Abstract
Background:
Symptoms from the gastrointestinal tract are highly prevalent in Parkinson’s disease (PD), but knowledge of the underlying pathology is incomplete and valid objective markers on regional gastrointestinal function are limited.
Objective:
The aims were to evaluate gastrointestinal transit time and motility in PD patients and controls.
Methods:
Twenty-two PD patients and 15 controls were included. Gastric-, small intestinal-, and caecum-ascending colonic transit times as well as colonic motility, defined as mass- and fast movements, were performed using the ambulatory 3D-Transit system. Gastrointestinal transit time with radio opaque markers, gastric emptying scintigraphy, and subjective non-motor symptoms were also evaluated.
Results:
Using the 3D-Transit system, the patient group displayed significantly longer small intestinal- and caecum-ascending transit times (
Conclusions:
Significantly increased small intestinal- and caecum-ascending 3D-Transit times were detected in PD patients. Also, time to first propagating colonic movement was increased. Radio opaque marker gastrointestinal transit time was significantly delayed, but no difference was seen in gastric transit time and gastric emptying time. The present findings highlight widespread intestinal involvement in PD increasing throughout the gastrointestinal tract.
INTRODUCTION
Parkinson’s disease (PD) is now known to be a multi-system disorder involving not only the central nervous system (CNS), but also the peripheral and enteric nervous systems. The majority of PD patients experience autonomic non-motor symptoms (NMS) from the gastrointestinal (GI) tract including constipation. These symptoms are often present at an early prodromal stage, which lends support to the hypothesis that PD pathology may initiate in the GI tract, and subsequently spread to the CNS via autonomic nerves [1, 2].
Symptoms from the upper and lower GI tract can overlap and be difficult to distinguish. Different objective methods are available for obtaining data on GI transit time and motility, although often restricted by lack of standardization, radiation exposure, cost, and invasiveness [3]. Furthermore, the small intestine is not very accessible to examination. However, improved objective markers of GI function may be essential to study small-intestinal medication absorption and to obtain knowledge of the underlying gut pathology and involvement in PD. Gastric emptying time (GET) and total GI transit time (GITT) have previously been studied in PD, but detailed data on regional intestinal transit and dysmotility are lacking, and specifically the small intestine has received very little attention [4, 5].
The 3D-Transit system is a novel method for assessing GI regional transit times and motility patterns based on the position of an ingested wireless electromagnetic capsule. This ambulatory system allows safe and well-tolerated GI examination under near-normal physiological conditions in the patient’s home environment [6]. The system provides unique data on segmental transit times throughout the GI canal, and can also assess certain colonic motility patterns. It has been validated in healthy controls (HC) and trialed in patients with various gastrointestinal disorders [6–8]. However, no data on PD patients have yet been published.
The study aims were to evaluate GI transit time and motility in a group of PD patients and control subjects using the novel 3D-Transit magnetic system.
METHODS
Ethics statement
The study was approved by the Central Denmark Region Committee on Health Research Ethics and Danish Health and Medicines Authority (No. 1-10-72-255-14 and 2014112300). All participants provided written informed consent.
Subjects
Twenty-two PD patients and 15 age- and sex-matched controls without neurological disorders were included. Gastric emptying scintigraphy was performed in a subset of subjects (19 PD, 14 HC). All participants were unselected according to presence or degree of constipation symptoms. Participants were excluded if meeting the following criteria: prior gastrointestinal surgery, gastrointestinal disorders besides constipation (i.e. malignancy, inflammatory bowel disorders, coeliac disease), significant systemic disorders or medical conditions, diabetes, endocrine disorders, psychiatric disease, substance abuse, and pregnancy.
All PD patients were diagnosed according to the UK Brain Bank criteria by movement disorder specialists [9]. All PD patients were on dopaminergic medication. Median levodopa equivalent daily dose (LEDD) was 626 mg (range 180–1750 mg). Specific details on medication are provided in the online Supplementary Table 1. Four patients were treated with laxatives before and during the study (Psyllium, Magnesium citrate, Macrogol 3350). Intake of proton pump inhibitors were prohibited >3 days prior to examinations.
