Abstract
Background:
Axial spondyloarthritis (axSpA) is a chronic inflammatory disease primarily affecting the axial skeleton, with tumor necrosis factor inhibitors (TNFi) as the standard first-line biologic disease-modifying antirheumatic drugs since the early 2000s. Interleukin-17 inhibitors (IL-17i) have emerged as effective alternatives, but the optimal sequencing after first-line TNFi discontinuation remains uncertain.
Objectives:
To compare real-world treatment persistence of IL-17i and TNFi as second-line therapies following initial TNFi discontinuation in patients with axSpA.
Design:
A nationwide retrospective cohort study using the Korean National Health Insurance Service-National Health Information Database (2010–2023).
Methods:
We included patients with axSpA who initiated TNFi as first-line targeted therapy and received IL-17i or TNFi as second-line therapy. The primary outcome was discontinuation of the second-line drug within 1 year. Drug retention was analyzed using Kaplan–Meier curves, and discontinuation risk was estimated using Cox proportional hazards models with multivariable adjustment, inverse probability of treatment weighting (IPTW), and IPTW with covariate adjustment. Subgroup analyses were conducted by type of prior treatment failure (primary or secondary).
Results:
Among 1,660 patients (IL-17i: 375; TNFi: 1285), overall treatment persistence and discontinuation risk were similar between groups. However, patients with primary treatment failure had significantly higher discontinuation risk with TNFi versus IL-17i, with adjusted hazard ratios ranging from 1.54 (95% CI, 1.14–2.07) to 2.11 (95% CI, 1.13–3.94). Results remained consistent across all analytic approaches.
Conclusion:
While overall persistence was comparable, IL-17i showed greater retention than TNFi among patients with primary treatment failure to initial TNFi therapy.
Plain language summary
Axial spondyloarthritis (axSpA), including ankylosing spondylitis, is a chronic inflammatory disease that affects the spine and sacroiliac joints. Biologic therapies such as tumor necrosis factor inhibitors (TNFi) are commonly used, but some patients discontinue these treatments because of insufficient response or side effects. After stopping the first TNFi, patients may switch either to another TNFi or to an interleukin-17 inhibitor (IL-17i). This study analyzed nationwide Korean health insurance data from 2010 to 2023 to compare how long patients continued their second biologic treatment after discontinuing the first TNFi. We evaluated treatment persistence (drug retention) and the risk of discontinuation within one year. The analysis included 1,660 patients with axSpA who received either IL-17i (375 patients) or TNFi (1,285 patients) as second-line therapy. Overall, treatment persistence was similar between IL-17i and TNFi users. However, in patients whose first TNFi failed to show sufficient effect (referred to as primary failure), those who switched to an IL-17i were significantly more likely to continue treatment than those who switched to another TNFi. These findings were consistent across several statistical analyses. In conclusion, both IL-17i and TNFi are effective options as second-line biologic therapies for patients with axSpA. However, IL-17i may provide better treatment continuity for those who did not respond adequately to their initial TNFi. These results may assist clinicians and patients in selecting optimal second-line biologic treatments for axial spondyloarthritis.
Keywords
Introduction
Axial spondyloarthritis (axSpA) is a chronic inflammatory rheumatic disorder that primarily affects the axial skeleton. 1 It is classified as radiographic axSpA (r-axSpA), or ankylosing spondylitis, when definitive structural changes such as sacroiliitis are present on X-ray. 2 Patients with axSpA require long-term pharmacological management throughout the disease course. The main therapeutic goals are to suppress inflammation, alleviate symptoms, and prevent structural progression, including fibrosis, calcification, and eventual spinal ankylosis.2–4
Tumor necrosis factor inhibitors (TNFi) have been recommended as first-line biologic disease-modifying antirheumatic drugs (bDMARDs) for patients with persistently active disease despite adequate non-steroidal anti-inflammatory drug (NSAID) trials, based on the 2003 Assessment of SpondyloArthritis International Society (ASAS) consensus and evidence from randomized controlled trials (RCTs) of agents such as etanercept.2,4–7 Since their introduction, TNFi use has increased, and they are now widely regarded as the standard of care for NSAID-refractory axSpA, with up to 41.6% of patients in Korea receiving TNFi therapy.8,9
Recently, interleukin-17 inhibitors (IL-17i) have emerged as effective options for axSpA, with demonstrated efficacy in RCTs and comparable real-world effectiveness to TNFi.2,10–12 These agents are now considered suitable as both first- and second-line bDMARDs. However, the optimal strategy following TNFi discontinuation remains uncertain, particularly regarding whether to cycle to another TNFi or switch to a bDMARD with a different mechanism, such as IL-17i, due to limited head-to-head comparative data.
