Abstract
This article gives a short summary of standardized documentation for pediatric diabetology from a European perspective. The approach chosen by the Austrian/German DPV (Diabetes Patienten Verlaufsdokumentation) group is detailed. The electronic health record used is briefly described, as are external benchmarking reports and national and international comparisons. Similar initiatives like the Hvidore study group, the SWEET initiative (Pediatric Diabetes: Working to Create Centers of Reference in Europe), and the T1DExchange (Type 1 Diabetes Exchange Registry) are compared to the DPV effort.
Keywords
An essential feature of standardization of measurement is that it allows for detection and learning from variation present in a health care system. Standardized documentation has the advantage to improve patient care due to better exchange of information among members of a medical team and, in addition allows both, internal and external quality control. This article addresses the latter aspect, based on 20 years of experience from the Austrian/German quality initiative DPV.
The aspect of quality of medical care has somewhat different challenges in chronic diseases, as opposed to acute medical disorders. For acute problems, QC (quality control) will focus on the question whether the problem has been solved to the complete satisfaction of patients, whether side effects did occur, for example infection or postsurgery bleeding following a surgical procedure, and what the costs arise for the patient (length of hospitalization, pain, number of control exams, direct financial burden for the patient) as well as for society (time not at work) or reimbursement companies. These aspects are often quite easy to cover with a limited number of items. 1 In contrast, the nature of chronic disorders provides specific challenges that have to be addressed when standardized documentation for chronic disorders like diabetes are implemented. 2
Historic Aspects: The St. Vincent Initiative
In October 1989, the WHO (World Health Organization) and IDF Europe (International Diabetes Federation) gathered diabetes experts and government officials in St. Vincent in the north of Italy to discuss actions required in view of the rising number of patients with type 2 diabetes. 3 The St. Vincent declaration identified 5 major goals to be achieved during the next 5 years (Table 1). 4
St. Vincent Declaration 4

The use of standardized documentation and information technology together with the application of quality control techniques developed previously in automobile manufacturing especially in Japan were listed among other tools to improve diabetes care. A specific data set and a documentation tool were developed in Europe (DiabCare or QUALIDIAB), with modifications for children. The St. Vincent initiative has had long-lasting effects on the public health aspects of diabetology 5 and has been adopted to other regions of the world like the “Declaration of the Americas” 6 or the Western Pacific Declaration on Diabetes in Kuala Lumpur in 2000 7 or the DiabCare project in India. 8
International Comparisons
The Hvidore Initiative
The Hvidore initiative was initiated in 1994 by Henrik Mortensen from Glostrup hospital, Denmark and supported by NovoNordisk over many years. Leading centers for pediatric diabetology from 18 countries in Europe, North America, and Japan contributed standardized information together with a central measurement of HbA1c on each of their patients. 9 Remarkable differences for average HbA1c at the participating centers were observed. In several subsequent projects the group demonstrated that these differences were stable over time, and that patients with good metabolic control during the early years of diabetes experience better metabolic control during the subsequent years as well. Different hypotheses have been tested on the nature of those center differences, including intensity of insulin therapy (which was not as closely related to metabolic control as expected by many participants) and structural aspects of pediatric diabetes care. In addition to HbA1c, quality of life of the patients and their families was a major focus of the group. Even as the majority of data were collected during an era where insulin pumps and technology were used by a small number of patients only, the work of the group was pioneering the field of international comparison of process and outcome indicators in an international environment and many subsequent joint projects were based on personal interactions among pediatric diabetologists from the Hvidore group. Major findings of the Hvidore research were recently summarized. 9
The SWEET Initiative—An International Effort of ISPAD
The SWEET initiative follows on the Hvidore results. 10 Again, major pediatric diabetes centers cooperate to improve outcomes of pediatric diabetes care. The project started as a European initiative based on a grant from the European Union, but since has spread out across the world including centers in Asia, North America, Africa, and Australia. The group uses a concise data set relevant for pediatric diabetology for standardized documentation of process and outcome of care, leading to regular benchmarking reports provided for the participating pediatric diabetes centers. In addition, audit visits, following a scheme developed by the UK NHS (United Kingdom National Health Services) are implemented, where each center is visited by a team consisting of a peer colleague from a different country together with the NHS audit teams. If a center fulfills the minimum requirements, the title of “SWEET pediatric diabetes center of reference (COR)” is awarded. Among the requirements are a minimum number of 150 patients seen at the center, a multidisciplinary team with pediatric diabetes expertise and experience, participation in quality circles, and a strong contribution to research. 11 Most recently, an ISPAD advantage invites smaller centers and centers from developing countries to participate in the SWEET initiative as collaborating centers.
