Abstract
Background
The complexity of gastrointestinal (GI) disorders associated with Parkinson's disease (PD) and the significant interactions between GI medications and the dopaminergic axis necessitates expert management. The integrated care model for disorders of the brain-gut interaction (DBGI) has advantages, however, has not been applied in concurrent DBGI and PD.
Objective
To test the hypothesis that our Parkinson's Research and Integrated Support Model (PRISM) will reduce symptom severity and improve the quality of life (QOL) in patients with GI symptoms associated with PD.
Methods
Patients with refractory GI symptoms referred to the PRISM clinic were evaluated and treated by the integrated efforts of movement disorder specialists, neurogastroenterologists, dietitians, occupational therapists, speech-swallow therapists, and neuroscientists. Patients underwent a battery of GI symptoms and QOL questionnaires and personalized actionable biomarkers (motility testing and swallowing studies). Inflammatory markers and stool tests were collected. An individualized standard of care treatment was established based on the specific DBGI diagnosis uncovered during the PRISM evaluation.
Results
44 adult PD patients with GI complaints were evaluated. The most common symptoms included constipation (97%), dysphagia (61%), and gastroesophageal reflux (34%). Actionable biomarkers were highly positive revealing esophageal dysmotility (20/21, 95%), slow-transit constipation (40/42, 90%), intestinal methanogen overgrowth (7/8, 87%), gastroparesis (17/20, 85%), oropharyngeal dysphagia (28/44, 63%), and dyssynergic defecation (27/42, 61%). GI symptom severity and QOL significantly improved (p < 0.05) as measured by all questionnaires.
Conclusions
More severely affected patients with Parkinson's treated with the Fixel PRISM approach showed significant improvements in GI symptom frequency, severity, and QOL.
Plain language summary
Many persons with Parkinson's disease experience severe gastrointestinal symptoms including constipation, difficulty swallowing, and bloating. These symptoms impact the quality of life for persons living with Parkinson's disease, however, are often overlooked and may require specialized care. Recognizing the need for more comprehensive gastrointestinal care for persons with Parkinson's disease, clinicians at the Norman Fixel Institute for Neurologic Diseases developed the Parkinson's Research and Integrated Support Model (PRISM). PRISM is novel clinic which uses an interdisciplinary team of movement disorder experts, neurogastroenterologists, dietitians, occupational therapists, speech-swallow therapists, and neuroscientists to address complex gastrointestinal symptoms associated with Parkinson's disease. The clinic is designed to be person-centric and to occur in a single visit. In this study, 44 persons with Parkinson's disease who were resistant to treatment of their GI dysfunction by a movement disorders neurologist were treated in the PRISM clinic between August 2021 and March 2024. This cohort experienced significant improvements in gastrointestinal symptoms including constipation, as well as overall quality of life, highlighting the importance of interdisciplinary gastrointestinal care for persons with Parkinson's disease.
Introduction
Parkinson's disease (PD) is a chronic neurodegenerative condition characterized by the loss of nigrostriatal dopaminergic innervation as well as degeneration in multiple motor and non-motor basal ganglia circuits. PD manifests with the development of classic motor symptoms including bradykinesia, rigidity, resting tremors, postural instability, and dystonia, though the patterns of symptoms are frequently variable between persons and may change with disease progression. Nonmotor symptoms including psychiatric disturbances, sleep disorders, mood disorders, and gastrointestinal (GI) symptoms are commonly present across all stages of PD and can significantly impact quality of life (QOL). 1 The entire GI system has been shown to be potentially affected in PD, and common manifestations include constipation, dysphagia, and gastroparesis. 2
Constipation has been described as the most frequent autonomic symptom of PD, occurring in up to 80%. 2 Constipation is a clinical diagnosis, but its symptoms do not always correlate with the underlying type—whether slow transit or defecatory disorder/dyssynergia—which is crucial for selecting the appropriate treatment, such as medications or pelvic floor physical therapy. The type of constipation can be confirmed by kidney, ureter, bladder abdominal X-ray (KUB), external anal sphincter electromyography, the balloon distension/expulsion tests, and anorectal manometry (ARM). 3 ARM and KUB are useful to triage rectal evacuation disorders and slow transit constipation.4,5 Dysphagia and gastroparesis are other common GI symptoms which may manifest in advanced PD, frequently heralding disease progression and potential future complications.6,7 Gastroparesis, in particular, can disrupt medication absorption, leading to erratic drug levels, which may increase the daily duration of OFF states or worsen the severity of OFF episodes in patients experiencing motor fluctuations.8,9
In recent years, there has been a shift toward providing persons with integrated care, where providers work together as a unified team on all aspects of treatment. By leveraging the expertise of providers from multiple health disciplines, integrated care models aim to provide more holistic and personalized care which can be tailored to the unique needs of a patients. 10 Integrated care has been utilized in a variety of medical specialties including oncology, surgery, primary care, internal medicine, and psychiatry, and it is associated with fewer hospital readmission rates, shorter hospital stays, improved overall survival, and improved patient outcomes.11–21 However, there has been a slow shift in recent years among experts toward integrated care for patients with severe GI symptomatology. This approach differs from multidisciplinary care, which involves a team of healthcare professionals from various specialties working independently to treat a patient, each focusing on their specific area of expertise. Integrated care, in contrast, is a more holistic approach where professionals coordinate efforts, share information, and create a unified treatment plan that addresses overall health needs. 22
The complexity and severity of GI dysfunction in PD frequently requires expert clinical management. However, there is a paucity of information on the outcomes of integrated care for patients with PD patients and GI symptoms. The Norman Fixel Institute for Neurological Diseases operates in a service and science hub model, 23 and the PRISM system was layered into the existing infrastructure to support interdisciplinary care. 23 The goal of development and deployment of the PRISM model was to establish an integrated clinic to comprehensively evaluate and to treat GI symptoms with an overarching goal of impacting QOL (Figure 1). The research aspect of the PRISM approach was focused on the use of data for quality improvement projects as well as the collection of microbiome data for future correlations. We hypothesized that our integrated holistic approach based on global GI inventories for unresponsive symptoms would improve GI symptom severity and overall QOL. We sought in this project to detail and to share critical aspects of the model including clinic development, clinic flow, impact on GI symptom severity, frequency of symptoms, and QOL. Finally, we endeavored to record and share the challenges associated with establishing a PRISM clinic, including future research directions.
