Abstract
Background:
Constipation is regarded as one of the prodromal features of Parkinson’s disease (PD) and there is emerging evidence linking gastrointestinal dysfunction and cognitive impairment (CI) in PD.
Objective:
We explored whether constipation is associated with development of CI in two independent cohorts of de novo PD patients (n = 196 from the Non-motor International Longitudinal Study [NILS] and n = 423 from the Parkinson’s Progression Markers Initiative [PPMI] study).
Methods:
Constipation was clinically defined using the Non-Motor Symptoms Scale (NMSS) item-21 [NILS] and Scales for Outcomes in PD-Autonomic (SCOPA-AUT) item-5 [PPMI]. We assessed baseline group differences (PD with or without constipation) in CI, global non-motor symptoms burden, motor dysfunction, and striatal dopaminergic denervation. Kaplan-Meier method estimated group differences in cumulative proportion of patients with incident CI over three years. In PPMI, we subsequently performed univariate and multivariate Cox survival analyses to evaluate whether constipation predicts incident mild cognitive impairment or dementia over a 6-year period, including constipation and other known predictors of CI as covariates.
Results:
Patients with constipation had greater motor and global non-motor burden in both cohorts at baseline (p < 0.05). Kaplan-Meier plots showed faster conversion to CI in patients with constipation in both cohorts (p < 0.05). In PPMI, 37 subjects developed dementia during a mean follow-up of 4.9 years, and constipation was an independent predictor of dementia onset (hazard ratio = 2.311; p = 0.02).
Conclusion:
Constipation in de novo PD patients is associated with development of cognitive decline and may serve as a clinical biomarker for identification of patients at risk for cognitive impairment.
INTRODUCTION
Constipation is highly prevalent across all stages of Parkinson’s disease (PD) and is recognized as a prodromal feature and risk factor for in-life manifest PD [1]. Neuropathological support of this observation comes from reports showing misfolded α-synuclein deposits in human gastrointestinal tissues up to two decades before the development of motor dysfunction [2], with recent findings supporting the involvement of the gut-brain axis in the pathogenesis of PD [3], particularly in the “body first” subtype of PD [4]. Although multifactorial in its origin, constipation in PD could be a clinical manifestation of dysautonomia, which has been associated with a more “malignant” PD subtype characterized by rapid motor and non-motor progression, including the development of dementia [5]. Mild cognitive impairment (MCI) is estimated to occur in up to 20% of PD patients at diagnosis and the CamPaIGN cohort study suggested that 46% of newly diagnosed PD patients may convert to dementia within 10 years [6–8]. Early identification of patients at risk of developing cognitive impairment (CI) in PD is still an unmet need and could help to stratify the early PD population for prognostic information, improvement of clinical management and personalized medicine. Although associations between gastrointestinal and cognitive dysfunction have been observed in recent clinical studies [9–11], whether constipation can independently predict the development of CI in de novo PD patients remains unclear. To test this hypothesis, we performed a longitudinal analysis on scale-based data capturing constipation and cognition collected from 196 de novo PD patients followed in a prospective multicenter cohort study: the Non-motor International Longitudinal Study (NILS). We further investigated our results for external validity in an additional and independent cohort of 423 de novo PD patients from the Parkinson’s Progression Markers Initiative (PPMI) database.
MATERIALS AND METHODS
Study design and subjects
Data on de novo PD patients from two independent multicenter cohorts were used for this study: the NILS and PPMI database (Supplementary Figure 1). NILS is an observational longitudinal study addressing the range, nature, and natural history of non-motor symptoms (NMS) in PD (https://www.gsttbrc.com/NILS). It involves 34 centers worldwide and has been adopted as a national study by the National Institute of Health Research in the United Kingdom (NRES SouthEast London REC3, 10084, 10/H0808/141). The PPMI is an observational study aimed at identifying biomarkers of PD progression using imaging, biologic sampling, as well as clinical and behavioral assessments. It involves 33 centers in the United States, Europe, Israel, and Australia. Documentation regarding the organization, aims, and methodology of the PPMI have been previously published [12]. Eligibility criteria for both studies are reported in Supplementary Table 1. For this analysis, only de novo subjects were selected from the NILS cohort, while the PPMI database recruited only de novo subjects as per protocol. Data for both cohorts were downloaded on December 1, 2019. In both studies, participants are longitudinally followed up on a yearly basis. These two studies were authorized by local ethics committees and all patients gave written consent prior to study procedures in accordance with the Declaration of Helsinki.
