Abstract
Silver nanostructures were successfully synthesized through a simple and “green” method using saccharides as reducing and capping agent. Transmission electron microscopy (TEM) and UV–Vis absorption were used to certify the quality of the silver nanoparticles obtained: firstly, size and dispersion.
In this work Silver NanoParticles (AgNPs) cytotoxicity related to saccharides capping (Glucose and Glucose-Sucrose) was explored in human epitheloid cervix carcinoma cells (HeLa). The cells were incubated with increasing AgNPs number/cell and HeLa cells viability was monitored for a period of 48 h compared with the positive and negative controls.
We observed that the toxicity increases with incubation time and with AgNPs number/cell. In particular, the different cytotoxic degree of the AgNPs, i.e. AgNP-G are more toxic than AgNP-GS, suggests that the cytotoxic effects are largely depended on the capping agent. The highest concentration of AgNP-G number/cell is able to induce extensive cell death of HeLa cells soon after 1 hr of incubation; conversely the lowest concentration of Ag NP-GS number/cell, surprisingly, is able to induce cell proliferation.
1. Introduction
Various types of nanomaterials exhibit new electrical, catalytic, magnetic, mechanical, photonic, and thermal properties to designnew device useful for a variety of scientific and technological sectors. The number of products containing nanomaterials and their possible applications continue to grow exponentially.
Nanomaterials are already being used or tested in a wide range of consumer products such as sunscreens, composites, and medical and electronic devices, and they are also being used as chemical catalysts.
Similarly to nanotechnology's success in consumer products and other sectors, nanomaterials also have promising environmental impact applications [1–3]. For example, nanosized cerium oxide has been developed to decrease diesel emissions, and iron nanoparticles can remove contaminants from soil and groundwater.
Most of the natural processes also take place in the nanometer scale regime. Therefore, a synergy of nanotechnology and biology could solve several biomedical problems and revolutionize the health and medicine fields[4].
Nanotechnology is currently employed as a tool to explore the darkest avenues of medical sciences in several ways like imaging [5], sensing [6], targeted drug delivery [7]. The new age drugs are nanoparticles of polymers, metals or ceramics, which can combat conditions like cancer [8] and fight human pathogens like bacteria [9].
There are, however, rising concerns about the adverse effects of these materials on human health and environment. Some nanomaterials have been reported to produce reactive oxygen species (ROS) and exert cytotoxicity
Data in literature suggest that silver coatings can be employedas antibacterialagent, for the treatment of wounds and burns or as a contraceptive and marketed as a water disinfectant and room spray [13].
Information on the toxicity of silver, which have been used hitherto as silver-impregnated dressing and pharmaceuticals, is available for a long time [14], and they are considered to have no adverse effects on the human body when they used in reasonable amounts. However, despite widespread use, there are still few data concerning the toxicity of Ag-NPs to humans at the cellular and molecular level [15–17].
Recent evidences report that Ag-NPs are highly toxic to various cultured cells: Ag-NPs exposure decreases viability, increases lactate dehydrogenase (LDH)-leakage, or inhibites mitochondrial function in rat liver cells [18], human fibroblasts [19], and rat adrenal cells [20]. Furthermore, Kim et al. [21] demonstrated the dose-dependent changes of alkaline phosphatase and cholesterol values, which might be a consequence of liver damage, in either male or female rats after 28-day oral exposure to Ag-NPs.
However, most of the studies evaluated the acute toxic effects of Ag-NPs at relatively high doses [22]. It is required to evaluate the chronic toxicity at low doses, which could be developed by the prolonged internal exposure because Ag-NPs may remain in target organs for a long time.
The toxicity of Ag-NPs at molecular level has not been reported yet so far.
In this study, we synthesize silver nanoparticles capped with two different glycans. Therefore, we investigated the possible toxic effects of different amounts of two glycans capped Ag-NPs on human epitheloid cervix carcinoma cells (HeLa) in terms of cell viability and morphology.
2. Materials and Methods
2.1 Chemicals
Silver nitrate (AgNO3, 99%), sucrose (99%), and α-D-Glucose (96%) was from Sigma-Aldrich and used as received. Deionized ultra-filtered water prepared with a Milli-Q water purification system was used throughout the experiments.
All glassware was washed by ultrasonication in a mixture of deionized water and nonionic detergent, followed by thorough rinsing with Millipore water and ethanol for many times to get rid of any remnants of nonionic detergent and dried prior to use.
