Abstract
Despite significant advances in the treatment of psoriatic arthritis (PsA) in the last two decades, remission remains elusive and there is no cure. Evidence from rheumatoid arthritis (RA) confirming enhanced response and outcome from earlier treatment intervention suggests the plausibility of the window of opportunity in the pathogenesis of RA. Yet, data are lacking in PsA. Although treatment response may be enhanced in shorter disease duration, it is unknown how this early intervention may impact long-term outcomes. Furthermore, it remains to be demonstrated whether there is a best treatment strategy and time of intervention. Crucially, the main hurdle when aiming for early treatment intervention is the ability to achieve a timely diagnosis that highlights the need to focus research efforts on characterizing the very early disease stages including the transition to PsA in the at-risk psoriasis population.
Introduction
Psoriasis (PsO) is a common inflammatory condition that most notably affects the skin but may present with other organ involvement, with an estimated world prevalence that lies between 0.09% and 11.4%. 1 Around 20% of patients suffering from PsO will develop psoriatic arthritis (PsA), with a reported PsA prevalence among PsO patients as high as 34.7%. 2
The clinical presentation of PsA is heterogeneous and includes multiple musculoskeletal features, such as peripheral and axial arthritis, enthesitis, nail dystrophy and dactylitis. PsA has been associated with reduced quality of life and increased disability and mortality. 3
The complex nature of PsA, its multiple disease phenotypes and the overlapping clinical features with other diseases such as osteoarthritis, make it challenging to diagnose especially early in the disease course. As a result, a diagnostic delay is common, 4 taking typically double that of rheumatoid arthritis (RA). This is of relevance since even a 6-month delay is associated with limitations of physical functioning and permanent disability despite the wide availability of efficacious drug therapies.5,6
Similar to other chronic conditions including RA, it is expected that a better understanding of the early phases of PsA may lead to an earlier, timely diagnosis opening up a window of opportunity to implement a tailored treatment intervention, that might benefit both joints and skin, minimizing irreversible joint damage or modifying the severity of the disease.
Yet, the lack of useful predictors of progression or biomarkers to allow for the early identification of PsA, especially in the PsO population at risk of developing PsA, is a significant unmet need and a research priority in this disease, as highlighted by researchers and patients alike. 7 This narrative review aims to summarize the latest evidence on the treatment of PsA, discussing the implications of early intervention and the need to address the lack of evidence and standardized definitions of what is early PsA.
Current treatment options in PsA
The development of advanced therapies such as the biological Disease Modifying Anti-Rheumatic Drugs (bDMARDs) in the early 2000s revolutionized the treatment of PsA. 8 The first class developed were the tumour necrosis factor inhibitors (TNFi), with the IMPACT trial in 2005 showing the efficacy of Infliximab, a monoclonal antibody, in patients with previous inadequate response to conventional Disease Modifying Anti-Rheumatic Drugs (cDMARDs). 9 Other TNFi followed including a TNF receptor inhibitor (etanercept), 10 two humanized antibodies (adalimumab and golimumab)11,12 and a pergolated antibody (certolizumab). 13 Importantly, all TNFis demonstrated good efficacy and acceptable overall safety profile including risk of infections such as tuberculosis.
The next generation of bDMARDs was aimed at specific targets within the interleukin (IL)-17/IL-23 pathway. Ustekinumab is a monoclonal antibody that binds to the p40 subunit that is found both in IL-12 and -23 leading ultimately to a reduction of IL-23, IL-17 and TNF levels by downregulating the T-helper (Th)17 response. Ustekinumab showed superior efficacy to TNFi for skin PsO and moderate response in the joints of TNF naïve and TNFi-experienced patients,14,15 including axial symptoms, as shown on a post hoc analysis of the PSUMMIT trial data, 16 despite negative data stemming from phase II trials in axial Spondyloarthritis (axSpA). 17 IL-17 blockade showed impressive efficacy in both skin and joint symptoms with two blockers of the IL-17A isotope (Secukinumab, Ixekizumab),18,19 including axial disease 20 and more recently a dual blocker of IL-17A and F isotopes (Bimekizumab). 21 Two head-to-head trials have been performed to date with Secukinumab and Ixekizumab utilizing Adalimumab as the comparator arm.22,23 Although neither was powered to demonstrate superiority, both studies showed numerically higher response rates for IL-17A blockade versus adalimumab in controlling skin outcomes with comparable efficacy in joint outcomes.
