Abstract
Background:
Late-onset psoriatic arthritis (LO-PsA) has been underexplored despite its growing prevalence in aging populations. Understanding its distinct clinical presentation and treatment patterns is essential to optimize care in this subgroup.
Objectives:
To describe the demographic, clinical, and therapeutic features of patients with LO-PsA compared to early onset PsA (EO-PsA) using data from the Argentine RECCAPSO registry.
Design:
Ambispective, multicenter analysis with a cross-sectional evaluation.
Methods:
Patients with PsA were categorized into EO-PsA (age of onset ⩽60 years) and LO-PsA (>60 years). Demographics, clinical characteristics, disease activity, and treatment variables were compared between groups using appropriate statistical tests. A multivariate logistic regression model was constructed to identify factors independently associated with LO-PsA.
Results:
A total of 271 PsA patients were included (EO-PsA: n = 184; LO-PsA: n = 87). LO-PsA patients had higher frequencies of hypertension (50% vs 21.4%, p < 0.001), diabetes (22.1% vs 7.9%, p = 0.007), and oligoarticular presentation (57.4% vs 40.5%, p = 0.03), and were less likely to receive b/tsDMARDs (42.6% vs 58.7%, p = 0.02). In multivariate analysis, hypertension (OR: 4.69, 95% CI: 1.83–12.03) and diabetes (OR: 14.83, 95% CI: 2.36–93.05) were independently associated with LO-PsA.
Conclusion:
LO-PsA presents a distinct clinical profile characterized by greater comorbidity burden and lower exposure to advanced therapies. These findings highlight the need for tailored management strategies in older adults with PsA.
Introduction
Psoriatic arthritis (PsA) is a chronic inflammatory disease that affects the joints and skin, belonging to the group of spondyloarthritis. 1 It is characterized by clinical heterogeneity across various domains, including peripheral arthritis, enthesitis, dactylitis, axial involvement, skin manifestations, uveitis, and inflammatory bowel disease.2,3 PsA significantly impacts patients’ quality of life due to pain, joint stiffness, and functional limitations. Its prevalence in the general population ranges between 0.02% and 0.42%, affecting approximately 10%–30% of patients with psoriasis.4,5
Late-onset PsA (LO-PsA) is traditionally defined as PsA that begins after the age of 60. However, different cohorts have utilized variable criteria to classify late-onset disease, as more than 50 years and 65 years. 6 These discrepancies highlight the lack of consensus on a universal cut-off point, potentially influencing the comparability of results and the global understanding of this phenotype. Moreover, these variations may reflect differences in population contexts and clinical characteristics across the studied cohorts.
This phenotype presents distinct clinical and prognostic features compared to early onset PsA, including greater structural damage, worse functionality, and a higher frequency of associated comorbidities. In addition, patients with LO-PsA face unique management challenges due to polypharmacy and therapeutic limitations in older individuals. Nonetheless, information regarding the prevalence and characteristics of this PsA subtype remains limited, particularly in regional contexts such as Latin America.7,8 LO-PsA poses specific clinical challenges due to its frequent association with comorbidities, polypharmacy, and age-related pharmacological limitations. These factors influence treatment strategies and may lead to therapeutic inertia or undertreatment, despite evidence of functional decline and joint damage in this subgroup.6,9,10 Understanding this phenotype is essential for optimizing care in older adults, a growing segment of the PsA population.
RECCAPSO (Red de Clinicas Colaborativas Argentinas en Artritis Psoriasica), the “Collaborative Network of Argentine Psoriatic Arthritis Clinics,” is a multicenter registry collecting clinical and demographic data from PsA patients treated at specialized centers across Argentina. This registry serves as a platform for studying PsA in the region, providing essential data for understanding phenotypic variations and management patterns in this population. The objective of this study is to describe the frequency of LO-PsA within the RECCAPSO cohort and analyze the differential characteristics between patients with disease onset before and after the age of 60.
Methods
This study is an ambispective, multicenter analysis with a cross-sectional evaluation. Data were obtained from the RECCAPSO registry, which integrates information from four university centers in Argentina through a RedCap-managed database. This platform captures demographic variables, family and personal history, and clinical and treatment characteristics of PsA. Patients included in this study had a confirmed diagnosis of PsA, fulfilled CASPAR classification criteria, 11 had at least one recorded consultation within the last 12 months, and were aged ⩾18 years at the time of consultation. Patients were classified as having early onset PsA (EO-PsA, age of onset ⩽60 years) or LO-PsA (age of onset > 60 years). The definition of age categories was agreed upon by the registry’s principal investigators based on existing literature and social characteristics, such as the average retirement age in Argentina. Patients with other rheumatic diseases (e.g., rheumatoid arthritis) and those with incomplete records were excluded. Both retrospective and prospective data were collected using a unified electronic case report form based on the RECCAPSO protocol. To ensure data consistency, only patients with complete core clinical variables and known age at PsA onset were included. Variables with missing data were excluded from the respective analyses; no data imputation was performed.