2.2 Synthesis of silver nanoparticles
Silver nanoparticles were prepared by a simple wet chemical method. A series of solutions were prepared dissolving 5 g of saccharides in 100 ml water and brought to boiling for some minutes; then 2.5 ml (102 M) of AgNO3 solution was added to it.
In the first preparation (AgNP-G), 2 ml aliquot of a 10−2 M aqueous solution of AgNO3 was added to 100 ml of 0.3 M aqueous β-D glucose solution.
In the second preparation (AgNP-GS), 2 ml aliquot of a 102 M aqueous solution of AgNO3 was added to 100 ml of 0.15 M β-D glucose and 0.15 M sucrose aqueous solution.
The mixture was kept on boiling for 30 minutes under vigorous stirring. After sometime, the colour of the solutions turned pale yellow, indicating the formation of silver nanoparticles.
2.3 Instrumentation
2.3.1 UV-vis spectroscopy
UV-visible spectra were recorded in the range between 320 and 800 nm using a T80 PG Instruments Ltd spectrophotometer. Optical spectra where obtained from freshly prepared solution and then within 30 days by measuring the absorption of the prepared as in a quartz cuvette with a 1 cm optical path.
2.3.2 TEM observations
Transmission Electron Microscopy (TEM) images and electron diffraction patterns were taken using an Hitachi 7100, at 100 KV representing the suitable acceleration voltage to obtain a good resolution and minimal radiation damage of the material. The specimens were prepared for transmission electron microscope observations by placing small droplets of NP solutions onto standard carbon supported 600-mesh copper grid and drying slowly in air naturally.
2.4 Cell culture
Human epithelioid cervix carcinoma cells (HeLa) were cultured in Eagle's minimum essential medium (EMEM) (Cambrex, Verviers, Belgium) supplemented with 10% fetal calf serum (FCS), 2 mM L-glutamine (Cambrex, Verviers, Belgium), 100 IU/ml penicillin and streptomycin solution (Sigma, St. Louis, MO) and 10000 U/ml nystatin (antimicotic solution) (Cambrex, Verviers, Belgium), in a 5% CO2 humidified atmosphere at 37°C. Cells were maintained in 75 cm2 flasks (concentration ranged between 2 × 105 and 1 × 106 cells/ml) by passage every 3 to 4 days.
2.5 Experimental design
15×104 HeLa cells were incubated for 24 and 48 hrs with different concentrations (50, 100, 150, 200, 250 and 500 μl/3 ml of culture medium) of AgNPs solutions (AgNP-G and AgNP-GS) corresponding to increasing AgNPs/cell number and different Ag and saccharides concentrations as summarized in table 1.
Ag, Ag/cell, NP/cell and saccharides concentrations used in cytotoxicity experiments

2.6 MTT cell viability assay
3- (4, 5-dimethylthiazol-2-yl)- 2, 5- diphenyltetrazoliumbromide (MTT) assay (MTT 98% Sigma–Aldrich) that is a cytotoxicity method, was performed according to the modified MTT method described by Sladowski et al [23]. After 24 and 48 hrs of incubation with in the nanoparticles, cells were extensively washed with 0.1 M phosphate buffer (PBS) pH 7,4 and then incubated with 1 mg/ml MTT in culture medium for 2 hours. After a further extensive washing in PBS, living cells were determined by MTT dye reduction. The amount of MTT-formazan produced is directly associated with cell viability and it can be determined spectrophotometrically (DU 640 B, Beckman Coulter, USA) at 570 nm after solubilization of crystals in 1 ml of DMSO.
Morphological evaluation of HeLa cells was performed with a light inverted microscope Eclipse TS100 (Nikon, Tokyo, Japan).
In control experiments, HeLa cells were cultured in the presence of saccharides solutions (Glucose or Glucose-Sucrose) or with 1.35 (μg) and 13.5 (μg) AgNO3. 1% Triton-X100 was used as a positive control.
2.7 Statistical Analysis
The two-tailed Student's t-test was used to analyze differences between controls and treated samples. Data are presented as mean value ±SD and the differences were considered significant at 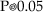 .
.
3. Results and discussion
It is well known that the main feature of the absorption spectra for metallic nanoparticles is the surface plasmon (SP) resonance bands. From one up to three SP bands can be observed corresponding to three polarizability axes of the metallic nanoparticles.
