Abstract
There is increasing evidence that microbial-based therapies can be useful in people with Parkinson’s disease (PD). In this viewpoint, we provide a state-of-the-art review of the clinical and pre-clinical evidence for probiotics and prebiotics in PD. Currently, short-term clinical studies, including double-blind placebo-controlled randomized clinical trials, have demonstrated safety, and efficacy primarily in improving constipation-related symptoms. Pre-clinical studies consistently reported improvements in a range of biological markers and outcomes, including evidence for attenuation of gut dysfunction and neuroprotection. Bacteria from the genus
INTRODUCTION
The role of the microbiome-gut-brain axis has garnered significant attention in Parkinson’s disease (PD), in which gastrointestinal dysfunction is a prominent feature.1–5 A growing number of clinical studies (>50) in the field have found an alteration of gut microbiome and metabolome in patients with PD, although specific microbial changes are quite heterogenous across different research populations.1,6–9, 1,6–9 Meanwhile, several pre-clinical studies have demonstrated that gut microbes and their microbial products (e.g., lipopolysaccharide or amyloid curli protein) can promote gut hyperpermeability and inflammation, leading to systemic inflammation and neuroinflammation, as well as increased
Excitingly, various microbial-directed therapies such as dietary modification, supplementation with probiotics and/or prebiotics, as well as fecal microbiota transplantation, are rapidly emerging as promising treatment strategies for PD.1,2,15,16, 1,2,15,16 Among them, probiotics and prebiotics which have been widely used as health supplements, are propitious and popular candidates as they are deemed to be affordable, accessible, safe, and effective. 17 Indeed, beliefs regarding the health-promoting attributes of microbial-based products date far back into early civilization, when humans started consuming fermented foods. 18 It is not surprising that their potential benefits in PD have been one of the commonly asked questions by patients in the clinic. This is also due in part to the high frequency of gastrointestinal symptoms in this patient population, particularly constipation with a prevalence of up to 70%. 19 In this Viewpoint, we summarize the clinical and pre-clinical evidence for probiotics and prebiotics (and their combination) as potential symptomatic or disease-modifying treatments in PD. Additionally, we discuss current recommendations and safety considerations for their usage in PD patients and offer perspectives on future development of these non-invasive gut-modulating therapies.
WHAT ARE PROBIOTICS AND PREBIOTICS?
Probiotics have been defined by expert consensus as “live microorganisms, which when administered in adequate amounts, confer a health benefit on the host”. 20 Since Metchnikoff’s observation on the potential link between consumption of fermented milk and extreme longevity in the early 1900s to the recent advances in microbiome science, there has been an exponential growth in the multibillion dollar probiotics industry, with an estimated value ∼USD70 billion in 2023. 18 Unfortunately, the term “probiotics” has been frequently misused and exploited, with products sold as such without manufacturers providing evidence for their health claims. While various national food and safety regulatory bodies have established guidelines, the International Scientific Association for Probiotics and Prebiotics (ISAPP) recommends that the term probiotic be used only on products that contain well-defined probiotic strain(s) with proof of delivery of viable strain(s) at efficacious doses, and convincing evidence of health effects in human studies. 20 Meanwhile, fermented foods with undefined microbial content should be described as “containing live and active cultures”, but should not be called probiotics. 20 More recently, to regulate and distinguish the use of live microorganisms as medicinal drugs vs. nutritional supplements, the United States Food and Drug Administration (FDA) and the European Pharmacopeia have released guidelines for the development and testing of “live biotherapeutic products (LBPs)” in clinical trials, which require stringent standards of safety, reliability, robustness, and consistency of each produced batch of probiotic strain(s). 21
The concept of prebiotics is relatively newer, where in the 1990s, microbial-targeted substrates such as non-digestible dietary oligosaccharides were recognized for their abilities to promote beneficial change in host microbial composition and/or activities.
22
The definition of prebiotics was updated by the ISAPP in 2017, as “a substrate that is selectively utilized by host microorganisms conferring a health benefit”.