The IL-23 inhibitors Guselkumab, 24 Risankizumab 25 and Tildrakizumab 26 all suppress the IL-23/IL-17 axis by inhibiting the p19 subunit of IL-23. Guselkumab demonstrated good efficacy on skin, joints, enthesis and dactylitis 24 and is currently undergoing trials in axial PsA. 27
The latest generation are the small molecules targeted synthetic (ts)DMARDs including the phosphodiesterase 4 (PDE4) and the Janus kinase (JAK) inhibitors. The PDE4 inhibitor Apremilast downregulates pro-inflammatory cytokines TNF and IL-23. Although the efficacy of Apremilast on the skin, joint, enthesis and dactylitis is modest, it is a well-tolerated oral agent with a remarkable safety profile.28–30
The JAK inhibitors target one or more of four intracellular signalling molecules, JAK1, JAK2, JAK3 and TYK2, which downregulates the JAK-STAT signalling pathway by inhibiting phosphorylation of target transcription factors that modulate immune cell function, leading to reduced secretion of pro-inflammatory cytokines IL-23, IL-6 and IL-15. Tofacitinib selectively inhibits JAK1 and JAK3 and was shown to have similar efficacy when compared to TNFi, but there are concerns regarding its safety profile which include serious infections, venous thromboembolism and lymphoma.31–33 Other JAK inhibitors entering the market are the JAK1 selective upadacitinib 34 and filgotinib 35 and the TYK2 inhibitor deucravacitinib. 36 Whether or not selectivity for different JAK molecules alters efficacy, toxicity and tolerability of the drugs in PsA and other rheumatic diseases remains to be seen. 37
Clearly, pharmacological options for PsA have come a long way, carrying an abundance of potent targeted agents, with a variety of modes of action and acceptable safety profiles.
However, most studies to date have focused on polyarticular disease of several years duration, often with established structural joint damage on conventional radiographic imaging.8,10,11 There are several reasons for this. Firstly, historical delays in diagnosis mean patients with shorter disease duration were rarely captured in clinical trials, which often included populations who were previously exposed and had inadequate response to conventional therapies, hence needing other treatment options. Moreover, until very recently clinical guidelines for the use of bDMARDs in PsA were modelled on a step-up approach whereby biologics or tsDMARDs could only be offered after previous exposure and failure to achieve optimal disease control or tolerability to at least two csDMARDs.38,39 Yet, real-world experience suggests that PsA presentation has changed over time with individuals being diagnosed at an earlier disease stage, often presenting with lower joint counts. 40 This has been reflected in clinical recommendations suggesting consideration of b/tsDMARD therapy after failure of at least one csDMARD in oligoarthritis or insufficient response to non-steroidal anti-inflammatory drugs (NSAIDs) or local glucocorticoid injections in enthesitis. 41 Furthermore, unlike the earlier clinical trials of TNFis, clinical trials of newer bDMARDs are more diverse in sex and race and generally include patients with shorter disease duration, lower swollen and tender joint counts (SJC and TJC) and C-reactive protein (CRP) levels at baseline. This is due, in part, to increasing awareness of the condition and earlier diagnosis, as well as advances in the availability and sensitivity of imaging such as ultrasound and Magnetic Resonance Imaging (MRI) that enable earlier detection of subclinical disease activity. While this is indeed a welcome advance in the care and management of PsA, it is an important confounder that must be considered when trying to directly compare efficacy from clinical trial data of TNFis versus newer b/tsDMARDs.