The optical properties of metallic nanoparticles ranging from microclusters to nanoparticles have been investigated mainly on the size effects concerning the shift of the SP resonance and the variation of the SP bandwidth. However, it has been demonstrated both theoretically and experimentally that the SP resonances of metal nanoparticles mainlydepend on the particle shape than on the size [24].
The UV-visible absorbance spectra of nanoparticles in aqueous β-D glucose solution and nanoparticles in aqueous β-D glucose-sucrose solution, displayed in Figure 1 (a) and (b) respectively, presented a strong extinction band with a maximum at 420 nm, characteristic for the spherical nanoparticles.

UV-visible absorbance spectra of AgNPs-G solution (picture a) and AgNPs-GS solution (picture b) obtained from freshly prepared solution, after 4 and 30 days
Typical transmission electron microscope images performed on freshly prepared solutions are shown in Fig. 2.

Transmission electron microscope images performed onto freshly AgNPs-G (a) and AgNPs-GS (b) preparations
It is evident that, in both cases, the nanoparticles were mostly spherical and well-dispersed. To determine the nanoparticles size distribution a lot of digitalized TEM images, performed onto 50 randomly chosen fields, were processed by Image Pro-Plus software. The observed nanoparticles, both for AgNP-G and AgNP-GS preparations, have a size distribution ranging from 5 to 40 nm and an average size d = 30 nm with a standard deviation σ = 5 nm.
Typical histograms showing the size dispersion of observed nanoparticles are reported in Fig.3.
Histogram of silver nanoparticles size distribution for AgNP-G (picture a) and AgNP-GS (picture b) together with the Gauss curve fitting
After 4 days, a significant difference in extinction spectra was observed. A second absorption band appears on the red side of the spectra, in the 600-700 nm range. This behavior is more evident in the aqueous β-D glucose solution (Fig. 1a). In addition, nanoparticles in aqueous β -D glucose-sucrose are more stable than nanoparticles in aqueous β-D glucose: after 30 day the shape of spectrum in Fig.1b is not significantly changed.
Established theoretical descriptions of Mie scattering from small aggregate clusters suggested that the plasmon resonance absorption of the linear aggregates would have an additional long wavelength component in the optical absorption spectrum relative to the absorption from isolated nanoparticles dispersed in solutions [25].
According to this theory, this new long wavelength band could be associated with the longitudinal mode of the electronic plasma oscillation along the long axis of nanoparticles chains. However, the absorption spectra of the solution after 4 day are also the typical spectra of nanorods, which are characterized by a dominant SP band at longer wavelength corresponding to the longitudinal resonance and by a much weaker transverse resonance at shorter wavelength [26].
The TEM images confirm the previous results. Figure 4 shows a tipical example obtained from nanoparticles in aqueous β-D glucose solutions after 4 days.

Tipical transmission electron microscope image obtained from AgNPs –G after 4 days
The cytotoxicity of AgNPs in aqueous β-D glucose (Fig. 4) and β-D glucose-sucrose (Fig. 4) solution was
Both for AgNPs in aqueous β-D glucose and β-D glucose-sucrose solution, the analyses showed a direct dose-response relationship, i.e. cytotoxicity increases with the AgNPs/cell number and incubation time.