Clinical assessments
Data extracted from the NILS database included demographics, such as age, age at PD diagnosis, disease duration, sex, and years of education, as well as Mini-Mental State Examination (MMSE), Non-Motor Symptoms Scale (NMSS) and its sub-domain 5 (Attention/Memory), Parkinson’s disease Sleep Scale (PDSS), Scales for Outcomes in PD (SCOPA) Motor, and dopamine transporter scan (DaTscan) specific binding ratios. Data extracted from the PPMI database included demographics (age, age at PD diagnosis, disease duration, sex, and years of education), Montreal Cognitive Assessment (MoCA), Movement Disorder Society-Unified Parkinson’s Disease Rating Scale (MDS-UPDRS), MDS-UPDRS part-I Item 1 (cognitive impairment), SCOPA-Autonomic (SCOPA-AUT), University of Pennsylvania Smell Identification Test (UPSIT), REM sleep Behavior Disorder (RBD) questionnaire (RBDQuest), DaTscan binding ratios and CSF amyloid-β 1–42 (Aβ1–42). In addition, data from neuropsychological tests were included, such as the Benton Judgment of Line Orientation 15-item (visuospatial functioning), Symbol Digit Modalities Test (processing speed), Semantic Fluency test (language/semantic fluency), Letter-Number Sequencing (working memory), and Hopkins Verbal Learning Test-Revised (HVLT-R, learning/immediate verbal memory and delayed verbal recall).
Constipation variables
At baseline, patients were classified according to the presence (PD-Const+) or absence (PD-Const-) of constipation. NMSS item 21 and SCOPA-AUT item 5 were used to assess constipation for NILS and PPMI respectively (cut-off score ≥1). Both NMSS item 21 and SCOPA-AUT item 5 define constipation as having less than three bowel movements per week over the last month.
Longitudinal cognitive assessment
Cognitive impairment (CI) was defined at two levels: (1) at the screening level, MMSE score of < 24 and MoCA score of < 26 were applied for NILS and PPMI, respectively [13, 14]; (2) using psychometric tests (only in PPMI), MCI categorization was reached through a cognitive test-based classification, requiring impairment (> 1.5 standard deviations below the standardized mean score, in the range recommended to establish a MCI diagnosis) on any two cognitive test scores (using both total immediate recall and recognition recall from the HVLT-R, and single scores from each of the other tests) and cognitive complaint. To mitigate the potential for misdiagnosis, patients with MCI classification on a single visit who were subsequently and consistently classified as normal cognition for at least two subsequent visits were deemed to be normal cognition at the end of their follow up visits. A diagnosis of dementia required impairment on at least one cognitive test score from at least two domains (> 1.5 standard deviations below the standardized mean score), cognitive complaints and evidence of functional impairment attributable to CI, sufficient to interfere with activities of daily life (a full description of these methods is available at http://www.ppmi-info.org/study-design/research-documents-and-sops/). The cognitive categorization in PPMI was implemented at a later stage, meaning that many patients only have cognitive categorization from 2-year follow-up visit onwards. Since this information was not available at earlier time points, patients with at least two cognitive tests of more than 1.5 standard deviations below normal at baseline were considered as “suspected MCI”, to reflect the lower level of confidence in this classification [15].
Statistical analysis
Statistical analyses were performed using the Statistical Package for the Social Sciences (SPSS), version 25.0 (IBM Corp., Armonk, NY, USA). To evaluate the clinical features associated with constipation, we first performed a cross-sectional analysis for each cohort at baseline. Normality of variables was tested with Shapiro-Wilk tests. Group differences in demographics, motor symptoms, global NMS burden, and specific NMS, including CI, were assessed using Mann-Whitney-U test or Pearson Chi-square where appropriate. Quade’s rank analysis of covariance was performed to assess age-corrected group differences.