When is the right time to start b/tsDMARDs in early PsA
Growing awareness among primary and secondary care physicians, the development of dedicated early inflammatory arthritis 42 and cross-specialty rheumatology/dermatology clinics 43 are some of the reasons contributing to earlier recognition and diagnosis of PsA. This, alongside the proven success of early and more intense management of RA,44,45 has spurred several clinical trials and observational studies in PsA over the past 5 years (Table 1) aiming to address the question as to whether early bDMARD use could indeed improve patient outcomes in this disease. This topic is now a priority for many clinical working groups and healthcare commissioners.7,46
Summary of interventional clinical trials of PsA of short disease duration published up to 2023.
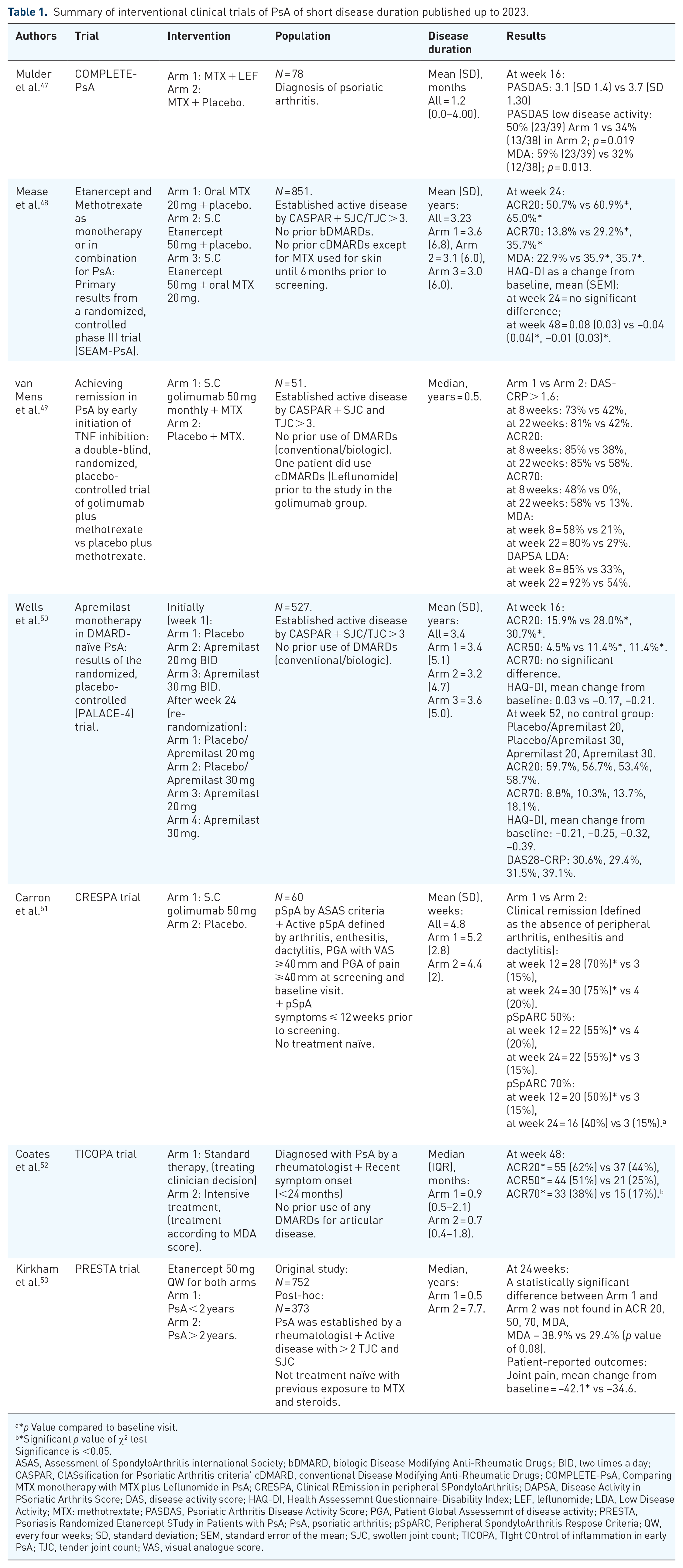
*p Value compared to baseline visit.
*Significant p value of χ2 testSignificance is <0.05.
Significance is <0.05.
