Abstract
We demonstrate the synthesis of silver nanoparticles by a potentially benign species of bryophilous
1. Introduction
Nanoparticles (NPs) are defined as having one dimension 100nm or less in size and due to their large surface area they tend to react differently than larger particles of the same composition, allowing them to be utilized in novel applications. Silver NPs are utilized for their optical, antibacterial, electrochemical and catalytic properties [1, 2]. Unfortunately chemical synthesis of silver NPs utilizes environmentally toxic or biologically hazardous reducing agents [3], so there has been a search for greener production alternatives [1, 2, 4–6].
Many microorganisms, plant extracts and fungi have been shown to produce NPs through biological pathways [1–4, 6, 7]. Synthesis of silver NPs has been investigated utilizing many ubiquitous fungal species including
Unfortunately, most fungal species that were recently studied (
Bryophytes are known to uptake and retain metal particles from their environment and many bryophytes have been shown to be metal-tolerant [14, 15]. Bryophyte extracts from
Our objective is to determine if an endophytic bryophilous fungus with sterile mycelium can synthesize metallic silver NPs in two different media; one that promotes and one that represses nitrate reductase. We will evaluate the subsequent NPs on their morphological characteristics, chemical composition and optical properties.
2. Experiment
2.1 Fungal Isolate, Growth and Synthesis of AgNPs
Isolate A3 (
The AgNO3+CN and Ag2SO4+CN media is consistent with the carbon/nitrogen ratio of Spezieller Nährstoffarmer agar, while utilizing the same concentration of silver ions as reported by other researchers [8, 10, 13]. The AgNO3+CN media supports fungal growth and supports the expression of nitrate reductase, the enzyme reported to be implicated in the production of silver NPs. Nitrate is an inducer of nitrate reductase and the carbon/nitrogen ratio supports better growth than a non-carbon containing medium. The Ag2SO4+CN media also supports fungal growth, but the nitrogen source (L-glutamine) represses the production of nitrate reductase, whereby silver NPs would need to be produced by other means.
An agar plug of actively growing mycelium was antiseptically placed into an autoclaved Erlenmeyer flask that contained 300ml of initial growth medium (8g Nutrient Broth [Difco] + 4g Yeast extract [Difco] + 10g dextrose [L−1 distilled water]) and placed on shake culture (123rpm at ca. 25°C) for 14 days. Under a laminar flow hood, fungal colonies were gravimetrically separated from the initial growth medium using autoclaved Whatman #1 filter paper. Approximately 20ml of autoclaved water was used to wash the excess growth medium from around the fungal mycelia. After three consecutive washes, sterile forceps were used to transfer ca. 5.0g (wet weight) of the washed isolate to 4 sterile Erlenmeyer flasks, each containing one of the following media: AgNO3 medium consisting of a fixed carbon/nitrogen (CN) ratio to support fungal growth and up-regulation of nitrate reductase (AgNO3+CN: 1.0mM AgNO3 + 0.83 g KNO3 + 0.2g glucose + 0.2 g of dextrose, L−1 distilled water) or Ag2SO4 medium, which consisted of the same fixed CN ratio to support fungal growth and L-glutamine to induce the repression of nitrate reductase (Ag2SO4+CN: 1.0mM Ag2SO4 + 1.0g L-glutamine + 0.2g glucose + 0.2g of dextrose, L−1 distilled water). The inoculated Erlenmeyer flasks were returned to shake culture for 96 hours at room temperature, at which time the production and characteristics of the silver NP formations were assayed.
2.1.1 Centrifuge procedure for AgNP isolation
Two cultures, one cultured in Ag2SO4+CN and one cultured in AgNO3+CN, were sonicated at 10W ultrasound with a cell disruptor to disrupt the mycelia, while another non-sonicated set was used as a control. The control test tubes were placed in a Clay-Adams centrifuge and spun at 2800rpm. The supernatants were collected and set aside (silver ion containing solution). Sterile water (10ml) was added to each control test tube containing the mycelium, shaken vigorously by hand for 30 seconds and spun at 15000rpm (30,996 g) for 10 minutes in a Beckman Model J2–21 centrifuge. The supernatant was collected in a separate clean test tube (unaltered wash solution).
The sonicated cultures were spun at 15000rpm (30,996g) for 10 minutes in a Beckman Model J2–21 centrifuge. The supernatant was collected in a separate clean test tube (silver ion containing solution). To each pellet, 10ml of sterile water was added, followed by vigorous hand shaking for 30 seconds to re-suspend the pellet. The test tubes containing the re-suspended pellets were centrifuged for a second time at the same settings. The supernatant was collected in a separate clean test tube (sonicated wash solution). The pellet was re-suspended in 10ml of sterile water (pellet solution) and all samples were assayed.
2.1.2 Media evaluation
The
2.1.3 Analysis
Imaging was carried out using a Vega TC SBII SEM equipped for simultaneous elemental analysis with an X-ray detector (X-MAX from Oxford Instrument) for Energy-Dispersive X-ray spectroscopy (EDX). Particle size was measured using Vega TC software. The optical transmission was done using a spectrograph USB2000 from Ocean Optics coupled with a Xenon light source.
3. Results and discussion
3.1 Media evaluation
Both media promoted good fungal growth for the

