Abstract
Dactylitis – a hallmark clinical feature of psoriatic arthritis (PsA) – that occurs in 30–50% of PsA patients, is a marker of disease severity for PsA progression, an independent predictor of cardiovascular morbidity and impairs the motor functions of PsA patients. There is a paucity of evidence for the treatment due to the absence of randomized controlled trials assessing dactylitis as a primary endpoint and current practice arises from the analysis of dactylitis as a secondary outcome. Corticosteroid (CS) injections for dactylitis in PsA patients are a therapeutic treatment option for patients with isolated dactylitis or for patients with flares in tendon sheaths, despite stable and effective systemic treatment. The aim of this narrative review is to briefly illustrate the clinical aspects of dactylitis in PsA, the imaging and clinimetric tools used to diagnose and monitor dactylitis, the current treatment strategies and principally to provide a comprehensive picture of the clinical efficacy and safety with ultrasound-guide and blind techniques of CS injections for dactylitis in PsA patients.
Introduction
Dactylitis, derived from the Greek word ‘daktylos’ meaning finger and also known as ‘sausage-shaped’ digit, is a diffuse finger or toe swelling due to an underlying inflammatory or infiltrative disorder.1,2 Dactylitis can also be defined as uniform swelling such that the soft tissues between the metacarpophalangeal (MCP) and the proximal interphalangeal (PIP) and the distal interphalangeal (DIP) joints and the digital tuft are diffusely swollen to the extent that the actual joint swelling can no longer be independently recognized.3,4 The different forms of dactylitis can be classified as inflammatory infectious (tuberculous dactylitis, syphilitic dacylitis, and blistering distal dactylitis), inflammatory non-infectious (spondyloarthritis (SpA) dactylitis, sarcoid dactylitis and gouty dactylitis) and non-inflammatory (sickle cell dactylitis). 2
In SpA dactylitis, there is a diffuse swelling of the entire digit due to synovitis, tenosynovitis, and soft-tissue inflammation. 2 Dactylitis, although is more frequent in psoriatic arthritis (PsA), is one of the clinical manifestations of all the SpA, a group of interrelated diseases which includes, ankylosing spondylitis, PsA, reactive arthritis, arthritis associated with inflammatory bowel disease, and the undifferentiated forms.1,2,5–7
Dactylitis occurs in 30–50% of PsA patients, often in early disease 8 and may sometimes occur for a long time in isolation as the only clinical manifestation of PsA and there are also patients with psoriasis who only exhibit, for years, dactylitis. 1 The point prevalence of dactylitis in PsA is >50% in some contemporary randomized controlled trials (RCTs) that include tumor necrosis factor inhibitors (TNFi) and interleukin (IL)-17–IL-23 axis cytokine inhibitors (i). 3
The aim of this narrative review is to briefly illustrate the clinical aspects of dactylitis in PsA, the imaging and clinimetric tools used to diagnose and monitor dactylitis, the current treatment strategies and principally to provide a comprehensive picture of the clinical efficacy and safety with ultrasound-guide and blind techniques of corticosteroid (CS) injections for dactylitis in PsA patients.
Clinical aspects, imaging, and clinimetric assessment
Clinical aspects
Dactylitis (‘sausage’-shaped fingers or toes) – considered a hallmark clinical feature of PsA – is clinically presented with sausage-like swelling of both digits of the hands (Figure 1(a) and (b)) and feet. Digits involved are presented with swelling and slight redness that are due to flexor tenosynovitis and marked adjacent soft-tissue swelling with a variable degree of small joint synovitis.1,9–11

(a) Dactylitis of the second digit of the right hand (frontal view). (b) Dactylitis of the second digit of the right hand compared to the contralateral (lateral view).