Population-Based Information—From Incidence Registries to Longitudinal Follow-Up
Early efforts focused on the epidemiologic aspects of diabetes, comparing incidence and prevalence. In Europe research groups from Scandinavian countries have been on the forefront on the establishment of population-based registries.12,13 In the United States the Pittsburg Registry 14 inspired many countries to start epidemiologic registries and compare the results: Eurodiab in Europe 15 or the IDF atlas, 16 comparing incidence and prevalence of diabetes on an international or even worldwide scope are results of this approach.
While comparisons of epidemiologic data from different regions might give some insight into the pathogenesis of both type 1 and type 2 diabetes, and quantitate the burden of diabetes for different societies, it quickly became evident that patients with diabetes have to be followed longitudinally and the development of acute and chronic complications during the disease have to be documented in a standardized way. The Allegheni County registry 17 and the EURODIAB Prospective Complications Study Group18,19 have been early examples for this effort. Results are encouraging and reflect the huge improvements in the outcome of long-term care for patients with type 1 diabetes. Comprehensive patient education and encouragement of self-management, improved insulin therapy, easy access to self-measurement of blood glucose (SMBG), and early treatment of concomitant vascular risk factors like hypertension, dyslipidemia, or smoking together with specialized medical care teams provided the basis for these remarkable improvements. In addition, reduced mortality for patients with long-standing type 1 diabetes has been reported by several groups.20,21
Why Implement Standardized Documentation?
Goals and benefits of standard documentation to interdisciplinary teams are:
▶ Comprehensive records documenting all relevant medical results and procedures are a standard requirement for doctors in most countries. With larger services and more patients covered, paper documentation gets difficult to handle, with respect to both storage space and readability of the notes.
▶ Use of computers for routine tasks like scheduling appointments, writing letters to referring physicians, informing patients, serial letters to doctors and families, administrating diabetes education groups, and so on.
▶ In-house transparency and quality management: To have key indicators of process and outcome easily available is an important feature to improve a large specialized diabetes service with several doctors involved. Questions like “How many patients use insulin pumps in our institution?,” “How many patients left our service during the last year?,” and “What is the average HbA1c for patients beyond remission in our service?” should be easily retrievable. This is only possible with standardized, electronic documentation.
▶ For adequate reimbursement of services comprehensive data are required. Despite differences, this is due for most health care systems, even for nationally funded systems, and becomes even more paramount when the quality of the service is directly related to reimbursement (“pay for performance”).
▶ Participation in regional or national programs, like registries, quality circles, continuous education.
▶ Identifying patients suitable for research protocols may also be a highly relevant goal, with positive financial implications for the service.
▶ Standardized data on an unselected subgroup or all patients seen during a certain period by a health care provider are often required for center certification, indicative of a high standard of care. As a certified center, financial reimbursement may be higher, and/or the center may attract more patients.
▶ Health care research is a further aim, which requires high-quality, standardized data. Ideally, health care providers are funded for the generation of these data.
How to Document: Paper, Computer, or Cloud?
Despite dramatic developments in information technology, the question of how to document the longitudinal course of a chronic disease is still challenging. Paper records are still used in many medical institutions, with the advantage of flexibility and easy implementation without the dependency on the continuous functioning of IT infrastructure, and with a relatively straight-forward concept for the protection of data privacy. However, paper records are often not standardized (free field documentation), and data are not readily available for aggregation and statistical analysis. Only few of the goals of documentation as described in the previous paragraph can be accomplished with paper records. Therefore, in many parts of the world electronic medical records have become routine, either as an exclusive documentation system or in combination with paper records. However, up to now, many systems are competing and standardization is lacking both nationally and internationally.
