Abstract
This study reported the synthesis of aqueous dispersions of highly stable silver nanoparticles (AgNPs) using gamma radiation with gum acacia (GA) as the stabilizing and protecting agent. The formation of nanosized silver GA-AgNPs at a gamma irradiation dose of 15 kGy was confirmed by the appearance of the characteristic surface plasmon absorption peak at 456 nm and in UV–vis spectra. The particle size distribution of silver nanoparticles that was determined by Dynamic Light Scattering (DLS) was approximately 22 nm. By using Transmission Electron Microscopy (TEM), mostly spherical GA-AgNPs particles with a mean diameter of 16.7 nm and crystalline in shape were confirmed by an X-ray diffraction (XRD) pattern. Fourier Transform Infrared (FTIR) spectroscopic analysis indicates the bonding of AgNPs with the COO- group of gum acacia. To increase our understanding of the mechanisms involved in plant responses to GA-AgNPs and AgNO3, in order to differentiate between particle-specific and ionic-silver effects, we determined the response of vegetative growth and the yield of two varieties (Bronco and Nebraska) of the common bean (Phaseolus vulgaris L.) to foliar applications of silver nanoparticles (GA-AgNPs 16.7 nm) and silver nitrate (AgNO3) each at 0.0, 5, 10, 20 and 60 ppm.
The foliar application of GA-AgNPs and AgNO3 significantly increased plant height, root length, number of leaves/plant, the leaves' area, total fresh and dry weights/plant and yield (i.e., number, fresh and dry weights of pods/plant and 100-seed weight). It also altered protein patterns in the two varieties and changed the phytohormone balance in the Nebraska variety by increasing the levels of growth-regulating substances, which explains the increase in both growth parameters and yield in GA-AgNPs and AgNO3 treated plants. An Atomic Absorption Spectroscopy (AAS) study unveiled the movement and residual accumulation of both forms of silver in different parts of the two bean varieties.
The results indicate the successful use of GA-AgNPs and AgNO3 in enhancing the growth and yield of the Bronco and Nebraska varieties under pot experiment conditions, and present a viable alternative to genetically modified (GM) crops for ensuring food security.
1. Introduction
Nanotechnology has become a growing concern in recent years, with the unlimited production of nanoparticles (NPs) and their successful applications in broad fields. Nanoparticles are atomic or molecular aggregates with at least one dimension that is approximately between 1 and 100 nm [1,2], which can drastically modify their physico-chemical properties as compared to the bulk material [3]. It is worth noting that nanoparticles can be made from a full variety of bulk materials and that they can explicate their actions, depending on the chemical composition, size and shape of the particles [4]. Nanoparticles are synthesized by chemical reduction [5], electrochemical reduction [6], photochemical reduction [7], gamma ray irradiation [8-13], UV irradiation [14] and microwave radiation [15]. In addition, ultrasonic organic solvents have been used for the green synthesis of nanoparticles, with water in particular being an important solvent for bioreduction [16].
Green synthesis is an alternative harmless and environmentally friendly method for producing nanoparticles [17]. Natural polymers such as sodium alginate [18], carboxymethyl cellulose sodium [19], heparin and extract of Memecylon umbellatum leaf have been used for the capping of silver and gold nanoparticles [20, 21]. Among the natural polymers, gum acacia (also known as gum arabic) is a well-known polysaccharide obtained from the stems and branches of the Acacia Senegal tree. Gum acacia is used as a stabilizer for capping silver nanoparticles mainly because it is a naturally water-soluble material and is non-toxic. Gum acacia-stabilized AgNPs offer compatibility for pharmaceutical and biomedical applications; the widespread availability of the naturally occurring polysaccharide makes this process amenable to large-scale industrial production and it has excellent emulsifying properties [22]. On the other hand, gamma irradiation has been proven to be a simple and efficient method for silver nanoparticles' synthesis. It requires an aqueous system, room temperature and ambient pressure [10-13, 23]. The Surface Plasmon Absorbance (SPA) properties of nanoparticles bear a direct relationship to the sizes, shapes and chemical compositions of nanoparticles. Shifts in intensity and wavelength of the SPA absorption spectrum followed the growth in particle size [24].
Common bean (Phaseolus vulgaris L.), is an herbaceous annual plant that is distributed worldwide for its edible fruit. The leaf is used as a vegetable and the straw for fodder. Beans are an important food among the Fabaceae family for people of all income categories as a source of dietary protein, vitamins, fibre, complex carbohydrates [25] and bioactive compounds with antioxidant capacities [26]. Higher plants interact strongly with their atmospheric and terrestrial environments, and are expected to be affected as a result of their exposure to nanoparticles due to the increase in the production of synthetic nanoparticles (NPs). Silver is an effective plant-growth stimulator, including silver nitrate (AgNO3), silicate and a water-soluble polymer [27]. Silver eliminates unwanted microorganisms in farmers' soils and hydroponics systems. It is being used as a foliar spray to prevent fungi, rot moulds and several other plant diseases. Moreover, silver ions in AgNO3 salt have been recognized to inhibit ethylene action [28]. The antimicrobial properties of silver have made it a major ingredient in several agricultural products and also in detergents, plastics, and textiles [29,30]. However, significant inputs of NPs on plants' foliage may be expected due to the deposition of atmospheric particles or the application of NP-containing pesticides [31]. Some plants are capable of taking up and accumulating engineered nanomaterials (ENs). The interaction of plant cells with the ENs leads to the modification of plants' gene expression and associated biological pathways, which eventually affect plants' growth and development [32,33]. The interactions of nanomaterials with plants have not been fully elucidated. There are different and often conflicting reports on the absorption, translocation, accumulation, biotransformation and toxicity of NPs on various plant species.
The impact of AgNPs on higher plants appears to depend on the species, the age of plants, the size and concentration of the particles and the experimental conditions [32]. AgNPs reduce seed germination in Hordeum vulgare and shoot length in Linum usitatissimum and Hordeum vulgare at levels of 10 mg [34]; increased root elongation of Eruca sativa seedlings occurs at 10 mg Ag L−1 of either PVP-AgNPs or AgNO3 [35]. A significant enhancement in the growth of Brassica juncea seedlings was recorded at 25 and 50 ppm silver nanoparticle [36]. However, AgNPs have no significant effect on seed germination in Cucumis sativus and Lactuca sativa at 100 ppm [37] or the seedling growth of castor beans, even at 4,000 mg L−1, while the silver in bulk form inhibited seed germination [38]. On the other hand, a rise in the concentration of nanosilver from 20 to 60 ppm improved the seed yield in Borage [39], increased the leaf surface area, shoot and root lengths in Phaseolus vulgaris and Zea mays [40] and rewarded the effects of flooding stress on saffron corms by increasing root numbers and increased leaves' dry weight at 80 ppm [41]. Moreover, the spraying of silver ions decreased the flower abscission of Alstroemeria plants by 100% within the two first days [42] and decreased the flowers and flower buds' abscission in orchid plants [43]. The mechanisms that mediate the effects of AgNPs on plants remain unknown. It is important to increase our knowledge of these mechanisms before implementing a large-scale agricultural utilization of AgNPs. Only a few studies are available on the effects of silver nanoparticles on higher plants.
