Abstract
Background:
Diabetes is one of the most prevalent chronic health conditions and diabetic neuropathy one of its most prevalent and debilitating complications. While there are treatments available for painful diabetic peripheral neuropathy (pDPN), their effectiveness is limited.
Method:
This retrospective, multi-center, real-world review assessed pain relief and functional improvements for consecutive patients with diabetic neuropathy aged ≥18 years of age who were permanently implanted with a high-frequency (10 kHz) spinal cord stimulation (SCS) device. Available data were extracted from a commercial database.
Results:
In total 89 patients consented to being included in the analysis. Sixty-one percent (54/89) of participants were male and the average age was 64.4 years (SD = 9.1). Most patients (78.7%, 70/89) identified pain primarily in their feet or legs bilaterally. At the last assessment, 79.5% (58/73) of patients were treatment responders, defined as having at least 50% patient-reported pain relief from baseline. The average time of follow-up was 21.8 months (range: 4.3 to 46.3 months). A majority of patients reported improvements in sleep and overall function relative to their baseline.
Conclusions:
This real-world study in typical clinical practices found 10 kHz SCS provided meaningful pain relief for a substantial proportion of patients refractory to current pDPN management, similar to published literature. This patient population has tremendous unmet needs and this study helps demonstrate the potential for 10 kHz SCS to provide an alternative pain management approach.
Introduction
Neuropathy is one of the most impactful consequences of diabetes mellitus and a cause of significant disability. 1 Despite its high prevalence in more than 50% of patients with diabetes mellitus 1 and the relative impact on quality of life considered higher than for other chronic pain conditions, 2 management options for peripheral neuropathy remain limited. 3 At present, no disease-modifying medications have been discovered, therefore treatment has been reliant upon improving glycemic control, management of cardiovascular risk via lifestyle modifications and management of neuropathic pain symptoms.3,4
Diabetic peripheral neuropathy typically presents distally and symmetrically with progression more proximally. 5 A diffuse “glove and stocking” pattern is characteristic of the condition and may present along with other typical neuropathic pain features (Figure 1). While needle electromyography and nerve conduction studies are the gold standard of diagnosing sensorimotor polyneuropathy, they are not required. As such, diagnosis may often be one of exclusion based on history, signs and symptoms without electrodiagnostic testing.6,7 Damage to the peripheral nervous system often affects both small and large fibers although some patients will have predominant disease of 1 type. While there is some correlation with length and severity of poor glycemic control, nerve damage can also occur in people with prediabetes. 8 In addition, peripheral neuropathy persists and can continue to progress despite optimal blood glucose management. The underlying pathogenesis is not completely understood, but the impact of the symptoms on patient quality of life has been well documented.9,10 Activities of daily living, work, sleep and mental wellbeing are all impacted by burning neuropathic pain in the hands and feet.9,10