DPV—Electronic Patient Record
In Germany and Austria a computer-based electronic health record specific for pediatric diabetes care has been developed and continuously updated during the last 22 years.22,23 It is the only documentation instrument that feeds data into the DPV registry and is used as electronic patient record in clinical settings. participation and usage of DPV software is voluntary as the software is not part of a national electronic medical record system in Germany or Austria. To improve participation of treatment centers, the software should provide many functions useful for both pediatric as well as adult diabetes centers, to ensure a wide usage:
The DPV software (Figure 1) includes all items required for the Austrian/German diabetes benchmarking, for certification of treatment centers in Germany (centers for type 2 diabetes only, centers for type 1 and type 2 diabetes, pediatric diabetes centers, centers certified for diabetic foot syndrome, centers certified for patients with diabetes who are admitted for nondiabetes reasons—orthopedic surgery, psychiatric comorbidity etc), for participation in the SWEET initiative, for the adult and the pediatric diabetes passport, for the German disease management programs type 1 and type 2 diabetes, as well as those items required to document high-level care according to the Austrian and the German national guidelines.

Key functions of the DPV documentation software.
Beside these mentioned national benchmarking tools the software allows the automatic generation of patient reports to be sent for referring physicians, graphical and tabular summary of previous results from the patient, as well as serial letters to doctors, patients and families. Best practice alert reminds the diabetes care team of pending routine examinations. Links to diabetes software DIABASS PRO allow download of data from insulin pumps performed during the clinical visit.
What to Document: Extensive Versus Focused Data Sets
There are many, yet inconsistent, recommendations for standardized documentation in diabetes. Items specific for diabetes care are usually not part of the standard data set of most clinic information systems. This has led to the definition of disease-specific minimal data sets for quality management and research. In Europe, the DiabCare basic information sheet 24 was an early adoption of this approach, which has been embraced by many groups, with a pediatric modification becoming part of the ISPAD (International Society of Pediatric and Adolescent Diabetes) guidelines, also used in the SWEET initiative. 10 Unfortunately, such standardized data sets are usually not part of electronic health records used in institutions providing care for pediatric or adult patients with diabetes, with the exception of data sets required for reimbursement. 25
A major mistake repeated by many groups embarking on multicenter standardized care is an extensive list of parameters to document during routine care (Table 2). The effort to comprehensively document as many aspects of diabetes care as possible may result in centers not participating at all, or providing incomplete data, or documenting a subset of patients only. It is preferential that the aims of the documentation are consented first, and only items directly related to these aims are included into the data set. Additional items may be added subsequently, after the infrastructure for the complete documentation of the limited data set has been established. It should also be possible to add items required to answer a specific research question only temporarily to the data set. For example, a current research in Austria addresses the role of parietal cell antibodies in pediatric diabetes patients, using a submask within the DPV documentation system visible only to centers in Austria. 26
List of Parameters Used in the DPV Diabetes Documentation System.