Unified Parkinson’s Disease Rating Scale part III (UPDRS-III) was performed in all patients after >12 hours withdrawal of parkinsonian medication. All other testing and questionnaires were performed in the “on” state. Eleven patients had a previous dopamine transporter SPECT (all pathological).
3D-Transit
The ambulatory 3D-Transit system (
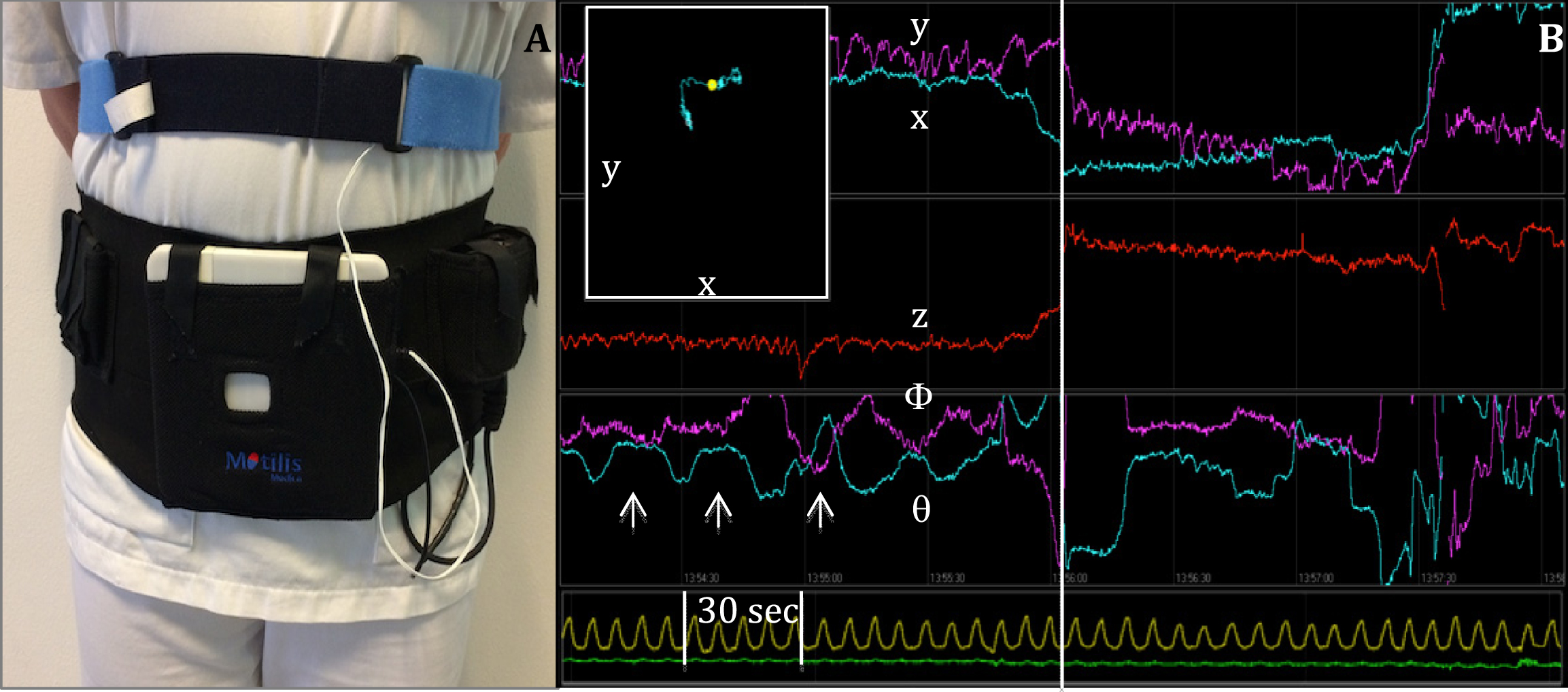
3D-Transit system. (A) Sensors in the detector plate register electromagnetic signals from the ingested capsule. A chest worn respiration belt registers artifacts due to respiration. (B) Graphs displaying position (
Transit time in each GI segment is determined by changes in contraction frequency as follows: gastric transit time (GTT): time from ingestion until frequency change from 3 contractions per minute (cpm) to 9–12 cpm; small intestinal transit time (SITT): time from pyloric to ileocaecal passage (frequency change from 6 to 3 cpm); caecum-ascending transit time (CATT): time from ileocaecal entry to passage of the hepatic flexure; colorectal transit time (CRTT): time from ileocaecal passage until capsule exit coincident with a bowel movement [6, 10]. Also, 2D spatial information obtained by the continuous position and orientation triangulation of the capsule was used to support the definition of segmental transit times (Fig. 1B).