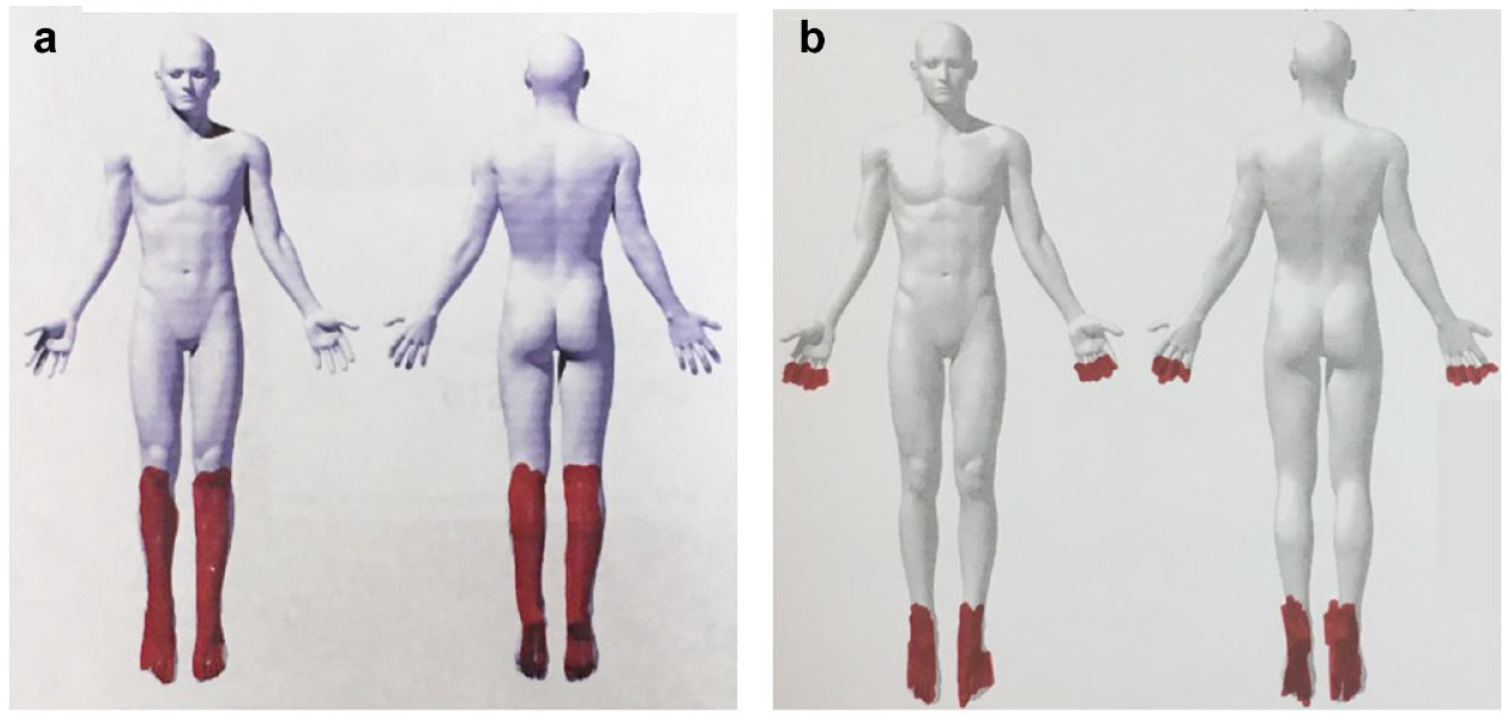
Examples of “stocking and glove” distribution of pain with peripheral diabetic neuropathy: Two patients with pDPN shaded the areas affected by pain in red, with (a) drawn by a patient with only lower limb symptoms that typically present earlier in the disease and (b) drawn by a patient whose symptoms have progressed to affect both upper and lower limbs. Abbreviation: pDPN, painful diabetic peripheral neuropathy.
Painful diabetic peripheral neuropathy (pDPN) can be difficult to treat. 3 Current clinical guidelines recommend pharmacological management options, including calcium channel alpha-2-delta ligands, serotonin-norepinephrine reuptake inhibitors, and tricyclic antidepressants followed occasionally by tramadol and other opioids, but many patients are unable to achieve meaningful pain relief. 2 ,11-13 More localized, topical treatments may also be administered, including capsaicin cream or a high-dose capsaicin patch. In addition to limited efficacy, side effects from these medications may also contribute to early treatment cessation. 14 Together, this may be reflected in the higher healthcare cost and utilization observed with pDPN. 15 A study in patients in the United States with pDPN spent between 20% and 31% more on medications than patients with diabetes and no neuropathy. They also had a significantly higher risk of opioid use (200%) and limb amputation (16.24 times higher). 15
Given the interest in non-opioid treatment options, there has been increasing interest in non-pharmacological alternatives. 5 Neuromodulation is one modality which has been investigated for pain management in pDPN. 5 Conventional spinal cord simulation (SCS) is a reversible, adjustable therapy that has been used for decades in a number of chronic neuropathic pain conditions.16,17 Despite modest outcomes in small cohorts of pDPN patients, the widespread adoption of the treatment is not without challenges.5,18 As pDPN primarily affects the extremities, there are technical challenges with conventional SCS that make pain relief difficult to obtain as this technique requires the induction of paresthesias overlapping the painful area.17,19,20
Alternatively, high-frequency SCS at 10 kHz uses a 10,000 Hz waveform that provides pain relief without inducing paraesthesia.21,22 The treatment has a growing body of randomized, controlled clinical trial and real-world observational evidence across a variety of pain conditions such as chronic refractory back pain, radicular leg pain, upper limb pain, axial neck pain, and now pDPN.5,23-29 As a paresthesia-independent mode of neuromodulation, 10 kHz SCS has shown potential for its application in pDPN. In 2020, in a case series of painful polyneuropathies, including 3 patients with pDPN, 10 kHz SCS delivered pain relief as well as sensory improvement and decreased reliance on pain medication. 30 A recent prospective multicenter, randomized, controlled trial (RCT) called the SENZA-PDN study evaluated 216 patients with pDPN assessed 10 kHz SCS in addition to conventional medical management.31,32 Results over 6 months demonstrated profound pain relief, improved function and quality of life, and further confirmed that a majority of pDPN patients treated with 10 kHz SCS experience improvements in sensation.
