Abstract
Background:
Tumor necrosis factor inhibitors (TNFi) effectively manage radiographic axial spondyloarthritis (SpA), and dose reduction is often used for stable patients. However, its long-term impact on radiographic progression remains unclear.
Objective:
To analyze the correlation between cumulative TNFi dose and radiographic progression in radiographic axial SpA.
Design:
Single-center retrospective chart review.
Methods:
Electronic medical records of patients with radiographic axial SpA from January 2001 to December 2018 were screened. The TNFi percentage dose was calculated as the total prescribed dose divided by the standard dose at 2-year intervals. The relationship between TNFi percentage dose and modified Stokes Ankylosing Spondylitis Spinal Score (mSASSS) changes was assessed using linear mixed models, separated into three baseline mSASSS groups: mSASSS ⩽24, mSASSS >24 to ⩽48, and mSASSS >48.
Results:
In the initial linear mixed model, radiographic progression, defined as the change in mSASSS over 2-year intervals, was examined in three baseline mSASSS groups. In the baseline mSASSS ⩽24 group, the cumulative TNFi dose showed a negative correlation with radiographic progression (β = −0.888, 95% confidence interval (CI): −1.793 to 0.017, p = 0.055). In the group with 24 < baseline mSASSS ⩽ 48, a positive but nonsignificant association was observed (β = 1.688, 95% CI: −2.119 to 5.495, p = 0.379). Similarly, for the baseline mSASSS >48 group, no significant correlation was found (β = 0.182, 95% CI: −0.832 to 1.196, p = 0.715). In the multivariable model of the baseline mSASSS ⩽24 group adjusted for age and sex, the cumulative TNFi dose was negatively correlated with the mSASSS change (beta = −1.871, 95% CI: −1.871 to −0.059, p = 0.037).
Conclusion:
In patients with radiographic axial SpA and baseline mSASSS ⩽24, the cumulative TNFi dose was negatively correlated with radiographic progression. Maintaining a standard TNFi dose may slow the progression of spinal structural changes in early stage SpA.
Background
Axial spondyloarthritis (SpA) is a chronic inflammatory arthritis that mainly targets the axial joints to cause stiffness and inflammation.1,2 In severe cases, spinal ankylosis may occur, significantly limiting mobility and reducing quality of life. Nonsteroidal anti-inflammatory drugs (NSAIDs) are the recommended first-line treatment for SpA. However, for patients who exhibit an inadequate response to NSAIDs, tumor necrosis factor inhibitors (TNFis), interleukin-17 inhibitors, and Janus kinase inhibitors are recommended as alternative therapeutic options.3 –5
TNFi has been the most widely used biological agent for axial SpA treatment for over 20 years. These drugs not only alleviate axial symptoms but also address extra-musculoskeletal manifestations. Additionally, extensive clinical experience with TNFi over the years has shown its effectiveness in slowing radiographic progression.6 –8 Despite these advantages, concerns exist regarding the long-term safety of TNFi, particularly with regard to infection risk and the associated economic burden.9,10
Tapering TNFi rather than discontinuing it may be beneficial for patients. Recent studies have explored tapering or dose reduction of TNFi to treat axial SpA.9,11 –15 Dose reduction of TNFi can reduce the risk of side effects while maintaining low disease activity. Moreover, it can reduce healthcare costs. However, dose reduction should be individualized, as it may exacerbate the disease in some patients depending on the disease state and underlying conditions. Current treatment guidelines suggest that tapering biologics should be considered in patients with maintained remission. 4 Nevertheless, evidence on the impact of reducing TNFi doses on structural damage remains limited. 14
Therefore, to assess whether TNFi dose reduction affects structural changes, we aimed to investigate the association between the cumulative dose of TNFi and radiographic progression using observational data from patients with radiographic axial SpA.
Methods
Participants
In this retrospective study, we reviewed the electronic medical records (EMR) of patients with radiographic axial SpA who were followed up from January 2001 to December 2018 at a single center.8,16 All patients documented and followed up in the EMR met the modified New York criteria for classifying radiographic axial SpA. Patients attended rheumatology clinics every 6 months to monitor disease activity and inflammatory markers, and they received cervical and lumbar spine radiographs every 2 years. Only patients treated with TNFi who underwent two or more radiographic assessments were included in this group.