The 2019 American College of Rheumatology (ACR) guidelines recommend a mechanistic switch in cases of primary nonresponse—defined as a lack of meaningful clinical improvement within 3–6 months of TNFi initiation—on the premise that early treatment failure may involve non-TNF-α–mediated pathways. 7 However, supporting evidence for this strategy remains limited. In contrast, the 2022 update from the ASAS and the European Alliance of Associations for Rheumatology (EULAR) adopts a pragmatic approach, supporting flexibility in subsequent treatment selection without requiring a change in therapeutic class. 2 The Korean guideline similarly recommends this flexible approach in choosing subsequent therapies. 13
This study aimed to evaluate real-world treatment persistence after discontinuation of initial TNFi, specifically comparing drug survival between patients who cycled to a second TNFi and those who switched to IL-17i.
Methods
Data source
We conducted a nationwide, population-based study using the Korean National Health Insurance Service-National Health Information Database (NHIS-NHID) from January 2010 to December 2023. The NHIS system covers about 97% of the population, with the remaining 3% receiving medical aid through Korea’s universal healthcare system. This comprehensive claim database includes demographic information, income-based insurance contribution levels, biannual health screening data, and detailed records on inpatient and outpatient visits, procedures, and prescriptions across all healthcare settings. 14
Study design and population
This retrospective cohort included patients newly diagnosed with axSpA between January 2013 and December 2023. To ensure inclusion of incident cases, patients with any recorded diagnosis of axSpA between January 2010 and December 2012 were excluded. Eligible individuals were required to (i) initiate TNFi as first-line biologic therapy and (ii) subsequently receive either another TNFi or an IL-17i as second-line therapy before 2023. Patients who initiated JAK inhibitors or biologic therapies in a non-sequential manner were excluded. Patients were assigned to the TNFi or IL-17i group according to their second-line agent. TNFi agents included adalimumab, etanercept, infliximab, and golimumab, while IL-17i agents included secukinumab and ixekizumab. The index date was defined as the initiation of the second biologic agent.
AxSpA was defined as at least two primary diagnoses using the International Classification of Diseases, 10th Revision (ICD-10) code M45. Patients with concurrent diagnoses of rheumatoid arthritis (M05, M06) or systemic lupus erythematosus (M32) were excluded. Diagnostic confirmation was strengthened by enrollment in the Korean Rare Intractable Disease (RID) program, which provides financial support based on physician-confirmed diagnoses using standardized criteria. RID codes were applied for r-axSpA (V140), RA (V223), and SLE (V136).8,15 RID registration for r-axSpA requires fulfillment of the modified New York criteria, which mandate radiographic sacroiliitis (bilateral grade ⩾II or unilateral grade III/IV) and at least two of the following: (1) chronic back pain lasting >3 months and improving with exercise, (2) limited lumbar motion, or (3) decreased chest expansion. 16
Outcome
The primary outcome was drug retention of the second biologic agent (TNFi or IL-17i) after discontinuation of initial TNFi during follow-up. Treatment episodes were defined from prescription data, with the start date as the prescription date and the end date calculated by adding the regimen-specific dosing interval (Supplemental Table 1). Consecutive prescriptions without a gap >90 days were combined into a single episode.
Discontinuation was defined as no subsequent prescription for the same agent within 90 days after the end of the previous episode, without switching to another targeted therapy. Switching was defined as initiation of a different targeted agent, marking the start of second-line therapy. Patients who switched were considered to have experienced first-line treatment failure. To minimize misclassification, cases with overlapping prescriptions for two targeted agents exceeding 8 weeks were excluded from the study population.
Covariates
Baseline characteristics were measured at second-line therapy initiation. Covariates included age, sex, employment status, income quartile, body mass index (BMI <18, 18–22, 23–25, and ⩾25 kg/m2, unknown), and smoking status (never, past, current, unknown). Healthcare facility type was classified as tertiary hospital or other. To account for potential temporal effects related to the approval of IL-17 inhibitors in 2017, the calendar period (before vs after 2017) was additionally included as a covariate.