Demographic variables included age, sex, body mass index (BMI), and family history of psoriasis or PsA. Clinical characteristics included disease duration, peripheral and axial involvement, enthesitis, dactylitis, and nail involvement. Comorbidities such as cardiovascular, metabolic, or other conditions were also recorded. Laboratory parameters included inflammatory markers such as CRP and ESR. Data were collected on treatment with conventional, biologics, or target synthetic disease-modifying antirheumatic drugs (cDMARDs, bDMARDs, tsDMARDs). This study adheres to the STROBE (Strengthening the Reporting of Observational Studies in Epidemiology) guidelines for observational research reporting (Supplemental Material). 12
Statistical analysis: Descriptive statistics were used to summarize demographic and clinical characteristics. Categorical variables were expressed as numbers and percentages, while continuous variables were reported as mean ± standard deviation or median and range, depending on the distribution. Comparisons between EO-PsA and LO-PsA groups were performed using Student’s t-test or Mann-Whitney U test for continuous variables and Chi-square or Fisher’s exact test for categorical variables. A significance level of 5% (p < 0.05) was established. A logistic regression analysis was performed to identify independent associations with LO-PsA. Variables with p < 0.05 in univariate analyses were entered into a multivariate logistic regression using backward stepwise selection. Multicollinearity was assessed using VIF values, and model fit was tested using the Hosmer–Lemeshow test. No corrections for multiple testing were applied, given the exploratory nature of univariate analyses.
Results
A total of 270 patients were included in the study, with their characteristics detailed in Table 1. Among them, 87 patients (32.1%; 95% CI: 26.4%–37.9%) were LO-PsA, while 184 patients (67.9%; 95% CI: 62.1%–73.6%) were classified as EO-PsA. The mean age for the EO-PsA group was 52.1 years (IQR: 26–82 years; SD: 11.3), compared to 73.9 years (IQR: 61–90 years; SD: 5.82) for the LO-PsA group. Disease duration was significantly longer in the EO-PsA group, with a mean of 9.25 years (IQR: 0–50 years), compared to 4.52 years (IQR: 0–21 years) in the LO-PsA group (p < 0.001). Demographically, 52.8% of LO-PsA patients were female, and 81% were white, with 97% having health insurance, none showing significant differences from EO-PsA (p: 0.2, 0.5, 0.8, respectively).
Baseline characteristics of the cohort.
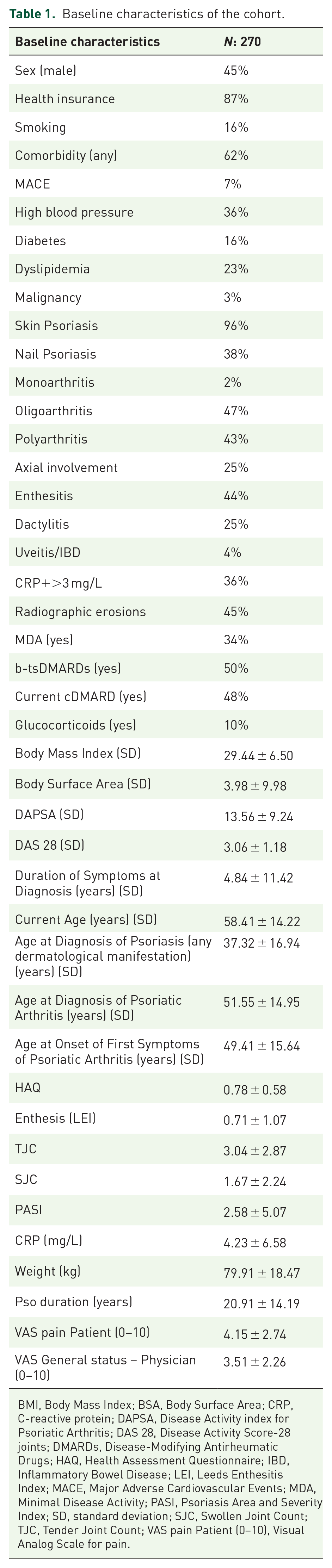
BMI, Body Mass Index; BSA, Body Surface Area; CRP, C-reactive protein; DAPSA, Disease Activity index for Psoriatic Arthritis; DAS 28, Disease Activity Score-28 joints; DMARDs, Disease-Modifying Antirheumatic Drugs; HAQ, Health Assessment Questionnaire; IBD, Inflammatory Bowel Disease; LEI, Leeds Enthesitis Index; MACE, Major Adverse Cardiovascular Events; MDA, Minimal Disease Activity; PASI, Psoriasis Area and Severity Index; SD, standard deviation; SJC, Swollen Joint Count; TJC, Tender Joint Count; VAS pain Patient (0–10), Visual Analog Scale for pain.