Mass movements in the colon and rectum, also known as high-amplitude propagated contractions (HAPCs) [11], was defined with the 3D-Transit methodology as movements covering one or more colorectal segments in ≤2 minutes (Fig. 3C) [10]. We also defined a subtype of fast movements as apparent propagating contractions covering less than one colorectal segment in ≤2 minutes. Both categories of movements were manually counted and registered for each participant. The reader was blinded to clinical category.
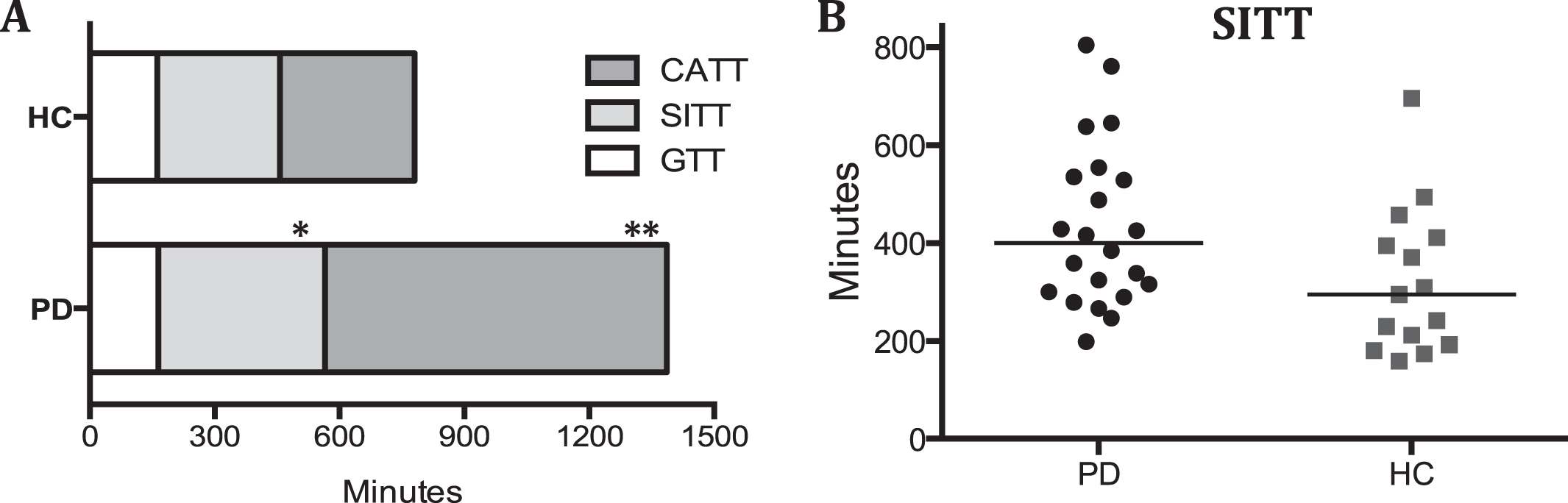
(A) Median 3D-Transit data (minutes) in PD patients and healthy controls (HC). Note that the CATT in PD patients is markedly underestimated (see main text). (B) Scatter plot of small intestine transit time in both groups. GTT = gastric transit time, SITT = small intestinal transit time, CATT = caecum-ascending transit time. *
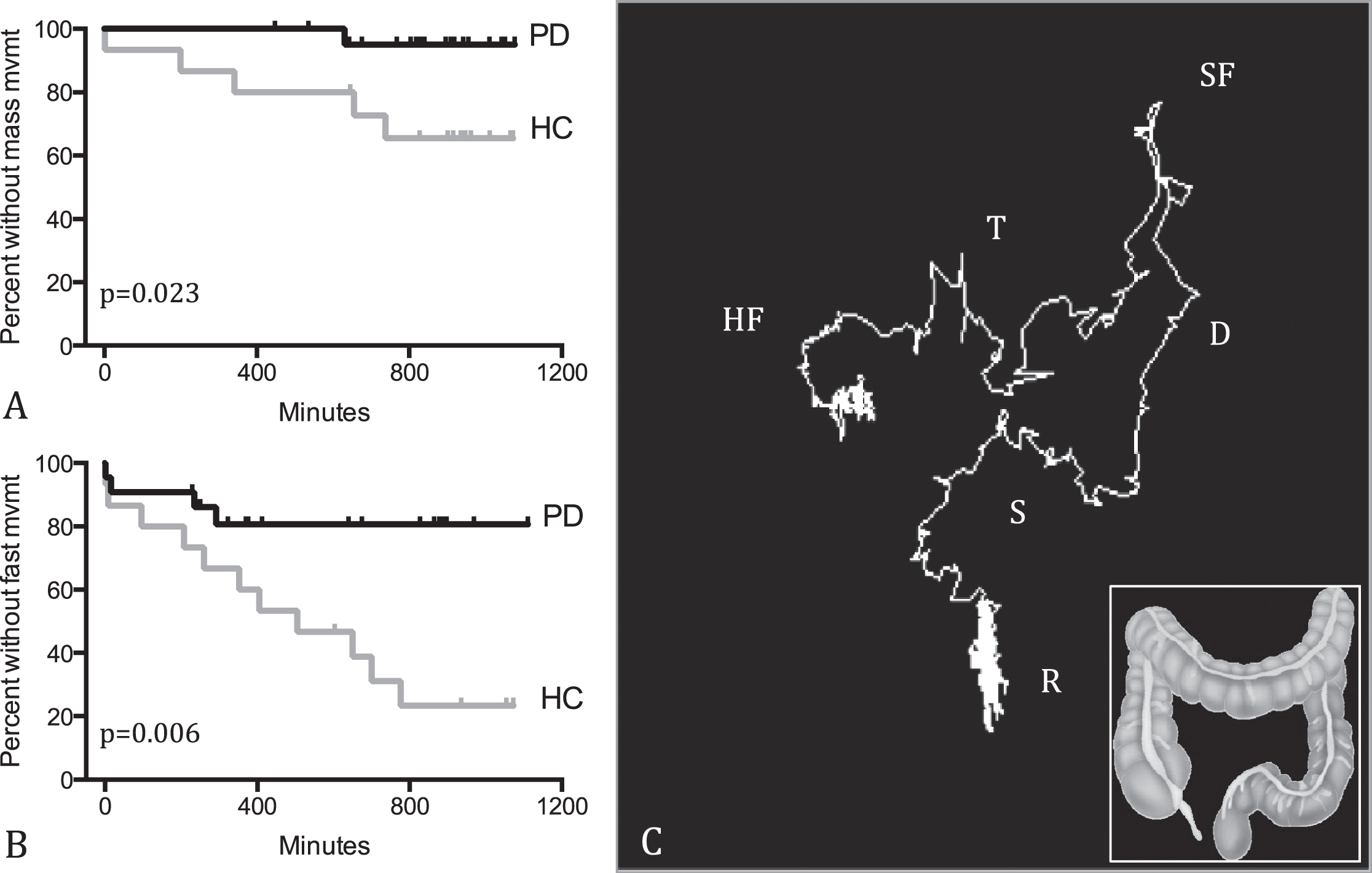
(A) Time to first mass movement in PD patients and healthy controls (HC) during mean 14.6 hours recording in the colon. (B) Time to first fast movement in PD patients and HC during mean 14.6 hours recording in the colon. (C) A mass movement in a control subject. The capsule passes from the hepatic flexure (HF) through the transverse (T), splenic flexure (SF), descending (D), and sigmoid (S) segments to the rectum (R) in ≤2 minutes. See inset for colon anatomy.
Gastrointestinal transit times
Gastric emptying time (GET) and total gastrointestinal transit time (GITT) were evaluated using scintigraphy and the radio opaque marker (ROM) techniques, as described previously [12]. In short, exponential gastric half-emptying time (T1/2) and time to emptying rate >90% (T90) were calculated from 1 min. abdominal scintigraphic images at 0, 30, 60, 90, 120, 150 and 180 min. after finishing a standardized solid meal containing 19–37 MBq 99mTc-labelled colloid [13]. Semi-automatic analysis was performed using dedicated software
GITT was defined as number of retained ROM on a CT scan performed on day seven, after ingestion of one capsule containing 10 ROM every morning for six consecutive days. GITT was calculated using the validated equation: GITT = (total ROM + 5)/10 [14]. A subset of the GET and GITT data was published previously [12].