Limited studies exist to corroborate RCT findings in typical real-world clinic settings. Therefore, this study aims to analyze the effectiveness of 10 kHz SCS in pDPN patients in a real-world, multicenter patient cohort.
Methods
Study Design
This study was a retrospective analysis of anonymised data extracted from a commercial real-world database (HFXCloudTM) of patients aged ≥18 years of age with diabetic neuropathy who were trialed and permanently implanted with a 10 kHz SCS device between May 2017 and November 2020. Patients provided consent to being included in the analysis before their data were downloaded in May 2021. Trial and permanent device implantation were performed according to each site’s standard practices. Briefly, patients who were candidates for the treatment were trialed with 10 kHz SCS (Senza System, Nevro Corp., Redwood City, California) by placing temporary leads anatomically at vertebral levels ranging from T8 to T11 (Figure 2). Given the paraesthesia-independent nature of the 10 kHz SCS therapy, intraoperative paraesthesia mapping was not required. Patients who reported at least 50% pain relief during the trial period were eligible to receive permanent device implantation. Stimulation was delivered at a frequency of 10 kHz and programmed according to standard algorithms for treating foot pain. Informed consent language for study participation was approved by the WCG Institutional Review Board (Puyallup, Washington).
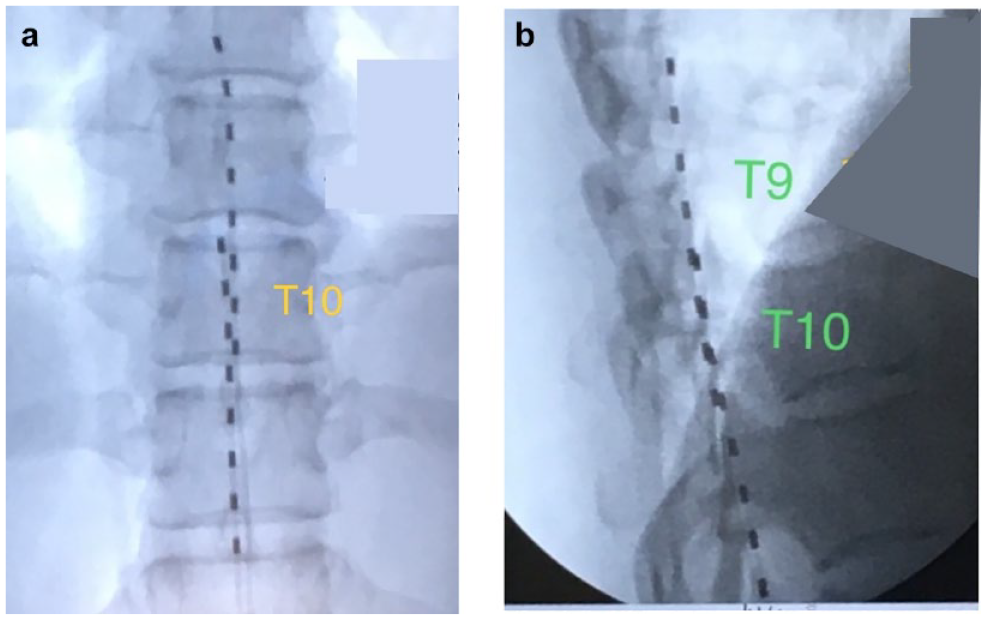
Typical dual octopolar lead placement along the midline T8-T11 epidural space shown in (a) anterior-posterior and (b) lateral x-rays.