The longitudinal analysis was carried out using Kaplan-Meier survival estimates and curves to determine group differences in cumulative proportion of patients with incident CI over three years. Comparisons were made using the log-rank (Mantel–Cox) test. For this purpose, only subjects with at least two research visits assessing cognition and baseline MMSE score ≥24 for NILS and MoCA score ≥26 for PPMI were included.
We further evaluated whether constipation may be independently associated with the development of MCI or dementia in the PPMI cohort where up to six years follow up data was available. A two-step approach with Cox survival analyses was performed: first, constipation and previously identified predictors of CI at baseline (age, sex, years of education, MDS-UPDRS III, RBDQuest, CSF Aβ1–42, UPSIT, DaTscan caudate uptake) as well as anticholinergic burden were evaluated at univariate level [16, 17]; second, multivariate Cox survival analyses were carried out including constipation as predictor and the above mentioned variables as covariates. Two models were explored: 1) conversion to dementia; 2) conversion to CI (defined as conversion to either MCI or dementia). The first occurrence of the event at follow-up was used as the time-to-event in the Cox models. For these analyses, only patients with full cognitive testing on visits following baseline and without missing covariates were included. As the majority of patients’ first cognitive categorizations were made at the 2-year follow up visit, there were 25 patients with MCI or PD dementia at their first categorization (up to the second year). As such, it is not possible to rule out that these patients did not already meet the criteria for MCI at baseline [15]. Thus, the Cox regression was repeated in a more restricted sample to assess whether constipation was predictive of future cognitive decline and did not depend on patients who may have had cognitively deteriorated at an earlier disease stage. In the repeated analysis, the criteria for “suspected MCI” (defined above) were applied at the baseline visit, and only participants without suspected MCI were included. The same approach was not needed for the dementia model as all PD patients were not demented at baseline, as per PPMI inclusion criteria. A p-value of < 0.05 was considered statistically significant. Benjamini-Hochberg procedure was used to correct for multiple testing.
RESULTS
Baseline analysis
The characteristics of the two independent samples are shown in Table 1. Constipation was present in 32.1% (63/196) in the NILS and in 32.4% (137/423) in the PPMI cohorts. In both cohorts, PD-Const+ groups were older and had been diagnosed with PD at a more advanced age than PD-Const- groups (p < 0.01). No statistically significant differences were observed in sex, years of education, and disease duration between PD-Const+ and PD-Const-. After correction for age and multiple testing, no significant differences were shown in cognitive screening tests in both NILS (MMSE) and PPMI (MoCA) cohorts, although PD-Const+ patients reported significantly higher cognitive complaints compared to PD-Const- (NMSS attention/memory domain scores for NILS and MDS-UPDRS part-I Item 1 for PPMI; p < 0.05) (Table 2). Likewise, constipated patients performed worse on motor assessments: in the NILS cohort, PD-Const+ group showed significantly higher scores in the SCOPA Motor scale (p = 0.012) than PD-Const-; in PPMI, similar results were observed for MDS-UPDRS Part II and III (p < 0.05). In relation to NMS, PD-Const+ patients in the NILS cohort showed significantly worse scores in NMSS (p = 0.001) and PDSS (p = 0.003) when compared to PD-Const- patients; similarly, in the PPMI dataset, PD-Const+ scored higher in MDS-UPDRS Part I (p < 0.001), SCOPA-AUT scale (p < 0.001) and RBDQuest (p = 0.030). Moreover, a higher MDS-UPDRS total score was observed in PD-Const+ in PPMI (p < 0.001). In both cohorts, DaTscan imaging showed no significant age-corrected differences in mean putamen and caudate uptake. No significant differences at baseline were observed in CSF measures or domain-specific cognitive testing for PPMI (Supplementary Tables 2 and 3).