Physical examination in acute dactylitis shows swelling, tenderness, and pain mostly along the flexor tendons of the involved digit1,12 and the digit swollen is so marked that the patient cannot flex his finger. Asymmetry versus the contralateral digit also favors dactylitis diagnosis. 10 Sometimes, there is also pain and swelling in the joints of the dactylitic finger or toe. There is no need to obtain imaging evaluation for the clinical diagnosis since physical examination has the same specificity and sensitivity as magnetic resonance imaging (MRI) for the diagnosis of flexor tenosynovitis.2,12–14
Diagnosis of PsA dactylitis by personal medical history is more difficult. In this case, there are only two possibilities for the diagnosis: (a) to show a photograph of finger or toe dactylitis to the patient for confirmation; (b) to examine the routine radiograph obtained by the patient’s physician at the time of the sausage-like swelling looking for the typical diffuse soft-tissue swelling on X-rays.2,15
Tender dactylitis, for yet unclear mechanisms, is a marker of disease severity for PsA progression in general. In fact, significantly greater joint damage and erosions occur in dactylitic in comparison to non-dactylitic digits and digits that show only arthritis.5,10,11 Furthermore, dactylitis can be an independent predictor of cardiovascular morbidity 10 and impairs the motor functions required for daily living. 16
Imaging
Ultrasound (US) and MRI have been used for diagnosing early PsA and have provided additional clues to the pathogenesis of the disease and permitting to differentiate PsA from rheumatoid arthritis (RA). 17
US can detect tenosynovitis, pseudotenosynovitis, extracapsular inflammatory changes, dactylitis, joint synovitis, subcutaneous edema, and abnormalities of the accessory pulleys 3 (Figures 2 and 3).

Tenosynovitis of the III right flexor tendon characterized by marked synovial proliferation, a moderate increase of the tendon thickness and a thickened and hypoecoic peritendineal tissue. Synovial sheath widening (circle) associated with soft-tissue edema (asterisk). Power Doppler function revealed diffuse and severe vascular signal inside and around the tendon sheath (Grade 3).
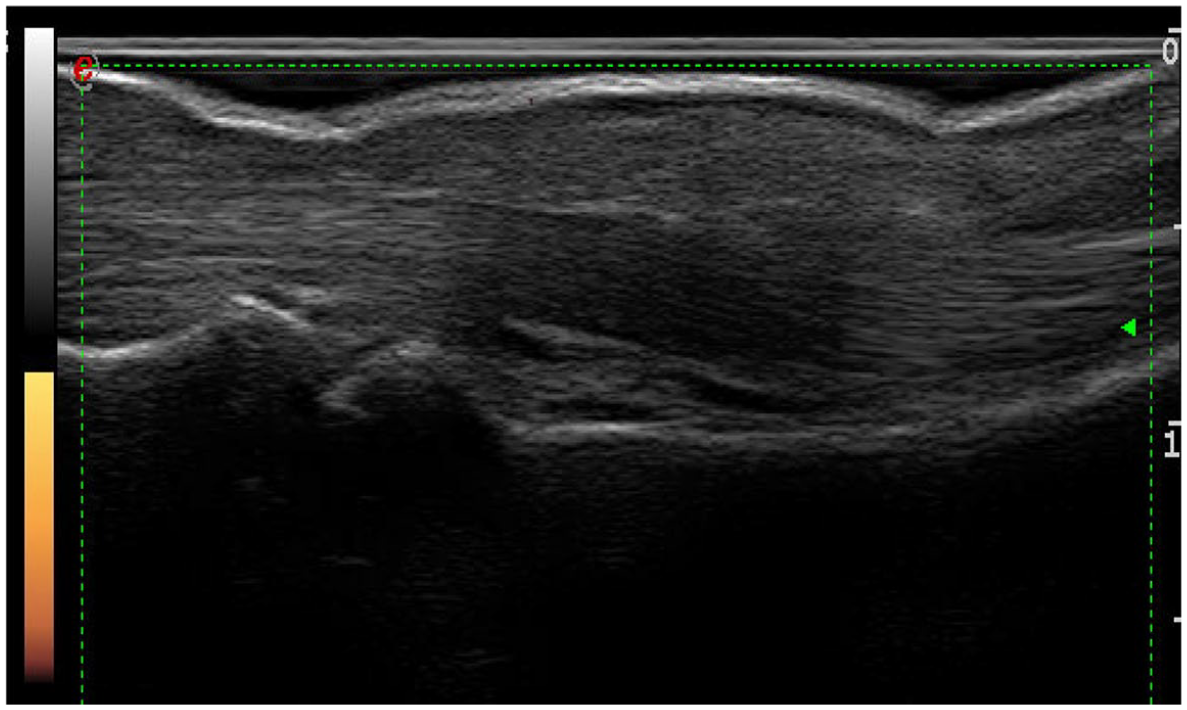
US follow-up after 4 weeks of the same tendon. Note the dramatic reduction of the power Doppler signal (Grade 0) and gray scale score (Grade 0) with the resolution of the flexor tenosynovitis and the soft-tissue edema.