Overview of how integrated care is achieved with the PRISM approach. Figure created with BioRender.
Methods
Clinic design
The PRISM Clinic was established in August 2021 at the Norman Fixel Institute for Neurologic Diseases at the University of Florida in Gainesville to care for people with PD presenting with severe or difficult to treat GI symptoms. Movement disorder physicians carefully screened patients and generated a referral to the PRISM Clinic when conservative measures performed solely by the movement disorder specialist including dietary modifications, hydration, exercise, and a trial of one or two first-line pharmacologic agents were insufficient or resulted in persistent or worsening GI symptoms. At each PRISM visit, patients were evaluated and treated by an integrated team consisting of movement disorder experts, neurogastroenterologists, dietitians, occupational therapists, speech-swallow therapists, and neuroscientists who each collected data for clinical and potential future research use (Table 1). Unlike traditional multidisciplinary care, the PRISM approach ensured that all team members actively collaborated during the same PRISM clinical session to formulate an organized, uniform, individualized, and standardized treatment plan. Meetings with the entire interdisciplinary team were conducted after each PRISM clinic to discuss patients assessed as well as upcoming patients for the next clinic. Additionally, monthly meetings with the entire team are conducted to resolve outstanding issues or clinical concerns. An overview of the PRISM clinic flow is presented in Figure 2. Six to eight patients were scheduled monthly using this integrated approach. A pragmatic point of care evaluation with KUB was done during the index visit to calculate the Leech score (LS)
24
and the rectal gas transaxial area (RGTA)
5
to screen for slow transit constipation and defecatory disorders respectively
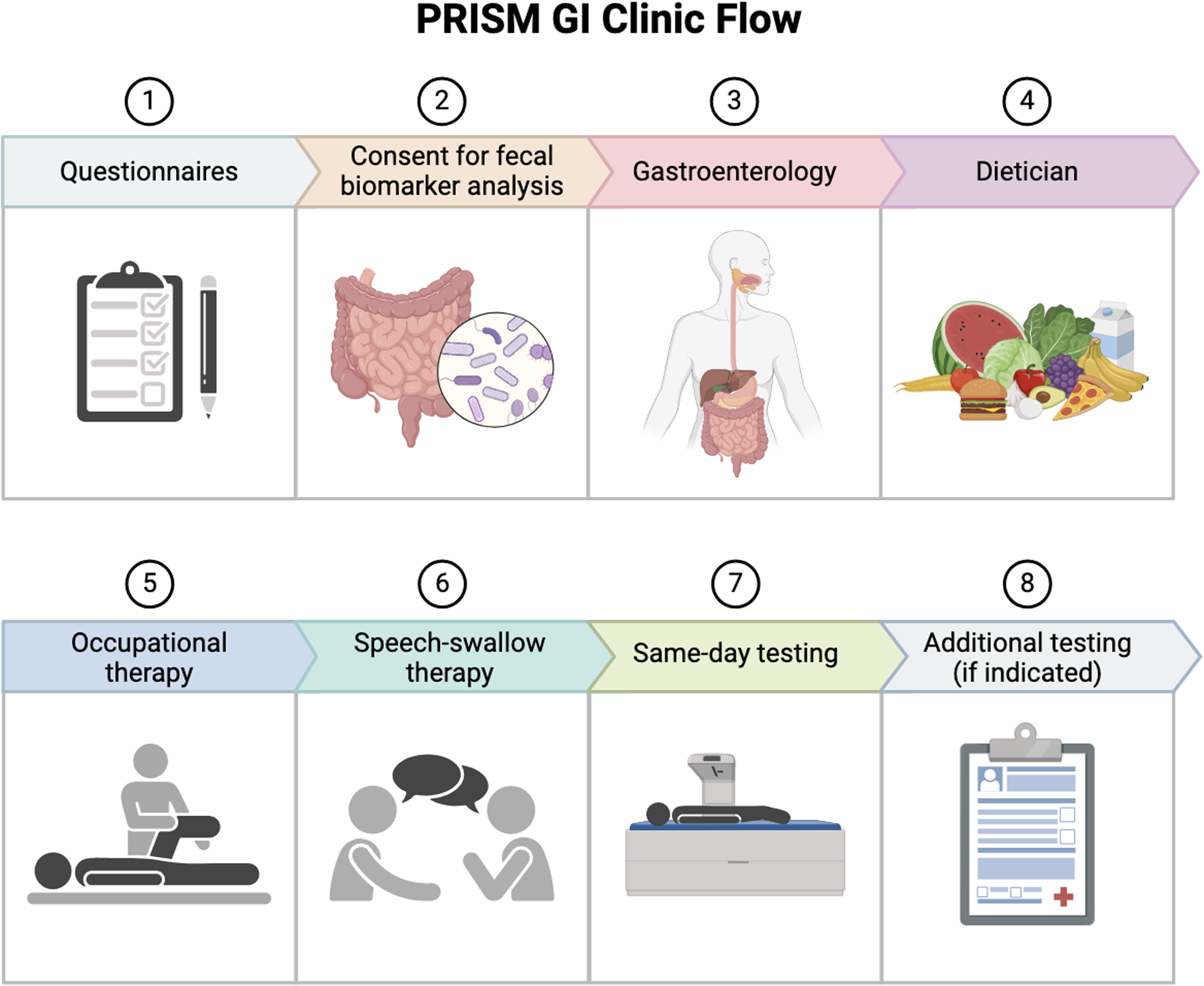
PRISM clinic flow.