Isolate A3 growth on nitrate (left) and L-glutamine (right) evaluation media after 4 days at 25°C.
3.2 Silver NP characteristics
The bryophilous

SEM image of bryophilous isolate A3 (
No significant change in the culture supernatant colour was observed in both media. This was in contrast to the appearance of a brown colour change in some previous fungal studies [8, 9], which was attributed to extracellular silver NP formation. The lack of supernatant colour change could have occurred due to the biosorption capacity of fungal mycelium to bind extracellular metals to their cell wall components [19, 20]. It is hypothesized that extracellular enzymatic NP formation occurred in the AgNO3+CN medium, as reported by [8, 9], followed by extracellular precipitation and subsequent NP binding to the hyphal cell wall. Our optical analysis of the various supernatants support this as the most likely conclusion since plasmon resonance was only detected in the wash solutions that were obtained after vigorous hand shaking of the washed fungal mycelia in distilled water.
Figure 3 demonstrates subglobose to broadly ellipsoidal silver NPs, ca. 25–50nm in width, embedded within a matrix, which were produced in the Ag2SO4+CN medium, while Figure 4 demonstrates the morphology of the silver NPs produced in the AgNO3+CN medium. They are of similar size and shape, but appear to have a different capping formation.

Silver NPs from Isolate A3 Ag2SO4+CN wash solution demonstrated that they are made up of individual NPs that appear to be embedded in a matrix.

Silver NPs from Isolate A3 AgNO3 +CN wash solution demonstrated that they are made up of individual NPs embedded within a matrix.
Larger silver particles within isolate A3 unaltered wash solutions where visualized as conglomerations that were on average 50 to 250nm. A conglomerate configuration [Figure 5] that was only visualized in the Ag2SO4+CN medium suggested their formation was created by nucleation centres on the hyphal cell wall.

