survey participants felt that CGM manufacturers were fairly or very responsive to replacement requests 79.2% of the time.
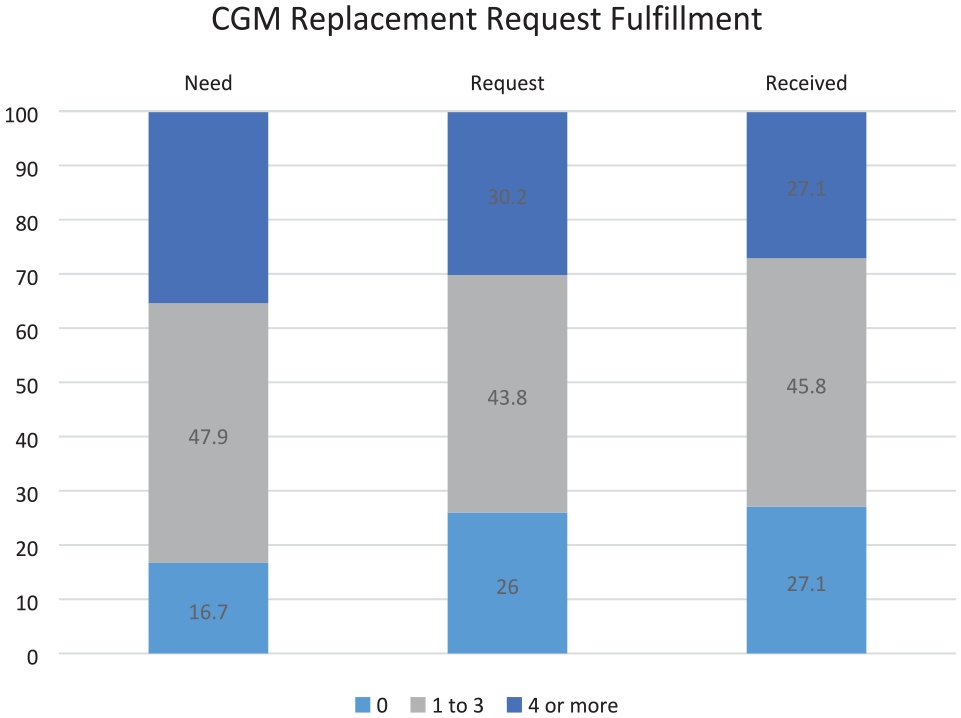
Frequency of CGM replacement requests and fulfillment.
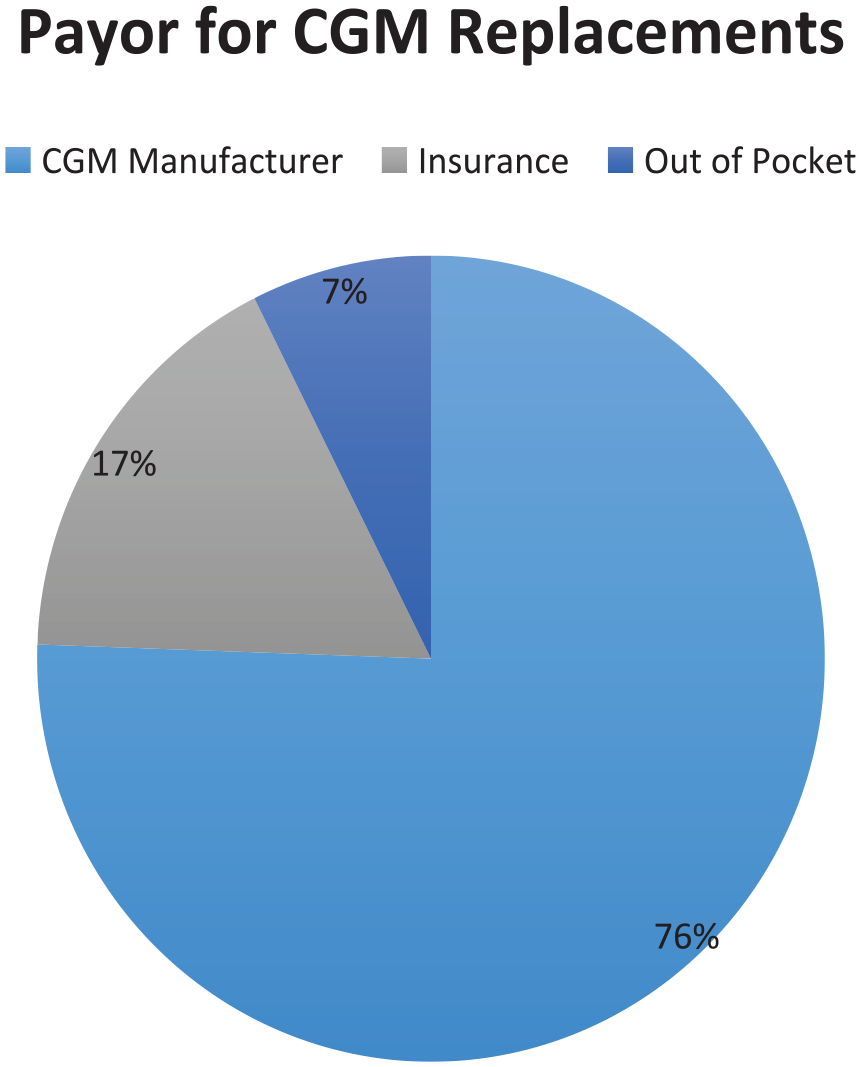
Payor for CGM replacements in the setting of device disruption.