Disease duration at index date was categorized as <12, 13–24, 25–48, and ⩾49 months. Any axSpA-related inpatient visits were assessed within 1 year prior to the index date. Comorbidities were identified using diagnostic codes from the 2 years preceding the index date, including dactylitis, enthesitis, fibromyalgia, psoriasis, inflammatory bowel disease (IBD) (Crohn’s disease and ulcerative colitis), uveitis, depression, anxiety, malignancy, and the Charlson Comorbidity Index. Episodes of pneumonia and serious infections requiring hospitalization during first-line TNFi therapy were also evaluated.
Statistical analysis
Baseline characteristics were compared using t-tests for continuous variables and χ2 tests for categorical variables. Median values were compared using the Mann–Whitney U test. Treatment transitions were visualized with a Sankey diagram.
Kaplan–Meier curves estimated drug retention, with group differences assessed by log-rank test. Patients were followed until discontinuation, switching to another targeted therapy, or for a maximum of 1 year, whichever occurred first.
To estimate the hazard ratio (HRs) and 95% confidence intervals (CIs) for treatment discontinuation, we used Cox proportional hazard model with three analytic approaches: (1) a multivariable-adjusted model, (2) inverse probability of treatment weighting (IPTW), and (3) IPTW with additional covariate adjustment.
Five multivariable models (Models 1–5) adjusted for sex, disease duration, type of treatment failure, comorbidities, healthcare setting, and year of second-line therapy initiation. To address potential confounding, IPTW based on propensity scores was applied. For variables that remained imbalanced after weighting, additional covariate adjustment was performed to enhance comparability between treatment groups.
Subgroup analysis
Treatment failure included discontinuation due to intolerance, primary nonresponse, or secondary nonresponse. In this study, subgroup analyses assessed drug retention according to the type of prior TNFi failure, comparing primary or secondary treatment failure. Primary treatment failure was defined as discontinuation within 4 months of TNFi initiation, based on the Korean National Health Insurance Service reimbursement policy requiring Bath Ankylosing Spondylitis Disease Activity Index (BASDAI) improvement at 3–4 months 17 ; secondary treatment failure referred to discontinuation after 4 months.
Sensitivity analyses
Sensitivity analyses were performed to specifically evaluate discontinuations due to nonresponse, excluding events likely related to adverse events or intolerance. Hospitalizations occurring within 3 months before discontinuation of second-line therapy were reviewed by investigators to determine probable cause (Supplemental Tables 2–5). Additional analyses assessed the robustness of findings by applying alternative definitions of primary treatment failure, using 3- and 6-month thresholds.
Results
Study population
Between 2013 and 2022, 1660 patients with axSpA who initiated a second-line targeted therapy after prior TNFi use were included (IL-17i group: 375 (22.6%); TNFi group: 1285 (77.4%); Figure 1).

Cohort selection algorithm and exclusion criteria for comparative analysis of second-line TNF inhibitor and IL-17 inhibitor use.
Treatment sequences
Adalimumab was the most frequently prescribed first-line TNFi, followed by etanercept and infliximab. For second-line targeted therapy, adalimumab remained the most common agent, followed by secukinumab and golimumab (Figure 2). Treatment patterns were similar across primary and secondary treatment failure groups. Secukinumab was more frequently selected following secondary treatment failure, whereas switches from adalimumab to etanercept were more common in the primary treatment failure group.
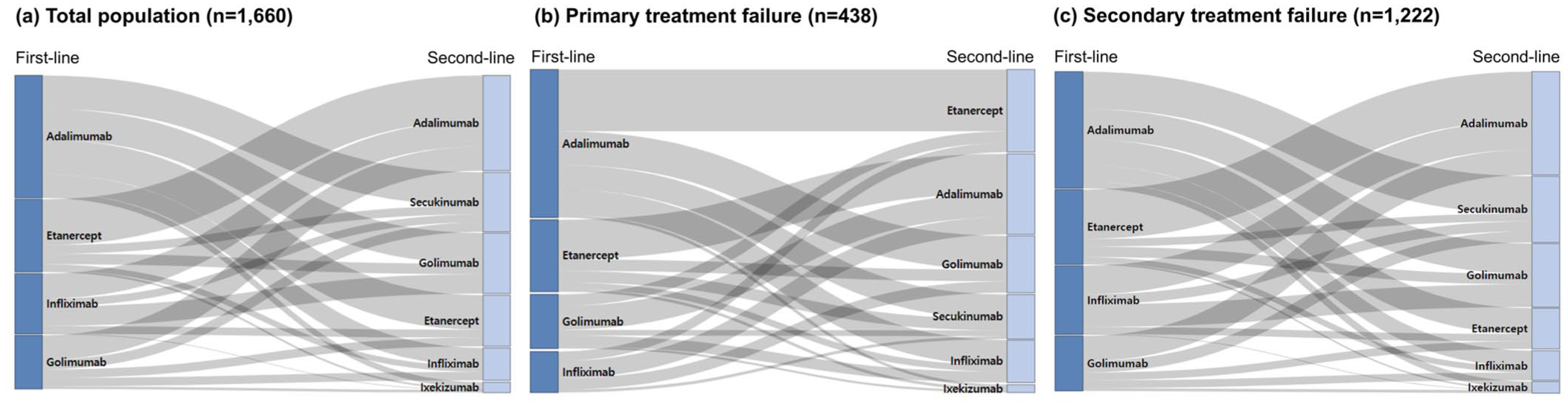
Treatment sequences from first-line TNF inhibitors to second-line targeted therapies.