This study was approved by the Institutional Review Board of Hanyang University Hospital (HYUH 2018-07-007). Owing to its retrospective nature, the Institutional Review Board of Hanyang University Seoul Hospital waived the requirement for informed consent. This study used anonymized patient data and complied with the principles outlined in the Declaration of Helsinki. The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement. 17
Clinical data
Baseline characteristics, such as age, sex, symptom duration, smoking status, human leukocyte antigen B 27 (HLA-B27) status, eye involvement, and peripheral involvement, were extracted from the EMR. TNFi prescriptions in rheumatology clinics followed the guidelines set by the Health Insurance Review and Assessment Service in Korea. If the Bath Ankylosing Spondylitis Disease Activity Index (BASDAI) score decreased by 50% or more than 2 points 3 months after administration of the first TNFi, an additional 6 months of treatment was permitted. Continuous administration was possible if the BASDAI score remained low, with evaluation performed every 6 months. Treatment was discontinued if no TNFi prescription was recorded for 6 months following the last prescription. For each patient, the total TNFi treatment duration was segmented into 2-year intervals, and the cumulative TNFi dose was calculated for each interval. Radiographic progression was measured by determining the change in the modified Stokes Ankylosing Spondylitis Spinal Score (mSASSS) between the beginning and end of each 2-year period. To verify the study using a 2-year interval, the data were analyzed using a 4-year interval.
The cumulative TNFi dose was calculated similarly to the defined daily dose as follows: (total number of grams of each TNFi used/number of weeks during the interval)/recommended weekly dose. 18 The recommended weekly doses were obtained from the Ministry of Food and Drug Safety’s drug approval documents as follows: 40 mg every 14 days for adalimumab, 50 mg every 7 days for etanercept, 50 mg every 28 days for golimumab, and 5 mg/kg every 56 days for infliximab. 18 For infliximab, the recommended weekly dose was calculated using the average body weight of South Korean men (71 kg) and women (57 kg).19,20
Spinal radiographic changes were assessed using the mSASSS. Two musculoskeletal radiologists (S.L. and K.B.J.) independently scored the radiographs, blinded to both patient information and the time points at which the images were obtained. To verify the results, we conducted reliability testing: intraobserver reliability was evaluated by having one radiologist repeat the scoring after a 6-month interval, and interobserver reliability was determined by comparing the independent scores. Any discrepancies were resolved by consensus. The intraclass correlation coefficients were excellent, with interobserver and intraobserver reliabilities of 0.978 and 0.946, respectively.8,21
Statistical analysis
Continuous variables are expressed as mean ± standard deviation, and categorical variables as frequencies and percentages. p-Values <0.05 were considered statistically significant.
First, we used a scatterplot to assess the relationship between the cumulative TNFi dose and changes in mSASSS. As the baseline mSASSS is a critical factor affecting the radiographic prognosis of radiographic axial SpA,16,22,23 participants were categorized into subgroups based on their baseline mSASSS (0–24, 24–48, and 48–72). A linear mixed model was developed for each baseline mSASSS group (change in mSASSS as the intercept + baseline mSASSS + cumulative TNFi dose). We identified the baseline mSASSS group with a significant relationship between the cumulative TNFi dose and mSASSS changes and proceeded with further analysis based on this group.
Second, a multivariate linear mixed model was constructed using the baseline mSASSS group identified in the previous step. The relationship between the cumulative dose of TNFi and the change in mSASSS was confirmed in a model that included the baseline characteristics of the inflammatory markers. The inflammatory markers, erythrocyte sedimentation rate (ESR) and C-reactive protein (CRP), were transformed into log values to normalize their distributions before being included in the model because of their right-skewed nature. Variables for the multivariable model were selected based on the results of univariate analyses, an assessment of multicollinearity, considerations of model parsimony to prevent overfitting, and their clinical relevance. We intentionally avoided using automatic variable selection methods, such as backward elimination or stepwise selection, as these approaches often include nonsignificant variables and can produce biased estimates of beta coefficients and R-squared values. CRP and ESR were included in the model because they demonstrated stronger correlations with changes in the mSASSS compared to the BASDAI and the ASDAS. Consequently, among the disease activity indices, ESR and CRP were incorporated into the model.
The relationship between the cumulative dose of TNFi and the change in mSASSS was analyzed at 2-year intervals, and a similar analysis was conducted for the 4-year interval. All statistical analyses were performed using the R programming language (version 3.6.1; R Foundation for Statistical Computing, Vienna, Austria; http://www.R-project.org/).