Comorbidities, including hypertension (50% vs 21.4%, OR: 4.69, 95% CI: 1.83–12.03, p = 0.001), obesity (29.4% vs 17.5%, OR: 1.92, 95% CI: 1.05–3.49, p = 0.04), and diabetes mellitus (22.1% vs 7.9%, OR: 14.83, 95% CI: 2.36–93.05, p < 0.001), were significantly more prevalent in the LO-PsA group.
Regarding disease activity, elevated acute phase reactants (CRP) were more commonly detected in LO-PsA patients compared to EO-PsA. EO-PsA patients exhibited higher PASI scores (3.7 ± 7.2 vs 1.9 ± 2.8, p = 0.07). BSA scores were also higher in EO-PsA patients (5.3 ± 12.2 vs 1.4 ± 2.9, p = 0.003). The prevalence of plaque psoriasis was similar between groups, with no cases of guttate or inverse psoriasis observed in the LO-PsA group. Nail disease was more prevalent in LO-PsA patients, affecting 56% versus 44.4% (OR: 1.57, 95% CI: 0.94–2.61, p = 0.07). In terms of articular involvement, an oligoarticular pattern was more frequently observed in LO-PsA patients (57.4% vs 40.5%, OR: 1.92, 95% CI: 1.14–3.21, p = 0.003), whereas polyarthritis was less frequent (33.8% vs 54%, OR: 0.43, 95% CI: 0.25–0.73, p = 0.01). In the LO-PsA group, 28 patients (39%) were in MDA, with no significant difference compared to the EO-PsA group (OR: 2.43, 95% CI: 1.38–4.27, p = 0.01).
Treatment patterns differed between groups. Advanced therapy medications were less frequently used in LO-PsA patients (43% vs 58%, p = 0.02). The most commonly used biologic DMARD (bDMARD) in the LO-PsA group was IL-17 inhibitors (20/37, 54%), whereas anti-TNF agents were more frequently used in the EO-PsA group (57/105, 54%) (p = 0.01). There was no significant difference in the use of non-steroidal anti-inflammatory drugs (NSAIDs), hydroxychloroquine (HCQ), corticosteroids (CS), methotrexate (MTX), or sulfasalazine (SSZ). However, COX-2 inhibitors were used less frequently in the LO-PsA group (7% vs 20%, p = 0.005).
Table 2 shows the most relevant differential characteristics between EO-PsA and LO-PsA, including demographic data, clinical features, and treatment patterns. The statistically significant variables were entered into a logistic regression model with LO-PsA as the dependent variable, and the results were as follows: Hypertension (OR: 4.69, 95% CI: 1.83–12.03, p = 0.001) and diabetes (OR: 14.83, 95% CI: 2.36–93.05, p = 0.004) were strongly and independently associated with LO-PsA (Table 3).
Differential characteristics between LO-PsA and EO-PsA: univariate analysis.

BSA, Body Surface Area; CRP, C-reactive protein; DAPSA, Disease Activity index for Psoriatic Arthritis; HAQ, Health Assessment Questionnaire; PASI, Psoriasis Area and Severity Index.
Multivariate logistic regression analysis for late-onset psoriatic arthritis (LO-PsA).
