Abstract
Introduction
Nanotechnology is a research area in constant growth, and its application varies from electronics, biomedicine, environment, food, textile, and biomedicine. 1 Metallic nanoparticles are a large and important component of nanotechnology. High surface area, low melting point, and optical and magnetical properties are just some of the characteristics that make metal nanoparticles appealing in nanotechnology.2,3
Silver nanoparticles (AgNPs) have attracted intensive research interest because of their advantageous applications, not only in biomedical 4 but also in drug delivery, 5 food industries, 6 agriculture, 7 and textile industries. 8 AgNPs show a unique antibacterial property; silver components are toxic to any microorganisms. 9 It is well-known that cell death can be provoked by structural and morphological changes induced by AgNPs. 9 Other typical properties of AgNPs are optical, thermal, and catalytic. When excited by a light source, a collective oscillation is generated on the conduction electrons on the surface. High thermal conductivity, high electrical conductivity, and high thermal stability are some of the thermal properties of AgNPs. 10 High surface area is the main reason for the excellent catalytic properties of AgNPs and, similarly, to other types of nanoparticles.10,11 These characteristics are highly influenced by particle morphology and size. 12 High control over the morphological properties of final properties is achieved by employing chemical procedures that can have hidden dangerous side effects for human health. 13 Several efforts are placed into developing green alternatives for the production of nanocomponents.14–17
Biological methods of nanoparticles synthesis using microorganisms, enzymes, fungus, and plants have been suggested as alternatives to chemical and physical methods. 1 Plant-mediated nanoparticle synthesis is preferred as non-toxic, cost-effective, eco-friendly, and safe for human therapeutic use. 18 Furthermore, the use of plant extracts to synthesize nanoparticles is advantageous over microorganisms due to the ease of scale-up, the less biohazard, and the elaborate process of maintaining cell cultures. 19
Several plant extracts are present in literature references focused on synthesizing AgNPs.20,21
Materials and methods
Materials
AgNPs were synthesized from fresh leaves of
Preparation of M. paniculata
Fully-grown fresh leaves of
Synthesis of AgNPs
About 10 mL of aqueous extract of
Characterization of AgNPs
The reduction of pure silver Ag+ ions was monitored by measuring the reaction mixture’s UV-Vis spectrum at wavelength from 200 to 800 nm. Moreover, 1 m
Screening of antimicrobial activity for silver nanoparticles
The antimicrobial effect of synthesized AgNPs was tested against a few human pathogens, such as Gram-positive
Antioxidant potential of AgNPs
DPPH radical scavenging activity
To assess the scavenging ability on 2,2-diphenyl-1-picrylhydrazyl (DPPH), 1 mL of DPPH (0.1 mM) was added to different concentrations (50, 100, 150, 200, and 250 μg/mL) of aqueous leaf extract and AgNPs.
38
The reaction mixture was shaken and incubated in dark for 30 minutes. The absorbance at 517 nm was measured using a UV-Visible spectrophotometer. Ascorbic acid was used as a standard. The lower absorbance of the reaction mixture indicated a higher percentage of scavenging activity. The percentage of inhibition or scavenging of radicals (RSA) was determined by equation (1), where 
Reducing potential activity
The reducing power assay was conducted based on the method of Oyaizu (1986).
39
Different concentrations of aqueous extract (1 mL) and AgNPs were separately mixed with 2.5 ml of phosphate buffer (0.2
Cytotoxicity of silver nanoparticles on human umbilical vein endothelial cell line
Cell line and drug preparation
The human umbilical vein endothelial cell line (HUVEC) was obtained from NCCS (National Centre for Cell Sciences), Pune, India. The cells were cultured in Dulbecco’s Modified Eagle’s Medium (DMEM) supplemented with 10% fetal bovine serum (Hi-media) and 2 mM glutamine (Sigma chemical co), 100 UI/ml penicillin, and 100 μg/mL streptomycin (GIBCO BRL). The cells were maintained at 95% air humidity in a biological incubator at 37°C with 5% CO2; then, the viability was assessed using MTT assay. The stock solution was prepared in DMSO and was stored at −20°C until use. The concentrations used for the study were freshly prepared for each experiment with a final DMSO concentration of 0.1%.
Assessment of cell viability using MTT assay
The cell viability of the AgNPs from
Cell migration analysis using AgNPs
The cell migration of HUVEC was evaluated using a scratch wound assay. 40 In brief, 1 × 105 cells/ml were added into 24-well culture plates and cultured overnight. The cells were starved with 1% v/v FBS medium for 24°hours. The cells were then scraped with a cross in the middle of the well with 200°μl pipette tips, and the medium was changed with a fresh medium containing IC50 concentration of AgNPs. The cells were then incubated for 48°hours. The cells around the wounds were visualized and imaged under an inverted microscope (×4 magnification). The percentages of the open wound areas were estimated under a microscope.
