Abstract
Introduction:
Enthesitis is a hallmark of psoriatic disease, but its clinical assessment is problematic in terms of diagnostic sensitivity and overlap with other comorbid conditions. Ultrasound is a useful tool that can give a more detailed assessment of enthesitis. Research demonstrates that those with persistent ultrasound entheseal disease are at risk of progressive articular damage. With limited data to guide choice between biologic therapy for psoriatic arthritis (PsA) patients, we wanted to assess the response of ultrasound-confirmed enthesitis to different forms of biologic therapies and study its utility in making more informed decisions.
Methods:
This was an open label observational study including patients aged ⩾18 years, who fulfil the classification criteria for PSA (CASPAR) and were due to commence on their first biologic therapy. The primary outcome was the change in MAdrid Sonographic Enthesitis Index (MASEI) score at 16 weeks of treatment. The MASEI score was also modified to assess the active elementary lesions (ActiveMASEI).
Results:
In all, 80 PsA patients were enrolled with 75 patients completing the study [secukinumab n = 23 and tumour necrosis factor inhibitor (TNFi) n = 52]. The mean reduction in MASEI score after 16 weeks of treatment was 3.42 with TNFi versus 1.74 with secukinumab (p = 0.097). There was a significant difference in the change in the MASEIActive score for TNFi versus secukinumab (4.37 versus 2.26; p = 0.030) and this difference was more pronounced when only power Doppler signal within 2 mm of the enthesis insertion was included (4.37 versus 2.00; p = 0.007). Clinical outcomes were similar for both classes of biologic apart from a significant reduction in regards to the Dermatology Life Quality Index and Psoriasis Area and Severity Index score with secukinumab versus TNFi.
Conclusions:
We have for the first time compared the effect of ultrasound-confirmed enthesitis between different forms of biologic therapies for PsA. We have seen an overall improvement in entheseal scores for both classes of medications and demonstrated a larger reduction in active entheseal disease for TNFi versus secukinumab that merits further exploration.
Plain language summary
Introduction
Enthesitis is implicated in the pathogenesis, diagnosis and prognosis of psoriatic arthritis (PsA).1–6 It is an important feature of this disease, resulting in considerable pain and disability. However, clinical assessment of enthesitis is problematic in terms of diagnostic sensitivity 7 and overlap with other comorbid conditions such as fibromyalgia. Studies have demonstrated that 20% of PsA patients suffer from concomitant fibromyalgia 8 and these patients, despite having higher clinical disease scores, have similar levels of objective inflammation on imaging to those with PsA alone. 8 Imaging has emerged as the preferred option to assess enthesitis, and research has demonstrated that those with persistent ultrasound entheseal disease are at risk of progressive articular damage. 5 The MAdrid Sonographic Enthesitis Index (MASEI) is a validated scoring system in Spondylarthropathy that is straightforward to perform. 9 It assesses both active (hypoechogenicity, bursitis, power Doppler signal and thickening) and chronic (calcification, enthesophytes and erosions) entheseal disease.
There is limited data to help clinicians select the most appropriate biologic therapy for PsA patients and its various musculoskeletal manifestations. The recommended first-line biologic agent is a tumour necrosis factor inhibitor (TNFi). However, recent studies suggest that IL-17 inhibitors including ixekizumab and secukinumab are equally effective compared to adalimumab in treating joint disease and enthesitis but this is based on clinical data alone, with its limitations.10,11 We wanted to assess the response on ultrasound-confirmed enthesitis to different forms of biologic therapies to study its utility in making more informed decisions and correlate clinical and imaging data. The primary outcome of the study was to assess the change in MASEI score at 4 months from baseline assessment.
Methods
The reporting of this study conforms to the strengthening the reporting of observational studies in epidemiology (STROBE) statement. 12 This was an open-label observational study in which scanning occurred blinded. Patients were referred from their rheumatology consultant within the Belfast Health and Social Care Trust if they were to be commenced on biologic treatment under their care. To be included, patients had to be aged ⩾18 years, fulfil the classification criteria for PSA (CASPAR) and were due to commence on their first subcutaneous biologic therapy. Exclusion criteria included the usual exclusions for biologic therapy including any other autoimmune rheumatic condition. Patients could not be included if they were currently on steroids or had received intramuscular steroid or had local steroid injections at an enthesis site within the preceding 6 weeks.