Sub-specialty providers and their clinical roles.

Clinical indications for GI testing.

Patients with PD were administered standard questionnaires at the start of each visit with guidance provided by a clinical research assistant. Care-partner assistance was used to answer questions only when needed. The standardized questionnaires included the Digestion-associated Quality of Life Questionnaire (DQLQ), 35 the Gastrointestinal Symptoms Rating Scale (GSRS), 36 and the novel in-house questionnaire, the Gainesville Gastrointestinal Symptom Inventory (GGSI). The GGSI was created to gauge GI symptom severity and the impact these symptoms have on QOL and the scale was constructed using Rome IV definitions. 37 The GGSI was composed of 7 arms to assess (1) dysphagia, (2) reflux symptoms, (3) dyspepsia/upset stomach, (4) constipation, (5) abdominal pain, (6) diarrhea, and (7) other miscellaneous information (Supplemental Figure 1). GI severity questions were answered on a scale of 0–3 points where never = 0 points, 1 day = 1 point, 2–3 days = 2 points, and 4 days or more = 3 points. QOL effect was evaluated by the following scale: 1 = never or not bothersome, 2 = Mild. not affecting daily activities/meals, 3 = Moderate. Limits some meals/daily activities, 4 = Severe. Limits most meals/daily activities. To minimize recall bias, only symptoms experienced 7 days prior to GGSI administration were considered. The total GGSI score was calculated by adding the total number of points for sections 1–6. A higher GGSI score reflected more severe symptoms and worse overall QOL. Patient responses were collected and stored in REDCap (Research Electronic Data Capture) and on the EPIC Electronic Medical Records.
Patients were additionally sent home with stool collection kits for fecal biomarker analysis to assess microbiota, inflammatory markers, permeability markers, and short-chain fatty acids. Fecal biomarker analysis data was the only test ordered solely for research purposes, and patients who declined to participate were still able to experience integrated care in our clinic. The MyChart patient portal powered by Epic (TM Epic Systems Corporation) was used in the short-term to follow-up with biomarker results and to develop a plan. Standard of care treatment was administered based on diagnosis, and follow-up appointments were scheduled based on acuity and response to treatment. Clinical research assistants contacted patients before and after their scheduled PRISM visit to answer questions and inform patients of care expectations.
Present study
A retrospective IRB approved (IRB202201172) chart review was conducted on patients evaluated in the PRISM clinic between August 2021 and March 2024. Patients were included in the cohort if they met the following criteria: (a) over the age of 18, (b) previous diagnosis of PD, (c) presented to our clinic with GI dysfunction, and (d) completed the GSRS, DQLQ, and the GGSI. Patients with atypical parkinsonism or any other movement disorder were excluded from the analysis. Demographic information, prevalence of GI symptoms, initial visit questionnaire scores, return visit questionnaire scores, and total number of specialized GI tests were collected from individual medical records. Key outcome variables for the analysis included the difference in total GI symptoms scores as measured by the GGSI and total QOL score as measured by the GGSI. Secondary outcome variables included the difference in initial and return visit GGSI individual arm scores, the DQLQ scores, and the GSRS scores.
Results were reported as means (SD), frequencies, and percentages unless otherwise stated. Paired t-tests were used for all comparisons. Normality was established with a Shapiro-Wilk test. For data that was not normally distributed, (GGSI abdominal pain scores, GGSI diarrhea scores, and all GSRS sub-scores) a Wilcoxon Signed Rank Test was utilized, and data was reported as median (IQR). All statistical analyses were conducted in SigmaPlot 15.
Results
There were 50 adult patients with PD and severe GI symptoms reviewed, and 44 patients were included in the analysis. Six patients were excluded because their GSRS, DQLQ, and/or GGSI records were incomplete. The mean age was 70 (±8.78) years, and 22 were female. The average PD duration was 6.8 years and the mean Hoehn and Yahr scale score was 2.45 (±0.91). Most patients (n = 31, 70.4%) did not receive deep brain stimulation (DBS)
Ancillary test results.