Expression of vascular endothelial growth factor using western blot analysis
The western blot analysis was carried out to detect VEGF expression on endothelial cells. HUVEC (1 × 105cells/well) were seeded in a 24-well culture plate and incubated for 24°hours. Various concentrations (3.925–1000°μg/ml) of AgNPs were added to the well plates and incubated for 48°hours. After the treatment, cells were harvested and washed twice with PBS. The cell pellets were then lysed with RIPA buffer (50°mM Tris-HCl pH 7.4; 150 m
Statistical analysis
All data were expressed as mean and standard error derived by the repetition of each test at least three times. All the analyses were calculated using the Statistical Package of Social Sciences (SPSS) software package version 16.0 program using analysis of variance (ANOVA) to investigate the effect. Results with
Results and discussion
Properties characterization
AgNPs produced by several techniques and sources characterized by few main properties, such as morphology, chemical composition, crystallinity, and stability. 14 Determining these properties estimates the quality of the AgNPs and their applicability to different purposes.
Stability
One of the most important properties of nanoparticles produced by a green procedure is stability. The easiest procedure to verify if in a solution AgNPs are present would be performing a visual check. It is well-known that the optical properties of spherical AgNPs depend on the AgNPs diameter and the refractive index near the nanoparticles.
41
Moreover, unaggregated AgNPs usually show a yellow–orange color in the solution; however, when the particles aggregate, the color of the solution would appear grey.
42
In our project, the AgNPs are formed by adding different concentrations of extracts (5°ml, 10°ml, 15°ml, and 20°ml) with aqueous AgNO3 (1°mM). After 24°hours incubation, the colors of the solutions with different concentrations of (a) Synthesis of silver nanoparticles using different concentrations of aqueous extracts from leaves of M. paniculata and different concentrations of AgNO3. (b) UV-Vis spectrum of 10 mL of AgNPs solution at different times, 1 and 30 days.
For the solution containing 10°ml of MP extract, the AgNPs appear very stable even after 30°days. The UV spectrum does not show major alterations from one to 30 days of incubation, as shown in Figure 1(b). At 10 mL concentration of extract, the characteristic absorption peak is observed at 438 nm in UV-Vis spectrum which confirmed the formation of AgNPs and their stability on the 30th day (Figure 1 (b)). According to the generalized theory, only a single SPR band peak is expected in the absorption spectra of spherical nanoparticles, whereas anisotropic nanostructures or aggregates of spherical nanoparticles could give rise to two or more SPR bands depending upon the shape of the particles. 43
Morphology
The surface morphology of AgNPs was investigated at the microscale level, using FESEM with EDAX, and at the nanoscale level, using an HR-TEM. The formation of AgNP and its agglomeration can be observed at the microscale level (Figure 2(a)). The FESEM micrograph shows a spherical shape with a particle diameter that ranges from 5–23°nm. In particular, the average projected equivalent diameter
44
of AgNPs is 14 ± 9 nm. This number is validated by analyzing the sample at the nanoscale level. In Figure 2(b) and (c), it is clear that AgNPs produced by MP are spherical; the average aspect ratio is 1.09. Examples of AgNPs generated from M. paniculata extract. The image is shown in (a) is obtained using an SEM, in (b) and (c) using an HR-TEM. In (d), an example of an EDAX analysis on AgNPs produced from leaves of M. paniculata is reported.
Chemical composition
AgNPs, regardless of the procedure used for the production, are supposed to show a strong aluminum peak in X-ray spectra. As expected, the presence of silver in the EDAX spectrum confirms the presence of elemental silver. The only elements detected using a FESEM-EDAX are aluminum or copper, oxygen, and silver. Aluminum (Al) and copper (Cu) are due to the aluminum foil and copper TEM grids used as substrate, respectively. Considering the above-mentioned elements and oxygen, silver shows an atomic percentage of about 30% with a standard deviation of 2 when analyzing three samples. Figure 2 (d) shows an example of an EDX sample where silver peaks are typical; metallic silver nanoparticles typically showed an absorption peak at 3 keV due to the surface plasmon resonance. 45
Solid-state
The crystallinity of AgNPs generated from MPs is analyzed by observing SAED patterns and deconvoluting an XRD spectrum. In Figure 3(a) , the SAED patterns obtained by analyzing AgNPs produced from MP extract show a high crystallinity. The same conclusions can be reached by observing the XRD spectrum, Figure 3(b). Several Braggs reflections with 2θ values of 38.0°, 44.1°, and 64.6° sets of lattice plan were observed which may be indexed to (111), (200), and (220) facets of silver, respectively, confirms the FCC crystalline structure. The values are in agreement with the JCPDS (Joint Committee on Powder Diffraction Standard) file No. 04-078. SAED (a) and X-Ray diffraction (XRD) (b) analysis of AgNPs from M. paniculata. In (d), an example of FTIR analysis of AgNPs from leaves of M. paniculata is shown.