Patients’ characteristics at baseline for the two independent cohorts (Non-Motor International Longitudinal Study and Parkinson’s Progression Markers Initiative cohorts)
Data are presented as median (25th and 75th percentiles) or number (percentage). Differences between groups were tested using Mann-Whitney-U test or Pearson Chi-square where appropriate (p<0.05 was considered significant). NILS, Non-Motor International Longitudinal Study; PD-Const+, Parkinson’s disease patients with constipation; PD-Const-, Parkinson’s disease patients without constipation; PPMI, Parkinson’s Progression Markers Initiative.
Baseline clinical features associated with constipation in de novo Parkinson’s disease patients from the Non-Motor International Longitudinal Study and the Parkinson’s Progression Markers Initiative study
Data are presented as median (25th and 75th percentiles) or percentage. p1, Differences between groups were tested using Mann-Whitney-U test or Pearson Chi-square where appropriate. p2, Quade’s rank analysis of covariance was performed to assess age-corrected group differences. *Significant p-values after Benjamini-Hochberg correction for multiple testing. p<0.05 was considered significant. aNMSS and SCOPA-AUT total scores were calculated excluding item related to constipation (NMSS: item 21, SCOPA-AUT: item 5). bAvailable for a subsample: PD-Const-: N = 27 (NILS), N = 284 (PPMI); PD-Const+: N = 10 (NILS), N = 135 (PPMI). DaTscan, dopamine transporter scan; MDS-UPDRS, Movement Disorders Society Unified Parkinson’s Disease Rating Scale; MMSE, Mini-Mental State Examination; MoCA, Montreal Cognitive Assessment; NILS, Non-motor international longitudinal study; NMSS, Non-Motor Symptoms Scale; PDSS, Parkinson’s Disease Sleep Scale; PD-Const+, Parkinson’s disease patients with constipation; PD-Const-, Parkinson’s disease patients without constipation; PPMI, Parkinson’s Progression Markers Initiative; RBDQuest, REM Sleep Behavior Disorder Questionnaire; SCOPA, SCales for Outcomes in PArkinson’s disease; SCOPA-AUT, SCales for Outcomes in PArkinson’s disease Autonomic; UPSIT, University of Pennsylvania Smell Identification Test.