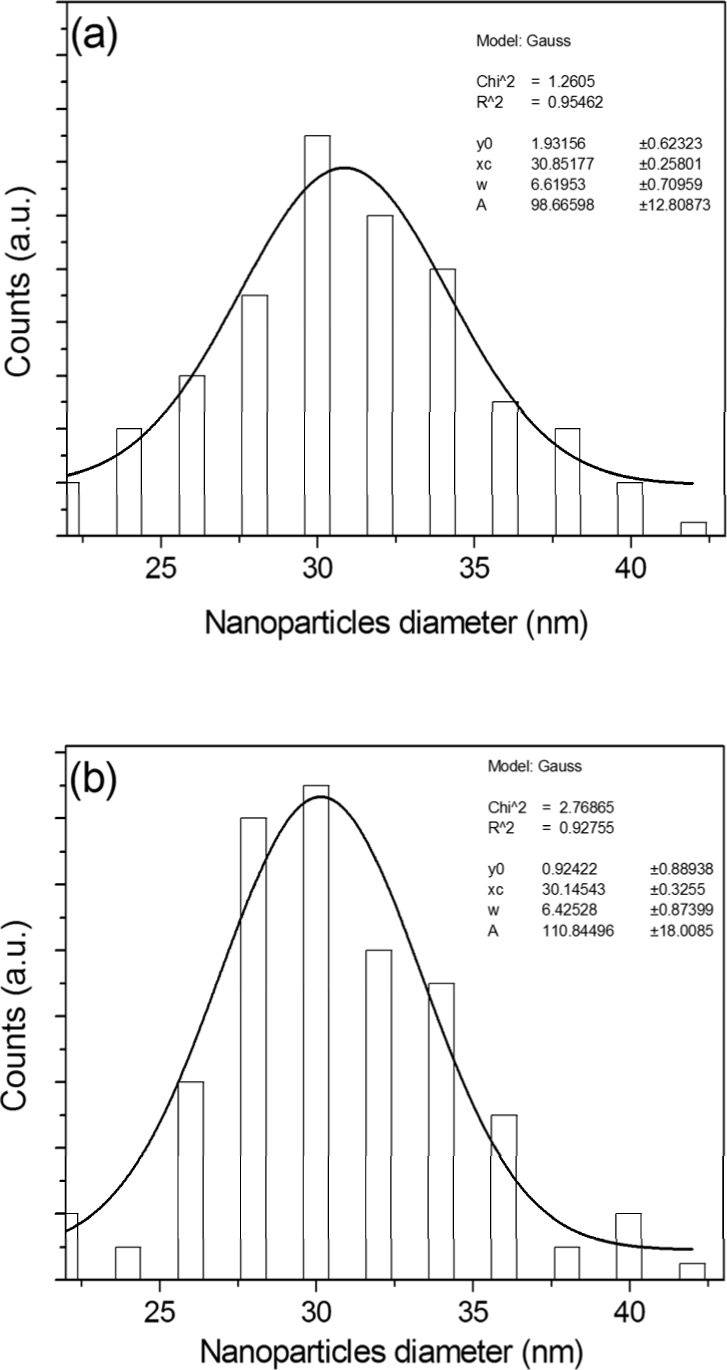
The AgNPs-G are more toxic than AgNPs-GS. In fact, AgNPs quantities necessary to produce 50% of cell viability decrement were 50 μl at 24 hrs for AgNPs-G (Fig. 5A) and 250 μl at 48 hrs for AgNPs-GS (Fig. 6A).

MTT assay performed in HeLa cells after incubation with AgNP-G (50, 100, 150, 200, 250 and 500 μl/3 ml of culture medium) for 24 and 48 hrs. Values (absorbance reported as percentage respect to the untreated cells at 0 hr of incubation, considered as 100%) are the average ± SD of three independent experiments.
MTT assay performed in HeLa cells after incubation with AgNP-GS (50, 100, 150, 200, 250 and 500 μl/3 ml of culture medium) for 24 and 48 hrs. Values (absorbance reported as percentage respect to the untreated cells at 0 hr of incubation, considered as 100%) are the average + SD of three independent experiments. All values of AgNP-GS are significantly different (p<0.05) with respect to the control value. The Ag concentrations related to different solutions volume used in experiments are reported in Table I.
Surprisingly, the lowest quantity of AgNP-GS (50 μl) induces cell proliferation of about 20% as suggested by increment of cell viability (Fig. 6A) and confirmed by anti-Phospho H3 immunostaining (Data not shown).