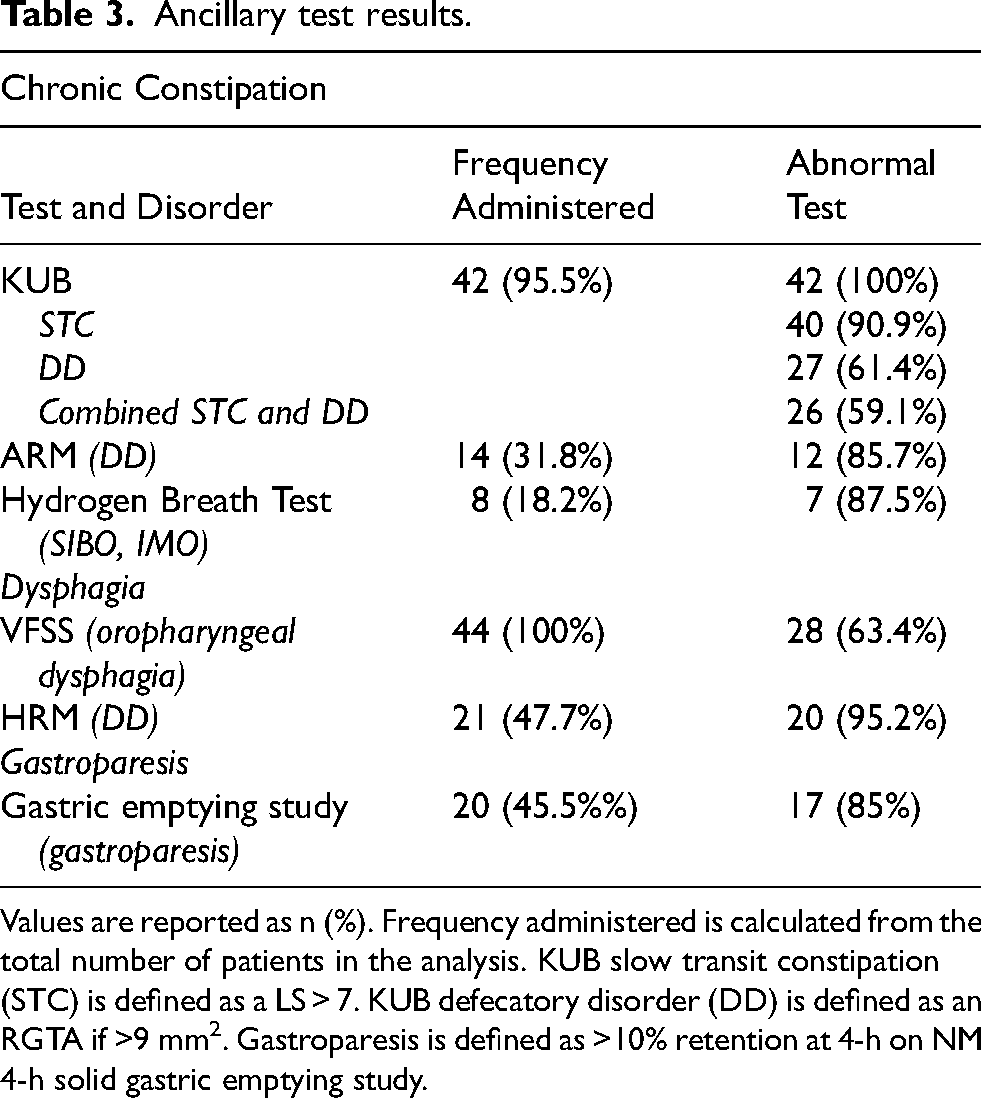
Values are reported as n (%). Frequency administered is calculated from the total number of patients in the analysis. KUB slow transit constipation (STC) is defined as a LS > 7. KUB defecatory disorder (DD) is defined as an RGTA if >9 mm2. Gastroparesis is defined as >10% retention at 4-h on NM 4-h solid gastric emptying study.
At the initial visit, the median number of hard stools per week was 2, median number of skipped bowel movements (BM) per week was 2, and median number of incomplete BMs per week was 2. At the return visit, the median number of hard stools per week decreased to 0.5, the median number of skipped BMs per week to 1, and the median number of incomplete BMs per week to 1.5. For the 15 patients who had a follow-up visit, the GGSI GI symptoms score (p = 0.006), the GGSI QOL score (p = 0.002), the GGSI dysphagia arm score (p = 0.049), and the GGSI constipation arm score (p < 0.001) all significantly decreased at the return visit (Table 4, Figure 3). The DQLQ (p = 0.021) and GSRS scores (p = 0.016) similarly decreased significantly at the return visit (Table 5).