National and Regional Analyses
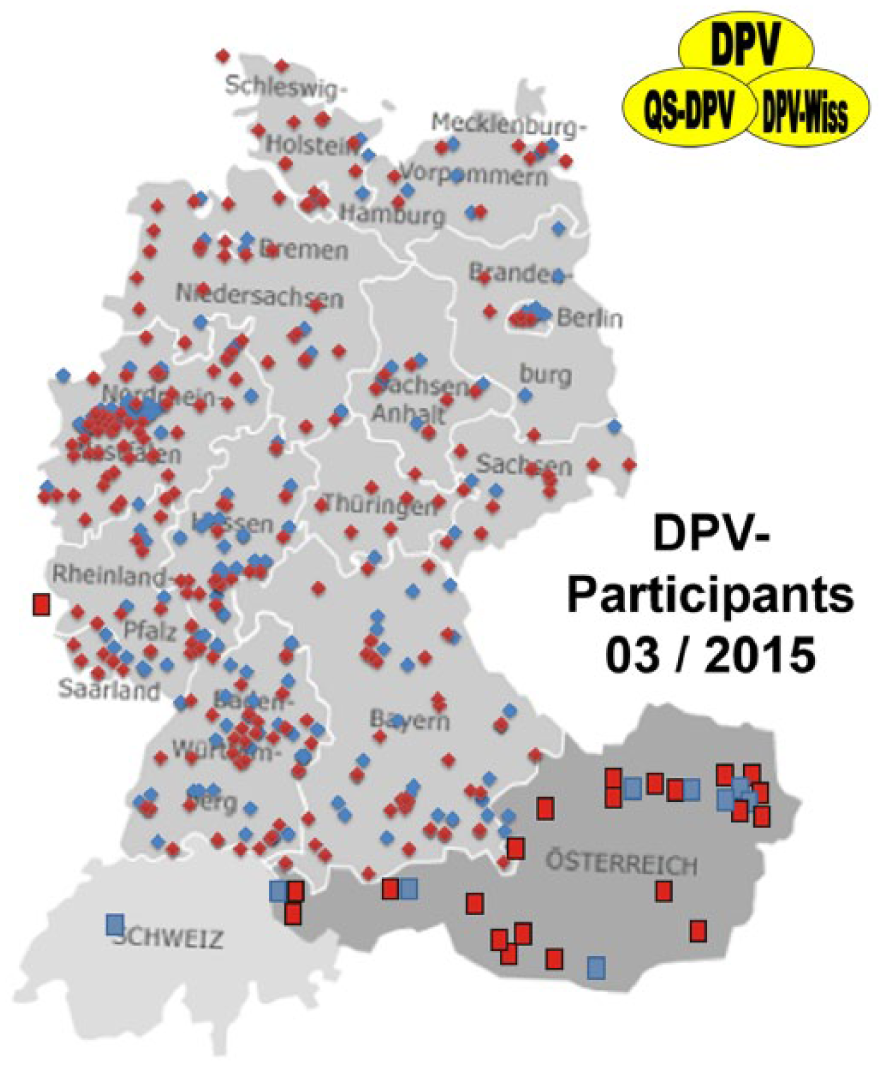
In Austria and Germany, we provide feedback in different ways. Analysis can be performed for all centers together (both nations analyzed together), for pediatric and for adult centers separately, for both nations separately if requested (Figure 2).
Map of Germany, Austria, Switzerland, and Luxemburg with participating specialized diabetes centers (pediatric centers in red, internal medicine centers in blue).
In addition, 14 regional pediatric quality circles use open analyses as a basis for discussion during there usually half-yearly meetings. This type of work is heavily recommended by the German Medical Society, however it requires a climate of openness and mutual trust. Differences among centers are much more meaningful and better to interpret if the identity of the respective centers is known. 27 However, there is agreement with all participating centers that national analyses in Germany and Austria are reported in anonymized form. This is in contrast to other national registries, such as the UK National Pediatric Diabetes Audit, which openly publishes results from each center, freely available on the Internet. 28
Participation in DPV is voluntary and centers have commonly agreed on anonymous evaluation modus. open reports would only be possible if all participating centers agree; this might be the case in some regional quality circles. In this special situation with agreement of all contributing centers open reports can be performed.
Although open reports might have the potential to further improve care and outcome there is also a possible risk, that centers discontinue sharing their data. This would lead to a major disadvantage as the population based character is a key strength of DPV. In both Austria and Germany, patients are free to choose their diabetes care team, and competition is present, especially in densely populated areas. Open reports therefore would get the potential to influence patient movements.
Documentation and Feedback: Benchmarking Reports
Feedback on the quality of a diabetes service, in relation to similar health care providers in the region or country, is an important component of a quality improvement initiative. Such benchmarking reports have been provided in Germany and Austria for more than 20 years. For each service, the results from the own institution are identified, while results from all other services are provided in anonymized form. So far, the step to entirely open reports has not been possible in Austria or Germany, in neither pediatric nor adult diabetology.