Baseline characteristics at second-line therapy initiation
At second-line therapy initiation, IL-17i users were significantly older (mean age 43.6 years (standard deviation; SD 13.4) vs 40.9 years (SD 13.5); p-value = 0.008; Table 1); however, this difference was not considered clinically relevant. IL-17i users also had longer disease duration (mean 38.9 months (SD 28.1) vs 30.6 months (SD 25.0); p-value < 0.001) compared with TNFi users. IL-17i users were also less likely to have been hospitalized for axSpA-related issues in the preceding year (10.1% vs 15.8%; p-value = 0.006) and had a lower proportion of nonsmokers (33.3% vs 41.9%; p-value = 0.007). Comorbidity patterns differed: psoriasis was more common in IL-17i users (21.1% vs 6.2%; p-value < .001), while IBD (1.6% vs 4.4%; p-value = 0.012), uveitis (17.1% vs 23.5%; p-value = .008), and anxiety (14.4% vs 19.1%; p-value = .039) were more prevalent in TNFi users.
Baseline characteristics of patients initiating second-line IL-17 inhibitor versus TNF inhibitor therapy.

Data are n (%), or mean (SD).
BMI, body mass index; IL-17i, interleukin-17 inhibitor; SD, standard deviation; TNFi, tumor necrosis factor inhibitor.
Regarding first-line TNFi, IL-17i users were more likely to have received monoclonal antibody therapies (85.1% vs 74.0%; p-value < 0.001) and had longer treatment duration (median 432 days (IQR 164–1089) vs 303 days (IQR 109–748); p-value < .001; Table 2). The IL-17i group also had a lower rate of primary treatment failure to first-line TNFi (19.5% vs 28.4%; p-value < .001).