Patients were assessed for baseline prior to 10 kHz SCS trial and at regular follow-up visits after device implantation. Intervals of patient follow-up after device implant were variable and performed according to each center’s standard protocol.
Patients were asked to report their percentage of pain relief from 0% to 100% at each follow-up visit compared to baseline. Successful response to 10 kHz SCS was defined as at least 50% patient-reported pain relief. Additionally, patients were asked about changes in sleep and improvement in overall function. All outcome data available in the database were aggregated for this report.
Results
Patients eligible for inclusion were contacted between 5 April 2021 and 23 April 2021 and a total of 89 consented to be included in the analysis. Two patients included in the analysis had their devices inactive. Patient characteristics are shown in Table 1. A majority of participants (60.7%, 54/89) were male and the average age of the group was 64.4 years (SD: 9.1, range: 41.9-85.3). Overall, the median time between implantation and the last visit was 18.4 months (SD: 11.0). All patients had pDPN listed as their primary pain etiology with most (65.2%, 58/89) reporting bilateral foot pain. A subset of patients (10.1%, 9/89) also reported significant low back pain in addition to pDPN. Some included patients (13.5%, 12/89) had prior experience with other types of SCS.
Baseline Demographic and Clinical Characteristic Criteria.
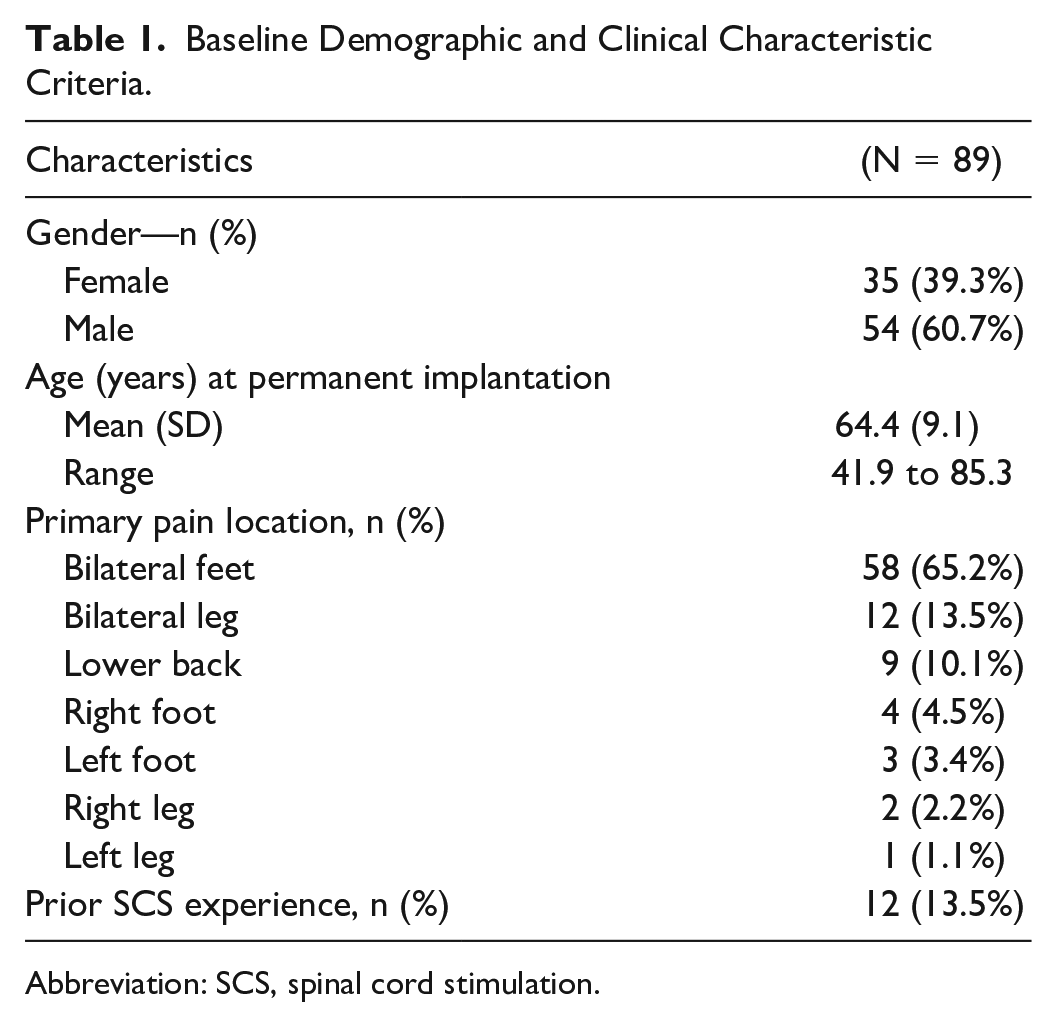
Abbreviation: SCS, spinal cord stimulation.