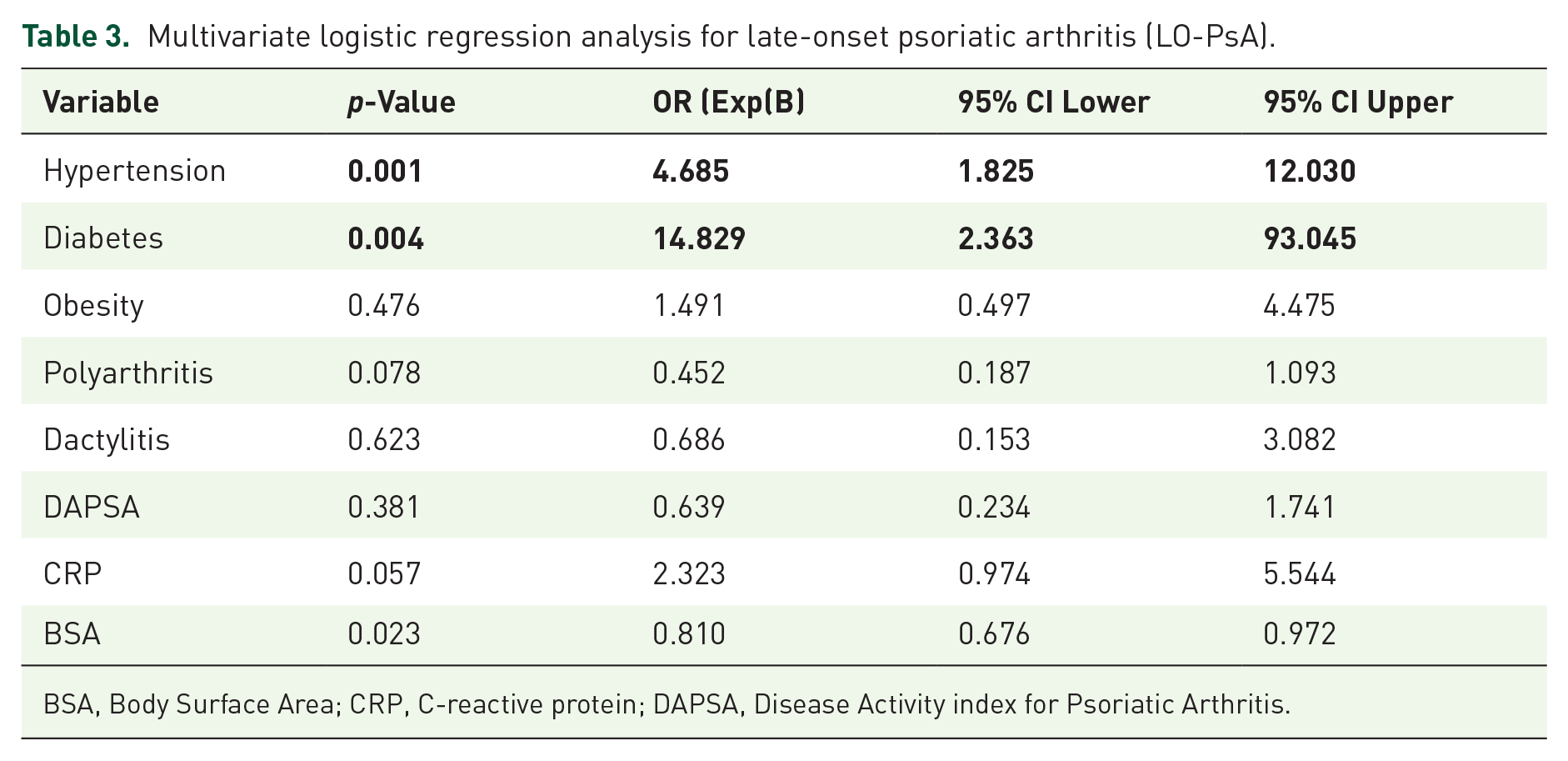
BSA, Body Surface Area; CRP, C-reactive protein; DAPSA, Disease Activity index for Psoriatic Arthritis.
Discussion
Our study identified that LO-PsA accounted for 32.1% of the cohort, highlighting its considerable prevalence among PsA patients. This group showed distinct clinical and demographic characteristics compared to EO-PsA. This has also been observed in patients with late-onset axial spondyloarthritis. 13 LO-PsA patients presented a higher prevalence of comorbidities, such as cardiovascular and metabolic diseases, and a predominance of oligoarticular patterns. In contrast, EO-PsA patients were characterized by more extensive skin involvement, higher rates of polyarthritis, and a greater overall disease burden. These findings underscore the heterogeneity of PsA presentations and emphasize the influence of age at onset on clinical outcomes. 14
Our findings align with those of Puche-Larrubia et al., who also reported that LO-PsA patients tend to present with less axial involvement and worse function. 9 We further highlight that undertreatment in older patients may stem from safety concerns, polypharmacy, or physician reluctance to escalate therapy, despite evidence of active disease. This may contribute to worse outcomes and requires tailored decision-making in older populations.
The concept of “inflammaging”—chronic low-grade inflammation associated with aging—may also play a role in amplifying immune dysregulation and influencing disease phenotype in LO-PsA.15,16 While our results are consistent with prior studies such as those by Yamamoto and Caso, others like Polachek et al. reported more modest differences between groups, underscoring the potential impact of regional healthcare access and cohort differences.6,10,17 These comparisons reinforce the need for local data to guide personalized approaches in Latin America.