The present study aimed at the synthesis of highly stable and densely dispersed silver nanoparticles, by using the gamma-radiolytic reduction method and gum arabic (GA) as the stabilizer. In addition, it aimed to characterize the growth response and yield of two varieties (Bronco and Nebraska) of the common bean (Phaseolus vulgaris L.) to different concentrations of gum arabic-coated silver nanoparticles (GA-AgNPs) and silver nitrate (AgNO3), compared to control plants. The outcome of this work may be useful to determine the biocompatibility of AgNPs and to identify silver uptake, optimum concentrations and varieties for further field applications.
2. Materials and Methods
2.1 Chemicals
All biochemicals employed in this study were of high purity, used without further purification and purchased from Sigma Chemical Co. (Sigma–Aldrich Gmb H, Sternheim, Germany), and all organic solvents were of AR grade. AgNO3 (>99%) was used for the preparation of the silver nanoparticles. Gum arabic (Acacia Arabica) was used as the stabilizer, while 2-Propanol was used as a scavenger of excess radicals.
2.2 Irradiation Source
The process of irradiation was carried out at the National Center for Radiation Research and Technology (NCRRT). The facility used was a 60Co-Gamma Chamber 4000-A-India. Irradiation was performed using 60Co-gamma rays at a dose rate of 2.08 kGy/h at the time of the experiment. In this process, 60Co-gamma rays interact with matter in the solution, mainly by photoelectric absorption and Compton scattering, in order to produce free electrons and also hydrated electrons induced from water radiolysis. Together, these electrons reduce the Ag+ into Ag0.
2.3 Synthesis of Silver Nanoparticles by the Gamma Irradiation Route
Gum arabic-coated silver nanoparticles (GA-AgNPs −16.7nm) were synthesized by mixing 10 mL of 5% gum arabic extract with 90 mL of 1 mM aqueous silver nitrate and 2-propanol 3% solution. The mixtures were exposed to gamma irradiation at doses 5, 10, 15, 20, 25 and 30 kGy at room temperature. The synthesized GA-AgNPs were separated by centrifugation at 15,000 rpm for 20 min in order to remove any excess gum arabic (GA), which acts as a stabilizing and protecting agent for silver nanoparticles, and pellets were dispersed in deionized water. The previous operation was repeated, and the GA-AgNPs solution was stored in cold conditions in brown bottles. After irradiation, the produced AgNPs were characterized by UV-Vis spectroscopy, Dynamic Light Scattering (DLS), Transmission Electron Microscopy (TEM), the Fourier Transform Infrared (FT-IR) spectroscopy and X-ray diffraction (XRD). The effects of GA-AgNPs and AgNO3 as foliar sprays on plants' growth and yield were brought about by using the different concentrations of 0 (control), 5, 10, 20, 60 ppm.
2.4 Characterization of Silver Nanoparticles
2.4.1 UV-Vis Spectral Analysis
UV-Vis spectra of AgNPs were reported as functions of wavelength using a JASCO V-560. UV/vis. spectrophotometer at 200–800nm, operated at a resolution of 1nm.
2.4.2 Transmission Electron Microscopy (TEM)
The size and morphology of the synthesized nanoparticles were detected by using the TEM model JEOL electron microscope JEM-100 CX. Coated silver nanoparticles were dropped onto carbon-coated TEM grids. The film on the TEM grids was allowed to dry. The excess solution was removed using blotting paper.
2.4.3 Dynamic Light Scattering (DLS)
The average particle size and size distribution was determined by using the PSS-NICOMP 380-ZLS particle sizing system, St. Barbara, California, USA.
2.4.4 Fourier Transform Infrared Spectroscopy (FT-IR)
FT-IR measurements were carried out in order to obtain information about chemical groups present around the AgNPs for their stabilization and to understand the transformation of functional groups due to the reduction process. The measurements were carried out using a JASCO FT/IR-3600 infrared spectrometer by employing the KBr pellet technique.
2.4.5 X-ray Diffraction (XRD) Analysis
For X-ray Diffraction (XRD) Analysis, the reaction mixture that was embedded with the silver nanoparticles was placed on a glass slide and dried at room temperature. XRD scans were obtained using a Rigaku model D/max2000PC X-ray diffractometer operating with a Cu anode at 40 Kv and 50 mA in the range of 2θ value between 20° and 100°, with a speed of 2°/min. Prior to the peak-width measurement, each diffraction peak was corrected for background scattering and was stripped of the Kα2 portion of the diffracted intensity. Crystallite size, L, was calculated using Scherrer's equation, D = K λ/β 1/2cosθ, for the peak broadening deriving from size effects, only. Where γ is the wavelength of X-rays used (1.5418′A), β is the full width at half-maximum (fwhm) intensity of the diffraction line, θ is the Bragg angle for the measured hkl peak and K is a constant that is equal to 0.9 for Ag0.
2.5 Spraying Treatments and Sampling
A pot experiment was carried out in clay loamy soil at the Experimental Farm of Helwan University, Cairo, Egypt during the autumn season of 2014. Uniform seeds of two varieties (Bronco and Nebraska) of the common bean (Phaseolus vulgaris L.) were obtained from the Horticultural Research Institute (HRI), Agricultural Research Center (ARC), Ministry of Agriculture, Giza, Egypt, and were sown on 14 September. Five seeds were sown per earthenware pot containing 15 kg of clay loamy soil (40 cm in diameter, 38 cm in depth). Selected soil samples had 52.9% clay, 28.0% silt, 13.2% fine sand and 5.9% coarse sand, and the texture of the soil was clay loamy. The seed indexes (i.e., the weight of 100 seeds) of the Bronco and Nebraska varieties were 14.44 and 46.47 g, respectively. The pots were arranged in completely randomized block design with nine treatments, four replicates per treatment and each replicate represented by three pots for each variety.
Fertilization was carried out for each pot in the proportion of 1 g ammonium nitrate (33.5% N), 1 g calcium superphosphate (15.5% P2O5) and 1 g potassium sulphate (48% K2SO4). These fertilizers were applied in two equal doses at 21 and 35 days after sowing (DAS).