Figure 3 shows the percentage of pain relief reported by each patient at their last follow-up. Responders are defined as those who reported at least 50% pain relief compared to baseline. At the last assessment, 79.5% (58/73) of patients were treatment responders over an average follow-up of 21.8 months (range: 4.3-46.3 months). The average reduction in pain during the assessment period was 60.5% (SD: 23.6%).
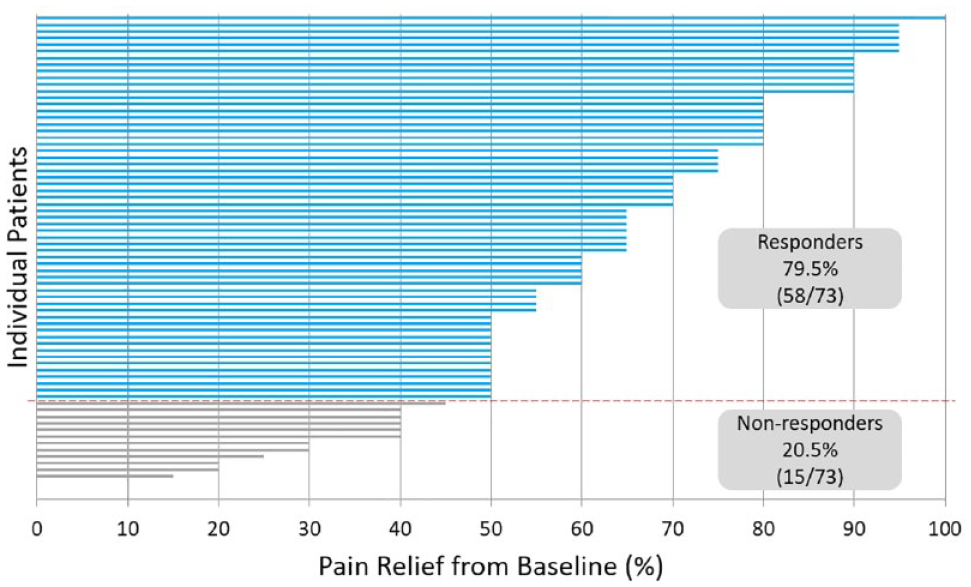
Patient-reported percentage pain relief at last visit compared with baseline.
Pain responders were also evaluated over time since permanent device implantation. Figure 4 illustrates the patient flow and number of evaluable patients at each follow up duration. Figure 5 shows the proportion of responders out of the total number of patients who had completed the designated follow-up at the time of the data extraction. Responder rates are stable longitudinally with 84.7% (50/59) of patients reporting at least 50% pain relief maintained over 12 months. Twenty-seven patients had completed 24-month follow-up post-implant and 88.9% (24/27) continued to report at least 50% pain relief compared to baseline.