Plate-like silver conglomerations that appear to be site of nucleation on
3.2 Elemental analysis of A3 (Rhizoctonia sp.) wash solutions
The elemental analysis clearly demonstrated that the bright objects in the SEM images are silver NPs and no silver was detected outside of them. The following elements were also routinely detected: carbons (C), chlorine (Cl), nitrogen (N), oxygen (O), phosphorous (P), potassium (K) and sulphur (S). After both sonicated wash solutions were annealed at 200°C for 3 hours in air, the EDX analysis did not indicate chlorine in any of the targeted NPs for the AgNO3+CN wash solution. Chlorine was still detected by EDX analysis after the same annealing time for the targeted NPs in the Ag2SO4+CN wash solution. For both unaltered samples and both wash solution samples, silver was always present when chlorine and sulphur were detected. Nitrogen, phosphorus and excessive carbon were also present for many silver-containing NPs [Table 1].
Energy-dispersive X-ray spectroscopy analysis of isolate A3 silver NPs.
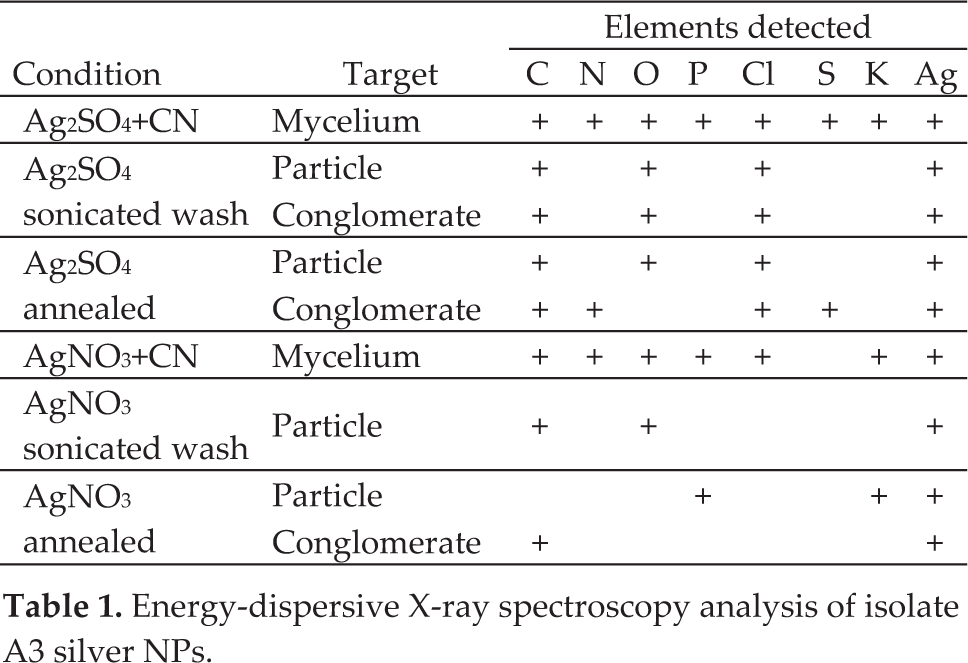
From the EDX analysis, we concluded that silver NPs produced using the AgNO3+CN medium produced silver NPs with less associated chemicals than by using the Ag2SO4+CN medium.
3.3 Optical transmission properties of A3 (Rhizoctonia sp.) solutions
The metallic state is characterized with charge carriers mobile enough to respond to electromagnetic fields. The measurement of this response is widely used for contactless detection of free charge carriers. In the case of NPs, their size and shape lead to formation of characteristic resonances, or absorption bands, which happen to be in the near-UV and visible spectral range for silver and gold NPs [22, 23]. Therefore, near-UV and visible light is a fine and precise probe for the detection of free charge carriers (resp. metallic state) in silver NPs and it has been widely used in the literature [6–9, 21–23]. Even a surface capping layer or an embedding dielectric matrix is not an obstacle to the detection of characteristic absorption bands, resp. the presence of metallic state.
Optical transmission measurements of the A3 unaltered wash solution from both media demonstrated a broad absorption band from 325nm to 600nm, with maximum absorption at 480nm [Figure 6]. Isolate A3 sonicated wash solutions from both media demonstrated a narrower absorption band, which shifted to shorter wavelengths with maximum absorption at 415nm. Such characteristic absorption bands for metallic silver NPs were not observed in any of the silver ion containing solutions for both media.

UV-vis absorption spectra for A3 unaltered supernatant wash and sonicated wash solutions after 96 hours.
The position of the narrower absorption bands in both A3 sonicated wash solutions were in agreement with the expected wavelengths [22, 23] for silver NPs within the size range of 25–50nm, which is also in agreement with the SEM images shown in Figures 2–5. The A3 unaltered wash solution obtained from both media contained larger NPs leading to maximum absorption at longer wavelengths, while the A3 sonicated wash solution from both media shows absorption peaking at shorter wavelengths. The silver NPs in the unaltered wash solutions from both media have a broad size distribution, as evidenced by the broad absorption extending in the UV range. These experimental observations demonstrate that the bio-synthesized silver NPs from both media contain enough mobile charge carriers to generate the characteristic plasmon resonance.
4. Conclusions
We have unequivocally demonstrated that a bryophilous
Footnotes
5. Acknowledgments
This work was made possible by a Lock Haven University Faculty Professional Development grant for promoting undergraduate research and by NSF grant #0923047. The authors would like to express their gratitude to Dr. Andrew Miller for technical support for the identification of the isolate.