A foliar spray was applied twice to the Bronco and Nebraska plants during vegetative growth (at 30 and 37 DAS), with either gum arabic-coated silver nanoparticles (GA-AgNPs) or silver nitrate (AgNO3) each at 5, 10, 20 and 60 ppm (i.e., singly), in addition to distilled water in the case of the untreated plants (control). The volume of the spraying solution was maintained just so that it completely covered the plant foliage until it dripped. Irrigation was regularly carried out at intervals according to the weather conditions, in order to maintain the moisture content of the soil to field capacity. Other agricultural practices were carried out as recommended for conventional bean planting.
At the time of harvest (75 DAS): Plants were drawn early in the morning from each treatment and carefully washed with distilled water. Plant growth characteristics that were measured (i.e., vegetative growth) were plant height (cm), root length (cm), leaf number/plant, total leaves' area (cm2) as described by [44] and total fresh and dry weights (g)/plant. In addition to the mean values of the yield and its related parameters (pods' number, pods' fresh and dry weights (g)/plant and 100-seed weight (100 -SW)) were determined. Representative plant samples were taken from each treatment and either dried in an electric oven with a drift fan at 70°C for 48 h until a constant dry weight was achieved, or fresh samples of leaves were used to determine some metabolic activities.
2.6 Chemical Analysis
2.6.1 Measurement of Total Ag Content
Concentration of Ag in plant samples (leaves, pods, roots and the complete plant) of the two bean varieties, after washing with distilled water, were quantified using Atomic Absorption Spectrophotometry (AAS) at 75 days after sowing. Briefly, 0.5 g of oven-dry samples was digested using HNO3 and HClO4 (25:1 ml). The clear digested liquid was filtered through a 0.45 m acid-resistant filter paper and the silver was quantified in the filtrate using AAS (model UNICAM939, equipped with deuterium background correction, England). All solutions were prepared with ultra-pure water with a specific resistance 18 e/cm.
2.6.2 Endogenous Phytohormones
Phytohormones were extracted in 10 g of fresh leaves of the Nebraska variety, sprayed with either GA-AgNPs or AgNO3, each at 0.0, 10 and 60 ppm as described by [45]. The samples were ground in cold 80% methanol, followed by triple extraction with fresh methanol for 2 h at 0°C. To estimate the amounts of acidic hormones, the plant hormone fractions and standards were methylated according to [46], so that they were ready for high performance liquid chromatography (HPLC) analysis. The retention time (RT) and the area of the peaks of authentic samples were used for the identification and characterization of the peaks of samples under investigation.
2.6.3 Sodium Dodecyl Sulphate-Polyacrylamide Gel Electrophoresis (SDS-PAGE) for Protein Analysis
Electrophoretic protein profiles were detected in the Bronco and Nebraska varieties that were foliarly sprayed with either GA-AgNPs or AgNO3, each at 0, 10 and 60 ppm. Proteins were extracted from the leaves' samples in liquid nitrogen. The tris buffer system used was the one originally devised by [47], as described by [48], with 2% (w/v) SDS and 5% (v/v) 2-mercaptoethanol to cleave the disulphide bonds. The slurry was centrifuged for 20 min at 12000 rpm. The samples were heated in a boiling water bath for 5 min before loading to ensure dissociation. Gel preparation was carried out according to [49], where 15% resolving gel was used. Bromophenol blue (0.001%) tracking dye was used for marking the buffer front during electrophoresis. The gel was electrophoretic at a 60 mA constant current at 120 V for 15 min, then at 200 V (for about 2 h). Staining was done using coomassie brilliant blue, and a solution of 10% acetic acid and 45% methanol was used for destaining. The analysis of the protein bands was carried out using Quantity One ® 4. 6.2.
2.6.4 Statistical Analysis
The analysis of data was carried out according to [50]. Means were compared using least significant difference (LSD at the 5% level) and Duncan's multiple range test at the significance P = 0.05.
3. Results and Discussion
3.1 Characterization of Silver Nanoparticles
3.1.1 UV-Vis Spectroscopy of Synthesized GA-AgNPs
3.1.1.1. Effects of the Gamma Irradiation Dose
UV-Vis spectral analysis confirmed the formation of a deep yellowish colour of gum-arabic silver nanoparticles in the gamma-irradiated gum acacia solution containing AgNO3. Different doses of gamma irradiation have the same effect on the plasmon resonance on the peak position at 5 kGy and 30 kGy gamma irradiation; a peak appeared at the SPR of silver nanoparticles at about 456 nm. However, as the gamma irradiation dose increased, the surface plasmon resonance remained at approximately the same position: between 454–457 nm in the synthesis of GA-AgNPs (Fig. 1a). The absorptions' spectra are due to the Plasmon excitations of particles [51]. It is a well-known fact that colloidal silver nanoparticles exhibit absorption in the wavelength range of 380–450 nm, depending on the complex dielectric constant of the metal, the cluster size and the environment [52].
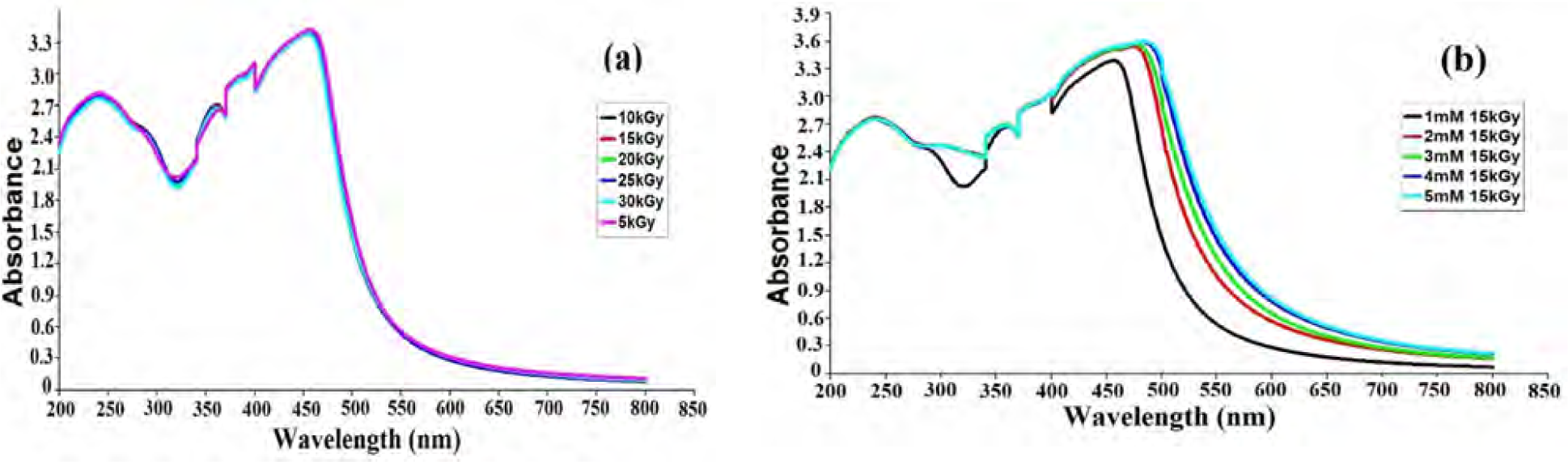
Characterization of the synthesized GA-AgNPs using UV-VIS spectra in an aqueous phase. (a) Effect of different doses of gamma irradiation and (b) Different concentrationsofAgNO3.