Extra-tendon soft-tissue inflammation and small joint enthesophytes at flexor tendon insertions seen by US has been shown in patients with PsA-related tenosynovitis but not with RA-related tenosynovitis. 18
US is a useful tool in PsA management for diagnosis, monitoring therapy and as an instrument guidance approach to steroid injections at the level of inflamed joints, tendon sheaths, and entheses. 17
The first US and MRI studies established that dactylitis is due to flexor tenosynovitis and marked adjacent soft-tissue swelling with a variable degree of joint synovitis occurring in 17–66% of the dactylitis.12,13,19,20
MRI in particular has contributed to better understand PsA pathogenesis by establishing that enthesis is the primary target of inflammation.17,21
In 2008, Healy and co-workers reported in their MRI study on PsA dactylitis that bone edema ranged from small areas adjacent to the joint capsule insertions to edema involving the whole phalanx supporting the hypothesis by McGonagle on a primary involvement of the entheses in dactylitis.2,22
In a comprehensive MRI study of PsA-related dactylitis nine features were found including synovitis, bone edema, osteitis, subcutaneous edema, flexor tenosynovitis, extensor tenosynovitis, inflammatory changes of the plantar and/or volar plates and/or the collateral ligaments, erosions and sesamoiditis at the thumb and great toe. 22
Finally, European League Against Rheumatism (EULAR) recommendations on the use of imaging in the management of PsA suggest the use of US or MRI (with the last considered secondary for the lower availability and high costs) when peripheral involvement (dactylitis, enthesitis, tenosynovitis, bursitis and arthritis) is suspected.17,23
Clinimetric assessment
The recognition that dactylitis is a distinct entity from arthritis led to the introduction of dactylitis digit counts as an outcome measure in some RCTs. Since it was difficult to make an objective score for the severity or extent of dactylitis, various methods have so far been used. 9 Initially, the most straightforward method was to count affected digits (score range 0–20). Afterwards, the Dactylitis Severity Score (DSS) was developed and frequently used.24,25 In 2005, Helliwell and colleagues developed a clinically objective and validated instrument for dactylitis called the leeds dactylometer. 26 This instrument led to the development of a more objective measure, the Leeds Dactylitis Index (LDI) that quantifies both the size and tenderness so that the score can differentiate between tender and non-tender dactylitis and which was able to identify improvements in dactylitis in RCTs where dactylitis was a secondary outcome measure.26–29
Current treatment strategies
The natural clinical course of dactylitis is largely unknown. In some patients, acute dactylitis is often the inaugural feature of PsA and may resolve without any treatment or it may remain the only manifestation of PsA for months to years. 10
Dactylitis is a critical domain in PsA clinical trials and treatment algorithms and is associated with more erosive forms and more active form of PsA thus, early diagnosis and therapy, favors better outcomes. Unfortunately, as highlighted in the Group for Research and Assessment of Psoriasis and Psoriatic Arthritis (GRAPPA) recommendation, 30 there is paucity of evidence in this field due to the absence of RCTs assessing dactylitis as a primary endpoint and current practice arises from the analysis of dactylitis as a secondary outcome.