Ultrasonography
The sonographer and primary investigator (PI) for the study was a trained rheumatologist with 6 years of experience in musculoskeletal ultrasonography and has European Federation of Societies for Ultrasound in Medicines and Biology (EFSUMB) accreditation. They are also part of the Group for Research in Psoriasis and Psoriatic Arthritis (GRAPPA) diagnostic Ultrasound Enthesitis Tool (DUET) group. 13 The sonographer was blinded to all clinical findings and treatment choice prior to scanning. Scanning took place in a darkened room and there was no discussion between sonographer and participant to prevent any clinical details being revealed. The patients were assessed within 2 weeks of commencing on biologic treatment and interpretation of the scans occurred as they were acquired. All patients were rescanned at least 16 weeks of treatment up to a maximum of 18 weeks. This was carried out by the same investigator blinded to treatment and clinical outcomes.
If a patient had not taken their medication during the treatment period for more 6 weeks, they were not included in the final analysis.
Inter- and Intra-observer variation assessment
Extensive inter-rater reliability exercises on enthesitis ultrasound scanning have been carried out by the PI as part of their role in the DUET study group and other multi-centre research.13,14 For completeness, 10 patients had their Achilles tendon rescanned by the primary investigator and another experienced sonographer locally. This was to ensure agreement in rating as per the MASEI protocol and its particular grading of the chronic and actives lesions of enthesitis as per the protocol noted below. In all, 46 patients had their baseline scans reassessed by the PI blinded to the initial result to evaluate intra-observer variability at the end of the study.
Entheses scanning protocol
Ultrasound was carried out with a Toshiba Xario 200 (Canon, West Sussex, UK) and a PLU-1204BT linear (5–18 MHz range) probe. Scanning of the entheses was as per the MASEI (Madrid Sonographic Enthesitis Index) protocol, 9 which examines six enthesis locations bilaterally: proximal plantar fascia, distal Achilles tendon, distal and proximal patellar ligaments, distal quadriceps and brachial triceps tendons). Ultrasound examinations of the knee enthesis (quadriceps and patellar sites) were performed with the patient in the supine position with the knees flexed 30°. For the Achilles tendon and plantar fascia, the patient was in the prone position with the feet hanging over the edge of the examination table at 90° of flexion. Triceps tendon entheses were evaluated with the subjects seated in front of the examiner, shoulders in internal rotation and elbows flexed 90°. The protocol for scanning is included in the Supplemental Material (Figure 1). In brief, the entheses were evaluated for the following: thickness, structure, calcifications, bursae, erosions and power Doppler signal in both the bursa and at the enthesis insertion. Thickness of the entheses was measured on the longitudinal plane as the maximum anteroposterior diameter in millimetres, at the proximal bony insertion.
A structure was defined as pathologic if loss of fibrillar pattern, hypoechoic aspect or fusiform thickening of the enthesis occurred; bone erosion was defined as a cortical interruption with a step-down contour defect witnessed on both longitudinal and transverse scan; and enthesophyte was defined as a step-up bony prominence at the end of normal bone profile. Calcifications were evaluated at the area of the enthesis insertion and classified according to size. Both ossifications and enthesophytes at the enthesis were also included as calcifications as per the protocol.
Blood flow was examined in each enthesis using power Doppler ultrasound, the settings of which were standardized with a frequency between 6.6 and 8.8 MHz, and a low wall filter. The pulse repetition frequency (PRF) is automatically set by the Toshiba Xario ultrasound machine based on the other parameters to allow for maximum sensitivity, and gain was adjusted to the point where power Doppler signal was not generated under the bone cortex. All power Doppler assessment was carried out with the joints relaxed.
The maximum MASEI score is 136. We further analysed the results to assess for the active components that make up the MASEI score including a thickened tendon, pathologic tendon structure, PD signal and bursitis, namely the MASEIActive which we presumed to be the lesions that would change with treatment. It includes any power Doppler signal that is within the length of the tendon at the entheses insertion and there is debate about a cut-off measurement for active enthesitis. The OMERACT group proposes to only include Doppler activity within 2 mm of the bony cortex. 15 The active score was therefore modified further to only include power Doppler signal within 2 mm of the enthesis (MASEImActive). As an exploratory outcome, we also included the common extensor origin at the lateral epicondyle (MASEIL) based on scoring from previous research, 16 the results of which are included in the Supplemental Material. All patients had all the sites scanned at baseline and at 4 months of treatment and scores were calculated.