First-line TNF inhibitor characteristics in patients initiating second-line IL-17 or TNF inhibitor therapy.
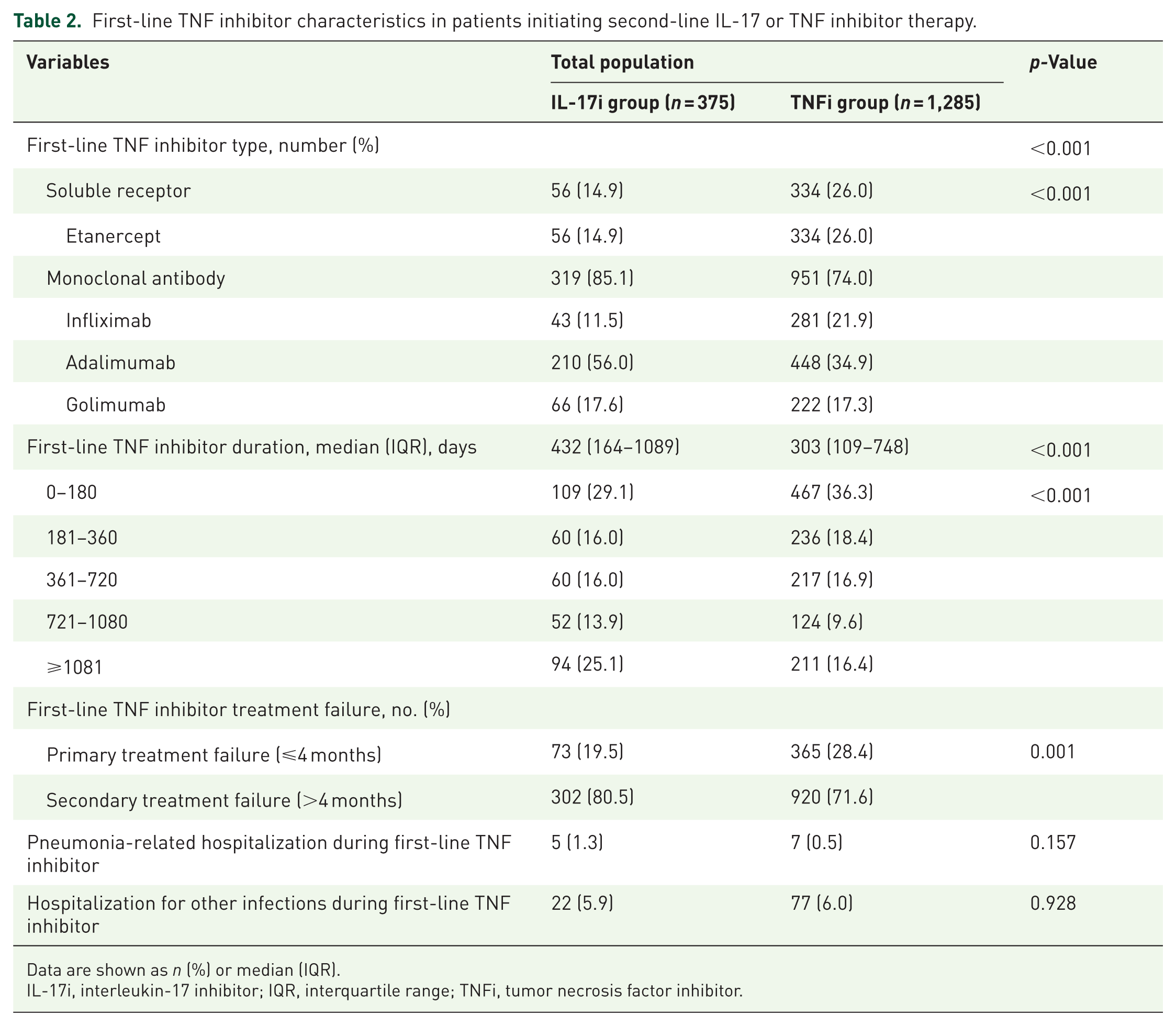
Data are shown as n (%) or median (IQR).
IL-17i, interleukin-17 inhibitor; IQR, interquartile range; TNFi, tumor necrosis factor inhibitor.
Comparative drug retention
Kaplan–Meier analysis showed no significant difference in overall drug retention between the IL-17i and TNFi groups in the entire cohort. However, among the primary treatment failure group, IL-17i was associated with significantly longer drug survival (log-rank p-value = 0.016; Figure 3).

Kaplan–Meier curves comparing drug retention between second-line IL-17 inhibitor and TNF inhibitors.
In Cox proportional hazards models, TNFi use was not significantly associated with treatment discontinuation in the total population (crude HR 1.27; 95% CI: 0.98–1.64), and this remained non-significant across adjusted models (adjusted HRs 1.20–1.24; Table 3). In IPTW analyses, however, TNFi use was associated with increased risk of discontinuation (HR 1.22; 95% CI: 1.04–1.41), and this result persisted after additional covariate adjustment.
Risk of second-line treatment discontinuation for TNF inhibitor versus IL-17 inhibitor users in the total population, primary treatment failure, and secondary treatment failure groups.