ASAS, Assessment of SpondyloArthritis international Society; bDMARD, biologic Disease Modifying Anti-Rheumatic Drugs; BID, two times a day; CASPAR, ClASsification for Psoriatic Arthritis criteria’ cDMARD, conventional Disease Modifying Anti-Rheumatic Drugs; COMPLETE-PsA, Comparing MTX monotherapy with MTX plus Leflunomide in PsA; CRESPA, Clinical REmission in peripheral SPondyloArthritis; DAPSA, Disease Activity in PSoriatic Arthrits Score; DAS, disease activity score; HAQ-DI, Health Assessemnt Questionnaire-Disability Index; LEF, leflunomide; LDA, Low Disease Activity; MTX: methotrextate; PASDAS, Psoriatic Arthritis Disease Activity Score; PGA, Patient Global Assessemnt of disease activity; PRESTA, Psoriasis Randomized Etanercept STudy in Patients with PsA; PsA, psoriatic arthritis; pSpARC, Peripheral SpondyloArthritis Respose Criteria; QW, every four weeks; SD, standard deviation; SEM, standard error of the mean; SJC, swollen joint count; TICOPA, TIght COntrol of inflammation in early PsA; TJC, tender joint count; VAS, visual analogue score.
A post hoc analysis of the Psoriasis Randomized Etanercept STudy in Patients with PsA trial was the first designed to investigate the effect of disease duration (⩽2 vs >2 years) on the outcome of treatment with etanercept 50 mg once weekly in patients with PsA and moderate to severe skin PsO. Significant improvements were seen in both groups in arthritis scores and patient-reported outcome (PRO) measures with a trend to better outcomes in the shorter disease duration group, although the study might be missing the power to detect small changes. 53
The SEAM-PsA study, reported by Mease et al. in 2019, 48 compared methotrexate and etanercept monotherapy versus combination therapy with both agents in patients with a mean PsA duration of 3 years at trial entry (although there was substantial variation, with a standard deviation of 6 years). Although numerically, combination treatment and etanercept monotherapy performed better than methotrexate alone leading to higher ACR20, ACR70 and minimal disease activity (MDA) responses at week 24, these differences were not statistically significant. Furthermore, HAQ-DI was unchanged between groups suggesting the efficacy of methotrexate in this population, although it has to be noted that there was no placebo arm in the trial. In a contemporary study, van Mens et al. reported on the efficacy of the combination of golimumab + MTX with MTX monotherapy on a much smaller population (n = 51) with a short disease duration of half a year (median). 49 Here, the combo arm significantly outperformed the methotrexate monotherapy arm for all endpoints at week 8 and week 22 (ACR20, ACR70, MDA and DAPSA LDA). An earlier study by Carron et al. in 2017 also showed excellent responses in undifferentiated Spondyloarthritis including PsA, to golimumab monotherapy with 75% of patients achieving remission at week 24 compared to placebo (15%), with the main characteristic being the very short symptom duration of just 4.8 weeks (mean) for the overall study population. 51 However, both these studies had relatively short interventional periods (24 weeks), with subsequent follow-up reports showing that remission was not maintained with a step-down approach, converting golimumab to MTX in most patients in both studies.54,55
Interestingly, a study by Wells et al. 50 in 2018 in early PsA, PALACE-4 (median ~3 years duration) comparing different dosing regimens of Apremilast showed that 52-week response rates were similar between all the different regimens for ACR20/70, HAQ-DI and DA28-CRP.
What is the best treatment strategy in PsA?