Clinical assessment
Enthesitis clinical assessment was carried out by PI after ultrasound scanning was completed and included the Spondyloarthritis Research Consortium of Canada Enthesitis Index (SPARCC) and Lead Enthesitis Index (LEI). Other clinical indices included the body mass index (BMI), 66 swollen joint count, 68 tender joint count, Psoriasis Area and Severity Index (PASI), Nail Psoriasis Severity Index (NAPSI), body surface area (BSA) and dactylitis count.
The C-reactive protein (CRP) was recorded from the electronic patient record. The validated disease activity measures such as Disease Activity in Psoriatic Arthritis (DAPSA) and Minimal Disease Activity (MDA) were calculated.
We also included patient-reported outcomes including the Health Assessment Questionnaire-Disability Index (HAQ-DI), patient global assessment visual analogue score (VAS), Dermatology Life Quality Index (DLQI), patient pain assessment VAS and the Bath Ankylosing Spondylitis Disease Activity Index (BASDAI).
Statistical analysis
Statistical analyses were performed using the IBM SPSS Statistic Version 26.0. Baseline characteristics were assessed for continuous variables by an independent t-test and for categorial variables by Fisher’s exact test. A change in ultrasound score for each participant was subjected to an independent samples t-test that did not assume equal variances in the group and comparisons made across treatments administered. Spearman’s correlation coefficients were utilized to assess clinical and imaging indices.
For intra-observer analysis, continuous variables were assessed using a coefficient repeatability score to demonstrate the smallest possible change which would be deemed to represent a true difference in the observations. Statistical significance was determined to be a p < 0.05.
Results
In all, 80 PsA patients were enrolled with the screening and recruitment process summarized in Supplemental Material 2. All patients received the dosing as per their drug licence approval with 24 patients commenced on secukinumab (150 mg, n = 18; 300 mg, n = 6) and 56 on TNFi (adalimumab n = 50, certolizumab pegol n = 4 and etanercept n = 2). In total, 75 patients completed the study (secukinumab n = 23 and TNFi n = 52) with one patient in secukinumab group and one in the TNFi group declining to attend follow-up and three patients in the TNFi group having over 6 weeks of gap in the treatment. Baseline characteristics are as per Table 1 and were similar to either class of biologic apart from the baseline DLQI score. The average age was 45.29 (12.74) years and 42 (52.5%) participants were female.
Baseline characteristics.

p
BASDAI, Bath Ankylosing Spondylitis Disease Activity Index; BMI, body mass index; csDMARD, conventional synthetic disease modifying anti rheumatic drugs; CRP, C-reactive protein; DAPSA, Disease activity in Psoriatic Arthritis; DAS-28, Disease activity score; DLQI, Dermatology Life Quality Index; HAQ, Health assessment Questionnaire Disability Index; LEI, Leeds Enthesitis Index; MASEI, Madrid sonographic enthesitis index; MASEIActive, Active elementary lesion of the Madrid sonographic enthesitis index; MASEImActive, MASEIActive score with modification to only include power Doppler <2 mm from enthesis insertion; NAPSI, Nail Psoriasis Severity Index; PASI, Psoriasis Area and Severity Index; SPARCC, Spondyloarthritis Research Consortium of Canada Enthesitis Index; TNFi, tumour necrosis factor inhibitor.