3.1.1.2. Effects of AgNO3 Concentration
The SPR band of silver nanoparticles was investigated at various concentrations of AgNO3, and they show that increasing the AgNO3 concentration (from 1 to 5 mM) shifted the surface plasmon resonance red band, indicating an increase of the silver nanoparticles' size (Fig. 1b). The intensity and width of the SPR depends on the size, morphology, spatial orientation, optical constants of the particles and the embedding medium [50, 54]. As the concentration of AgNO3 increased, the intensity of these peaks increased significantly and red shift occurred. The increase in particle size with an increase in the concentration of initial AgNO3 at 15 kGy gamma irradiation dose is presented in (Fig. 1b). The probable reason for this is that the Ag+ ions increase within the nanoscopic domains (loops) with the increasing AgNO3 concentration, and the collision frequency of silver particles increases significantly [55]. In principle, peak shift can be assigned to changes in particle size, shape and the dielectric environment [52].
3.1.2 TEM Analysis and Dynamic Light Scattering (DLS)
The TEM micrograph of GA-AgNPs, Fig. (2a), confirms the spherical shape of the particles and their uniform distribution without any significant aggregation at a mean diameter of 16.7 nm. The synthesis of nanoparticles was performed at a 1 mM AgNO3 concentration using 15 kGy of gamma irradiation dose. At the same conditions, as Fig. (2b) shows an average particle size distribution of approximately 22 nm, which was established by the DLS instrument. Distribution and particle sizes were mainly dependent upon spectral analysis [56].

Characterization of the synthesized GA-AgNPs at irradiation doses of 15 KGy and 1 mM AgNO3. (a) TEM micrograph indicates a mean diameter of 16.7 nm and (b) DLS indicates a particle size distribution of approximately 22 nm
3.1.3 X-ray Diffraction Analysis of Silver Nanoparticles
Intense X-ray diffraction (XRD) peaks were observed corresponding to the (111), (200), (220) and (311) planes for silver at 2θ angles of 38°, 46°, 65° and 78°, respectively (Fig. 3a). Only 2θ values of 81.5° correspond to the reflections from the (222) planes that were specific to GA-AgNPs' synthesis. These diffraction peaks represent the face-centred cubic (fcc) crystalline phase of the silver nanoparticles [57, 58]. The XRD spectrum of GA-AgNPs confirmed that the AgNPs formed using the gum arabic-extract coating agents in our experiment were crystalline in form.
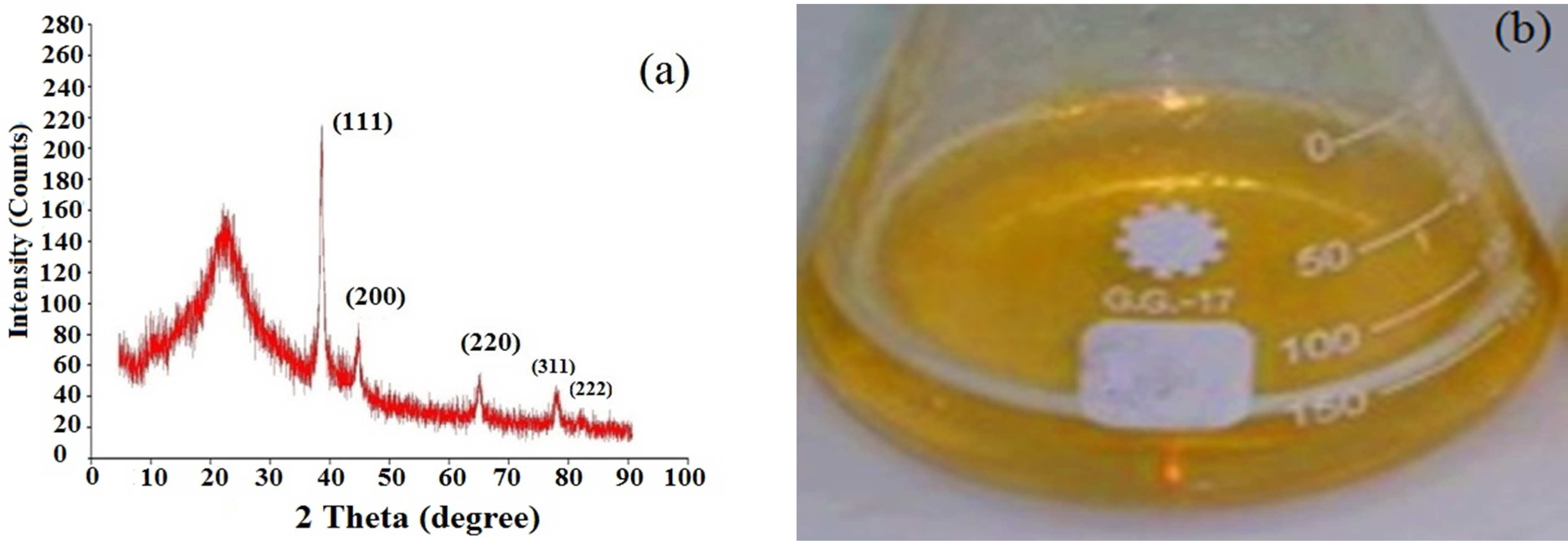
Characterization of synthesized GA-AgNPs at irradiation doses of 15 kGy and 1 mM AgNO3. (a) XRD spectrum and (b) Deep yellowish colour of fabricated GA-AgNPs
3.1.4 Fourier Transform Infrared Spectroscopy
The FTIR spectrum for dry gum acacia powder showed peaks centred at 3416 cm−1 (OH stretching), 2924 cm−1 (C–H stretching), 1364 cm−1 (OH deformation) and 1071 cm−1 (C–OH stretching) in the raw material (Fig. 4a), which are typical for polysaccharides. The presence of numerous hydroxyl and carboxyl groups and hydrogen bonding between them resulted in appreciable broadening of the peaks at 3400 cm−1 and 1000 cm−1 [59]. Gum arabic-capped silver particles showed disappearance of two peaks at 1422 cm-1 and 1364 cm−1 and the appearance of one peak at 1385 cm−1, which may be attributed to the contribution towards the stabilization of the AgNPs by the –OH bond deformation by a silver metal surface (Fig. 4b). In the case of the FTIR spectra of carboxylic acids, the range 500 cm−1 to 560 cm−1 represents -COO rocking. Comparing the FTIR spectra, it can be seen that the changes in the –COOH group/–OH, the peak appeared at 531 cm−1 in the raw material, but after encapsulation of nanoparticles, the peak shifted to 559 cm−1, and the peak intensity for the –C– of the carboxylic group also reduced after the encapsulation of nanoparticles. Therefore, the shift in the peak position from 531 cm−1 to 559 cm−1 in the gum arabic-capped AgNPs supporting the bridging was followed by linkage involving the silver surface and oxygen atoms of carboxyl groups [60]. More appropriately, there was clearly a shift in the transmittance because of the interaction of nanoparticles with plant biocompounds (GA) that act as stabilizing agents and prevent the direct contact of particles [61].