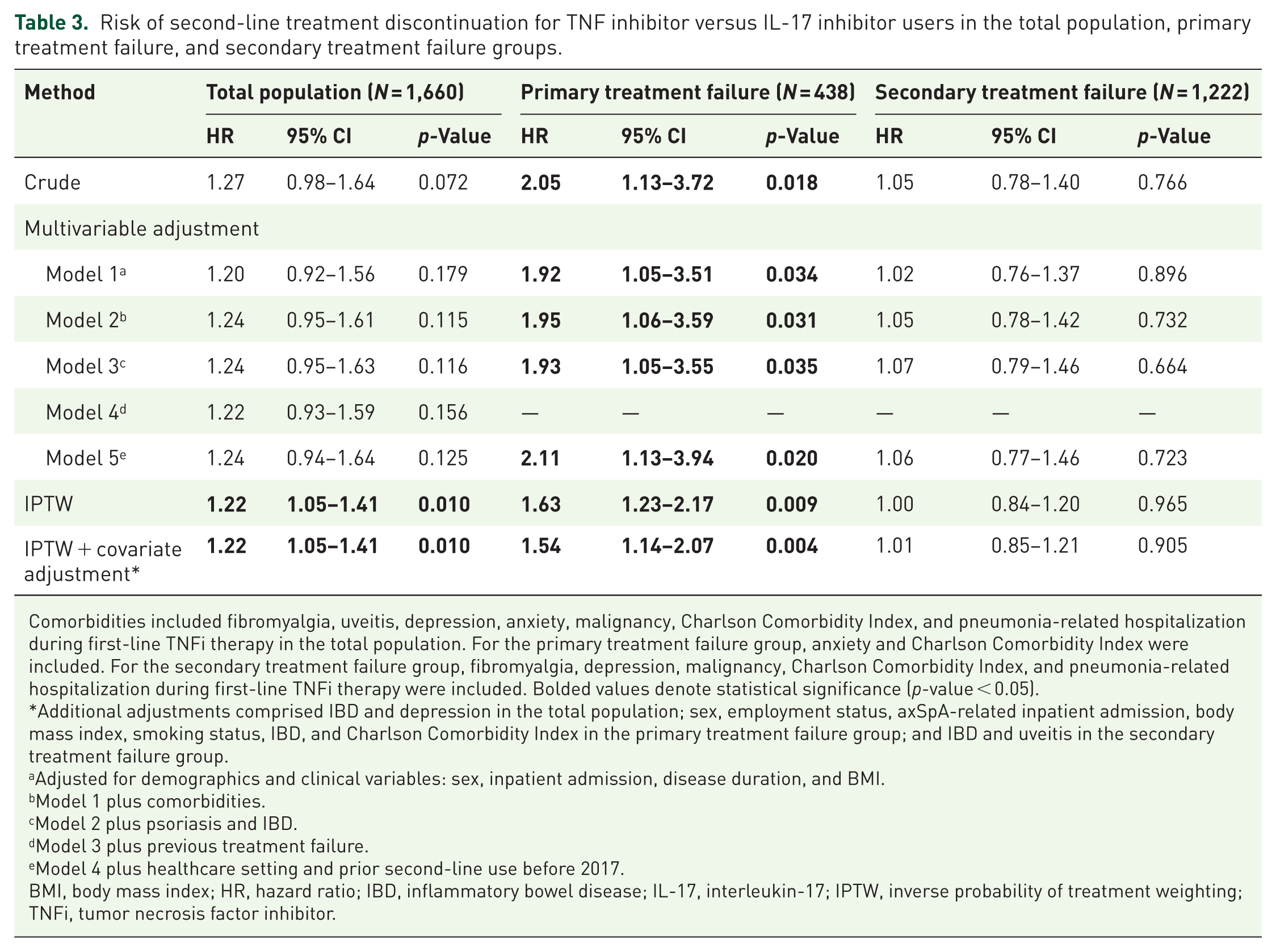
Comorbidities included fibromyalgia, uveitis, depression, anxiety, malignancy, Charlson Comorbidity Index, and pneumonia-related hospitalization during first-line TNFi therapy in the total population. For the primary treatment failure group, anxiety and Charlson Comorbidity Index were included. For the secondary treatment failure group, fibromyalgia, depression, malignancy, Charlson Comorbidity Index, and pneumonia-related hospitalization during first-line TNFi therapy were included. Bolded values denote statistical significance (p-value < 0.05).
Additional adjustments comprised IBD and depression in the total population; sex, employment status, axSpA-related inpatient admission, body mass index, smoking status, IBD, and Charlson Comorbidity Index in the primary treatment failure group; and IBD and uveitis in the secondary treatment failure group.
Adjusted for demographics and clinical variables: sex, inpatient admission, disease duration, and BMI.
Model 1 plus comorbidities.
Model 2 plus psoriasis and IBD.
Model 3 plus previous treatment failure.
Model 4 plus healthcare setting and prior second-line use before 2017.
BMI, body mass index; HR, hazard ratio; IBD, inflammatory bowel disease; IL-17, interleukin-17; IPTW, inverse probability of treatment weighting; TNFi, tumor necrosis factor inhibitor.