To date there is only one study addressing dactylitis as a primary endpoint, the GO-DACT study, 31 a multicentre, randomized, double-blind, placebo-controlled, phase-3b trial of golimumab plus methotrexate (MTX) versus placebo plus MTX, in MTX-naïve and biologic disease-modifying anti-rheumatic drugs (bDMARDs) naïve patients with PsA and active dactylitis. The primary endpoint was DSS change from baseline to week 24. The GO-DACT trial showed that the combination of golimumab plus MTX is associated with significantly greater clinical improvements in dactylitis in comparison with MTX monotherapy. 31
Dactylitis has been treated with non-steroidal anti-inflammatory drugs (NSAIDs), CS injections, conventional synthetic (cs) DMARDs, targeted synthetic (ts) DMARDs and bDMARDs.32–34 Compared with bDMARDS, csDMARDS alone are less effective. 9 tsDMARDs such as apremilast and tofacitinib have also been shown to be effective in the treatment of dactylitis. 3
According to the recommendation by GRAPPA 30 most physicians use NSAIDs and CS injections as first-line therapy, although they have not been formally studied in RCTs. 30 According to GRAPPA recommendation, for resistant cases, csDMARDs such as MTX, leflunomide (LEF), sulfasalazine (SSZ) or a tsDMARDS such as apremilast (a phosphodiesterase 4 inhibitor (PDE-4i)) are recommended. For resistant cases, biologics such as TNFi or IL-12/23i should be initiated. In case of TNFi failure or IL-12/23i failure, it is recommended switching to another bDMARDs (TNFi, IL-12/23i, IL-17i) or to a PDE-4i.
In the updated 2019 EULAR recommendations, 35 local CS injections are proposed as initial therapy or adjunctive therapy in PsA.
CS injections
CS injection for dactylitis in PsA patients is a therapeutic treatment option for patients with isolated dactylitis or in patients with flares in tendon sheaths, despite stable and effective DMARDs treatment.
Local treatment with steroid injection into the tenosynovial sheath is widely used by many clinicians in everyday clinical practice despite this therapeutic strategy is largely empirical because there is lack of data on dactylitis therapy and because randomized, double-blind placebo-controlled trials have not been conducted. 35 While intra-articular CS gives positive results for a few weeks, injections into tendon sheaths often give months of relief. 36
Steroid injection could be performed also into the joint (MCP, PIP and DIP) at the same time of intratenosynovial sheath injection because synovitis and tenosynovitis are integral features of dactylitis. 3
Contraindications and side effects
Absolute contraindications for local CS injection are local infection at dactylitis site and allergy to CS preparation or anesthetics (if used); relative contraindications are anticoagulation therapy or bleeding diathesis, systemic infection and uncontrolled diabetes. 37
Adverse reactions 37 of dactylitis CS injection include the following:
Acute reactions (during injection–within 1 hour): vasovagal reaction, pain, local ecchymosis and bleeding and anaphylaxis for anesthetic.
Intermediate reactions (hours–days): post-injection flare of pain, facial flushing, menstrual irregularity, pain at the finger, hyperglycemia usually with no clinical risk also in patients with diabetes mellitus. 38
Late reactions: tendon rupture, infection, skin depigmentation and skin atrophy.
Tendon rupture is a recognized complication of CS injections,39–42 but no instance of tendon rupture after more than 300 CS injections for idiopathic and rheumatoid digital flexor tenosynovitis was found by Gray and colleagues. 43
Type of CS preparations
There is little evidence to guide medication selection for CS injections for dactylitis, and most recommendations are based on a combination of clinical experience and personal preference.