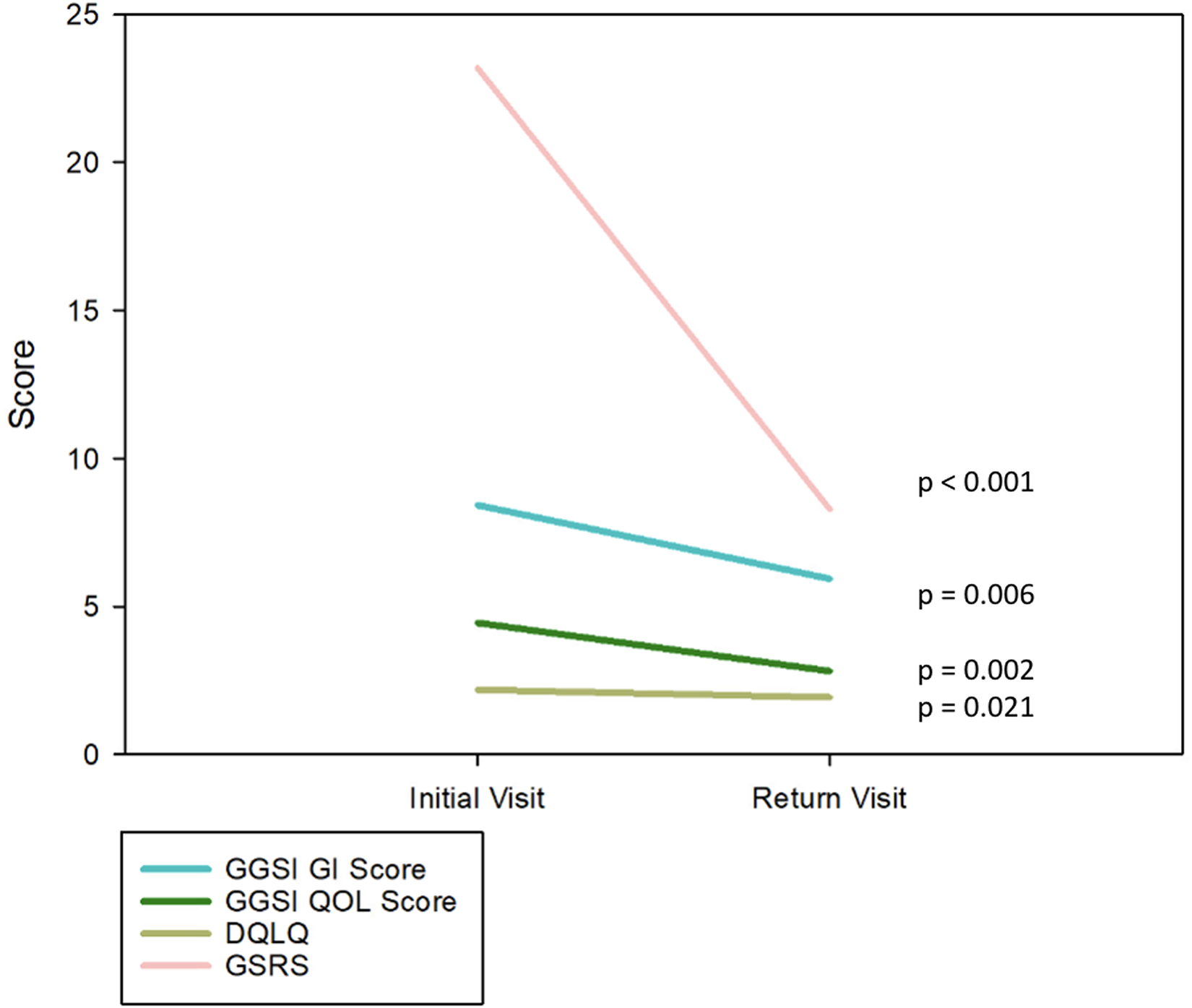
Line graph of initial and return visit questionnaire scores.
Initial and return GGSI scores.
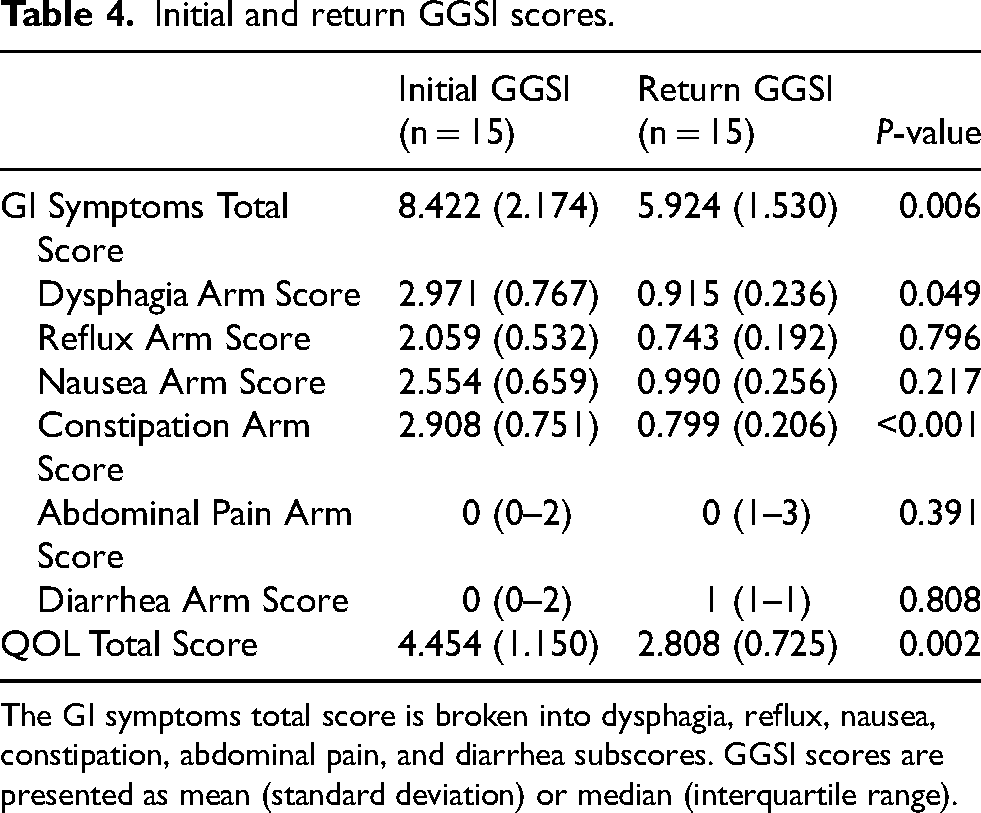
The GI symptoms total score is broken into dysphagia, reflux, nausea, constipation, abdominal pain, and diarrhea subscores. GGSI scores are presented as mean (standard deviation) or median (interquartile range).