Longitudinal analyses of cognitive impairment
For the Kaplan-Meier survival analysis, 124 patients from NILS and 314 patients from PPMI were included (Supplementary Figure 1). Kaplan-Meier curves and log-rank (Mantel-Cox) tests showed that, in both databases, the PD-Const- group had longer CI-free survival times over three years when compared to the PD-Const+ group (Log Rank, NILS: Chi-square = 4.663, p = 0.031; PPMI: Chi-square = 6.515, p = 0.011) (Fig. 1).
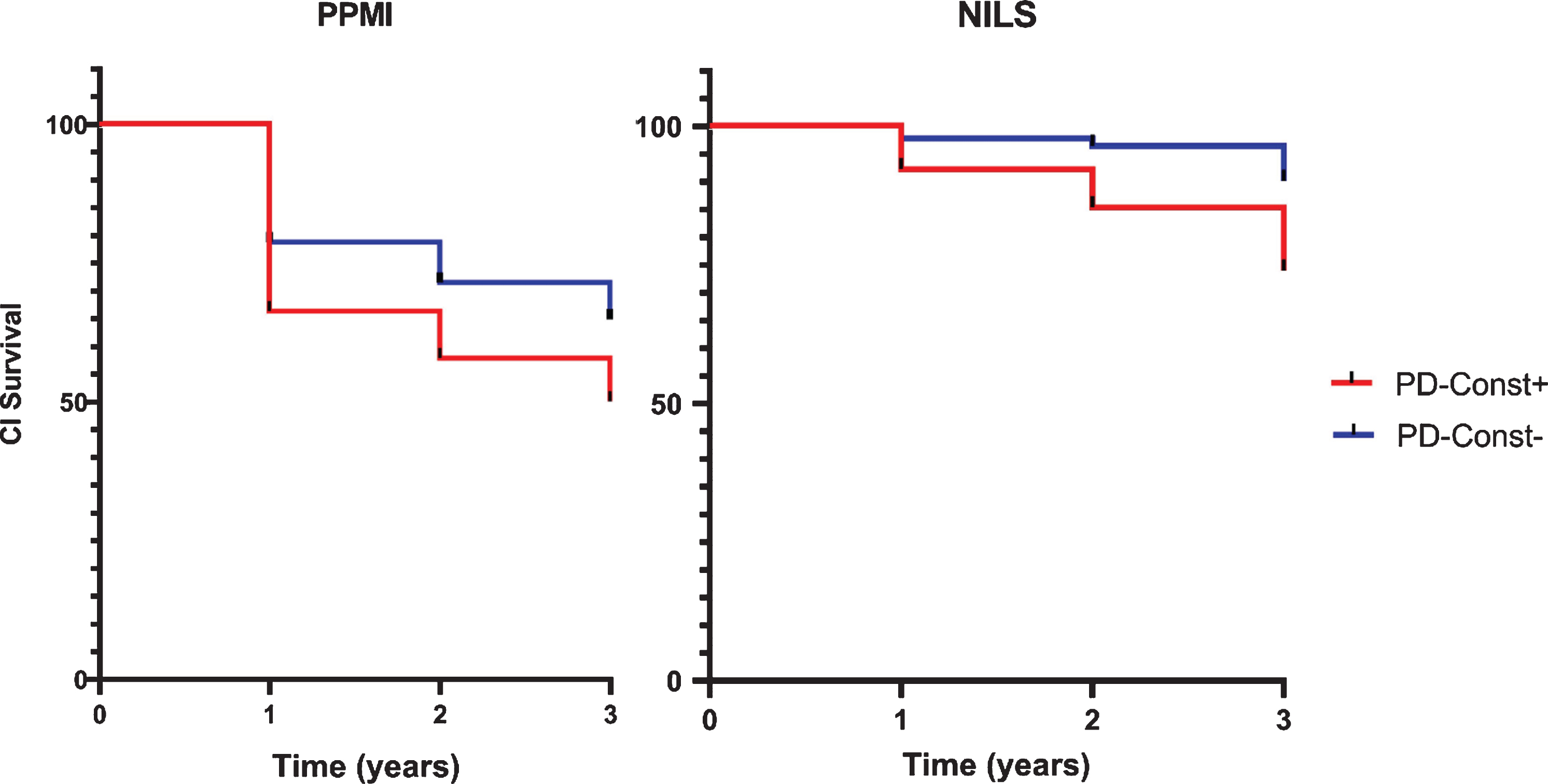
Kaplan-Meier curves for cognitive impairment-free survival in the two cohorts. (Non-Motor Longitudinal International Study cohort - Log Rank, Mantel Cox: Chi-square = 4.663; p = 0.031; Parkinson’s Progression Markers Initiative cohort - Log Rank, Mantel Cox: Chi-square = 6.515; p = 0.011). CI, cognitive impairment; NILS, Non-Motor-International Longitudinal International Study cohort; PD-Const+: Parkinson’s disease patients with constipation; PD-Const-: Parkinson’s disease patients without constipation; PPMI, Parkinson’s Progression Markers Initiative cohort.
For Cox regression survival analysis, 399 patients from PPMI were included (Supplementary Figure 1). During a median follow-up of 6.0 years (mean 4.9 years), 75 (18.8%) subjects developed CI (MCI or dementia), of which 37 (9.3%) developed dementia. Cox proportional-hazards analyses showed that conversion to dementia was associated with constipation in both univariate (hazard ratio [HR] = 3.493; 95% confidence interval [CI] = 1.810–6.744; p < 0.001) and multivariate models (HR = 2.311; 95% CI = 1.141–4.683; p = 0.020) (Table 3). Constipation predicted conversion to cognitive impairment (MCI or dementia) in both univariate (HR = 2.956; 95% CI = 1.872–4.667; p < 0.001) and multivariate Cox regression (HR = 1.722; 95% CI = 1.057–2.805; p = 0.029) (Table 4). When patients with “suspected MCI” at baseline were excluded from the analysis (n = 66), constipation predicted conversion to MCI in the univariate Cox regression (HR = 3.333; 95% CI = 1.824–6.090; p < 0.001), but the result did not reach significance in the multivariate analysis (HR = 1.717; 95% CI = 0.898–3.285; p = 0.102) (Table 5).