Common local injectable CS preparations for dactylitis include triamcinolone acetonide, triamcinolone hexacetonide, methylprednisolone acetate, dexamethasone sodium phosphate and betamethasone sodium phosphate (see Table 1). Long-acting insoluble CS have been developed to enhance anti-inflammatory effect and have a longer duration of action resulting in a prolonged clinical benefit despite a higher incidence of cutaneous side effects.36,44 Triamcinolone hexacetonide is the least soluble – as well as the least rapid for onset of action – of the commonly used CS, followed by triamcinolone acetonide.44,45 In our experience, the most commonly used CS for PsA dactylitis injections is triamcinolone acetonide followed by methylprednisolone acetate.
Steroid dosing and equivalents.

CSs are listed in order of prevalence of use in our experience.
Therapeutic scheme
For stenosing tenosynovitis, single CS injection is considered to be the best method of care, and in the event of full reduction of symptoms and then recurrence, a second injection may be used.46,47 Single local injection with triamcinolone acetonide is effective and safe for treating trigger finger and the effects of steroid injections last up to 12 months. 48 For trigger finger, in patients with partial improvement, optimal timing of the next injection appears to be after 69 days.49,50
In a 10-year single-center retrospective study of US CS tendon sheath injections in juvenile idiopathic arthritis, only 58/926 (6.3%) tendon sheaths required a repeat injection and only 2 minor – with no major complications – were observed, exhibiting a high technical success rate and safety. 51
There are no data for the optimal timing for a further injection for dactylitis in PsA, but in our experience, a second injection – 4 weeks after the first – could be necessary in the limited cases of patients with partial response. Moreover, CS injection should be guided by the response to previous injections, the availability of other therapies, clinical judgment and patient preferences but we rarely inject a single digit more than two or three times.
Adding anesthetics
Reasons to mix local anesthetics with CS for dactylitis injection (lidocaine 1%-2%, xylocaine 1% or bupivacaine 0.25%) are diagnostic (relieved pain immediately indicates accurate injection), therapeutic (to bridge the interval before the CS yields effect) and preventive (diminishing post-injection pain and flare, diminishing the risk of skin atrophy injecting a dose of anesthetics after CS injection to clear the needle track). As with the choice of CS, the choice of local anesthetics is based more on personal preference and experience than on evidence.37,44,50 In our clinical practice, we do not mix local anesthetics for the increased risk of allergy and vasovagal reactions.
General injection technique