Subgroup analyses by type of prior TNFi failure
Among the secondary treatment failure group, risk of discontinuation did not differ significantly between treatment groups. In contrast, among the primary treatment failure group, TNFi use was consistently associated with a higher risk of discontinuation compared to IL-17i. The crude HR was 2.05 (95% CI: 1.13–3.72), with adjusted HRs from 1.92 to 2.11. This subgroup effect largely explained the overall risk elevation observed in IPTW models. Associations were consistent in both covariate-adjusted and IPTW-weighted analyses.
Sensitivity analyses to assess robustness of findings
To assess the robustness of the primary findings, we conducted multiple sensitivity analyses. First, we evaluated whether hospitalizations potentially attributable to adverse events influenced discontinuation outcomes (Supplemental Tables 2 and 3). The frequency of such events was similar between groups, with infections being the most common cause. Diagnostic codes for these events are provided in Supplemental Tables 4 and 5.
A sensitivity analysis using a stricter outcome definition—excluding patients with hospitalizations suggestive of treatment-related adverse events—yielded results consistent with the main analysis (Supplemental Table 6).
Further analyses using alternative definitions of primary treatment failure at 3 and 6 months also corroborated the main findings (Supplemental Tables 7 and 8).
Discussion
The findings from this large, nationwide cohort study provide real-world evidence on the comparative persistence of IL-17i and TNFi as second-line therapies in axSpA following TNFi discontinuation. The principal finding is that, while overall drug retention is similar between IL-17i and TNFi, IL-17i demonstrates significantly greater persistence in patients with primary TNFi failure. Although IL-17i users were older and other baseline differences existed, covariates statistically significant in univariable analyses were adjusted for multivariable models and were balanced using IPTW. Results remained consistent across these analyses, indicating that baseline differences did not account for the observed differences in treatment persistence. The robustness of these findings across multiple analytic approaches and subgroup analyses supports a mechanism-based approach to biologic sequencing in axSpA.
TNFis are recommended as first-line biologic therapy for axSpA in both EULAR and ACR guidelines, reflecting their established efficacy and safety in patients with persistent disease activity despite NSAID treatment.2,7 However, a substantial proportion of patients require a change in therapy due to inadequate response or intolerance. 18 The mechanisms underlying TNFi resistance are heterogeneous. Primary nonresponse is often attributed to disease driven by patient characteristics and alternative inflammatory pathways, such as the IL-17/Th17 axis, while secondary loss of efficacy is more commonly related to immunogenicity or pharmacokinetic factors.19,20
Previous real-world studies have also demonstrated similar effectiveness between IL-17i and TNFi in patients with prior TNFi exposure. A cohort study using the Swiss Clinical Quality Management registry showed comparable drug retention and no significant difference in BASDAI50 response at 1 year between secukinumab and TNFi. 12 Similar results were observed in the Korean College of Rheumatology Biologics and Targeted Therapy (KOBIO) registry, which additionally assessed response outcomes including ASAS20, ASAS40, and Ankylosing Spondylitis Disease Activity Score (ASDAS), and found no significant difference between treatment groups. 21 These studies provide important insights into the comparative effectiveness of secukinumab and TNFi in real-world populations, both in terms of drug retention and clinical response. However, differences in prior bDMARD exposure likely underestimated secukinumab’s efficacy, as more patients had ⩾2 prior bDMARDs compared to those on TNFi in both studies. The present study extends this evidence by analyzing a larger, nationally representative cohort restricted to patients who received TNFi as first-line therapy, allowing for a more accurate comparison of second-line treatment outcomes.
Although prior studies have shown similar effectiveness between IL-17i and TNFi, variability in individual treatment responses persists. Such inadequate responses can be subcategorized into primary and secondary nonresponse, a distinction recognized by the 2019 ACR guidelines, which recommend different strategies based on response type. 7 The importance of this distinction was further highlighted in a prospective cohort study at a tertiary hospital in Korea, which found that patients with primary treatment failure to a first TNFi had a higher risk of discontinuing a second TNFi, whereas secukinumab was not associated with this risk. 22 Although this study was underpowered to draw statistically robust conclusions due to a small sample size, it supports the hypothesis that switching to a therapy with a different mechanism may be more effective in patients with primary treatment failure. This may reflect an underlying non-TNF-α–driven disease process that responds better to IL-17 inhibition.