Clinical assessment
Clinical disease stage and motor function were evaluated using the Hoehn and Yahr scale (H&Y) [15] and UPDRS-III [16]. Subjective NMS were assessed by the NMS questionnaire (NMSQuest), defining constipation as presence of <3 bowel movements per week and/or straining during the past month [17]. Olfactory function was tested with Sniffin’ Sticks 16-item identification test
Protocol
To maximize valid comparison of methods, 3D recordings were started immediately after the CT scan and simultaneous to gastric scintigraphy. The 3D monitor is worn throughout the day and night, which gives rise to some discomfort. This is particularly true for PD patients with a poor quality of sleep. Moreover, many PD patients have severely prolonged total GITT, and would need to wear the monitor for up to one week to obtain complete data sets. For this reason, the present pilot study was restricted to a 24-hour (range 22–26.5 h) recording period in the PD group. The main objective was to assess the tolerability of the system in PD, obtain data on small intestine transit time, and get preliminary data on perturbations in colonic mass movements. In the control group recordings were performed until exit of the capsule from the body. The participants were instructed not to eat or drink for six hours after capsule ingestion to prevent prolonged gastric emptying.
All participants kept a diary on defecation time, time of food ingestion, sleep, and daily activities. Time of meals were restricted to 8 a.m., 2 p.m., and 6.30 p.m., and intake of coffee, alcohol, and soft drinks were prohibited throughout the recording period. Also, sports and hard physical work were not allowed. The detector was only removed for a short period each day to allow the participants to shower.
Statistics
Statistical analyses were performed using Prism 6
RESULTS
Demographic and clinical data are listed in Table 1. PD patients and controls were matched on gender, age, and body mass index (BMI). No significant difference was seen in cognitive performance between groups. The patients showed significantly lower olfaction scores and significantly higher levels of subjective constipation symptoms. A positive correlation was seen between 3D GTT and scintigraphic GET in the control group (
Demographic and clinical data of patients with Parkinson’s disease (PD) and control subjects
Data given as mean±SD or median (range). *The “24 h” entry for controls reflects the caecum-ascending transit time during the first 24 h recording. **
3D-Transit
3D-Transit examinations were well tolerated without side effects. The patient group displayed significantly longer SITT and CATT compared to controls (Fig. 2). No between-group difference was seen in GTT.
After 24 hours, the capsule had reached the colon in all participants, and was present in the caecum-ascending segment (17 PD, 2 HC), transverse (2 PD, 4 HC), descending (2 PD, 1 HC), and recto-sigmoid segment (2 HC). The capsule had passed the total GI tract in one PD and six HC. Thus, after 24-hour monitoring, the capsule was situated in the caecum-ascending segment in 17 PD and 2 HC and more distally in 5 PD and 13 HC, amounting to a highly significant difference (
The mean CATT in the PD patients during 24-hour recording period was 14.6 hours. We therefore restricted the analyses of time to first mass movement and fast movement to the 14.6 hours colonic recording period in both groups. During this period, the time to first mass- and fast movement were significantly increased in the patient group (
The prevalence of subjective constipation symptoms was 41% in PD and 7% in controls (Table 1). No significant difference was seen in SITT and CATT between constipated and non-constipated patients (see Table 2). Note that full CATT was only obtained in five PD patients.
3D-Transit and NMSQuest subjective constipation data in patients with Parkinson’s disease
Data given in minutes as median (range). NMSQuest, non-motor symptoms questionnaire; SITT, small intestinal transit time; CATT, caecum-ascending transit time.
DISCUSSION
Using the ambulatory 3D-Transit system we report exact SITT data in PD for the first time. The SITT was significantly increased compared to control subjects. We also demonstrated a highly significant increase in proximal colonic transit time, and a decrease in colonic mass- and fast movements in the PD patients. As previously reported, no difference was found in gastric transit times with the 3D-Transit system or emptying time with gold standard scintigraphy [20]. Most studies of early-to-moderate disease stage PD patients support this finding of unaffected gastric emptying when measured by scintigraphy, whereas the 13C breath test methodology tends to show a more profound difference between early stage patients and controls [4].