Ultrasound outcomes
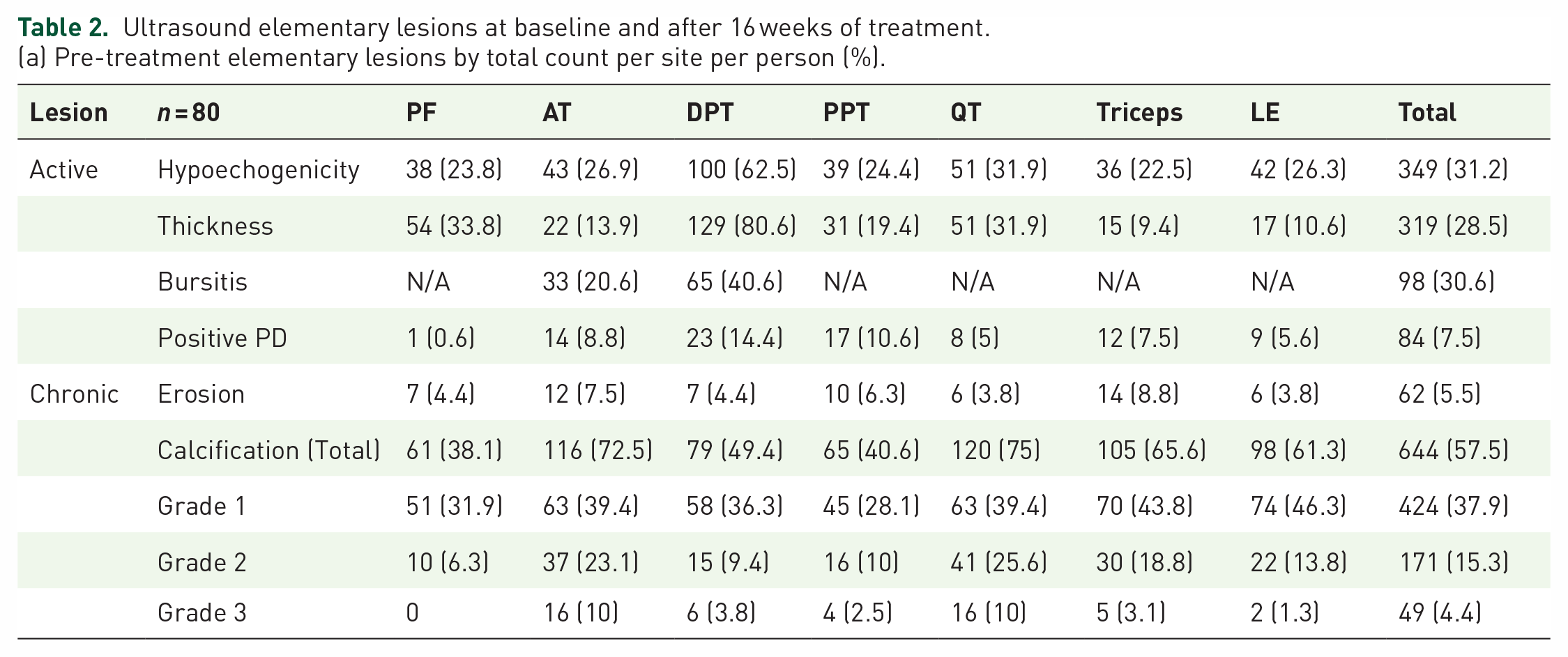
A breakdown of the elementary lesions demonstrated on scanning is shown in Table 2 both by site and by patient. In terms of active lesions, the most common finding was hypoechogenicity seen in 31.2% of entheseal sites examined at baseline, and the distal patellar tendon was the most common site for active entheseal changes. Positive power Doppler signal was seen at 84 out of 1120 (7.5%) entheses examined. In terms of chronic lesions, erosions were most commonly witnessed at the triceps insertion. Calcification or enthesophyte formation was seen at 57.5% of entheseal sites with grade 1 being the most common findings. The ultrasound outcomes are shown in Table 3. For the primary outcomes, the mean difference in the change of the overall MASEI score was 3.42 with TNFi versus 1.74 with secukinumab (p = 0.097). When counting only the active entheseal lesions, there was a larger mean difference in the change for the MASEIActive score for TNFi versus secukinumab (4.37 versus 2.26 p = 0.030), and this difference was more pronounced when only power Doppler signal within 2 mm of the enthesis insertion was included (4.37 versus 2.00 p = 0.007). Similar results were obtained when we compared only adalimumab with secukinumab (Supplemental Table 4).
Ultrasound elementary lesions at baseline and after 16 weeks of treatment.
(a) Pre-treatment elementary lesions by total count per site per person (%).
(b) Post-treatment elementary lessons by total count per site per person (%).

r site per person (%).
(c) Post-treatment elementary lesions present per patient (%).

(d) Post-treatment elementary lesions present per patient (%).

AT, Achilles tendon; DPT, distal patellar tendon; LE, lateral epicondyle; PF, plantar fascia; PPT, proximal patellar tendon; QT, quadriceps tendon.
Ultrasound outcomes with change in ultrasound score (SD) by treatment administered.

p < 0.05 in bold.