Another retrospective cohort study using the KOBIO registry compared drug retention and clinical efficacy between secukinumab and TNFi, including a subgroup analysis by primary and secondary nonresponse. 17 This study found no significant difference among primary non-responders in the risk of drug discontinuation between groups. However, among secondary non-responders, secukinumab was associated with a higher risk of discontinuation and lower odds of BASDAI50 response. This finding may be explained by differences in prior treatment exposure: in the secukinumab group, 93% of patients had received more than two prior TNFi, compared to only 21% in the TNFi group. Since later-line use of either TNFi or IL-17i is generally associated with lower response rates, this imbalance in treatment sequencing likely influenced the observed outcomes. 23 Similar findings have been reported, with higher drug survival in patients receiving TNFi compared to those receiving IL-17i; however, this difference was not statistically significant after adjustment for the number of prior bDMARDs. 24 Therefore, the effectiveness of secukinumab may have been underestimated due to greater prior bDMARDs in that group. While relatively better outcomes with TNFi in secondary non-responders warrant further investigation, these results should be interpreted cautiously given baseline imbalances.
Strengths and limitations
This study adds to the growing body of real-world evidence by examining drug retention among axSpA patients who, after discontinuing a first TNFi, initiated second-line targeted therapy, comparing those cycling to a second TNFi with those switching to an IL-17i.12,17,22 The use of a large, representative dataset from the Korean National Health Insurance Service system, where biologic use is strictly regulated, enhances generalizability in this healthcare setting. Mandatory BASDAI assessments for reimbursement likely reduce misclassification of treatment response, supporting drug retention as a valid proxy for effectiveness.
Although the 4-month cut-off for defining primary treatment failure may be affected by documentation or administrative delays, sensitivity analyses using 3- and 6-month thresholds yielded consistent results, supporting this definition. Analytically, the study employed rigorous methods, including multivariable adjustment and IPTW, to address confounding. Clinical adjudication of potential adverse event-related discontinuations by independent physicians further strengthened outcome classification. Consistency across sensitivity analyses supports the robustness of these findings.
However, several limitations should be considered when interpreting these findings. Non-radiographic axSpA could not be identified in Korean administrative claims data and was therefore not included. Disease duration reflected time since radiographic diagnosis rather than symptom onset. However, this inherent limitation of claim-based data does not affect internal comparability. In addition, the absence of detailed clinical variables such as ASDAS and inflammatory markers (e.g., CRP) limits direct assessment of disease activity. While inflammatory burden may influence biologic selection and response, this limitation does not undermine the observed differences in treatment retention by primary and secondary failure status and highlights the need for studies incorporating clinical data.
Reliance on claims data also prevents definitive determination of discontinuation reasons, despite adjudication efforts. Although previous studies indicate that reasons for first TNFi discontinuation do not reliably predict second-line outcomes,25,26 residual confounding—including unmeasured factors such as radiographic progression—cannot be ruled out. A formal sample size calculation was not performed because the cohort size was determined by the retrospective study design and the available eligible population. Although the sample size of the primary treatment failure subgroup was limited, the consistency of findings across multiple analytic approaches supports the reliability of our results. Finally, because treatment selection in real-world practice is influenced by multiple patient- and physician-level factors that cannot be fully captured in claims data, these findings should be interpreted with appropriate caution.
Conclusion
This real-world cohort study demonstrated that, following TNFi treatment failure, overall drug retention was similar between IL-17i and TNFi. However, among patients with primary treatment failure, IL-17i showed significantly higher retention than TNFi, suggesting potentially greater effectiveness of IL-17i in this subgroup. These findings highlight the importance of considering treatment response type when selecting second-line biologic therapy. Nevertheless, head-to-head RCTs are necessary to confirm these results, and further studies should explore optimal treatment sequences following IL-17i use to preserve long-term therapeutic options.
Supplemental Material
sj-docx-1-tab-10.1177_1759720X261424367 – Supplemental material for Persistence of IL-17 and TNF inhibitors following initial TNF inhibitor use in axial spondyloarthritis: a nationwide retrospective cohort study
Supplemental material, sj-docx-1-tab-10.1177_1759720X261424367 for Persistence of IL-17 and TNF inhibitors following initial TNF inhibitor use in axial spondyloarthritis: a nationwide retrospective cohort study by Yu-Seon Jung, Soo-Kyung Cho, Se Rim Choi, Sun-Young Jung and Yoon-Kyoung Sung in Therapeutic Advances in Musculoskeletal Disease
Footnotes
Acknowledgements
During the preparation of this work, the authors used ChatGPT in order to enhance language clarity and EssayReview for professional English editing. After using these services, the authors reviewed and edited the content as needed and take full responsibility for the content of the publication.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