Transit times
Total and segmental transit times were generally easily determined. Only minor interference on transit data was caused by body movements. This was considered acceptable compared to the advantage of ambulatory examination under near-normal physiological conditions. Also, previously published data showed acceptable day-to-day variation and inter-observer agreement in determining segmental transit times with the 3D-Transit system [6].
In support of our findings, a recent scintigraphy study also reported significantly increased SITT in PD patients compared to controls after ingestion of an isotope filled capsule [21]. However, due to methodological limitations and radiation exposure, the capsule location on scintigraphic images were determined only at a few time points precluding an exact determination of SITT. In contrast, 3D-Transit data allows a specific determination of the regional transit in each GI segment. Also, Su and colleagues recently reported delayed SITT in 20% of PD patients measured by the wireless motility capsule, although the study did not include control subjects for comparison and CTT data was not published [22]. Overall, delayed SITT in PD may have clinical implications and could contribute to malabsorption of parkinsonian medication and the development of small intestinal bacterial overgrowth (SIBO) [23, 24].
In six of the current control subjects, the capsule had passed the entire GI tract within 24 hours. This was the case for only one PD patient. The study showed generally delayed CATT in the PD group, but we cannot comment on transit times in the transverse, descending, and rectosigmoid colon, since only 24-hour recordings were performed in the patient group. In the present study, 17/22 (77%) PD patients and 2/15 (13%) controls, the 3D capsule was situated in the caecum-ascending colonic segment after 24 hours, but based on the present data it is not possible to evaluate the transit time in the more distal colonic segments measured by the 3D-Transit system. Nevertheless, recently published ROM data suggested that transit times in the transverse and rectosigmoid colon is even more delayed as compared to the ascending colon in PD patients [12]. Put together, these findings indicate that colonic function in PD is universally affected. The present study also demonstrated that total GITT measured with ROM was significantly increased in the patient group (see Table 1). Correlation analyses between 3D and ROM data for total GITT were not possible in this study, as total 3D colonic transit was not obtained in the patients. However, a good correlation between ROM and 3D-Transit total GITT has previously been published in healthy controls [6].
Fast- and mass movements
The time to first fast- and mass movement during the initial 14.6 hours of colonic recording was significantly longer in the PD group. Of note, the results may be somewhat biased by the limited recording period in the patient group. Nevertheless, it has been shown that mass movements are most often initiated in the caecum-ascending part of the colon [25, 26], giving rise to movement of colonic content throughout the more distal segments. This means that even if the capsule was situated in a more distal part of the colon in the control group, the registered mass movements would most probably have started proximally. Also, both fast- and mass movements in the present study were registered for a comparable time period of 14.6 hours in both groups. Supporting these findings, a previous study reported decreased colonic motility in patients with slow transit constipation, possibly due to parasympathetic denervation [27].
Contributing factors
The relatively wide GITT range in the present control group signifies considerable physiological variation in non-symptomatic healthy subjects. A day-to-day variation in GI transit measured by 3D-Transit of up to 45% was previously demonstrated in healthy subjects [6]. Therefore, multiple capsule monitoring or consecutive monitoring of the PD patients would provide more exact and specific GI transit data. Moreover, treatment with dopaminergic drugs in PD may contribute to constipation [28, 29]. We recently showed a positive correlation between LEDD and colonic volume in a group of PD patients, indicating an impact of dopaminergic treatment on GI function [12]. In contrast, Krogh et al. showed a decrease in unsuccessful attempts at defecation in PD patients using levodopa compared to patients not using it, measured by a constipation questionnaire [30]. Such discrepancies in the literature can now be investigated further using this real-time capsule system, and further studies are needed in populations of
Functional gradients
Interestingly, Fig. 2A indicates a gradient of increasing GI dysfunction from the proximal to the distal GI tract. Early and severe damage to the parasympathetic nervous system is well characterized in PD [31], and recently we demonstrated decreased PET signal in the small intestine and particularly the colon in newly diagnosed PD patients using the acetylcholinesterase marker 11C-donepezil [20, 32]. In other words, a similar gradient of pathological signal decrease was seen on this PET tracer, which may be a marker of the parasympathetic nervous system. These gradients are reversed compared to published studies of pathological α-synuclein deposition, which demonstrated the greatest number of pathological inclusions in the proximal GI tract [33, 34]. The authors interpreted these histological findings to mean that the proximal gut is most severely affected by disease-specific pathology. However, the relative scarcity of pathological α-synuclein inclusions in more distal segments of the gut could also be caused by a “dying-back” phenomenon, i.e. that the colon shows fewer pathological inclusions because of severe parasympathetic terminal loss [35, 36]. Similar inverse gradients of terminal loss and α-synuclein inclusion density have been reported in the heart and skin in PD patients [37, 38]. For the moment this hypothesis remains speculative and is very difficult to prove, due to the challenges of demonstrating parasympathetic nerve terminal loss on a background of intrinsic enteric neurons, which are also mainly cholinergic.