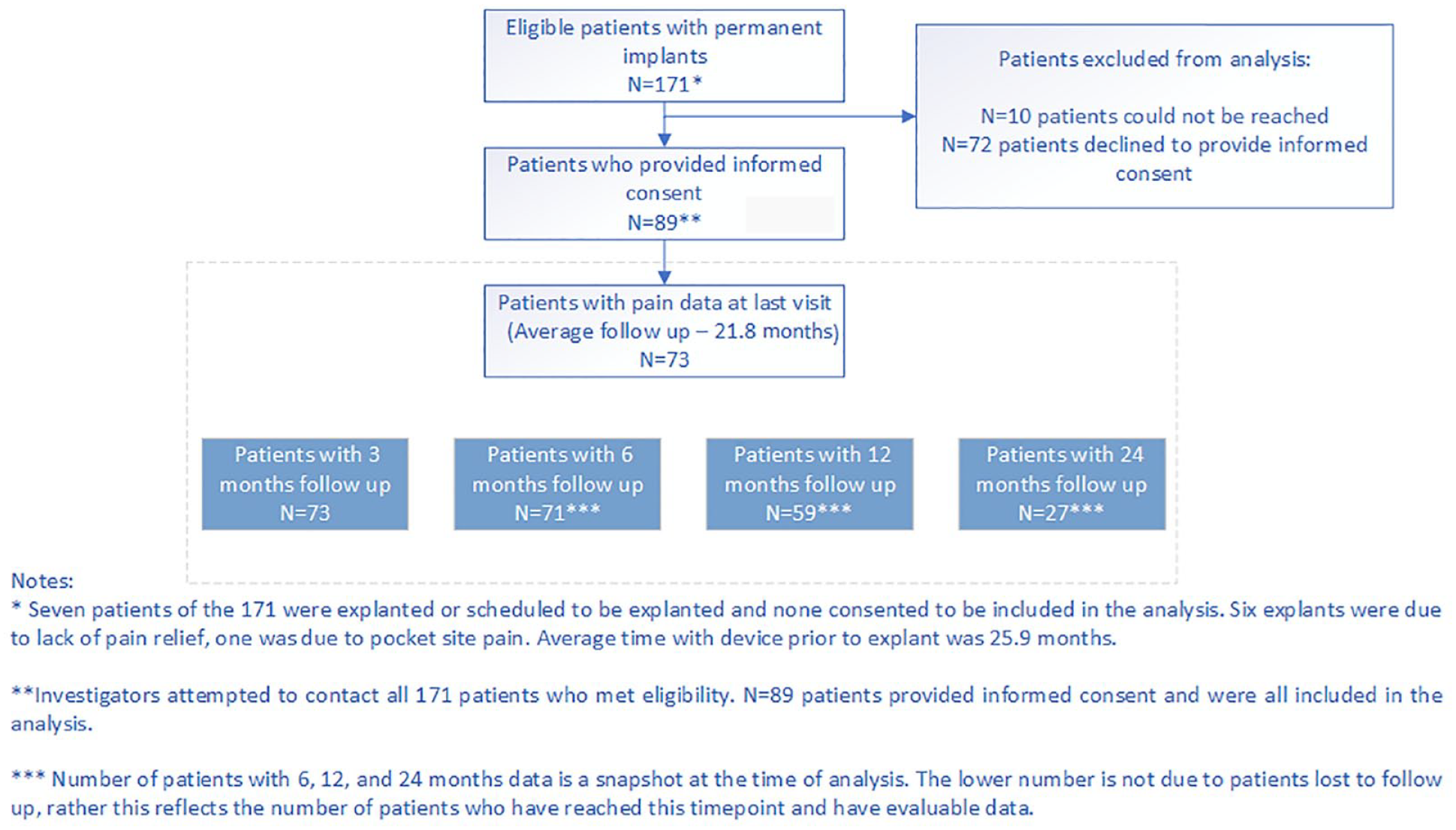
Patient flow diagram to illustrate evaluable patients at different follow up durations.
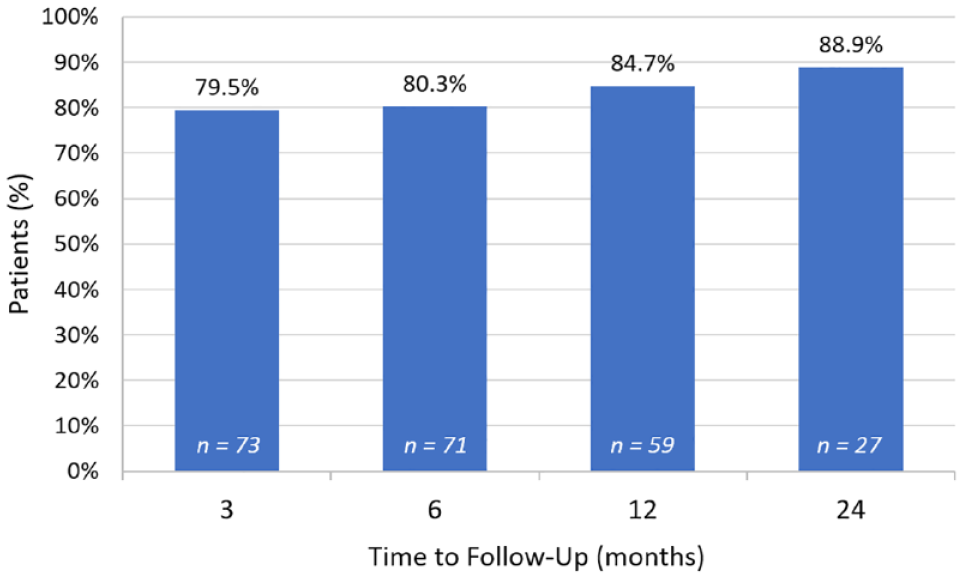
Proportions of pain responders over time, defined as at least 50% pain relief reported.