22
Selectivity to microbial fermentation is considered central to the prebiotics concept, and distinguishes prebiotic compounds such as fructooligosaccharides (FOS) and galactooligosaccharides (GOS) known to enrich
Probiotics and prebiotics act through diverse mechanisms, in part, mediated through modification of the microbiota and/or its function.15,17,24, 15,17,24 Their potential beneficial effects on system-wide metabolic and physiological functions include modulation of immune function, defense against pathogens, production of organic acids, and improvement of gut barrier function.15,17,24, 15,17,24 Notably, their benefits on human health may be tied to specific strains and specific diseases, e.g., a particular probiotic strain or mixture of strains may be effective for one disease but ineffective for other diseases.17,25, 17,25 Given the complex inter-individual variability in the gut microbiome, these microbial-based therapies may also require personalization based on host microbiome profiles. 26
EVIDENCE FOR PROBIOTICS IN PARKINSON’S DISEASE
Probiotics have been most extensively studied to assess their efficacy in managing gut-related symptoms. Several meta-analyses have demonstrated that certain probiotics (e.g.,
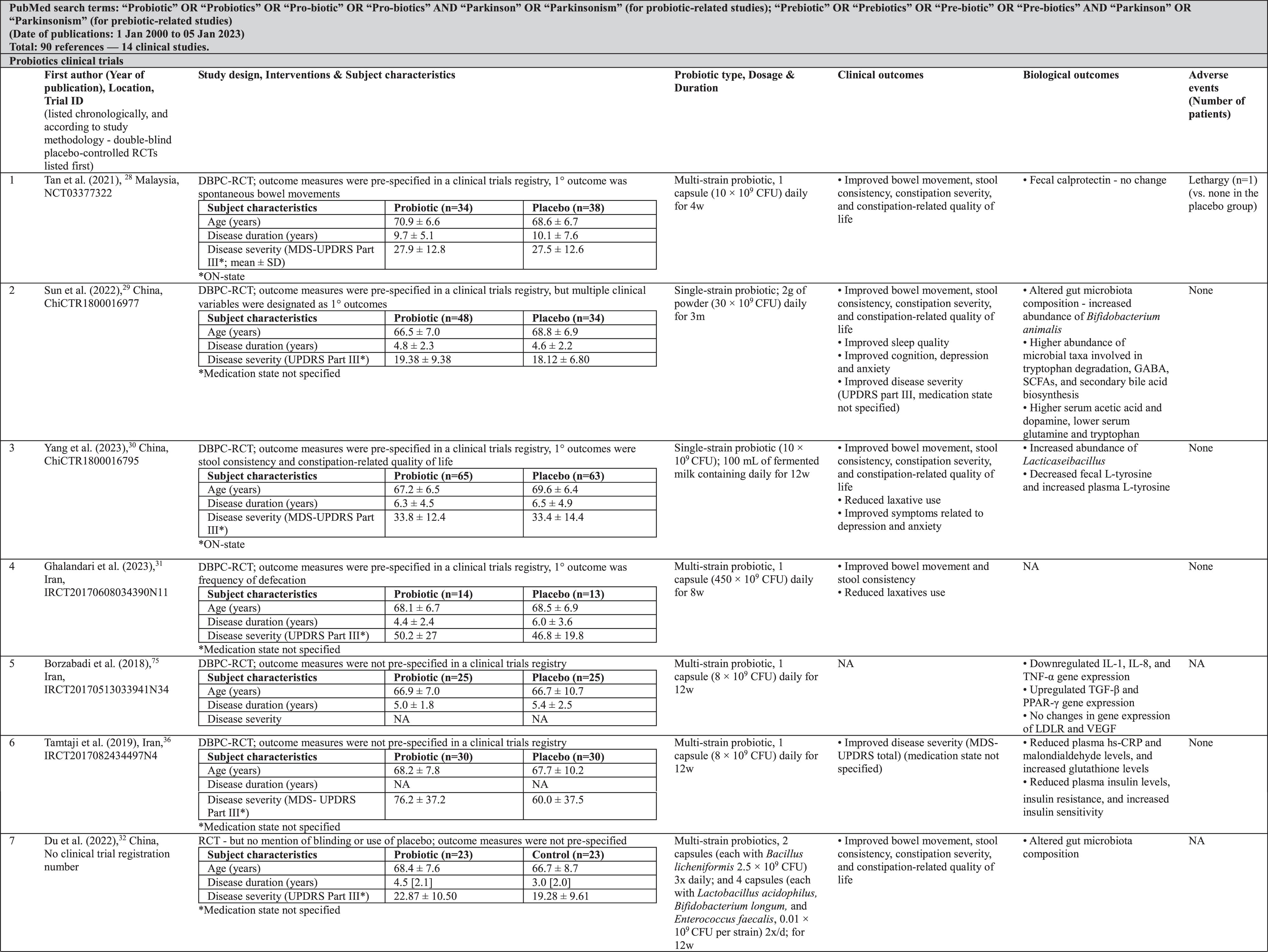
In PD, five double-blind placebo-controlled randomized clinical trials (RCTs), with sample sizes ranging from 27 to 128, showed that single-strain or multi-strain probiotics significantly improved bowel movement, stool consistency, constipation severity scores, constipation-related quality of life, and/or laxative usage, for up to 12 weeks (Table 1).28–32 Two other double-blind placebo-controlled RCTs (
Probiotics, Prebiotics, and Probiotics with Prebiotics Clinical Studies in Parkinson’s Disease
Probiotics clinical studies also noted improvements in PD severity,29,35,36, 29,35,36 as well as in the duration of the ON and OFF periods. 35 However, major limitations of these studies were the lack of specification whether motor evaluations were done in the medication-ON vs. medication-OFF state,29,36, 29,36 reporting of only total scores of the Movement Disorder Society-Unified Parkinson’s Disease Rating Scale (MDS-UPDRS) without separate reporting of subscores (parts I–IV) as has been recommended (raising concerns about selective reporting 36 ), 37 and open-label methodology with a high risk of bias. 35 Investigators also reported improved sleep quality, 29 cognition, 29 depression,29,30, 29,30 anxiety,29,30, 29,30 and PD-related quality of life. 35 Adverse events in these clinical trials appear to be very infrequent (Table 1).
The commonly used probiotics bacteria in these studies were from the genus
Commonly used probiotic species in probiotic clinical studies in Parkinson’s disease