MASEI, Madrid sonographic enthesitis index; MASEIActive, active elementary lesion of the Madrid sonographic enthesitis index; MASEImActive, MASEIActive score with modification to only include power Doppler <2 mm from enthesis insertion.
The intra-observer coefficient repeatability (CR) measurement for the MASEI score and MASEI Active score was r = 1.87 and 0.65, respectively. In summary for the MASEI score, which has a maximum value of 136, the true difference on intra-reader measurements is less than 1.87. For the 10 patients who had their Achilles tendon assessed again by an independent sonographer and the PI, 60% of cases had the exact same score and for the remaining four patients the difference was only by 1 point out a maximum of 12.
Clinical outcomes
Clinical outcomes are shown in Table 2. There was a significant reduction for secukinumab versus TNFi in certain skin outcomes, namely the PASI score [3.44 (3.50) versus 1.03 (2.30); p = 0.001] and DLQI score [5.57 (7.52) versus 1.35 (4.18); p = 0.005]. Otherwise, there was similar clinical findings seen for both classes of biologic therapy.
Outcomes by treatment administered.

BASDAI, Bath Ankylosing Spondylitis Disease Activity Index; CRP, C-reactive protein; DAPSA, disease activity in psoriatic arthritis; DAS-28, disease activity score; DLQI, Dermatology Life Quality Index; HAQ, Health assessment Questionnaire Disability Index; LEI, Leeds Enthesitis Index; MDA, Minimal Disease Activity; NAPSI, Nail Psoriasis Severity Index; PASI, Psoriasis Area and Severity Index; PsARC, Psoriatic Arthritis Response Criteria; SPARCC, Spondyloarthritis Research Consortium of Canada Enthesitis Index.
Correlation with clinical and ultrasound enthesitis assessment
The Spondyloarthritis Research Consortium of Canada Enthesitis Index (SPARCC) had a positive mild correlation with the baseline MASEIActive score (r = 0.23, p = 0.042) and a change in SPARCC significantly, albeit mildly correlated with a change in MASEIActive (r = 0.28, p = 0.014). This was also seen with the MASEImActive and SPARCC scores both for baseline score and change in score (r = 0.24, p = 0.03; r = 0.30, p = 0.01). The SPARCC score did not significantly correlate with the baseline MASEI score but a change in overall MASEI score did mildly correlate with a change in SPARCC score (rs = 0.17, p = 0.13; rs = 0.32, p = 0.005). There was a positive but not significant correlation with the LEI score at baseline and with change in the three ultrasound assessments. Full details of the correlation scores are noted in Table 5.
Correlation between clinical enthesitis scores and ultrasound assessment both at baseline and change with treatment.

p<0.05 in bold.
LEI, Leeds Enthesitis Index; MASEI, Madrid sonographic enthesitis index; MASEIActive, Active elementary lesion of the Madrid sonographic enthesitis index; MASEImActive, MASEIActive score with modification to only include power Doppler <2 mm from enthesis insertion. SPARCC, Spondyloarthritis Research Consortium of Canada Enthesitis Index.
With regards to the baseline clinical characteristics, there was a mild correlation with BMI and increasing age and a higher overall MASEI score (rs = 0.23, p = 0.041 and rs = 0.31, p = 0.006). Active entheseal scoring (MASEImActive) had a mild correlation only with a baseline higher HAQ score (rs = 0.22, p = 0.049) and baseline pain VAS score (rs = 0.24, p = 0.031) but overall there were no strong clinical correlations with active ultrasound enthesitis. In terms of correlation for changes in clinical scores and ultrasound scoring again, there were no strong relationships. There was a mild correlation between the change in MASEImActive and reduction in BASDAI and swollen joint count (rs = 0.25, p = 0.031 and rs 0.23, p = 0.046, respectively), but otherwise there were no other significant correlations.
Outcomes for those with fibromyalgia
Of the 80 patients at baseline, 15 patients were also diagnosed by a consultant rheumatologist with comorbid fibromyalgia (FMS). In those with fibromyalgia, there was a statistically higher mean tender joint count, pain VAS, global VAS, HAQ and DAPSA score (p < 0.05). There was however no statistically significant difference in swollen joint count, CRP value, DAS28 or clinical entheseal scores. There was higher mean entheseal ultrasound scoring for those without FMS compared with those with FMS. For the MASEI, MASEIActive, MASEImActive, the mean value for those with or without FMS was 20.4 versus 24.51 (p = 0.132), 8.73 versus 12.42 (p = 0.027) and 8.53 versus 11.63 (p = 0.050), respectively. There were 13 patients with FMS who were included in the final analysis but none reached MDA compared with 49.2% of those without FMS (p = 0.001) and there was greater change in pain VAS score for those without FMS (26.61 versus 8.38, p = 0.01). Otherwise between both groups there was no significant difference in degrees of change in CRP values, composite disease scores, clinical outcomes and ultrasound entheseal scores.