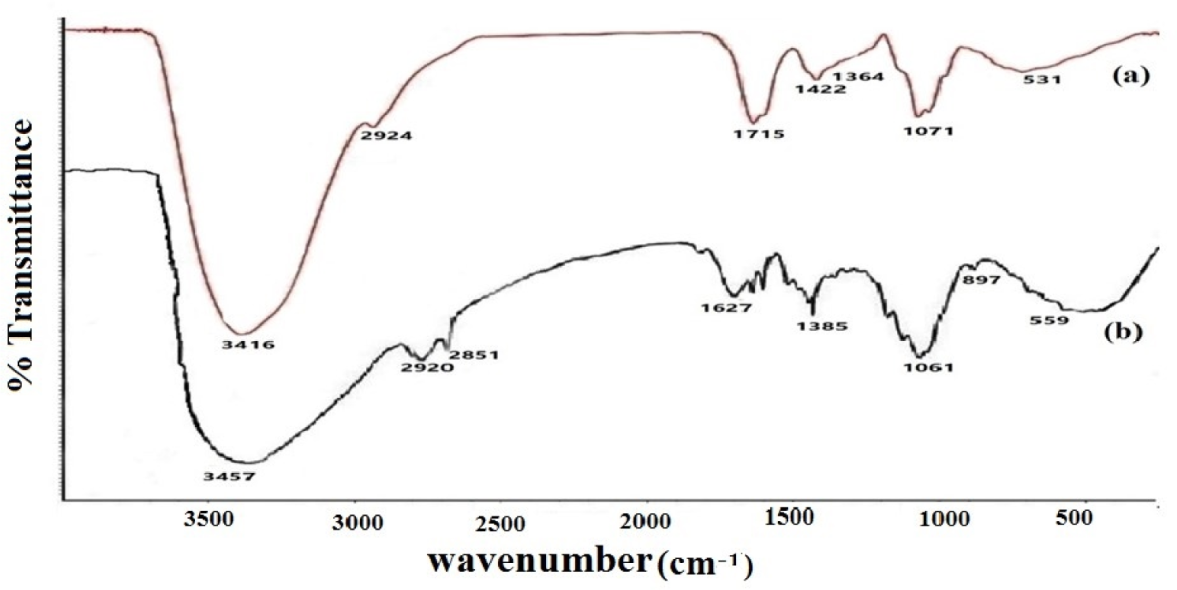
FTIR spectra of (a) Gum arabic extract and (b) Gum arabic-capped silver nanoparticles formed at irradiation doses of 15 kGy and 1 mM AgNO3
The results of the FTIR spectra of gum arabic and gum arabic-capped silver nanoparticles confirm the bonding between AgNPs with -OH/COO- groups, thereby stabilizing the nanoparticles formed. Thus, plant extracts (GA) can be used to stabilize the metallic nanoparticles owing to the existence of flavonoids, nitrate reductase and reducing sugars [62].
This novel method consists of a controlled reduction without any undesired oxidation products, extremely stable colloids and very pure silver ions reduced to AgNPs in the high gamma irradiation doses. A stabilizer was used as the protective colloid, preventing the AgNPs from aggregation. With different gamma irradiation, the particle size of the AgNPs decreased or increased at different irradiation doses. Using this method, the researchers were able to obtain AgNPs of different sizes by controlling the gamma irradiation dose.
3.2 Mechanism of Silver Reduction by Gamma Irradiation Process
When aqueous solutions are subjected to gamma-radiolysis, it produces the following species [63]:
The solvated electrons and H. atoms are strong reducing agents, and can reduce Ag+ ions to neutral Ag0 atoms:
The reduction of Ag+ ions is the main process for the formation of nanoparticles under gamma-radiolysis. Therefore, the oxidizing OH. radicals produced in the radiolysis of water (Eq. (1)) should be scavenged, and this can be performed efficiently by adding 2-propanol. Besides this, H. Radicals are also scavenged by these molecules:
Since the electrochemical potential of the organic radical is more positive than that of the Ag+/Ag0 system [7], the reaction of (CH3)2 C.OH that is formed from 2-propanol with Ag+ ions is relatively slow. Thus, during the process of irradiation, Ag+ ions are primarily reduced by solvated electrons and lead to Ag0. The growth of silver nanoparticles by reduction of Ag+ to Ag0 is stepwise [64]. These neutral Ag0 atoms at first dimerize when associated with excess Ag+ ions.
The charged dimer clusters Ag2+ may further react with excess silver cations by a cascade of coalescence processes, to form trimer, tetramer and higher-order silver ion clusters (Ag+n+1) [65], besides the doubly charged Ag32+, Ag42+, etc. [66]. The aggregation of these clusters into higher metallic clusters and nanoparticles occurs as the nucleation in the solution increases. In the case of gum arabic being used as a stabilizing agent, the neutral Ag0 atoms at first dimerize when they associate with the excess Ag+ ions trapped in the individual loops of gum acacia polymer chains. After the aggregation of the clusters into higher metallic clusters and nanoparticles occurs, as the nucleation in the solution increases in agglomerated clusters are trapped in the nanolevel domains of gum acacia (which are formed by the intramolecular and intermolecular H– bonds of –COOH and –OH groups of this polymer providing a network), they convert themselves into nanoparticles on continuous irradiation. These metal clusters surfaces are likely to be anchored through strong association between the Ag nanoparticle's surface and the ‘O’ atom of the functional groups (–COOH and –OH) of gum acacia [67].
3.3 Growth Parameters
Data presented in Tables 1 and 2 show that foliar application of either GA-AgNPs or AgNO3 at 5, 10, 20 and 60 ppm promoted almost all growth criteria of two bean varieties (Bronco and Nebraska) compared to the corresponding untreated control plants (i.e., untreated with either GA-AgNPs or AgNO3). In all cases, GA-AgNPs (5–60ppm) showed, in this study, a much better ability than AgNO3 (5–60 ppm) to promote growth of the two bean varieties, with the most effective treatment occurring at 10 ppm GA-AgNPs, possibly through participation in the synthesis of auxin and/or cytokinin, the enhancement of cell division, chlorophyll accumulation and nutrient uptake.