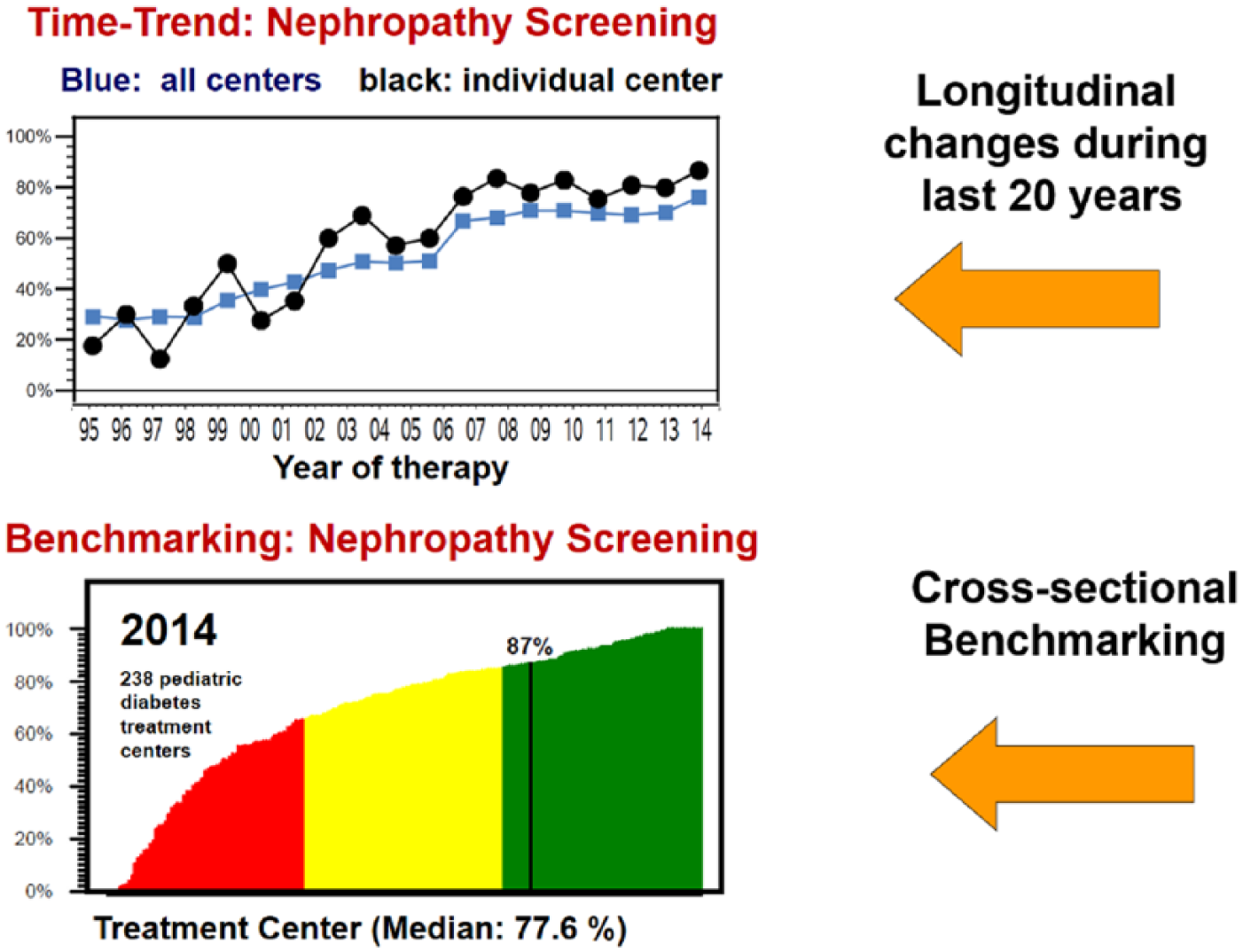
Most benchmarking reports provide histograms comparing the results among centers. The DPV benchmarking reports do this as well, using the traffic light scheme to differentiate centers with good, intermediate, or “substandard” results. In addition, for most quality indicators a longitudinal view is provided, with the change of the indicator at the respective institution in comparison to the change in the entire group. It is our firm belief that this dynamic view on medical quality is even more important than the static one: a center that improved during the last year from “poor” to “average” quality achieved a dramatic improvement, which would not be visible in the cross-sectional analysis.