Conclusions
Few studies have assessed enthesitis as a primary outcome and its response to treatment. This is to our knowledge the first paper that has assessed a change in ultrasound-confirmed enthesitis in PsA between different classes of biologic therapies. 17 Ultimately, we have seen an overall decrease in ultrasound confirmed entheseal disease for both classes of biologic therapy. In terms of comparing ultrasound response for the treatments administered, there was a larger reduction in the TNFI group but for the overall MASEI score this did not reach significance. The study did suggest that there was a significant mean difference in improvement for TNFI versus IL-17i in terms of active ultrasound entheseal disease but this was a secondary outcome. We have shown in terms of clinical outcomes that secukinumab has greater improvement on skin outcomes as per other trial data. The MASEI score when modified, to only include active entheseal elementary lesions, was an effective tool to monitor treatment response. There was some correlation with the SPARCC score but there was not a significant correlation with the LEI score which is validated in PsA. This may be due to the fact that the MASEI score covers similar sites to the SPARCC score and the LEI and MASEI only overlap assessing the Achilles tendon. As part of this study, we also included the common extensor origin at the lateral epicondyle (LE) which is part of both the SPARCC and LEI scores and the results are included in the Supplemental Material. Essentially, the addition of LE to the MASEI score did not demonstrate a significant correlation with LEI either. Other studies have demonstrated a weak correlation between the MASEI and the clinical LEI scores 18 but interestingly that research group when only assessing the clinical sites of the MASEI did not see a correlation with ultrasound MASEI scores. Other studies have suggested a disparity in clinical and overall ultrasound entheseal changes. 19 It makes sense however to attempt to compare active entheseal changes on ultrasound assessment with clinical scores to ascertain which clinical indices are most accurate. This was a focus of a well-designed study by Sapsford et al. 20 and they demonstrated a higher correlation between an ultrasound enthesitis activity scores and the SPARCC (r = 0.44) score as opposed to the LEI score (r = 0.36). We also do know that ultrasound gives us more information about what is going on sub-clinically and residual active ultrasound changes are associated with poorer outcomes 21 so in order to fully assess enthesitis imaging is important.
An increasing BMI and age corresponded to a higher overall MASEI score but this was not the case if only assessing the active entheseal disease, highlighting the possible contribution of mechanical damage and a potential pitfall in ultrasound entheseal assessment. We also demonstrated that those with comorbid FMS can have higher patient-reported outcome scores despite objective ultrasound assessment demonstrating lower active entheseal disease. We have also demonstrated that achieving disease remission is complicated by comorbid FMS with no patients reaching MDA in this cohort.
Currently, we have no effective biomarkers to predict response to the various musculoskeletal manifestations of PsA. This is a heterogeneous condition and if we can better define disease activity in each of the core domains with the aid of imaging we may be able to provide stratified decision-making. Further randomized controlled trials with similar objectives maybe able to confirm the findings from this article and demonstrate the feasibility of ultrasound confirmed enthesitis as a tool to stratify patients.
We recognize the limitations of this study in that it was a single site open-label study. Clinical assessment was carried out by the same examiner who had completed the ultrasound assessment reflecting a real-life clinical scenario. Furthermore, although both treatments were broadly matched for demographics more patients were on a TNFi and we did not reach a target of 50 patients on secukinumab for which the study was originally powered for.
Supplemental Material
sj-docx-1-tab-10.1177_1759720X231179524 – Supplemental material for Effects of TNF-α inhibition versus secukinumab on active ultrasound-confirmed enthesitis in psoriatic arthritis
Supplemental material, sj-docx-1-tab-10.1177_1759720X231179524 for Effects of TNF-α inhibition versus secukinumab on active ultrasound-confirmed enthesitis in psoriatic arthritis by Ashley Elliott, Gary Wright, Adrian Pendleton and Madeleine Rooney in Therapeutic Advances in Musculoskeletal Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