Results of the Cox proportional-hazards analysis for the predictors of dementia over up to six years in the Parkinson’s Progression Markers Initiative cohort
aCategorical value based on SCOPA-AUT item 5; bCategorical value, cut-off 5 points; cLog-transformed value; dcategorical value; p<0.05 was considered significant. ACB, anticholinergic burden score; CI, confidence interval; CSF, cerebrospinal fluid; HR, Hazard Ratio; MDS-UPDRS, Movement Disorders Society Unified Parkinson’s Disease Rating Scale; RBDQuest, REM Sleep Behavior Disorder Questionnaire; UPSIT: University of Pennsylvania Smell Identification Test.
Results of the Cox proportional-hazards analysis for the predictors of cognitive impairment (mild cognitive impairment or dementia) over up to six years in the Parkinson’s Progression Markers Initiative cohort
aCategorical value based on SCOPA-AUT item 5; bCategorical value, cut-off 5 points; cLog-transformed value; dCategorical value; p<0.05 was considered significant. ACB, anticholinergic burden score; CI, confidence interval; CSF, cerebrospinal fluid; HR, hazard ratio; MCI, mild cognitive impairment; MDS-UPDRS, Movement Disorders Society Unified Parkinson’s Disease Rating Scale; RBDQuest, REM Sleep Behavior Disorder Questionnaire Score; UPSIT: University of Pennsylvania Smell Identification Test.
Results of the Cox proportional-hazards analysis for the predictors of cognitive impairment (mild cognitive impairment or dementia) over up to six years in the Parkinson’s Progression Markers Initiative cohort. Subjects with “suspected MCI” at baseline were excluded
aCategorical value based on SCOPA-AUT item 5; bCategorical value, cut-off 5 points; cLog-transformed value; dCategorical value; p<0.05 was considered significant. ACB, anticholinergic burden score; CI, confidence interval; CSF, cerebrospinal fluid; HR, hazard ratio; MCI, mild cognitive impairment; MDS-UPDRS, Movement Disorders Society Unified Parkinson’s Disease Rating Scale; RBDQuest, REM Sleep Behavior Disorder Questionnaire; UPSIT, University of Pennsylvania Smell Identification Test.
DISCUSSION
In this study, we infer that the presence of constipation, clinically defined by the use of the validated NMSS and SCOPA-AUT, was associated with development of cognitive impairment in two independent multi-center international cohorts of de novo PD patients. The use of two independent cohorts spanning different populations in the United States, Latin America, Europe, Australia, and Asia, is a unique aspect of this work.
In NILS we found that de novo PD patients with constipation developed cognitive impairment earlier than patients without constipation over a 3-year follow up period. These results were confirmed in the independent PPMI cohort and, additionally, in this dataset, we showed that constipation was associated with the development of cognitive impairment over a 6-year follow up period. Our results are of interest in light of a recently published study that highlighted the relationship between gastrointestinal symptoms (reduced gut motility, straining, and fecal incontinence) and the risk of developing cognitive decline in PD [9]. However, constipation was not specifically discussed and other recognized predictors of cognitive impairment in PD, such as RBD, hyposmia, CSF Aβ1–42, and reduced caudate dopamine transporter uptake were not taken into consideration [16], whereas we demonstrated that even after including these factors, constipation remained a significant predictor of dementia. In addition, the results remained statistically significant even after adjusting for the use of anticholinergic medication, motor impairment, and age, which are all known risk factors for the development of cognitive decline [16, 17]. In our model using PPMI data, other significant predictors of dementia were years of education, UPSIT scores as well as CSF Aβ1–42 levels and mean caudate uptake, in agreement with previous studies [14, 18].