Likewise, the study by Queiro et al. reinforces these observations by identifying more limited clinical patterns and a greater burden of comorbidities in LO-PsA patients, consistent with our findings. 18 Furthermore, the review by Caso et al. highlighted that LO-PsA patients experience a more aggressive evolution of peripheral disease and a significant increase in joint damage over time. 6 They also noted differences in therapeutic response, emphasizing the need for more personalized approaches. In addition, our results coincide with those of Polachek et al., who described a higher burden of metabolic comorbidities in older patients with PsA. 17
Therapeutic strategies also varied between groups. In our study, LO-PsA patients were less likely to receive advanced therapies, possibly due to polypharmacy and the comorbidities commonly found in this group. IL-17 inhibitors were more frequently used in LO-PsA, reflecting their efficacy in controlling peripheral disease, while anti-TNF agents predominated in EO-PsA. 19 This is consistent with trends observed in younger cohorts. Finally, we believe that the results of the “ORAL Surveillance” study may influence the reduced use of JAK inhibitors in older patients.20,21
One of the challenges in interpreting the clinical phenotype of LO-PsA is the potential confounding effect of coexisting osteoarthritis (OA), which becomes more prevalent with aging. Features such as oligoarticular involvement and structural changes may overlap between OA and PsA, potentially influencing diagnostic and therapeutic decisions. Although our study did not include specific data on OA prevalence, this limitation should be considered when interpreting our findings. Future studies should incorporate imaging criteria to better delineate the contribution of OA in late-onset cases. 22
In the multivariate model, hypertension and diabetes remained independently associated with LO-PsA. In contrast, obesity, which showed a significant association in univariate analysis, lost statistical significance, suggesting a possible confounding effect. In addition, the wide confidence interval observed for diabetes reflects limited precision and likely stems from the small number of diabetic patients in the LO-PsA subgroup. These findings should be interpreted with caution and warrant confirmation in larger cohorts.
The strengths of this study include its large, well-characterized cohort from the RECCAPSO registry, with data obtained in Argentina, a region with limited information on PsA.23,24 In addition, the multicenter design enhances the generalizability of our findings. However, the cross-sectional nature of the study limits causal inferences. Moreover, the absence of consensus on the age cut-off for defining LO-PsA introduces variability in comparisons with other studies. Although articular damage is a key aspect of disease burden in PsA, standardized imaging data were not consistently available in our cohort to allow a robust comparison between early- and late-onset groups. While radiographic erosions were reported in 45% of the overall sample, the lack of uniform imaging assessment across centers precluded further subgroup analysis. This represents a limitation of our study and highlights the need for future longitudinal research with standardized radiographic protocols to better characterize structural progression in LO-PsA. In addition, data on socioeconomic status and polypharmacy were not captured in the registry, limiting the assessment of potential confounders in treatment decisions for older patients. The wide confidence interval observed for diabetes likely reflects limited sample size in this subgroup, and results should be interpreted with caution. In addition, no formal correction for multiple comparisons was applied, as univariate analyses were exploratory.
Conclusion
Our findings emphasize the need for personalized approaches to PsA management, particularly in older patients where comorbidities and age-related immune changes play a critical role.
LO-PsA may represent a distinct clinical profile characterized by greater comorbidity burden and lower exposure to advanced therapies. These findings suggest the need for tailored management strategies in older adults with PsA, which should be further explored in prospective studies.
Supplemental Material
sj-docx-1-tab-10.1177_1759720X251356206 – Supplemental material for Distinctive clinical patterns and management trends in late-onset psoriatic arthritis: data from Argentina’s RECCAPSO registry
Supplemental material, sj-docx-1-tab-10.1177_1759720X251356206 for Distinctive clinical patterns and management trends in late-onset psoriatic arthritis: data from Argentina’s RECCAPSO registry by Gustavo A. Medina, Maximiliano Fenucci, Sebastián Magri, Marcelo Abdala, Tamara Arias, Brian Abdala, Leila Abbas, Vinod Chandran and Rodrigo García-Salinas in Therapeutic Advances in Musculoskeletal Disease
Footnotes
Acknowledgements
None.
Declarations
Supplemental material
Supplemental material for this article is available online.
Use of artificial intelligence
Use of Artificial Intelligence: ChatGPT (OpenAI, 2024 version) was used to assist with grammar correction and language editing. All authors reviewed and approved the AI-assisted revisions to ensure their accuracy and consistency with the scientific content of the manuscript.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