Results
Baseline characteristics
Among the 1280 patients reviewed, 372 met the inclusion criteria for this retrospective study (Figure 1). Baseline patient characteristics are shown in Table 1. Of the 372 patients, 10.8% were women, and the mean age at diagnosis was 33.63 (10.00) years. The mean baseline mSASSS was 16.37 (17.24). When the TNFi administration period was divided into 2-year intervals, 871 intervals were obtained, and 317 intervals were obtained when divided into 4-year intervals.

Flowchart depicting patient inclusion and exclusion in this study.
Baseline characteristics.

BASDAI, Bath Ankylosing Spondylitis Disease Activity Index; HLA, human leukocyte antigen, mSASSS, modified Stokes Ankylosing Spondylitis Spinal Score.
Relationship between cumulative dose of TNFi and changes in mSASSS over a 2-year interval
Supplemental Figure 1(A) shows the frequencies of cumulative TNFi doses over a 2-year interval. The mean and median cumulative TNFi doses were 0.6036 and 0.5753, respectively, ranging from 0.1247 to 1.4535. Figure 2 presents a scatterplot illustrating the relationship between the cumulative dose of TNFi and the change in mSASSS in all patients and the three baseline groups at 2-year intervals. Considering each trend line, no significant relationship was observed between the cumulative dose of TNFi and the change in mSASSS. A multivariable linear mixed model was developed to evaluate the relationship between the cumulative dose of TNFi and the change in mSASSS among all patients. No statistically significant association was observed between the cumulative dose of TNFi and the change in mSASSS (Supplemental Tables 1 and 2).
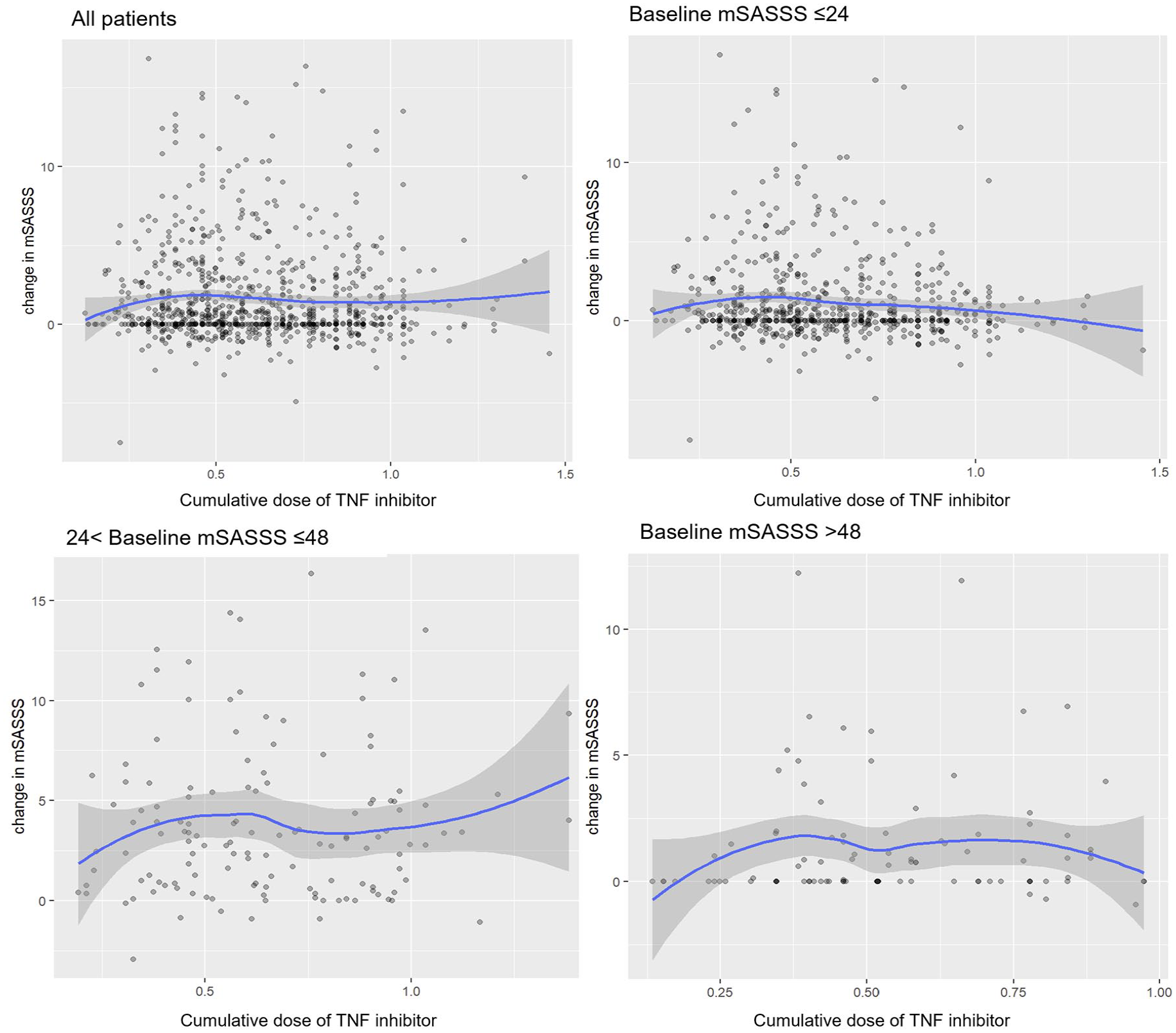
Scatterplot of the cumulative dose of TNF inhibitor and the change in mSASSS at 2-year intervals.