Patients were asked about improvements in sleep and overall ability to function since treatment with 10 kHz SCS. A majority reported improved sleep (78.5%, 51/65) as well as improved function (76.0%, 57/75, Figure 6).
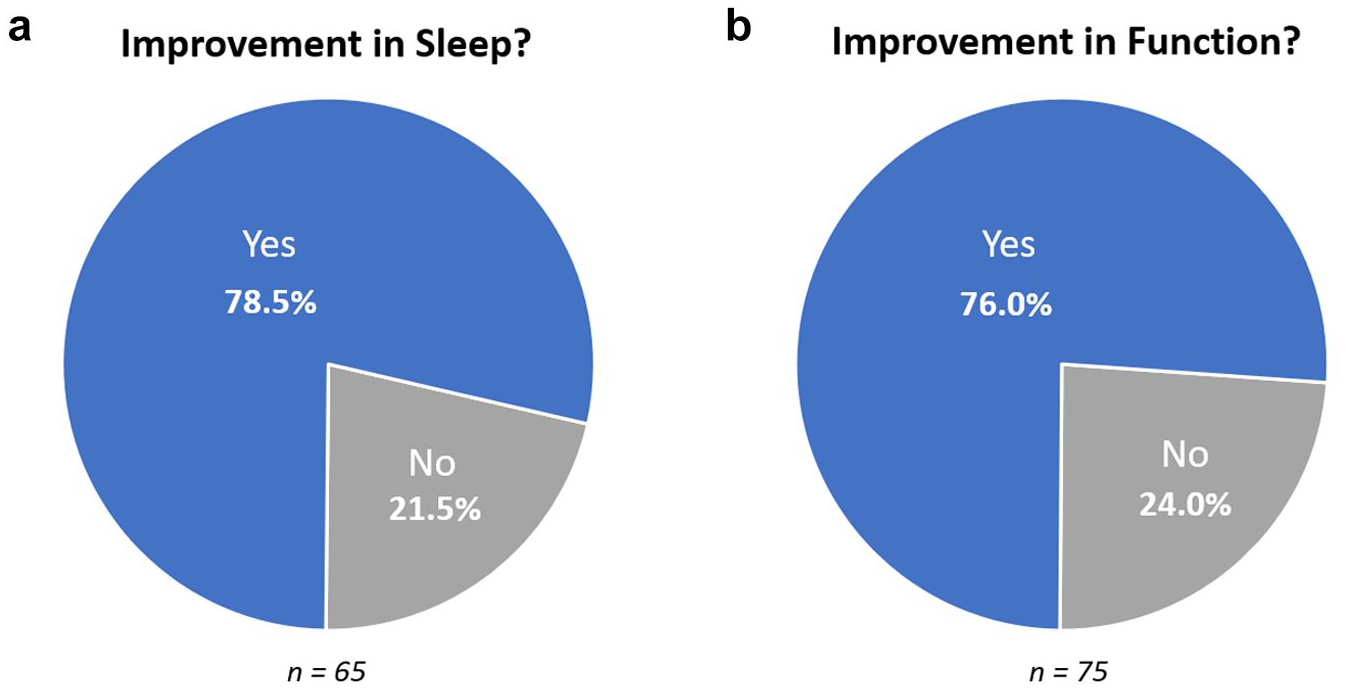
Evaluation at last visit of (a) changes in sleep and (b) overall function.