An FTIR measurement can determine the biomolecules responsible for the reduction and capping of synthesized AgNPs. The sharp and strong different peaks are observed at 3429°cm−1 (O-H stretch, H-bonded alcohols, phenols groups), 2923°cm−1 (C-H stretch alkanes), 1626°cm-1 (N-H bend 1° amine), 1583°cm−1 (C-C stretch in-ring aromatic), 1039°cm−1 (C-N stretch aliphatic amines), 728°cm−1 (C-Cl stretch alkyl halides), and 589°cm−1 (C-Br stretch alkyl halides), respectively (Figure 3(c)). The results indicated that the interaction of amino groups, keto, and aldehyde
46
is responsible for AgNP synthesis. These data indicate that the involvement of phenols, alkanes, amines, aromatic, aliphatic amines, and alkyl halides residues present in
Biological applications of silver nanoparticles
Antimicrobial activity
The synthesized AgNPs tested for their efficacy against human pathogens ( Antimicrobial activity of synthesized silver nanoparticles from leaves of M. paniculata.
The effects of AgNPs on bacterial cells are unclear and complicated. 49 However, there are various mechanisms on the action of AgNPs on the bacterial cell. 50 Some of these mechanisms were summarized and presented as follows: (i) the ability of AgNPs to anchor to the bacterial cell wall and then penetrate it , 51 (ii) the formation of free radicals by the AgNPs which can damage the cell membrane and make it porous ,52, 53 (iii) releasing the silver ions by the nanoparticles which can interact with the thiol groups of many vital enzymes and inactivate them, 54 and (iv) the nanoparticles can modulate the signal transduction in bacteria which stops the growth of bacteria. 55
All the human pathogens are resistant to standard drug ampicillin, but they are susceptible to the synthesized nanoparticle from leaves of
Antioxidant activity
In light of the difference among the wide range of assays available, a single antioxidant assay results can give only a reductive suggestion of the antioxidant potential. Moreover, the chemical complexity of samples with a mixture of different functional groups and chemical behavior could lead to a scattered result. Therefore, an approach with multiple assays in screening work is highly desirable. Thus, to better compare the results and cover a wide range of possible applications,
DPPH radical scavenging activity of AgNPs
Free radical scavenging activity is the most extensively used method to understand the potentiality of AgNPs toward their bioactivity. DPPH (purple) is a protonated radical which has a characteristic absorbance at 517 nm using a spectrophotometer, which decolorizes (yellow) upon activation with antioxidants. Various researchers use DPPH scavenging activity as a fast and reliable parameter to assess the In (a), DPPH radical scavenging activity of synthesized silver nanoparticles from aqueous extract of 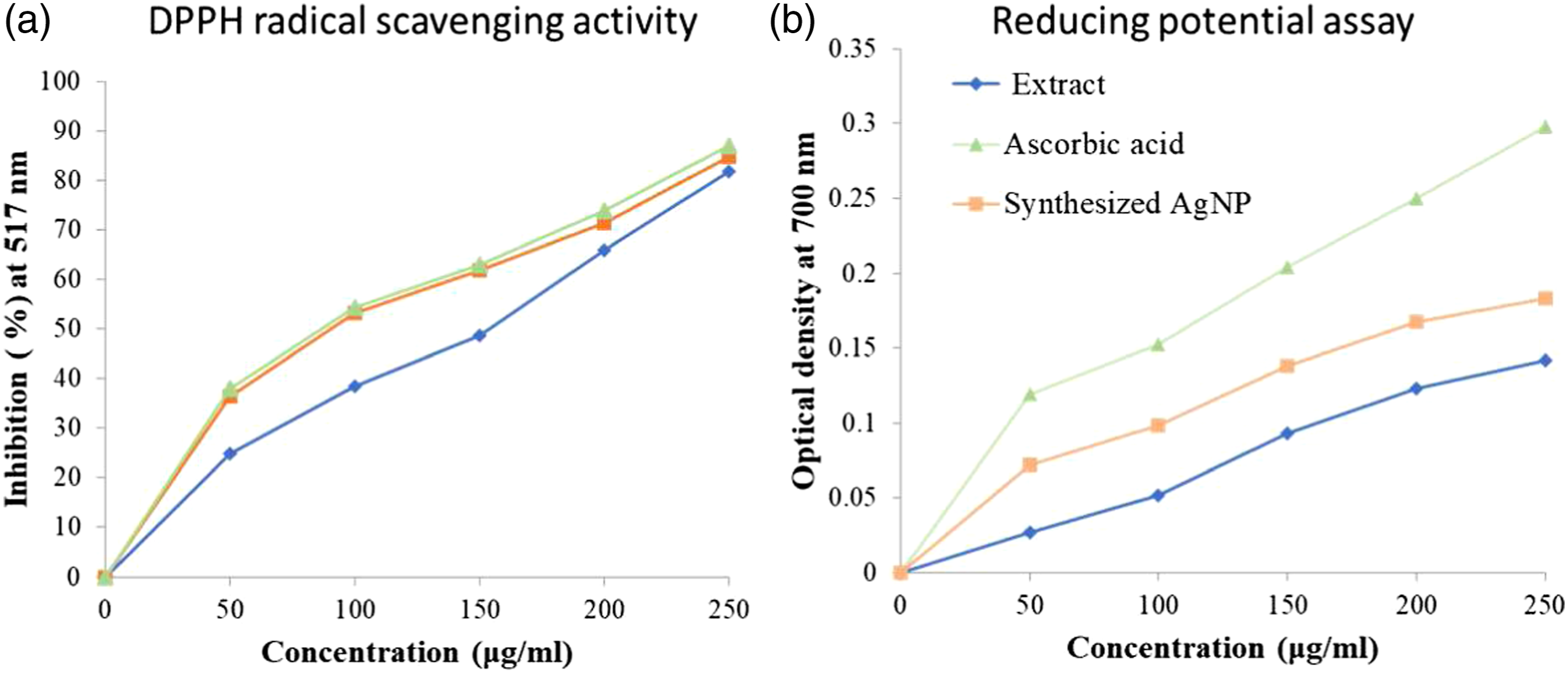
Reducing power assay using silver nanoparticles
Reducing power is associated with antioxidant activity, 56 and the compounds with reducing power indicate that they are electron donors and can reduce the oxidized intermediates of the lipid peroxidation processes; thus, they can act as primary and secondary antioxidants. The presence of antioxidants can result in the reduction of Fe3+ to Fe2+, and the amount of Fe2+ complex is monitored by measuring the formation of blue color at 700°nm. The increased absorbance at 700°nm indicates an increase in reductive potential.