Using the initial linear mixed model, the relationship between the cumulative dose of TNFi and the change in mSASSS was investigated for each baseline mSASSS group (Table 2). In this model, the intercept represents the average natural increase in mSASSS every 2 years. In our dataset, the average baseline mSASSS is 18.14, which differs from the mean presented in Table 1 because each patient is recorded multiple times at 2-year intervals. Consequently, the “natural” or predicted increase in mSASSS over a 2-year period for a patient with this average baseline is 1.551. The subgroup estimates, however, are derived from regression models that use specific baseline values and include interactions or nonlinear adjustments. For example, for a patient with a baseline mSASSS of 20, the predicted increase is calculated as 2.389 + 0.135 × (20 − 18.14), which equals approximately 2.64. For a patient with a baseline mSASSS of 36, the predicted increase is given by 8.835 − 0.171 × (36 − 18.14), yielding about 5.78. In the case of a patient with a baseline mSASSS of 70, due to limitations in the scoring range, we applied a cap by using 60 instead of 70, and the formula becomes 11.264 − 0.208 × (60 − 18.14), resulting in roughly 0.48. In the baseline mSASSS ⩽ 24 group, the cumulative dose of TNFi showed a negative correlation (beta = −0.888 (95% confidence interval (CI): −1.793 to 0.017, p = 0.055)). This suggests that the mSASSS may increase more slowly as the cumulative dose of TNFi increases in the group with a baseline mSASSS ⩽24 than in the other baseline groups. Therefore, we focused on models in the group with baseline mSASSS ⩽24.
Initial linear mixed model for each baseline mSASSS group at 2-year intervals.

CI, confidence interval; mSASSS, modified Stoke Ankylosing Spondylitis Spinal Score; TNFi, tumor necrosis factor inhibitor.
Multivariable linear mixed models for 2-year intervals
Table 3 shows the univariate and multivariate models for the subgroup with baseline mSASSS ⩽24. In univariate analysis, baseline mSASSS, age, and smoking history were significantly positively associated with changes in mSASSS, whereas female sex and peripheral involvement were significantly negatively correlated with these changes. Multivariate Model 1 included only age and sex. In this model, baseline mSASSS was positively correlated with changes in mSASSS (beta = 0.129, 95% CI: 0.101–0.156, p < 0.001). In contrast, the cumulative dose of TNFi was negatively correlated with changes in the mSASSS (beta = −1.871, 95% CI: −1.871 to −0.059, p = 0.037). In other words, a higher baseline mSASSS was associated with a faster increase in mSASSS, whereas a higher cumulative TNFi dose was associated with slower mSASSS progression. In multivariable model 2 (including log CRP), the cumulative TNFi dose exhibited a negative correlation with mSASSS changes but did not quite reach statistical significance (p = 0.096). In contrast, in Model 3 (including log-ESR), the correlation again failed to reach statistical significance (p = 0.130).
Multivariable linear mixed model for baseline mSASSS ⩽24 groups at 2-year intervals.

CI, confidence interval; CRP, C-reactive protein; ESR, erythrocyte sedimentation rate; HLA, human leukocyte antigen; mSASSS, modified Stoke Ankylosing Spondylitis Spinal Score; TNFi, tumor necrosis factor inhibitor.
Verification of results in 4-year intervals
To verify whether similar results could be obtained in the 4-year intervals, we constructed a model following the procedure used in the 2-year interval. In the 4-year interval, the frequencies of cumulative doses of TNFi are shown in Supplemental Figure 1(B). The mean and median cumulative TNFi doses were 0.577 and 0.529, respectively, and the minimum and maximum values were 0.144 and 1.373, respectively.