Discussion
CGM use has revolutionized diabetes management due to highly accurate and reliable systems. CGM has been shown to improve glycemic management in individuals using continuous subcutaneous insulin infusion and multiple daily injection of insulin.6,7
To date, much thought and research has focused on the accuracy and reliability of CGM10,11 with fewer studies examining the realities of daily use. Limited data are available on sensor durability and failure rates. The US Food and Drug Administration approval documentation for popular CGM devices note that 70% to 80% of sensors last through the end of their wear period.12,13 Our study, on the contrary, shows that CGM device disruptions are the rule rather than the exception with a minority of individuals achieving uninterrupted wear time over a 12-month period. Intrinsic device factors, including malfunction, insertion issues, and displacement, are the most common etiology for disruption to wear time. Only a limited number of other studies to date have investigated the rates of these phenomena.
CGM utilization is the standard of care for individuals with T1D and for people with T2D treated with insulin. As a result, medical systems are challenged to create and implement policies for an ever-changing landscape of diabetes devices. Consensus guidelines have emerged to guide CGM use in the inpatient setting with recognition of the benefits that these devices confer to inpatient glycemic control and patient satisfaction. 14 We are encouraged by the relatively low rate of device removal in the setting of hospitalization, 4.1%.
However, in the setting of medical care, device removal for the purpose of imaging significantly contributes to an already large preexisting disruption burden. At present, most CGM devices are not labeled for use during radiation exposure (X-ray or computed tomographic [CT] scan) or during magnetic resonance imaging (MRI). The only CGM approved to be safely worn in certain MRI systems is the implantable Eversense CGM. However, emerging evidence suggests that device use may be safe during these circumstances without significant decline in sensor reliability.15,16
Only 35% of participants in our cohort reported checking capillary blood glucose in the last year. When their devices are displaced or removed, patients are at increased risk of adverse glycemic events as evidenced by the significant rates of hypoglycemia and hyperglycemia that were attributed to device interruption. Given these findings, it is critical that patients have access to replacement sensors. In addition, our data highlight the importance of having readily available blood glucose meters and strips to quickly supplant CGM devices in the event of disruption. Overreliance on CGM technology may predispose individuals to periods devoid of glycemic monitoring when these devices fail. This issue is made worse by inconsistencies in payor coverage for glucose monitoring supplies in patients using CGM, particularly with Medicare.
Current prescription and dispensing practices provide an exact number of sensors per month without redundancy to account for the realities of daily CGM use. Patients are faced with the choice of depending on manufacturer-driven replacement programs or waiting until it is time for their next prescription refill. Our data suggest that most replacement requests are fulfilled with a minority of patients incurring additional out-of-pocket expenses, 6.6%. However, this comes at the cost of prolonged CGM interruptions for many with 19.6% of our cohort going seven or more days without their devices. This is problematic for all patients, but particularly problematic for those using automated insulin delivery systems, and those patients with hypoglycemia unawareness. CGM devices are somewhat unique among replaceable types of durable medical equipment in that once removed, use is curtailed and there is no clear recourse for replacement. Consider the fallacy of providing an exact or insufficient number of ostomy bags if some do not adhere properly, or an inadequate supply of oxygen. Redundancy or a clear pathway for urgent replacement such as allowing prescribing providers to order replacement CGM supplies through a local pharmacy should be considered by payers.
Our study has several methodological limitations given that all data collected were self-reported and are subject to recall bias and nonresponder bias. Our results may be skewed by responses from individuals more dissatisfied with their CGM. Furthermore, by selecting for individuals with active CGM use, we are unable to capture those who may have given up on CGM technology altogether due to a poor user experience secondary to disruption or malfunction events. In addition, all patients were recruited from a single site with a demographic skew to an academic endocrine practice and not necessarily representative of the general population.
Further research is necessary to better estimate the appropriate amount of device replacements needed to mitigate the health consequences and cost associated with poor diabetes control. This can be addressed with a future prospective study design closely tracking CGM disruption frequency, etiology, and duration. Participant CGM data can be analyzed to provide quantitative data on the duration of sensor interruption.
The lack of redundancy of CGM supplies with current prescription and supply practices contributes to care disruptions and results in hyperglycemic and hypoglycemic events. Patients need to have access to replacement devices for continuity of care to prevent adverse events.
Conclusions
CGM is the standard of medical care for individuals with diabetes treated with insulin. Increasing numbers of individuals, primarily those with T1D, are being managed on HCL insulin systems that rely on CGM data to function. Based on our participant survey, a minority of individuals experience the full CGM wear time, and most experience numerous disruptions in CGM wear over a 12-month period. The lack of redundancy of CGM devices is a feature of current prescribing and supply limitations, and it leads to adverse hyperglycemic and hypoglycemic events. Advocacy is needed to motivate device and insurance companies to provide more than the exact numbers of monthly devices to account for the realities of daily wear more appropriately.
Footnotes
Abbreviations
CGM, continuous glucose monitoring; T1D, type 1 diabetes; T2D, type 2 diabetes.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article. A.M.M. has participated on advisory boards for Medtronic Inc and Novo Nordisk. J.B.M. has served on consulting and advisory boards for Bayer, Boehringer Ingelheim, MannKind, Novo Nordisk, and Thermo Fisher and received grant support from Beta Bionics, Medtronic Inc, NIH, Novo Nordisk, and JDRF. Other authors have no conflicts of interest to disclose.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