The reducing potential of synthesized AgNPs was found to surge with an increase in concentrations, indicating that the reducing potential of AgNPs was found to be lesser than that of the ascorbic acid (Figure 5(b)). It is evident from Figure 5 (b) that the reducing powers of the aqueous extract of
Cell viability assay using silver nanoparticles
The viability of HUVEC cells is checked using different doses (1000–3.906°μg/mL) of AgNPs from leaves of Cell viability of HUVEC by MTT assay and cytotoxicity were expressed as the concentration of 50% (IC50) cell growth inhibition. The experiments were performed in triplicates. Images were taken by Inverted Phase contrast microscope magnification ×20 scale bar 50 μm.
Cell migration assay using AgNPs
Angiogenesis is associated with several pathologies including cardiovascular diseases, chronic inflammation, cancer, and wound healing, and depending on the circumstances, it can be beneficial and deleterious. To explore the angiogenic potential of AgNPs from Cell migration potential by scratch wound assay. Photos were taken 48 hours after scratch wounding at ×4 magnification.
Wound healing involves the regeneration of specialized cells by the proliferation of surviving cells and connective tissue response characterized by the formation of granulation tissue. 57 It is also characterized by hemostasis, reepithelialization, and remodeling of the extracellular matrix. Thus, the effect of ethanolic extract and the acetone fraction on wound contraction and epithelialization suggests it may enhance epithelial cell migration and proliferation, as well as the formation, migration, and action of myofibroblasts.
Expression of VEGF by western blotting analysis
VEGF is an important proangiogenic cytokine and improves angiogenesis during wound healing by stimulating the migration and proliferation of endothelial cells through the extracellular matrix.
58
The VEGF levels in HUVEC cells are upregulated after 24 hours of incubation (Figure 8(a)). The highest upregulation of VEGF protein is detected at the concentration of 15.625°μg/ml (Figure 8(b)). (a) Effect of silver nanoparticles from 
Angiogenesis is a critical component in wound healing. Delayed or absent revascularization at the wound site contributes to the etiology of chronic wounds. Induction of angiogenesis by VEGF can be considered as a factor to improve wound healing. Western blotting analysis of HUVEC upregulates the VEGF expression similar to that of β-actin.
Conclusion
Biosynthesizeing nanoparticles are very common and have been extensively used in various biomedical applications. We developed a simple green chemistry approach for the synthesis of AgNPs by
Footnotes
Acknowledgments
The authors greatly acknowledge the University Grant Commission (Grant Number GCCO/A-2/UGCBSR/2012/384 to P. Rama) and the Natural Sciences and Engineering Research Council of Canada (Grant Number RGPIN-2018–04735 to Anubhav Pratap-Singh) for financial support of this work. The authors are also thankful to the Director, CAS in Botany, University of Madras, Chennai, Tamil Nadu, India, for providing lab facilities.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