First, we examined the relationship between the cumulative dose of TNFi and the change in mSASSS for each baseline mSASSS group using a scatterplot (Supplemental Figure 2) and created an initial linear mixed model for each group (Supplemental Table 3). Similar to the model in the 2-year interval, in the group with baseline mSASSS ⩽24, a significantly negative correlation was observed between the increased cumulative dose of TNFi and the slow increase in mSASSS (beta = −3.106, 95% CI: −6.205 to −0.006, p = 0.050).
A multivariable model was constructed for patients with a baseline mSASSS of ⩽24 over a 4-year interval (Table 4). In Model 1, which included age and sex as variables at 4-year intervals, there was a negative correlation between the cumulative dose of TNFi and the change in mSASSS (beta = −3.308, 95% CI: −6.410 to −0.205; p = 0.037). This finding indicates that, as in the 2-year intervals, an increase in the cumulative dose of TNFi was associated with slower changes in the mSASSS. In Models 2 and 3, the cumulative TNFi dose demonstrated a negative correlation with changes in mSASSS (p = 0.065 and p = 0.062, respectively).
Multivariable linear mixed model for baseline mSASSS ⩽24 groups at 4-year intervals.

CI, confidence interval; CRP, C-reactive protein; ESR, erythrocyte sedimentation rate; HLA, human leukocyte antigen; mSASSS, modified Stoke Ankylosing Spondylitis Spinal Score; TNFi, tumor necrosis factor inhibitor.
Discussion
In this study, we examined the relationship between the cumulative dose of TNFi and changes in mSASSS among patients with radiographic axial SpA. Overall, our analysis did not demonstrate a statistically significant correlation between cumulative TNFi dose and radiographic progression when considering the entire patient cohort. However, in a multivariable model adjusted for age and sex, a negative trend was observed in patients with a baseline mSASSS of ⩽24 at the 2-year interval (β = −0.888, 95% CI: −1.793 to 0.017, p = 0.055), although this association did not meet conventional thresholds for statistical significance. In contrast, no correlation was observed in patients with a baseline mSASSS exceeding 24, suggesting that once structural damage has advanced, any potential benefit of TNFi may be attenuated. It is important to note that the univariable models did not reveal a significant association, which, together with the borderline significance in the subgroup analysis, underscores the need for cautious interpretation. The discrepancies between the univariable and multivariable results may be attributed to confounding by baseline mSASSS and other patient characteristics, as well as potential sample size limitations. These findings lend partial support to recent guidelines advocating a “window of opportunity” for early aggressive treatment in radiographic axial SpA.24,25 However, further validation with larger datasets is needed.
When patients are treated with a TNFi and their disease activity is stable, dose reduction may be recommended for safety and cost-effectiveness in long-term treatment.4,5 This approach can prevent recurrence and sustain low disease activity more effectively than discontinuing TNFi in patients with radiographic axial SpA. 26 Additionally, it has been reported that reducing the dose of TNFi is not inferior to maintaining a standard dose.10,12 According to Korean health insurance data, 17.8% of patients with axial SpA reduce their TNFi dose to half of the standard dose within 2 years. This practice has also been associated with reduced medical costs, indicating that TNFi dose reduction can be beneficial for patients with axial SpA who require long-term treatment. 20
Therefore, the benefits of dose reduction should not be overlooked. A meta-analysis examining TNFi dose reduction assessed its effectiveness and safety in axial SpA. This analysis included six randomized trials with 747 patients, encompassing axial and nonaxial SpA. 9 This study reported that the standard dose of TNFi was more effective in achieving and maintaining low disease activity. However, the included randomized controlled trials were few, which may introduce bias, highlighting the need for methodologically well-designed studies to better understand the benefits of TNFi dose reduction. Additionally, the study emphasized the difficulty of evaluating significant radiological changes associated with TNFi dose reduction in randomized controlled clinical trials.
A 2-year study investigating TNFi tapering according to a predefined algorithm reported that 52% of the patients received a reduced dose, which was maintained for 2 years without clinical deterioration. 14 Notably, imaging changes, including radiograph and magnetic resonance imaging, were followed for 2 years. Although the magnitude of change over 2 years was minimal, there was a significant increase in the mSASSS (11.7 (18.7) to 12.2 (19.0)). However, the 2-year observation period was insufficient to fully evaluate the effect of TNFi dose reduction on radiological progression.