Informed consent must be obtained before performing the procedure discussing risks (risk of pain, bleeding, cutaneous atrophy or skin discoloration, steroid flare reaction, and rare risk of infection, tendon rupture, and possible failure to improve), benefits and alternatives with the patients. To start, wash your hands because injections must be performed using aseptic technique or with no touch technique. Prepare equipment for CS injection: 1 mL syringes with fine needle (25 Gauge, 0.5 mm × 16 mm), triamcinolone acetonide 40 mg/mL (0.5–1 mL) or methylprednisolone acetate 40 mg/mL (0.5–1 mL), local anesthetic if needed (0.5 mL of bupivacaine 0.25%, 0.5 mL of xylocaine 1% or 0.5 mL lidocaine 1–2%), isopropyl alcohol, chlorhexidine or povidone iodine, topical analgesia with ethyl chloride cooling spray and bandage. Position the patient with the hand supine on the table with supplemental lighting if available. Identify and mark the site of injection with a skin pencil or by a pressure mark with tip of an ear speculum to guide needle placement. Clean overlying skin with povidone iodine, chlorhexidine or isopropyl alcohol. Use ethyl chloride spray which gives immediate anesthesia. Put on sterile gloves or do not touch the injection site. The patient and the physician should be in a comfortable position and the syringe should be in the dominant hand. Enter the skin with an angle of 45° with proximal to distal direction and advance until the tip of the needle is at the injection site (Figures 4 and 5). Drawback the plunger to make sure the needle tip is not located within a vessel and inject CS into the tendon sheath and if resistance is met, withdraw slightly the syringe and reposition before further injection of CS because injection should not be given into a tendon for the risk of rupture. We place more or less 1 mL of CS in the affected tendon sheath and then we remove the needle applying bandage or a sticking plaster. After the injection, the patient has to flex and extend the digit to help the spreading of the CS within the flexor tendon sheath. The patient is then again advised for absolute rest of the injected area for 1 day and limited use for 2 weeks and reminded that, for the characteristic of the CS preparation, the full benefit of the procedure will be experienced in 1 or 2 weeks. If the patient experiences a local reaction starting after 48 hours – despite the risk is very low using no touch technique or aseptic technique – there is a need for evaluation for infection.37,44,50,52
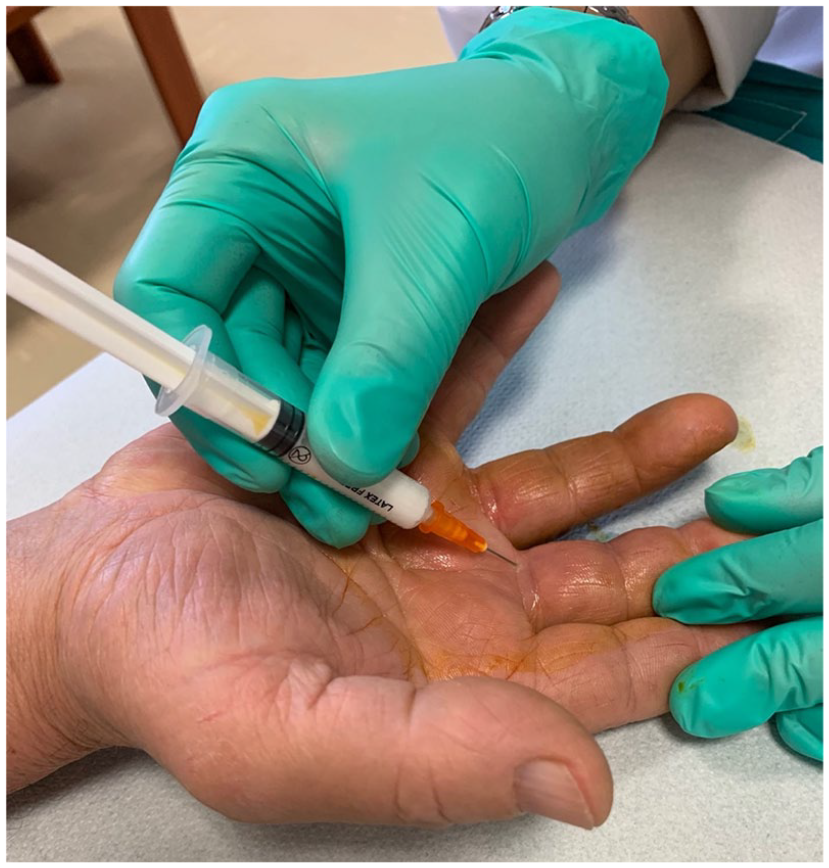
Blind corticosteroid injection for PsA dactylitis.

US-guided corticosteroid injection for PsA dactylitis.
Blind CS injection technique
A blind CS injection (Figure 4), if given correctly, is an effective method to treat dactylitis. For CS injection into digital flexor sheath there is a need of knowledge of the local anatomy to minimize risk and maximize benefit.