In addition, 2-dimensional graphs give the relationship between competing quality indicators, for example frequency of nephropathy screening per center as shown in Figure 3.
Anonymized external comparisons, cross-sectionally and longitudinally, as provided by the Austrian-German DPV quality improvement initiative.
A Challenge: Following Patients Across the Transition From Pediatric to Adult Care
A unique patient identifier, like a social security number, is required to follow-up patients after the institution providing diabetes care has been changed. In many European countries, legal requirements and privacy rules prohibit such unique patient identifiers. Using a hash key (asymmetric encryption) based on name and birth date may provide a partial solution compatible with legal requirements. Transition from pediatric to adult diabetes care is an important step in the life of young patients with diabetes, and patient-centered research should allow to combine information from the pediatric and the adult treatment centers. For example, a recent study from Austria and Germany reported that patients with prepubertal diabetes onset who achieved better metabolic control during the early years of diabetes achieved better control during adulthood as well, despite the metabolic and psychosocial disturbances usually present during puberty. 29 This “tracking” of metabolic control across puberty is important for an integrated lifetime view on a chronic disease like type 1 diabetes.
Multinational Comparisons of Multicenter/Population-Based Registries
Both Hvidore 30 and SWEET, 31 as well as other similar initiatives, focus on large, well-known centers for pediatric diabetology in different countries. However, patients attending these centers, as well as the care provided, are most likely not representative for all patients from the respective country. Comparison among longitudinal regional/national registries documenting the process and outcome of care would therefore provide additional information on the current state of pediatric diabetology, and point to strengths and weaknesses on a national level.
Since the year 2000, the Austrian and the German working groups for pediatric diabetology have joined efforts and use the same documentation system, as well as benchmarking reports. This has been a major step for the 2 German-speaking countries, however it required continuous awareness of the sensibilities as well as the characteristics of both groups of pediatric diabetologists. There are clear differences, for example in the relationship between academic and nonacademic institutions, and the availability of pediatric diabetes specialists in private practice, between Austria and Germany. Meanwhile most pediatric diabetes centers in Austria, including all university institutions, participate in this joint effort.
Benchmarking reports are provided for all centers from both countries together, as well as for German and for Austrian centers separately. Despite some variability, there are profound similarities between both countries, as illustrated in the following graph comparing pump use in pediatric patients since the year 1995 (Figure 4).
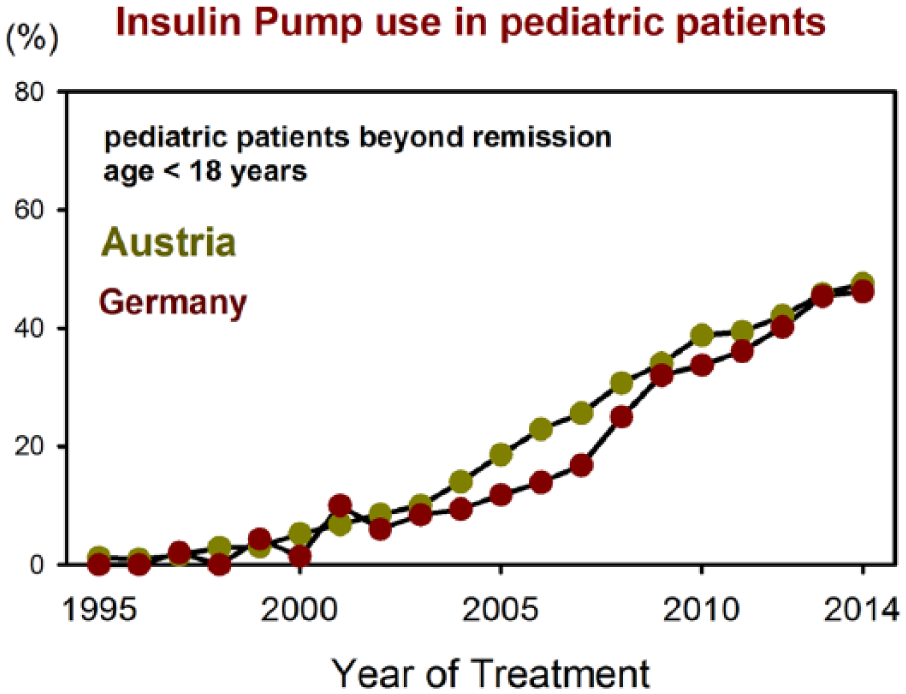
Comparison of pump use in pediatric patients during the past 20 years in Austria and Germany.