We designed this study to address the shortcomings of previous studies using extensive retrospective data. The study period was divided into 2-year or 4-year intervals, and the doses used for the four types of TNFi were converted into cumulative doses compared to the standard dose. This method allowed us to utilize the available data to overcome the limitations of previously introduced methods and detect subtle radiological changes due to dose reduction. In particular, we converted the therapeutic dose of TNFi into a cumulative dose because the use of the four types of TNFi varied over the 2- or 4-year period for various reasons. This method effectively generated statistical values by processing the cumulative dose of TNFi over a certain period, thus addressing complex problems such as TNFi switching, drug rest periods, and irregular outpatient visits by patients using subcutaneous injections. However, these methods cannot fully capture the prescriber’s intent. The cumulative dose metric used in our study does not reliably differentiate between complete treatment discontinuation and a reduction in dose (e.g., receiving 50% of the standard dose or extending dosing intervals). This limitation may introduce heterogeneity in our analysis, as the clinical implications of discontinuation versus dose reduction are distinct. Moreover, we lack detailed information on the reasons behind these adjustments (e.g., infection, concomitant diseases, remission). Therefore, our analysis was conducted under the assumption that patients continued to meet the criteria for maintaining low disease activity, as required for medication maintenance under the National Health Insurance. Future studies should aim to capture these distinctions in order to better elucidate their impact on radiographic progression.
However, this study had certain limitations. First, we used various TNFi at different doses and formulations to treat axial SpA. These effects were expressed as indicators of the cumulative TNFi dose. Second, the number of patients treated for >4 years was limited; therefore, the relationship between the cumulative dose of long-term TNFi and radiographic progression could not be fully established. Third, the cumulative dose of TNFi may be tapered by a doctor due to low disease activity but may be reduced due to infection or patient factors. Therefore, we analyzed long-term TNF inhibitor use in terms of cumulative dose, acknowledging that tapering may have occurred for heterogeneous reasons, some of which may be related to ongoing disease activity, potentially confounding the observed relationship with structural progression. Fourth, because this was a retrospective study, replacing randomized controlled trials with tapering or dose reduction was difficult. Fifth, the potential influence of chronic NSAID treatment on structural damage progression could not be fully assessed in our study due to the lack of detailed medication histories. While most patients reported pro re nata (PRN) use, the absence of standardized data on NSAID exposure represents a limitation that should be addressed in future research.
Conclusion
Our overall analysis did not reveal a statistically significant association between cumulative TNFi dose and radiographic progression in the entire patient cohort. However, in subgroup analyses, we observed that among patients with a baseline mSASSS ⩽24, a negative correlation was present over a 2- to 4-year period. In contrast, no significant association was found in patients with more advanced baseline damage. These findings suggest that, at least in patients with minimal structural damage, maintaining a standard TNFi dose rather than tapering it may help prevent further progression of radiographic damage. Nonetheless, because this association was observed only in a specific subgroup and did not reach conventional statistical significance in the overall analysis, further validation in larger cohorts is warranted.
Supplemental Material
sj-docx-1-tab-10.1177_1759720X251358022 – Supplemental material for Association between cumulative TNF inhibitor dose and spinal radiographic progression in radiographic axial spondyloarthritis in patients with modified stoke ankylosing spondylitis spinal score ⩽24
Supplemental material, sj-docx-1-tab-10.1177_1759720X251358022 for Association between cumulative TNF inhibitor dose and spinal radiographic progression in radiographic axial spondyloarthritis in patients with modified stoke ankylosing spondylitis spinal score ⩽24 by Tae-Hwan Kim, Seo Young Park, Ji Hui Shin, Seunghun Lee, Kyung Bin Joo and Bon San Koo in Therapeutic Advances in Musculoskeletal Disease
Supplemental Material
sj-docx-2-tab-10.1177_1759720X251358022 – Supplemental material for Association between cumulative TNF inhibitor dose and spinal radiographic progression in radiographic axial spondyloarthritis in patients with modified stoke ankylosing spondylitis spinal score ⩽24
Supplemental material, sj-docx-2-tab-10.1177_1759720X251358022 for Association between cumulative TNF inhibitor dose and spinal radiographic progression in radiographic axial spondyloarthritis in patients with modified stoke ankylosing spondylitis spinal score ⩽24 by Tae-Hwan Kim, Seo Young Park, Ji Hui Shin, Seunghun Lee, Kyung Bin Joo and Bon San Koo in Therapeutic Advances in Musculoskeletal Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