Initial and return DQLQ and GSRS scores.
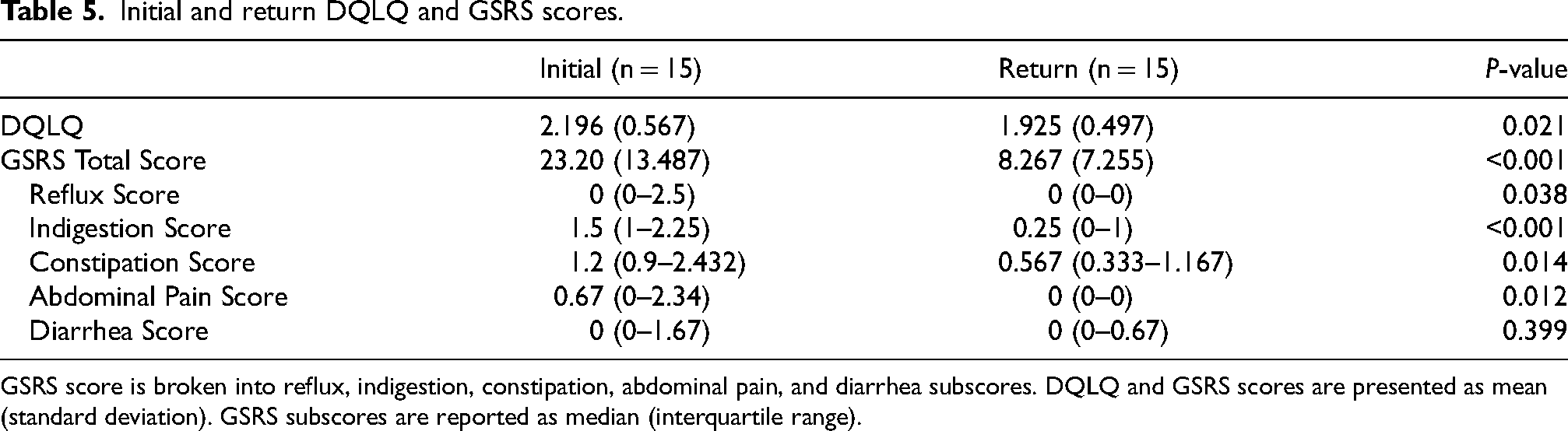
GSRS score is broken into reflux, indigestion, constipation, abdominal pain, and diarrhea subscores. DQLQ and GSRS scores are presented as mean (standard deviation). GSRS subscores are reported as median (interquartile range).
Discussion
The complexity and severity of GI disorders associated with PD, especially constipation, significantly affects QOL and in severe cases require expert clinical management. 1 We have developed an integrated clinic that combines specialized expertise, innovative application of GI diagnostic tools and tests, as well as patient-centered care in a deliberate effort to address the complex needs of those with severe GI symptoms. Our data demonstrated that patients with severe GI symptoms treated with the PRISM approach had significant improvements on GI symptom frequency, severity, and overall QOL which was confirmed by using multiple questionnaires.
The most common symptoms presented in our cohort included constipation (97%), dysphagia (61%), and GERD (34%). The most common actionable biomarkers applied included VFSS (100%), KUB (96%), and esophageal HRM (47%). The pragmatic point of care approach of the index visit (standard questionnaires, KUB, VFSS) and the effectiveness of the personalized PRISM approach highlights its potential for replication in other healthcare settings to benefit a broader population of PD patients.
The findings of this study suggest patients treated in our clinic experience significantly worse symptoms and GI dysfunction than the general population of patients with PD. 38 The KUB performed in 95% of patients showed abnormalities in 100% of cases. 87% of patients tested in our clinic demonstrated delayed gastric emptying on NM 4-h solid GES. While some studies have reported delayed gastric emptying in up to 50% of PD patients,39,40 others have found no significant differences compared to age-matched controls.41–43 Interestingly, the cardinal symptoms of gastroparesis including nausea and vomiting were not as prevalent in this cohort. This may reflect the heterogenous nature of PD, individual compensatory mechanisms to reduce unwanted GI symptoms, medication interactions, or severity of other common, more bothersome GI symptoms such as severe constipation. However, the diagnosis of gastroparesis is critical because the efficacy of PD medications can be affected by gastric emptying times. 42 VFSS detected oropharyngeal dysfunction in 63% of tested patients, exceeding prior estimates of 35–55% in PD.44–46 ARM findings were abnormal in 85% of tested patients, suggesting that defecatory disorders are common in patients with severe constipation, with prevalence exceeding literature estimates by up to 19%.47–50 Lastly, HBT results were abnormal in 87% of patients, exceeding previously reported small intestinal bacterial and methanogen overgrowth prevalence rates in PD, ranging from 25.3% to 70%.51–53 It is likely that the prevalence of these symptoms is greater in our cohort compared to the general population of PD because the PRISM clinic is designed for PD patients with severe GI dysfunction failing first line therapies. These findings reinforce the importance of comprehensive GI evaluations in PD patients, as motility disorders, defecatory disorders, and bacterial/methanogen overgrowth appear to be more widespread than previously reported. Furthermore, the results of this analysis highlight how GI symptoms rarely present alone but rather, as complex upper and lower syndromes. Many patients in this cohort presented with a constellation of symptoms that include constipation, dysphagia, and bloating. This finding suggests that the entire GI system is affected in PD rather than isolated organs and clinicians should consider addressing GI symptoms in a systemic, holistic manner.