When we included MCI in our data analysis, constipation appeared to have a greater association with the risk of dementia. Indeed, after inclusion of strict criteria to define MCI in PPMI at baseline (excluding “suspected MCI”), constipation remained a significant predictor of cognitive impairment (dementia or MCI), but only at univariate level. On the one hand, this could be partially due to the exclusion of 66 participants with “suspected MCI” from data analysis, but on the other hand, this might be related to the fact that PD-MCI is a heterogeneous condition, including both subjects who will convert to dementia and subjects who will remain cognitively stable or could potentially revert to normal cognition [6].
We found an almost identical prevalence of constipation in de novo PD in both cohorts (32.1% in NILS and 32.4% in PPMI) despite the use of two different outcome measures (NMSS item 21 for NILS and SCOPA-AUT item 5 for PPMI). In both cohorts the presence of constipation was associated with older age at baseline and, after correction for this possible confounder, we found that PD patients with constipation had higher motor and non-motor symptoms burden, including RBD, and autonomic dysfunction, confirming previous findings [19, 20]. Among non-motor features, it appeared that RBD is associated with constipation and could also predict cognitive impairment in PD [21, 22]. It is possible, therefore, that RBD and constipation, which are also clinical biomarkers of prodromal PD [22, 23], might signpost a specific non-motor PD endophenotype associated with cognitive dysfunction and propensity to dementia. Indeed, the recently proposed cholinergic subtype of PD can fit into this category [24–26], and an independent cross-sectional cohort study from Moscow, Russia, also suggested a link between constipation and cognitive dysfunction in de novo PD [27]. While there is some literature to support the association of central dopaminergic dysfunction with constipation [28, 29], this was not evident in our analysis from baseline data. Non-dopaminergic mechanisms underpinning constipation have also been reported previously and our results support this observation [20, 31]. Our data is also in line with Fereshtehnejad et al., who reported the association of cognitive impairment with RBD and autonomic dysfunction, identifying a more malignant phenotype which progresses faster to dementia probably due to an underlying more diffuse neurodegenerative process [5].
There are of course several limitations of our study which we need to highlight, and these include the use of different and subjective outcome measures to assess constipation and other motor and non-motor features of PD in the two longitudinal cohorts. For instance, we recognize that the MoCA is a more sensitive tool to measure cognitive impairment in PD than MMSE [32]. Despite that, our results were consistent across both cohorts and we tried to identify constipation as accurately as possible in the context of there being no PD-specific validated constipation scales. We are, therefore, unable to comment on more granularity related to the pathophysiology of constipation such as slow transit versus outlet constipation. Although the definition of constipation did not meet the ROME IV criteria, the definition we used in this study is routinely applied in clinical practice. Finally, we acknowledge that constipation in PD is multifactorial, and our study did not consider all possible non-PD related causes of constipation, such as dehydration and diet. Nevertheless, the understanding of pathophysiological mechanisms underlying the association between constipation and development of cognitive impairment is beyond the purpose of this study and we believe that our results are meaningful, as they were replicated in two independent international cohorts of de novo PD patients. This also implies that our results are not influenced by common confounders, including PD medication. Other potential factors associated with cognitive impairment in both PD and general population have not been included in our analyses for two main reasons. First, for some of them, such as physical exercise [33], degree of physical frailty [34] or novel plasma biomarkers [35, 36] for cognitive impairment in PD, data were not directly available. Second, we chose not to include data regarding other clinical features that have previously been associated with cognitive decline in PD, such as orthostatic hypotension or diabetes mellitus [35, 37], in order to focus our analysis on the currently most reliable predictors of cognitive impairment in PD [16].
In summary, we report that constipation is associated with the development of cognitive impairment in two independent and multicenter cohorts of de novo PD, suggesting a common pathophysiological background. Constipation could prove useful, possibly in combination with other biomarkers, as a predictor of future cognitive decline with implications for clinical management, clinical trial enrolment and design, as well as for our understanding of PD pathophysiology.