The flexor tendon sheath is a membranous structure that surrounds the flexor tendons from the metacarpal neck to the volar plate of the DIP. It is attached longitudinally to the underlying bony structures. The sheath is thick over the bones and thin in areas overlying the joints, to allow digital flexion. The A1 pulley marks the proximal border of the flexor sheath and overlies the metacarpal neck. The proximal border can be roughly defined by the distal palmar crease. 53
The injection is placed at the base of the digit (Figure 4), through the flexor crease where the digit meets the hand. Place the needle in the midline of the finger, through the finger flexion crease at the base of the finger, and angle it approximately 45° distally, with the bevel of the needle facing distally (see Figure 4). Advance the needle through the skin until the resistance encountered by the needle is decreased, indicating that the needle is within the flexor sheath. The plunger of the syringe should depress easily, indicating that the needle is most likely in the flexor tendon sheath.
Proper needle placement requires attention to anatomical landmarks and lack of resistance during injection. 43 Injection from a midlateral approach at the proximal phalangeal level is also been described. 52
Toe dactylitis
In the feet, the most commonly affected toe is the fourth. 3 Like finger dactylitis, toe dactylitis is due to flexor tenosynovitis, and extensor tenosynovitis may be present in addition. 13
CS injection for toe dactylitis is placed at the plantar base of the involved toe entering the skin with an angle of 45° and advancing the tip of the needle with proximal to distal direction. The remaining procedure is the same as for finger dactylitis.
US-guided CS injection technique
US-guided CS injection (Figures 2, 3 and 5) can be performed using a high-frequency linear transduces (6–18 MHz). The digit to be injected is prepared in a sterile fashion, and the US probe is swept along the tendon in both transverse and longitudinal planes to identify a location along the tendon sheath that permits a safe injection. The needle that appears as a hyper-reflective line is advanced under direct US visualization into the tendon sheath in the transverse or longitudinal approach and is directly visualized ensuring that the tip is located within the tendon sheath. The transverse approach (out of plane) is favored because it provides better visualization and permits to direct and place the needle tip away from the tendon fibers.51,54 At the injection, CS is visualized spreading within the tendon sheath verifying the correct placement of the drug. This could be followed by injection of local anesthetic to clear the needle track and help prevent cutaneous atrophy. 51
US-guided or blind CS injection for dactylitis
An early RA study, comparing US and clinical examination in the detection of flexor tenosynovitis, found that clinical examination is a valuable tool for detecting flexor tenosynovitis for its high specificity and positive predictive values, but a negative clinical examination does not exclude inflammation and an US should be considered. 55
Blind intra-articular injections in peripheral joints achieve good accuracy with a satisfactory clinical response in RA when performed by a trained physician. 56
Luz and colleagues 57 demonstrated that US did not increase the accuracy of wrist injections for RA when they were performed by an experienced rheumatologist.
In a systematic review with meta-analysis, 58 based on one trial (moderate quality), injection into the tendon sheath of the long head of the biceps brachii resulted more accurately performed using US guidance than by the blind method 59 and based on another trial (low quality), the US group had a greater reduction in pain and greater improvement in function with no significant difference between groups for adverse events. 60
As for trigger finger treatment, true intra-sheath injection offered no apparent advantage over subcutaneous injection in the treatment of trigger digits 61 and the use of US-guided injection of CS showed no superior clinical benefits compared to the blinded technique. 62 US guidance offers no advantage over landmark guidance, other than greater accuracy of intra-sheath placement of CS for trigger finger. 50
In the triple-blinded randomized clinical trial of Mardani-Kivi and co-workers, it was found that extra-sheath CS injection and intra-sheath CS injection of the finger flexor tendon under US guidance in patients with trigger finger are equally effective. 63
Utilizing US guidance for CS injections into tendon sheaths has shown clinical advantage to conventional blind injections in the adult RA population in terms of accuracy and effectiveness of the therapy. 54
In the randomized comparative study of Gutierrez and colleagues, 54 the short-term efficacy of local CS injection for tenosynovitis – in patients with inflammatory chronic arthritis (including 32 PsA patients) – was demonstrated the superiority of US-guided CS injection over conventional blind injection in terms of better outcomes measured by functional, clinical and US scores. Furthermore, US-guided CS injection resulted less painful than the conventional blind injection. Despite US-guided CS injection is more time-consuming than the blind approach (6–10 minutes), on the light of these results, the US-guided approach should be considered the standard approach for tenosynovitis in rheumatological practices. 54
In our experience (data not published), US resulted a valuable tool for diagnosis, CS injection and treatment follow-up of dactylitis in PsA.