Effect of foliary spraying with silver nanoparticles (GA-AgNPs) and silver nitrate (AgNO3) on growth characteristics and yields of the Bronco variety at 75 days after sowing. Different letters indicate significant differences between treatments (Duncan test, P ≤ 0.05). Means in each column followed by the same letter are not significantly different.

Effect of foliary spraying with silver nanoparticles (GA-AgNPs) and silver nitrate (AgNO3) on the characteristics and yields of the Nebraska variety at 75 days after sowing. Different letters indicate significant differences between treatments (Duncan test, P ≤ 0.05). Means in each column followed by the same letter are not significantly different.

Foliar application of GA-AgNPs of up to 60 ppm significantly increased plant height, root length, number of leaves per plant, the leaves' area and total fresh and dry weights (g/plant) compared to corresponding untreated control plants for the Bronco and Nebraska varieties. GA-AgNPs at 10 ppm recorded the highest plant fresh (36.62, 17.70 g/plant) and dry weights (5.97, 3.29 g/plant) for Bronco and Nebraska varieties, respectively, compared with (14.97, 4.21 g/plant) for the fresh weight and (2.22, 0.71 g/plant) for the dry weight of their respective controls. In Borage, plant height, number of leaves and dry weight were improved by increasing the nanosilver concentration up to 60 ppm [39]. In Brassica juncea seedlings, sharp increases of 15 and 25% in shoot length and 167 and 277% in root length were observed at 25 and 50 ppm silver nanoparticle treatments, respectively [36]. The increases were mediated via plant growth regulators like cytokinins and gibberellins, which are involved in cell division and cell elongation, respectively [68,69]. Moreover, the GA-AgNPs treatment, especially at 10 ppm, increased the number of leaves per plant and the leaves' area/plant as compared to the control plants and AgNO3 treatments in the two bean varieties. In this respect, increasing the concentration of silver nanoparticles from 20 to 60 ppm increased the number of leaves, leaf area, shoot and root lengths of both common bean (Phaseolus vulgaris L.) and corn (Zea mays L.) crops [40]. Both leaf number and leaf area are regulated by a complex interaction of various genes whose expression is modulated by growth hormones [70]. In Arabidopsis seedlings, AgNPs antagonized the aminocyclopropane-1-carboxylic acid (ACC) and reduced the expression of ACC synthase 7 (ACS7) and ACC oxidase 2 (ACO2), suggesting that AgNPs acted as inhibitors of ethylene (ET) perception and could interfere with ET biosynthesis [71]. In Crocus Sativus, AgNPs promoted root growth by blocking ET signalling under flooding conditions [41]. Ethylene controls the leaf number by regulating leaf abscission. Inhibition of ET action reduces the event of abscission [39], which is one of the important reasons for the recorded increase in leaf number. Therefore, the antagonistic action of GA-AgNPs could slow down the effect of ET, resulting in an increase in leaf number in both the Bronco and Nebraska varieties. In addition, the permeability of nanosilver is far greater than that of silver nitrate, due to the small size of the nanoparticles, which causes the greater adhesion of nanoparticles to plant tissues [39].
Furthermore, AgNO3 (5–60 ppm) was less effective than GA-AgNPs (5–60 ppm) in increasing the vegetative growth of the Bronco and Nebraska varieties (Tables 1 and 2). The increment in growth characteristics (i.e., plant height, root length, number of leaves per plant, the leaves' area and total fresh and dry weights (g/plant) was greater at 10 ppm AgNO3 for two common bean varieties, possibly through the effect of silver in preventing the ET action. Similarly, silver ions cause an increase in the stem heights of tulips and rose plants [72]. In vitro conditions, silver nitrate-inhibited the biosynthesis of ET and caused the regeneration of multiple shoots from hypocotyl sections of cotton [73]. In addition, silver nitrate was also effective at increasing the wet weight of tobacco [74]. The application of silver ions can displace copper ions from the receptor proteins and, consequently, block ET action by preventing it from connecting to its receptors in plant cells [75-77]. However, in the Bronco variety, most growth parameters showed a low response at 60 ppm AgNO3, with a minimum increase in total fresh and dry weights (which increased by only 6.15 and 9.01% more than the control, respectively). In this connection, the number of leaves, plant height and dry weight of Borage plants were reduced by increasing the silver nitrate concentration from 100 to 300 ppm in the Borage plants [39].
Moreover, GA-AgNPs and AgNO3 treatments accelerated flowering and, consequently, pod formation. The Bronco variety (as a small seed) recorded higher values in most studied growth traits, i.e., the highest number of pods and the fastest production of flowers (i.e., it took the smallest number of days for the complete transformation of flowers than did Nebraska (as a big seed). In Borage, a rise in the concentration of nanosilver from 20 to 60 ppm increased the dry weight of inflorescence, while the opposite result was obtained by increasing the concentration of silver nitrate from 100 to 300 ppm [39].
3.4 Yield
The application of GA-AgNPs (5–60 ppm) and AgNO3 (5–60 ppm) significantly enhanced the yield (i.e., the number, fresh and dry weights of pods/plant (g/plant), 100-SW (g)) and, consequently, may increase the seed yield/plant of two common bean varieties (Bronco and Nebraska) when compared to the corresponding untreated control plants as presented in Tables 1 and 2. GA-AgNPs was more effective than AgNO3 at increasing yields, with the most promising results for the fresh and dry weights of pods (increased by 141.64 and 239.8% more than the control) that were obtained at 10 ppm in Bronco and 100-SW (increased by 66.25 and 52.95% more than their controls) at 20 ppm in Bronco and Nebraska, respectively. It did so by stimulating IAA, inhibiting ABA contents, enhancing photosynthetic activity, accumulating dry matter and, consequently, by increasing the translocation and accumulation of certain metabolites in plant organs, which affected their yield (Tables 1 and 2). In Borage (Borago officinalis L.), the foliar application of either nanosilver or silver nitrate increased the seed yield due to the increase in the amount of inflorescence in the unit area, the number of seeds in the inflorescence, the weight of the seeds and a decrease in seed abscission [39]. In Brassica juncea, an optimal increase in seed yield, due to the higher rate of photosynthesis, was recorded at a 10 ppm concentration of Gold-nanoparticle (AuNP) [78].