Biological processes investigated in these trials included probiotics’ ability to alter host gut microbiota composition and metabolic capacity, modulate host metabolism, and reduce host inflammatory pathology and gut barrier dysfunction. Among the various biological markers assessed were short-chain fatty acids (SCFAs), dopamine, tryptophan, glutamine, insulin, calprotectin, interleukins, hs-CRP, and zonulin, measured in faeces or in biological fluids such as blood or urine (Table 1). While alterations in biological markers were often (although not always) observed with probiotics treatment, their biological and clinical significance, and the mechanisms underlying these effects, are currently uncertain.
In this regard, pre-clinical animal and cell models provide valuable opportunities to further dissect the mechanistic effects of probiotics, particularly those mechanisms that cannot be easily investigated in human subjects. We have previously discussed these in detail and provide an updated compilation of these studies in Supplementary Table 1.
15
This is evidently an active field of research with a relatively large number (∼40) of studies published. The vast majority were in murine models, and in line with human studies mostly commonly involved the administration of bacteria from the genus
EVIDENCE FOR PREBIOTICS IN PARKINSON’S DISEASE
In general, the evidence on prebiotics in PD has lagged behind that for probiotics. As discussed above, prebiotics administered together with probiotics have demonstrated efficacy in managing constipation-related symptoms in PD (Table 1).33,34, 33,34
There are thus far only two clinical trials specifically studying prebiotics in PD (Table 1). Although this research has provided valuable insights into the possible mechanistic effects of prebiotics interventions in PD patients (discussed further below), the studies were non-randomized and open-label and thus have a high risk of bias. The study by Hall et al., using a prebiotics bar (containing a mixture of resistant starch, rice bran, resistant maltodextrin, and inulin) for ten days, reported improved gastrointestinal symptoms in the subgroup of patients on PD medications, but not in the
In these studies, prebiotics significantly altered the gut microbiome by reducing the abundance of pro-inflammatory bacteria and increasing the levels of SCFA-producing bacteria.56,57, 56,57 This finding was further corroborated by the restoration of fecal butyrate levels, 57 and increased plasma SCFA levels post-intervention. 56 Additionally, reduced levels of plasma zonulin, 56 and fecal calprotectin seen after prebiotics administration suggested improvement in intestinal barrier integrity and gut inflammation,56,57, 56,57 respectively.