Current evidence for steroid injection
The anti-flogistic effect of local CS injection in para-articular disorders was first reported by Howard in 1953. 64
A systematic review of the efficacy of CS injections of tendon sheaths, excluding stenosing tenosynovitis of the wrist and hand, found that low-quality studies suggest that tendon sheath injections may provide at least short-term improvement in pain. 65
In a model to estimate the costs and outcomes associated with treatment strategies for trigger finger was found that offering 3 CS injections before surgery is a cost-effective strategy. The results were sensitive to the probability of injection site fat necrosis, success rate of steroid injections, time to symptom relief after a steroid injection, and cost of treatment. 66 Non-rheumatoid flexor tenosynovitis resolved in 100% of a small group of patients treated with CS injection and persistent benefit was noted in 93%. 67 In a recent comprehensive literature review on common chronic pain conditions of the hand was found that when the etiology of the pain is inflammatory, CS injections provide significant pain relief and increased functionality and that 10–20 mg of methylprednisolone or triamcinolone injections are the most common steroids used for small joint pain. One study in particular showed that triamcinolone injections compared to methylprednisolone had greater improvements in patient-reported pain and functionality. 46
In a randomized double-blind study, tenosynovitis remission in RA was achieved more frequently in the US-guided intratenosynovial CS injection group than in the intramuscular CS injection group. 68 In the study of Gray and colleagues, 43 reviewing the results of intratendon sheath CS injection in 173 episodes of flexor tenosynovitis in 46 RA patients, was found that 93% of the episodes resolved completely for 3 or more months (median: 25 months), the likelihood of a favorable response did not diminish with treatment of recurrent flexor tenosynovitis in a given digit and that tenosynovitis did not recur in 59% of cases.
To the best of our knowledge, there is only one observational study addressing the effectiveness of steroid injection in psoriatic dactylitis. 69 Girolimetto and colleagues found that one local CS injection of 20 mg of methylprednisolone acetate was able to lead to an improvement of dactylitis-related symptoms and digit swelling for at least 3 months in about 90% of cases. 69
In our experience (data not published), US-guided triamcinolone acetonide injection for dactylitis in PsA patients, resulted in a complete resolution of 8 out of 10 dactylitis at 4 weeks.
Another study investigated the short-term efficacy of CS loco-regional treatment performed under US guidance in tenosynovitis of patients with chronic inflammatory arthritis – including 11 PsA patients – found a significant improvement for the VAS pain, the tenderness score and the sonographic scorings for Gray Scale (GS) and Power Doppler (PD). 70 Similar results were reported by Girolimetto and colleagues showing that clinical improvements of active dactylitis are linked to the regression of US evidence of extracapsular inflammation (particularly flexor tenosynovitis and soft-tissue edema) at 1 and 3 months after local CS injection. These findings suggest the pivotal role of tenosynovitis and extra-articular disease for symptoms in PsA dactylitis. 71
Conclusion
CS injections can be recommended as a safe and effective treatment for dactylitis in PsA, especially if long-lasting CS preparation are injected in the flexor tendon sheath with a US-guided technique.
Future studies using dactylitis as a primary outcome measure will determine the most appropriate treatment for this painful and damaging condition. Given the importance and frequency of dactylitis in PsA, such studies should include both robust and quantifiable clinical indices (e.g. LDI and DSS), as well as imaging tools, the latter of which are particularly promising as valid and sensitive measure for diagnosis, treatment and follow-up of dactylitis in PsA.
Footnotes
Conflict of interest statement
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