The AgNO3 significantly increased the two beans' yields; AgNO3 at 10 ppm increased the fresh and dry weights of pods (increased 39.44 and 145.2% more than the control) in Bronco and 100-SW increased 64.62 and 46.72% more than the control at 20 ppm in Bronco and Nebraska, respectively (Tables 1 and 2). In wheat, silver nitrate improved seed yield [79] as a result of reducing seed abscission due to the inhibitory effect of silver on ethylene action [43]. According to several authors, the abscission process is regulated by phytohormones such as auxin (IAA), ethylene and abscisic acid (ABA). The latter induces the abscission process through stimulation of ethylene biosynthesis, while auxin is effective in delaying the abscission process by reducing the sensitivity of cells to ethylene [75]. Ethylene activates the biosynthesis genes of hydrolytic enzymes, e.g., cellulose and polygalacturonase, which induces the separation of plant organs from the main plant [80]. Silver ions decreased 100% of the flower abscission of Alstroemeria plants, as compared to untreated flowers, within the two first days [42] and decreased both the flowers' and flower buds' abscission in orchid plants [43]. Nanosilver particles have a larger surface area in contact with the environment due to their small size. Thus, the amount of adhesion to the cell surface increases, which leads to their higher efficacy [81]. On the contrary, the highest weight of 100 seeds was observed in the control Borage sample, while this weight decreased when either nanosilver or silver nitrate was applied [39].
Moreover, GA-AgNPs and AgNO3 treatments accelerated flowering and, consequently, pods' formation in the two bean varieties. The Bronco variety (as a small seed) recorded higher values in most studied growth traits, i.e., the highest number of pods and the shortest time to flower (i.e., it took the shortest period of days for the complete transformation of flowers when compared to Nebraska as a big seed). In Borage, a rise in the concentration of nanosilver from 20 to 60 ppm increased the dry weight of inflorescence, while the opposite result was obtained by increasing the concentration of silver nitrate from 100 to 300 ppm [39].
3.5 Uptake of Silver in Nano- and Bulk Forms
Atomic absorption spectroscopic studies show negligible increases in the bioaccumulation of both forms of silver (either nanosilver or silver nitrate) in different parts (i.e., roots, leaves, pods and the whole plant) of the two common bean varieties with an increase in the concentrations of GA-AgNPs and AgNO3 from 5 to 60 ppm, as compared to the corresponding untreated control plants (Tables 3, 4 and Fig. 5).
Atomic absorption spectroscopy measurement of Ag residual concentration (μg g−1 dry weight) in unequal dry weights of different parts of the Bronco plant, as affected by foliar spraying with silver nanoparticles (GA-AgNPs) and silver nitrate (AgNO3) at 75 days after sowing

Atomic absorption spectroscopy measurement of Ag residual concentration (μg g−1 dry weight) in unequal dry weights of different parts of the Nebraska plant as affected by foliar spraying with silver nanoparticles (GA-AgNPs) and silver nitrate (AgNO3) at 75 days after sowing


Atomic Absorption Spectroscopy measurement of Ag content (μg/g dry wt.) in different parts of (a) Bronco and (b) Nebraska varieties, as affected by foliar spraying with silver nanoparticles (GA-AgNPs) and silver nitrate (AgNO3) at 75 days after sowing
Increasing the applied concentrations of GA-AgNPs and AgNO3 up to 60 ppm increased the uptake of both forms of silver (either nanosilver or silver nitrate) in different parts, especially the leaves, followed by the roots of the two common bean varieties. GA-AgNPs at 60 ppm recorded high silver uptakes of 0.35 and 0.49 μg/g dry weight for whole plants of the Bronco and Nebraska varieties, respectively, compared with 0.25, 0.29 μg/g dry weight for their respective controls (Fig. 5). Similarly, increasing the concentration of applied AuNPs up to 100 ppm increased the uptake of AuNPs to 21.36l μg/g fresh weight in the leaf tissues of Brassica juncea [78] and the intracellular CuO nanoparticle concentration in wheat roots with an increase to the CuO nanoparticles' exposure [82].
Furthermore, the silver content (μg/g dry weight) was often higher in bulk (AgNO3) (5–60 ppm) in comparison with GA-AgNPs (5–60 ppm) in different parts of the two common bean varieties. The highest silver contents (0.44 and 0.53 μg/g dry weight) in whole plant tissues were observed at 60 ppm AgNO3 in Bronco and Nebraska varieties, respectively (Fig. 5). In maize seedlings, silver uptake was 12.2 fold higher in AgNO3 treatment than for citrate-coated AgNPs, which suggests minimal dissolution of the NPs by AgNPs coating [83]. In castor seedlings, the silver content was high in AgNO3-exposed plants than in the AgNP stabilized with PVP, when compared to untreated controls, due to the slow release of Ag ions from AgNPs and depending on the concentrations employed, up to 4000 ppm [38]. On the contrary, the silver content in the Cucurbita pepo stem was four to seven times greater with the solution containing AgNPs when compared with their corresponding bulk form, due to the greater ion release from AgNPs [69]. The studies on the dissolution and aggregation of silver nanoparticles (gallic acid-coated, PVP-coated) and AgNO3 were reported to cause lower dissolution of PVP–AgNP in water to form Ag+ when compared with GA-AgNPs or AgNO3 [84].
Moreover, analysis of 1 gm dry weight of different parts (i.e., roots, leaves, pods and whole plants) of the Bronco variety indicates less bioaccumulation of both forms of silver (either nanosilver or silver nitrate) than in the Nebraska variety. GA-AgNPs and AgNO3 at 60 ppm increased silver uptake to 39.7 and 76.1% more than in the control Bronco plants, while they increased silver uptake to 55.6 and 83.5% more than in the control Nebraska variety, respectively. Brassica napus is known to be a metal hyper-accumulator, with significant metal accumulation being reported for Zn [85] and nanogold [86]. These accumulated nano-gold particles move through the vasculature system of the plant [87].