Discussion
Painful DPN is a progressive and disabling condition that can be difficult to manage.1,3,4 While medical management options exist, there can be a trade-off between adequate pain relief and unacceptable treatment-related burden. 15 For a number of years, neuromodulation has remained an enticing, yet elusive treatment option due to challenges for conventional SCS providing acceptable coverage of the periphery without causing uncomfortable paresthesias.33-35 Studies with conventional SCS in pDPN were small and demonstrated limited success.33-35 Given the success of 10 kHz SCS in other chronic, intractable pain states,36-45 the randomized, controlled, clinical trial, SENZA-PDN set out to examine the utility of 10 kHz SCS in pDPN.31,32 Not knowing how 10 kHz SCS translates in clinical practice, this retrospective, real-world analysis of pain relief and selected quality of life measures reported in 89 patients with pDPN demonstrated a high level of effectiveness and concordance of responder rates reported in the SENZA-PDN randomized controlled trial. 32
Similar to results reported for 10 kHz SCS in randomized, controlled trials and real-world evidence in other pain states such as trunk and limb pain and upper limb and neck pain,42-44,46 this study reports 79.5% of patients achieved at least 50% pain relief compared to baseline. This is impressive given the limited lines of pharmacological therapy and the high rate of treatment failure of the current options available.2,47 Typically, only around 30% of patients are expected to achieve even a 30% reduction in pain with conventional medical management.2,47 It is also highly aligned with the results in the SENZA-PDN clinical trial population, where 79% (n = 75/95) patients achieved a treatment response (≥50% pain relief plus no worsening of baseline neurological deficits) compared to only 5% (n = 5/94) on medical management alone (P < .001). 32 At 6 months, mean VAS scores reduced by 76.3% (versus no change in the conventional medical management group). 32 Furthermore, 62% of patients in the device arm had improvement in neurological function, including sensory perception of 10-g monofilament and pinprick, at 6 months compared to only 3% in the medical management arm.
While SENZA-PDN assessed 10 kHz SCS in patients with pPDN over a 6-month follow-up, the current analysis not only provides an insight into treatment performance at 6 months in a real-world setting, but it also provides longer-term follow-up data on 59 patients implanted for at least 12 months and 27 patients implanted for at least 24 months as of May 2021. While the patient numbers are currently limited over the longer-term follow-up due to a majority of patients not yet reaching 24-month follow-up, it is reassuring to see a durability of pain response over longer follow-up in a real-world setting.
Limitations
This analysis had limitations due to use of a commercial database. First, the retrospective nature of this study limited the systematic collection of patient data, including clinical characteristics, medication use, implantation details and quality of life measures. Additionally, given the last visit approach of the data analysis, patients were at varying timepoints since permanent device implantation. Finally, analyses included in the study were limited to available data which were not collected uniformly for all patients. The lack of randomization plus need for insurance approval could also introduce selection bias for the patients who receive treatment and may not be representative of the broader population.
Conclusions
This real-world study concurs with findings from the SENZA-PDN randomized, controlled trial at 6 months, finding a majority of patients reported significant pDPN pain relief that conferred improvements in function and sleep. These outcomes demonstrate durability in a real-world setting as patients included had treatment for 21.8 months on average and up to a maximum of 46.3 months. Therefore, this study provides additional evidence of the utility of 10 kHz SCS as a treatment option for patients with pDPN. However, the results have limitations and should be interpreted with caution, despite their concordance with previous reports of 10 kHz SCS effectiveness in pPDN.31,32
Footnotes
Acknowledgements
The authors would like to thank Cat Panwar, PhD, of Panwar Health for help with drafting this manuscript.
Abbreviations
pDPN, painful diabetic peripheral neuropathy; SCS, spinal cord stimulation; kHz, kilohertz; SD, standard deviation; RCT, randomized controlled trial
Declaration of Conflicting Interest
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Drs. Chen and Sills have received research funding from Nevro Corp.; Dr. Hesseltine has received consulting fees from Nevro Corp.; Dr. McJunkin has received consulting fees from Nevro Corp. and Omnia Medical; Mr. Patil and Drs. Bharara, Caraway, and Brooks are employees of Nevro Corp.; Dr. Nashi has nothing to disclose.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Nevro Corp. (Redwood City, CA) provided funding for medical writer assistance in drafting this manuscript.