Germany and Austria are not the only countries implementing population-based national comparisons. Multinational comparisons, based on high level diabetes registries, were started in 2008 for the Scandinavian countries, including Denmark, Iceland, Norway, and Sweden. 32 Records from 6523 patients younger than 15 years of age from 89 centers were available, and differences in metabolic control, rate of severe hypoglycemia and pump use were present. Such comparisons allow to identify strengths and weaknesses of diabetes care in different countries, and thereby offer an opportunity to openly discuss und understand such differences, potentially leading to change in care and improved outcome.
Furthermore comparison with data from the T1DExchange in the United States and the National Diabetes Audit (NPDA) and Australasian Diabetes Data Network (ADDN) as performed recently show huge potential to compare care and outcome in young patients with diabetes is different parts of the world.33-36
While comparisons so far focus on countries with developed health care systems with universal health care coverage and easy access to the system for all children, the mission of ISPAD as an international society is to improve the outcome of care for every child with diabetes. Some centers from developing countries are currently starting to implement a standardized documentation. It is a major challenge to develop items suitable for these environments, and to implement benchmarking formats that stimulate the improvement among such centers, rather than leading to disappointment due to inferior outcomes.
These thoughts and examples on standardized documentation in pediatric diabetology together with regional, national and international benchmarking and comparison of quality indicators of pediatric diabetes care should motivate care teams from other regions of the world to implement similar initiatives, and build on the experience already available. The authors of this article are happy to support any initiative that tries to implement similar or superior registries.
Footnotes
Acknowledgements
The authors acknowledge the long-standing collaboration with all participating treatment centers in Germany, Austria, Switzerland, and Luxemburg, as well as statistical and data managing support by E Bollow, K Fink, J Hermann, A Hungele, and R Ranz. We enthusiastically thank Prof Edith Schober from Vienna for initiating the Austrian-German collaboration on quality improvement in pediatric diabetes.
Abbreviations
BMBF, German Ministry of Education and Research; BMI, body mass index; CNDM, German Competence Network Diabetes Mellitus; COR, center of reference; DDG, German Diabetes Association; DKA, diabetic ketoacidosis; DPV, diabetes patient follow-up registry; DZD, German Center for Diabetes Research; EFSD, European Foundation for the Study of Diabetes; HbA1c, glycated hemoglobin A1c; IDF, International Diabetes Federation; ISPAD, International Society of Pediatric and Adolescent Diabetes; IT, information technology; NPDA, National Pediatric Diabetes Audit; QC, quality control; SMBG, self-measurement of blood glucose; SWEET, Pediatric Diabetes: Working to Create Centers of Reference in Europe; T1DX, Type 1 Diabetes Exchange Registry; UK NHS, UK National Health Service; WHO, World Health Organization.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The DPV initiative is funded by the German Ministry of Education and Research (BMBF) as part of the German Competence Network Diabetes Mellitus (CNDM), which has been integrated into the German Center for Diabetes Research (DZD) as of January 2015. Additional funding was available from the European Foundation for the Study of Diabetes (EFSD) and the German Diabetes Association (DDG).