3.6 Endogenous Phytohormones
Data presented in Table 5 show that foliar application of either GA-AgNP or AgNO3 at 10 or 60 ppm, respectively, has a bioregulatory effect on phytohormones balance in the leaves of the Nebraska variety. They have a stimulatory effect on the detected amounts of IAA, GA3, GA3/ABA and total cytokinins, and an inhibitory effect on ABA contents as compared with untreated (control) plants. Changes in phytohormones may be involved in gene expression regulating the signalling activities or levels of growth regulating substances through having a direct impact on the activities of oxidoreductive enzymes related to hormonal metabolism. In turn, such changes lead to an increase of the metabolic compounds, which can also explain the increased growth parameters and yield in GA-AgNP- and AgNO3-treated plants, as compared with untreated ones. In this connection, the abscission process could be regulated by phytohormones such as auxin (IAA) and abscisic acid (ABA). The latter induces the abscission process through stimulation of the ethylene biosynthesis, while auxin is effective in delaying abscission by reducing the sensitivity of cells to ethylene [75,88]. Several reports have indicated the interaction of AgNPs with plant-signalling pathways. For example, in Arabidopsis, AgNPs induce root growth promotion and activate gene expression involved in cellular events, including cell proliferation, metabolism and hormone-signalling pathways. AgNPs induced the gene expression of the ABA-signalling pathway, indole acetic acid protein 8 (IAA8), 9-cis-epoxycarotenoid dioxygenase (NCED3), dehydrated cis-epoxycarotenoid dioxygenase (NCED3) and dehydration-responsive RD22. In addition, AgNPs acted as inhibitors of ET perception and could interfere with ET biosynthesis [71]. Moreover, cells that were treated with AgNPs exhibited an up-regulated auxin-responsive gene, IAA8, whose over expression results in the suppression of lateral root development [89]. AgNPs down-regulate genes that are involved in systemically acquired resistance (SAR) responses to pathogens and hormone signals, including auxin and ET [90]. In Eruca sativa, AgNP treatment promotes root growth and induces the expression of jacalin-related lectin (JAL), a gene that has been implicated in the release of nitrile, a precursor of auxin synthesis [35]. On the other hand, application of silver ions can displace copper ions from the receptor proteins, consequently blocking ethylene perception, since copper ions play a critical role in ethylene binding upon receptors [76,91]. These results suggest that physiological responses mediated by ABA, auxin and ET signals are most affected by AgNPs and AgNO3, resulting in the enhancement of growth phytohormones levels that have highly positive impacts on the vegetative growth and yield of Bronco and Nebraska varieties.
HPLC analysis for endogenous hormone concentrations (mg 100 g−1 F. Wt.) of the Nebraska variety as affected by foliar spraying with silver nanoparticles (GA-AgNPs) and silver nitrate (AgNO3) at 55 days after sowing

3.7 SDS-Protein Electrophoresis
The SDS- electrophoresis protein banding patterns indicate that application of either GA-AgNP or AgNO3 at 10 and 60 ppm, respectively, altered protein patterns in the leaves of the two bean varieties, as compared with their corresponding controls. It does so via the expression of new polypeptides of the molecular weight of 25–26 kDa (in Bronco), 50.72 and 47.00kDa (in Nebraska), and the disappearance of control polypeptides with the molecular weight of 28.40 in the two bean varieties. These circumstances may increase the adaptation of the two bean varieties to a diverse array of stresses (Fig. 6).

SDS-PAGE of relative molecular weights of M: Protein marker and proteins extracted from the leaves of two 55-day-old bean varieties [var. Bronco lanes (1–5) and Nebraska lanes (6–10)] as affected by foliary spraying with distilled water lanes (1, 6); silver nanoparticles' (GA-AgNPs) lanes (2, 7) at 10 ppm and lanes (3, 8) at 60 ppm; silver nitrate lanes (4, 9) at 10 ppm and lanes (5, 10) at 60 ppm
In the Bronco variety, nine different bands with the molecular weights of 70.27, 59.70, 52.69, 44.20, 41.89, 35.02, 28.29, 14.14 and 9.47 kDa were detected in control plants. GA-AgNPs and AgNO3 treatments induced the appearance of one band with the molecular weight of 25–26 kDa and the disappearance of one band with the molecular weight of 28.29 kDa. In this connection, the prolonged adaptation of cells to salinity resulted in increasing the induction of protein of Mr 26 kDa (called osmotin) [92] and 25 kDa protein in the cells of Citrus sinensis [93]. Plants over expressing osmotin neutralize the reactive oxygen species (ROS) by producing more compatible solutes or by the expression of specific antioxidative enzymes [94]. Moreover, GA-AgNPs induced the appearance of three polypeptides with the molecular weights of 73.71, 29.95 kDa at 10 ppm and 37.08 kDa at 60 ppm. Only the AgNPs' exposure causes the alteration of some proteins related to the endoplasmic reticulum and the vacuole in rocket (Eruca sativa) seedlings [35]. On the other hand, AgNO3 treatments at 10 and 60 ppm induced the appearance of one polypeptide with the molecular weight of 49.63 kDa and the disappearance of two polypeptides with the molecular weights of 44.20 and 52.69 kDa in the Bronco variety. In this respect, the electrophoretic pattern of soluble proteins of sorghum (Sorghum bicolour L.) cv. Jambo showed the expression of a new polypeptide (a marker protein for salt) of the molecular weight 50 kDa, via the expression of a stress-resistant gene [95].
In the Nebraska variety, a number of detected bands ranging between nine and 11 in number with the approximate molecular weights of 7.97–69.99 kDa were detected. Application of GA-AgNPs and AgNO3 treatments at 10 and 60 ppm induced the appearance of two bands with the molecular weights of 51.27 and 47.00kDa and the disappearance of control proteins with the molecular weights of 61.35 kDa, except in the 10 ppm GA-AgNPs treatment. In this respect, Ag treatments (PVP-AgNPs and AgNO3) over five days with 10 mg Ag L−1 cause changes in proteins involved in the redox regulation and in the sulphur metabolism in rocket seedlings by activating some common enzymatic and non-enzymatic pathways of ROS-detoxification machinery, including superoxide dismutase (SOD) and Type 2 peroxiredoxin (PRX) — a thiol peroxidase [35]. Moreover, AgNO3 treatments induced the appearance of three polypeptides with the molecular weights of 69.99, 13.29 and 11.82 kDa in the Nebraska variety. In this connection, AgNO3 increased the abundance of several major latex proteins, four glutathione-S-transferase, an isoflavone reductase and a universal stress protein. These proteins are leitmotifs in metal detoxification [96] and caused a specific increase of several proteins involved in energy production, including plastidial and mitochondrial ATP synthase subunits, carbonic anhydrase and aconitate hydratase. This up-regulation might help cells to produce more reducing power in order to facilitate the response to AgNO3 stress [35].
4. Conclusion
The present study demonstrates the biological mechanism for biosynthesis and the rapid production of high yield, stable gum arabic-capped silver nanoparticles (GA-AgNPs) with a mean diameter of 16.7 nm using 1 mM AgNO3 and gamma irradiation at 15 kGy doses. GA-AgNPs have a narrow distribution, a small size and a uniform shape. GA-AgNPs are biocompatible because of the surface capping with the natural polymer, non-toxic and eco-friendly Gum acacia. GA-AgNPs could be efficiently utilized in agricultural research to obtain the better health of crop plants, as showed by our study.
Foliar application of GA- AgNPs and AgNO3 especially at 10 ppm, significantly increased every morphological attribute, particularly those of seed yield, altered proteins and phytohormone balance, and indicated low residual accumulation of nanoparticles in different parts of the two bean varieties. The results obtained in the present findings are novel from an application point of view, and may help in standardizing an alternate technology to GM crops in order to ensure global food security and provide an easily applicable solution for sustainable agriculture around the world.