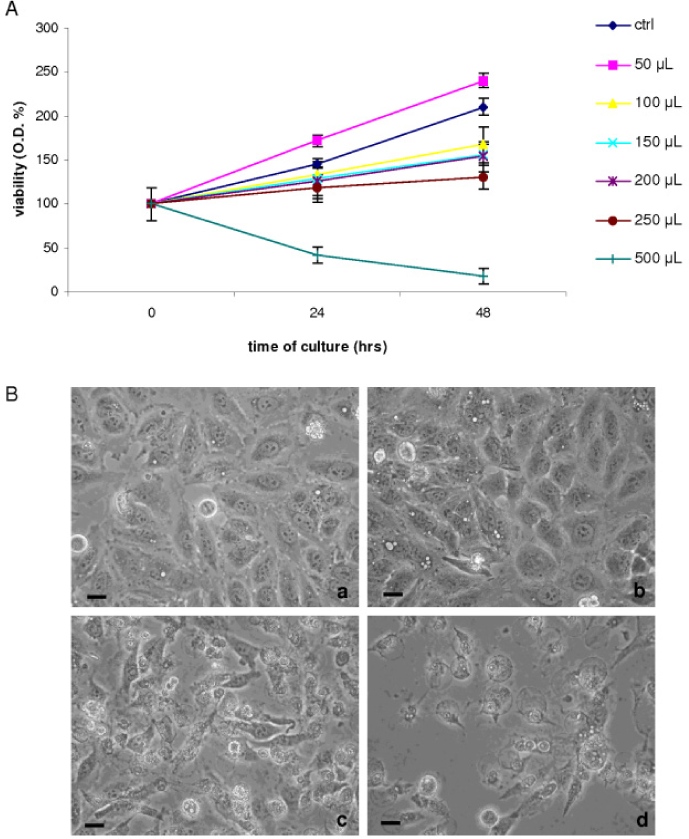
The cellular changes upon exposure to AgNPs were also monitored at light inverted microscope (Fig. 5B, 6B). Both with the highest AgNPs-G and AgNPs-GS number/cell (as reported in table 1), HeLa cells detach from substrate and show morphological features of cell death soon after 1 hr of culture in the presence of nanoparticles.
All values of AgNP-G are significantly different (p<0.05) comparing with the control value. The Ag concentrations related to different solutions volume used in experiments are reported in Table I.
a) 24 hrs of culture in the presence of 50 μl of AgNP-G;
b) 48 hrs of culture in the presence of 50 μl of AgNP-G;
c) 24 hrs of culture in the presence of 500 μl of AgNP-G;
d) 48 hrs of culture in the presence of 500 μl of AgNP-G. Bars = 10 μm
In order to verify if silver ions could have a particulate effect on HeLa cells, a further check was made by adding silver ions to the culture medium, doing so the silver amount is the same to colloidal solutions ones.
Results suggest that silver ions are more toxic than glycans capped nanoparticles. Indeed, cell viability is rapidly affected by the highest concentration (13.5 μg) of Ag ions. 50% of cell death is reached as fast as after 15 min of incubation.
a) 24 hrs of culture in the presence of 50 μl of AgNP-GS;
b) 48 hrs of culture in the presence of 50 μl of AgNP-GS;
c) 24 hrs of culture in the presence of 500 μl of AgNP-GS;
d) 48 hrs of culture in the presence of 500 μl of AgNP-GS. Bars = 10 μm
At 4 hours of incubation with 13.5 μg of Ag ions all cells died (Fig. 7). Conversely, cell viability is not affected by the presence of saccharides only, i.e. glucose and saccarose-glucose, neither at 24 or 48 hrs. These results strongly suggest that cytotoxicity depends on the NPs presence.

Light inverted microscope micrographs of HeLa cells incubated with 13.5 μg of Ag.
a) 30 min of culture;
b) 1 hr of culture;
c) 4 hrs of culture;
d) 24 hrs of culture.
Bars = 10 μm
4. Conclusions
In this paper we have shown that AgNP-G are more toxic than AgNP-GS and the toxicity increases with the incubation time and the AgNPs number/cell. In HeLa cells incubated with the highest concentration of AgNP-G or AgNP-GS, cell death is observed as soon as after 1 hr. Thus, cytotoxicity of AgNPs is exerted also on tumor cells, that in this context behave as normal cells when challenged with AgNPs [27,28].
Surprisingly, the lowest concentration of Ag NPs-GS number/cell induces cell proliferation of about 20%.
These results can be explained according to Kawata et al [22] suggesting that metal ions, including silver act as a catalyst and exhibit the ability to produce toxic reactive oxygen species (ROS) in the presence of oxygen species. Recent studies indicate that Ag-NPs increase production of intracellular ROS [29]. The ROS can act as signal molecules to promote cell cycle progression by affecting growth factor receptors, AP-1, NFkB, and soon induce the oxidative DNA damage. These mechanisms have been speculated to play important roles in carcinogenesis and tumor progressing actions of carcinogenic chemicals.
The knowledge of the interaction of nanoparticles with cells is crucial to address the engineering of nanostructured materials towards biocompatibility. All this could be useful in the design of new therapeutic strategies as for example in the drug delivery of anticacncer compounds as seen from the very recent novel therapy like PDT.