Our paper represents one of the early attempts at implementing an integrated care model specifically tailored to PD patients with severe refractory GI symptoms. The PRISM Clinic model was meticulously designed following the guidelines outlined by Radder et al. 54 to minimize delays in receiving specialized PD care, while also improving patient access and leveraging the Fixel Service and Science hub model. 23 The streamlined referral process, guided by movement disorders specialists, prioritizes individuals with the most pressing needs, and thus minimizes delays in receiving crucial treatments. Proper evaluation and testing facilitated an enhanced understanding of the multifactorial nature of GI dysfunction in PD with the aim of implementing a combination of dietary, pharmacological, and psychological therapies. Constant communication between patients and clinical research staff before and after PRISM clinic visits facilitated a supportive and collaborative relationship between patients and providers, which may have reduced appointment cancellations and improved compliance to treatment regimens. Furthermore, monthly meetings involving the entire integrative team ensured regular updates on patient progress, refinement of treatment strategies, and interdisciplinary feedback to align care plans across specialties. The integration of multiple same-day appointments and diagnostic procedures additionally optimized efficiency while minimizing inconvenience. This pathway facilitated a timely diagnosis and intervention strategy and enabled more effective management.
The novel GGSI questionnaire we developed has yet to be validated, however we chose the items to be more PD specific and to provide information to aid clinicians in decision making.35,55 The GGSI was unique compared to the other questionnaires as it combined evaluation of GI symptom severity and QOL metrics into one objective questionnaire. This combined approach minimized the need to administer multiple questionnaires and thus may have the potential to reduce fatigue. The GGSI additionally presented clinical questions in simple language to enable comprehension and accurate documentation of symptoms. Future research aims to validate the GGSI to improve reliability and sensitivity in assessing GI symptoms and QOL in PD patients. Validation studies will also provide insight in GGSI interpretation, including what scores are correlated with mild, moderate, and severe symptoms in PD patients.
In our experience, the creation of a novel, integrated specialty clinic presented clinicians and researchers with unique challenges. The process of scheduling had to accommodate the patient with the disease and provider availability, minimize wait times, and effectively manage unexpected changes or cancellations. 56 Depending on availability, it may not in every case be possible to schedule the entire team or anticipate the procedures which will be required on a single visit. Patients traveling from distant locations, those working, those with other health problems or family obligations had to be accommodated. The future use of telemedicine and remote assessment may be a potential solution to some of the logistical challenges.
The optimal frequency of PRISM clinic dates per month to maximize access to care while efficiently utilizing clinic resources will need to be better defined. Holding one clinic day a month led to increased waiting times, which created barriers to access. These barriers were mitigated by encouraging a direct channel of communication with clinical coordinators and providers by remote access using the MyChart patient portal powered by Epic (TM Epic Systems Corporation).
Collecting fecal samples for fecal biomarker analysis created unique challenges for both clinical and research aims. Obtaining consent for fecal biomarker analysis and providing stool collection kits was initiated in August 2023. Fecal collection kits were mailed or provided to patients in the PRISM clinic. While no conclusions can be drawn at this stage regarding this ancillary investigation, it has been difficult to obtain a sufficient sample size due to issues with constipation and other confounding factors. Stool samples can prove difficult to collect, patients frequently forget to collect their stool, and some perceive that handling stool may be unhygienic and embarrassing. 57 Additionally, patients with severe constipation may not produce enough stool for collection or may be in pain while producing stool and thus disregard collection. These challenges, combined with the lack of time to collect stools, led to delays in obtaining fecal biomarker data to share with clinicians and to use for research. To address these challenges, we use convenient, at-home microbiome kits for collections and plan to educate patients on the timing of collection to ease these issues. We will continue to collaborate with our multidisciplinary team (e.g., nurses, dietitians, and physiotherapists) to manage constipation more effectively and facilitate stool collection. Furthermore, we are hopeful that in 2025, with increased staffing, we will be able to collect more samples to run microbiota and biomarker analyses. Future studies will aim to evaluate the results of our fecal biomarker analyses as well as strategies to improve recruitment rates to better understand the efficacy of our service and science hub model.
Despite these challenges, we found that the integrative approach, guided by standardized protocols, facilitated a more comprehensive and structured evaluation for our patients. This likely improved diagnostic accuracy and treatment consistency, making the process more efficient and accessible for patients. Moreover, we believe that the expertise of individual providers in PD-related GI issues—when applied within a shared protocol—resulted in better outcomes compared to independent assessments by non-specialists. The integrated nature of the clinic allowed for a holistic approach, incorporating nutritional, psychological, and rehabilitative support which not only enhanced diagnostic precision, but also contributed to overall improvements in patient QOL.