Subjective symptoms
The patient group presented significantly more subjective NMS compared to controls, and significantly higher constipation prevalence. No correlation was seen between constipation and SITT. Correlations with colonic transit were not tested, because only 24-hour monitoring was performed, but previous publications have reported only minor or absent correlations between subjective and objective GI data in PD [5, 12].
Limitations
Only 24-hour 3D-Transit data was obtained in the patient group. Thus, total colonic transit is not available. However, the primary goal of this study was to trial the tolerability of the capsule system in a PD population, and specifically to quantify SITT in PD and also obtain initial data on colonic dysmotility patterns. Moreover, total ROM-GITT data was recently published showing highly significant increased colonic transit time in the patient group, so it was deemed less important to obtain full colonic data sets in the PD patients [12].
The intake of food and beverage was not standardized, laxatives were not paused during the study, and intake of coffee was prohibited. Also, the participants were allowed to live their normal daily life, all of which might have affected the results. However, the purpose was to examine the near-normal everyday GI function, which requires preservation of daily life routines. Also, laxative intake results in faster GI transit time, and a restriction would only have further prolonged the capsule GI passage, resulting in even more significant group differences.
The current study participants were not tested for SIBO, which theoretically could affect small intestinal transit time. However, it is unclear whether SIBO is caused by delayed transit or may induce delayed transit [23, 24]. As such, studies are needed to establish the causal relationship between SIBO and delayed SITT.
Colonic motility measured by fast- and mass movements were manually determined, as an automated procedure is not presently available. This might have biased the results. Thus, all colonic movements were carefully analyzed in relation to accelerometer and respiratory belt data, discarding changes not explicitly related to colonic movement. Also, the data were analyzed by an expert in the 3D-Transit system, blinded to subject categories. Furthermore, the capsule measures motility dynamically in relation to intestinal content. Thus, we speculate that the capsule may travel a shorter distance during a movement, therefore being categorized as a fast movement, although it may be part of a mass movement. The different movements remain to be defined more accurately. Nevertheless, time to first propagating movement was in every case prolonged in the patient group.
CONCLUSION
Using an ambulatory GI-Transit system, PD patients showed significantly prolonged transit times in the small intestine and in the proximal colon. Furthermore, time to both first mass- and fast movement in the PD patients were significantly increased. No between-group difference was seen in gastric transit time obtained with the 3D-Transit system and with solid meal scintigraphy.
In conclusion, these first published results on real-time 24-hour GI transit times in PD patients suggest widespread slow transit times, which exhibits an increasing gradient of dysfunction most significant in the proximal colon, while gastric transit time appeared normal. Further studies are needed to evaluate patients in earlier disease stages and the influence of anti-parkinsonian medication on GI motility.
FINANCIAL DISCLOSURES
Karoline Knudsen, Anne-Mette Haase, Tatyana D. Fedorova, and Anne C. Bekker have nothing to disclose.
Karen Østergaard:
Klaus Krogh:
Per Borghammer:
CONFLICT OF INTEREST
The authors have no conflict of interest to report.
Footnotes
ACKNOWLEDGMENTS
This work is supported by the Parkinson’s disease foundation, Denmark and the foundation of July 2, 1984, Denmark.