Footnotes
ACKNOWLEDGMENTS
We acknowledge data collection efforts by all contributors of the NILS. The following centers participate in the NILS (with principal investigator): King’s College Hospital, London (Prof. K. Ray Chaudhuri); Lewisham University Hospital, London (Prof. K. Ray Chaudhuri); Lund University Hospital, Lund (Prof. P. Odin); Reinkenheide Hospital, Bremerhaven (Prof. P. Odin); IRCCS San Camillo, Venice (Prof. A. Antonini); IRCCS San Raffaele, Rome (Prof. F. Stocchi); Leiden University Medical Centre, Leiden (Prof. J.J. van Hilten); University Medical Centre Groningen (Prof. T. van Laar), Groningen; Transilvania University Hospital, Braşov (Dr. C. Falup-Pecurariu); Hospital Ruber Internacional, Madrid (Dr. M. Kurtis); Aristotle University of Thessaloniki, Thessaloniki (Prof. S. Bostantjopoulou); Assaf Harofeh Medical Center, Tel Aviv University, Zerifin (Prof. J.M. Rabey); Le Terrazze Hospital, Cunardo (Dr. G. Riboldazzi); Hospital Clinico San Carlos, Madrid (Prof. M.J. Catalan); Medway Maritime Hospital, Gillingham (Dr. C. Ellis); Princess Royal University Hospital, Orpington (Dr. B. Kessel); Macclesfield District General Hospital, East Cheshire (Dr. M. Silverdale); Norfolk and Norwich University Hospital, Norwich (Dr. P. Worth); Yeovil Hospital, Somerset (Dr. R. Sophia); United Lincolnshire Hospital, Lincoln (Dr. J. Sharma); Salford Royal Hospital, Manchester (Prof. M. Silverdale); Forth Valley Royal Hospital, Edinburgh (Dr. S. Pal), University of Kansas Medical Centre, Kansas City (Prof. K.E. Lyons), Carlos Andrade Marín Hospital, Quito (Dr. M. Serrano-Duenas); Universidade Federal de São Paulo, São Paulo (Prof. V. Borges); Hospital Dr. Domingo Luciani, Caracas (Prof. M. Gallardo); Hospital de Clinicas Jose de San Martin, Buenos Aires (Prof. F. Micheli); Instituto Nacional de Neurologia y Neurocirugía, Mexico City (Prof. M. Rodriguez-Violante), All India Institute of Medical Sciences, New Delhi (Prof. M. Behari); Hyogo Medical College, Nishinomiya (Prof. K. Abe); University of Calcutta, Calcutta (Prof. K. Bhattacharya). The views expressed are those of the authors and not necessarily those of the NHS, NIHR or Department of Health. The authors acknowledge the support of the Movement Disorder Society Non-Motor PD Study Group, the NIHR London South Clinical Research Network and the NIHR Biomedical Research Centre. This article represents independent collaborative research part funded by the NIHR Biomedical Research Centre at South London and Maudsley NHS Foundation Trust and King’s College London. Data used in the preparation of this article were also obtained from the Parkinson’s Progression Markers Initiative (PPMI) database (http://www.ppmi-info.org/data). For up-to-date information on the study, visit ![]() . PPMI (a public–private partnership) is funded by the Michael J. Fox Foundation for Parkinson’s Research and funding partners, including AbbVie, Avid, Biogen, Bristol-Myers Squibb, Covance, GE Healthcare, Genentech, GlaxoSmithKline, Lilly, Lundbeck, Merck, Meso Scale Discovery, Pfizer, Piramal, Roche, Servier, Teva, and UCB. This study was not supported by additional funding.
. PPMI (a public–private partnership) is funded by the Michael J. Fox Foundation for Parkinson’s Research and funding partners, including AbbVie, Avid, Biogen, Bristol-Myers Squibb, Covance, GE Healthcare, Genentech, GlaxoSmithKline, Lilly, Lundbeck, Merck, Meso Scale Discovery, Pfizer, Piramal, Roche, Servier, Teva, and UCB. This study was not supported by additional funding.
CONFLICT OF INTEREST
The authors have no conflict of interest to report.