While it is possible that the improvements in our patients’ symptoms may have been due to the utilization of medical interventions available outside of an integrated care model, this clinic's success was likely facilitated by the combination of unique resources including funding to create integrative centers, neurogastroenterology testing availability, clinical expertise with PD, and the use of clinical coordinators. Smaller and non-university based medical centers with less access to funding and to specialists may not be able to recapitulate this model. Neuro-gastroenterologists in particularly are especially scarce worldwide, representing a minority of all GI providers.58,59 Future studies should evaluate long term complications, morbidity and mortality rates, and hospitalization records of PRISM patients. We should thus examine associated healthcare costs before recommending this model to a larger number of centers.
A limited number of PD patients with refractory GI symptoms were referred to our clinic, inducing a selection bias and effectively limiting the sample size of this analysis. The small sample size in this analysis reflects the early stage of our clinic's development rather than the potential patient population, as our clinic is unique in focusing on GI symptoms in PD. However, this relatively small number of patients facilitated an opportunity to refine our clinic flow, optimize diagnostic tools, and to evaluate longitudinal preliminary patient outcomes. Given the results of this pilot study, we plan to expand our clinic to a larger number of patients.
Furthermore, interview-style questionnaires can introduce bias, which may affect the accuracy and reliability of results. 60 Interview-style questionnaires, while valuable for data collection, are prone to several biases that can impact the reliability and validity of the results including interviewer bias (the interviewer's behavior, tone, or phrasing can influence the respondent's answers), response bias (respondents may provide answers they think are socially desirable or acceptable rather than their true feelings), recall bias (respondents may have difficulty accurately remembering past events or experiences), sampling bias (if the sample is not representative of the larger population, the results may be skewed), confirmation bias (interviewers may unintentionally seek out information that confirms preconceived notions), and cultural bias (cultural differences can affect the interpretation and responses to questions). Addressing these biases through careful design and implementation of interview-style questionnaires will be essential.
One key area for future research will be to validate the GGSI constructed for this clinic as the field needs more diagnostic and screening instruments validated in PD populations. Because levodopa has the potential to affect gastrointestinal motility and growing evidence suggests that GI health influences drug pharmacokinetics, it would be valuable to investigate whether improvements in quality of life observed in patients treated with this interdisciplinary approach are associated with enhanced dopa-responsiveness, potentially mediated by changes in levodopa equivalent daily dose or absorption. Additionally, the effect of DBS on GI symptoms in PD is under-investigated. Current studies lack rigorous design, show weak retrospective correlations, and do not assess GI motility or systematically evaluate the effects of medications and DBS settings. To bridge this gap in the literature, a thorough assessment of postoperative GI symptomatology through analysis of GI symptom questionnaire and biomarkers following DBS therapy will be necessary.
Conclusion
Patients with severe GI symptoms and PD cared for using the PRISM approach experienced positive impacts on the overall severity of gastrointestinal symptoms and QOL. The model is a promising approach to enhance care and outcomes in this population, though scalability, cost and long-term benefits in reducing GI related hospitalizations will require further study.
Supplemental Material
sj-pdf-1-pkn-10.1177_1877718X251335047 - Supplemental material for The fixel GI Parkinson's research and integrated support model (PRISM)
Supplemental material, sj-pdf-1-pkn-10.1177_1877718X251335047 for The fixel GI Parkinson's research and integrated support model (PRISM) by Grace Hey, Manuel Amaris, Matthew Beke, Nur Walker-Pizarro, Candice Rogers, Vinata Vedam-Mai, Rachael Dorsey, Nicole Herndon, Michael S Okun and Adolfo Ramirez-Zamora in Journal of Parkinson's Disease
Footnotes
Acknowledgments
The authors have no acknowledgments to report.
ORCID iDs
Ethical considerations
IRB approval was obtained to perform the research outlined in this manuscript (IRB202201172). We confirm that we have read the Journal's position on issues involved in ethical publication and affirm that this work is consistent with those guidelines.
Consent to participate
Informed consent was obtained from all research participants.
Consent for publication
Not applicable
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr Ramirez-Zamora has received research support from the Parkinson's Foundation and consulting honoraria from Medtronic Inc, Boston Scientific, Cerevel therapeutics, Blue Rock Therapeutic, Iota Inc, and Signant Health in the past 12 months. Dr. Okun serves as Medical Advisor the Parkinson's Foundation, and has received research grants from NIH, Parkinson's Foundation, the Michael J. Fox Foundation, the Parkinson Alliance, Smallwood Foundation, the Bachmann-Strauss Foundation, the Tourette Syndrome Association, and the UF Foundation. Dr. Okun's research is supported by: R01 NS131342 NIH R01 NR014852, R01NS096008, UH3NS119844, U01NS119562. Dr. Okun has received royalties for publications with Hachette Book Group, Demos, Manson, Amazon, Smashwords, Books4Patients, Perseus, Robert Rose, Oxford and Cambridge (movement disorders books). The institution and not Dr. Okun receives grants from industry. All other authors have no additional financial disclosures to report.
Data availability
Our data cannot be shared because it contains Protected Health Information which we are not permitted to disclose outside of individuals who are listed on the approved research protocol.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