Pre-clinical studies of prebiotics in PD are also relatively fewer (7 published), compared to probiotics studies (Supplementary Table 3). Various types of fibers, including wheat bran, resistant maltodextrin, inulin, and nutriose, and potential prebiotics like flavanol and dioscin, have been tested on different animal and cell models to explore the effects and mechanisms of prebiotics in PD. Consistent with the findings from clinical trials, prebiotics supplementation was found to remodel the altered gut microbiome towards an increased abundance of taxa associated with potentially protective effects.58–60 A range of other benefits similar to those observed with probiotics was also reported, including evidence for neuroprotection and “clinical” motor and non-motor improvements.58,60–66, 58,60–66 Interestingly, one study reported much better neuroprotective effects when prebiotic was combined with probiotic (in this case, polymannuronic acid and
WHAT WE TELL PEOPLE WITH PARKINSON’S IN OUR CLINIC?
While pre-clinical and clinical evidence suggest promising benefits of probiotics and prebiotics in PD, careful consideration of indications and safety issues, adherence to regulatory guidelines, and individualized risk assessment are essential to ensure their safe and effective use. Here, we summarize recommendations for patients based on the limited but growing studies in the PD field, as well as general guidelines and knowledge on these supplements. The discussion with patients should obviously also take into account their level of understanding, and desire for knowledge, regarding PD and its management.67–70
Multi-strain probiotics are hypothesized to confer greater benefits through additive or synergistic actions, but some strains may compete for nutrient sources and inhibit each other’s growth. It is therefore important that multi-strain formulations have Data are even more scarce for prebiotics, where the exact ingredients of prebiotic fiber are infrequently reported. Resistant starch and FOS are among the prebiotics used in PD studies. Probiotics/prebiotics are frequently packaged in capsule, powder, or liquid forms. Freeze-dried probiotics, delivered in capsules or powder, generally contain higher doses of probiotics, are more convenient for storage (usually in cool, dry, and dark places), and have a longer shelf-life.
74
Meanwhile, fermented milk containing probiotics requires cold-chain preservation. Regulatory bodies have established safety criteria for the assessment of probiotics for human use (encompassing records of isolation history, taxonomic identification, and absence of virulence, infectivity, toxicity, and transferable antibiotic resistance genes). In the USA and Europe, strains that have passed safety criteria are designated as “Generally Recognized as Safe (GRAS)” or have “qualified presumption of safety (QPS)” status, respectively.
77
Marketing regulatory guidelines for prebiotics are less clear and vary between countries. Most prebiotics products are currently marketed as dietary fiber, or as complementary ingredients in probiotics and/or nutritional milk supplements. Nevertheless, prebiotics product labels should contain standard information for dietary supplements including serving size and suggested dosage, quantity, and percentage composition of active ingredients per unit dose, other ingredients, and expiration date. Additionally, stamps of approval from local regulatory bodies, good manufacturing practice (GMP) certification and/or other accreditations from recognized third party testing laboratories, signify that the product manufacturer has conformed to local safety standards and best practices to ensure quality-controlled production and delivery of these supplements. Importantly, health claims on the product label should be interpreted carefully; probiotics/prebiotics products in the USA carry a disclaimer statement from the FDA that the health claim statements have not beenevaluated.
CONCLUSION AND FUTURE PERSPECTIVES
Notably, existing probiotics and prebiotics products in PD trials have yet to meet regulatory requirements to be marketed as a drug (i.e., intended for use in the diagnosis, cure, treatment, or prevention of disease). In this regard, two novel probiotics strains,
Genetically-engineered probiotics bacteria with specific desired phenotypic traits that can be transferred to the host, such as the expression of glucagon-like peptide-1 (GLP-1, which plays key functions in neurogenesis, neuronal metabolism, and synaptic plasticity) have shown promise in two pre-clinical studies in PD (Supplementary Table 1).15,81,82, 15,81,82 Meanwhile, in a mouse model study, administration of a human probiotic strain
Importantly, as with management of PD overall,68,70,90, 68,70,90 a key principle with microbial-directed therapeutics in PD is that “
Footnotes
ACKNOWLEDGMENTS
The authors gratefully acknowledge the funding support from the Ministry of Higher Education Malaysia (FRGS/1/2018/SKK02/UM/02/1) and the Malaysia Science Toray Foundation, for their previous and ongoing research work on probiotics in Parkinson’s disease.
FUNDING
The authors did not receive funding support for the preparation of this article.
CONFLICT OF INTEREST
The authors have no conflict of interest to report.
DATA AVAILABILITY
The data supporting the findings of this study are available within the article and/or its supplementary material.