An important consideration for clinicians treating PsA, beyond which drug(s) to use and when to treat, is what is the optimal treatment strategy to induce prompt and sustained remission. The TIght COntrol of inflammation in early PsA trials sought to address this by randomizing early PsA patients (median 0.6 months duration) to either standard care or targeting aggressively towards MDA. 52 Although the short-term, primary results of the study pointed towards a treat-to-target (T2T), intensive treatment regime as leading to superior outcomes at 6 months, 5-year follow-up data from the study cohort showed comparable outcomes between both treatment arms, with decreased MTX use in all groups, and similar numbers of patients on the T2T arm more likely to still be on bDMARD. 56 A recent strategy study reported the combination of Leflunomide plus methotrexate alone showing greater improvements in disease activity according to PASDAS scores at week 16, although the combination appeared to be less well tolerated than methotrexate monotherapy. 47
Overall, these studies all targeted a newly diagnosed, cs and b/tsDMARD naïve population. Taken together and notwithstanding the heterogeneity of study designs, varied inclusion criteria and disease duration cut-offs, together with differing modes of action of each drug, these data suggest that the enhanced efficacy seen in these studies may not be related to the individual drugs but to their use in early, treatment-naïve PsA populations. However, it should be noted that selection bias can occur in clinical trial populations leading to reduced applicability of findings to real-world clinical settings. Furthermore, the longevity of response and the possibility of whether or not early treatment might enable long-term remission with the withdrawal of bDMARD therapy remains uncertain, and the current European League Against Rheumatism (EULAR) and Group for Research and Assessement of Psoriasis and Psoriatic Arthritis (GRAPPA) recommendations advise caution with drug tapering/withdrawal even in patients who achieve early and sustained responses.41,57
Besides randomized clinical trials, there are few observational studies reporting on real-world outcomes for the use of advanced therapies in early PsA. One observational study published in 2023 is the APROACH study, 58 a non-interventional prospective 52-week cohort study exploring the effect of Apremilast in patients with early PsA (disease duration of 1.1 years (0.7–1.8) at enrolment). The primary outcome was PsA disease activity at week 24 as measured by cDAPSA. Authors report cDAPSA response rates of 59.7% at 24 weeks (primary endpoint) with response rates of 42.5% and 69.8% at 16 and 52 weeks, respectively. Improvements in secondary endpoints (reduction in PsO body surface area [BSA], resolution of enthesitis/dactylitis, resolution of nail PsA and improvement in EQ-5D-5L score) were also observed at 24 weeks and maintained/improved at 52 weeks. 58 It is difficult to compare this directly with PALACE-4 and the original Apremilast trials, however, due to differences in primary and secondary endpoints. However, it is encouraging that response for cDAPSA was maintained and even improved at 52 weeks compared with the 24-week primary endpoint, suggesting longevity of Apremilast in early PsA.
Overall, despite growing research in this area, much remains to be understood about when is the right time to start b/tsDMARDs, and particularly whether drug-free remission can be achieved in early PsA. Likewise, the long-term efficacy, tolerability and longevity of b/tsDMARD response when treatment is started in early disease is not known. The potential ramifications for this are far-reaching given that the average age at disease onset/diagnosis is lower for PsA/axSpA when compared to RA, for example, and so patients are facing longer periods of their lives on these medications than would be the case in the context of other rheumatological diseases. The question of if, how and when to reduce or even withdraw biological treatment in patients achieving sustained remission remains unknown. Furthermore, there may be no hard or fast rules and it is likely that the answer will vary depending on each patient’s preference as well as their individual underlying disease phenotype. Dactylitis, for example, is a poor prognostic factor for disease progression suggesting the need for earlier treatment intervention. 59 Other factors such as uveitis, inflammatory bowel disease, erosive or rapidly progressive radiographic disease or axial involvement may be deemed higher risk for biologic withdrawal or tapering. 60 This is of particular relevance since the evidence suggests that it can be difficult to regain response to a b/tsDMARD once treatment is interrupted, for example, for infection, pregnancy or surgery. Furthermore, real-world evidence shows that responses to second-/third-/fourth-line b/tsDMARD agents are consistently reduced compared with first line. 61 Thus, there is a significant need for biomarkers to both identify early PsA and assist with these key treatment decisions as we seek to personalize patient care. Recent advances in genomics and hybrid approaches combining clinical, PROs and genomics/bioinformatics data could enable these in the future; however, for now these technologies are only in use in the research setting. 62
How early is early
To date, a standardized definition of early PsA is lacking. Bio-pathological models of established PsA have evolved over the past 25 years and have indirectly led to the theorization of the stages preceding the clinical onset of this condition. In particular, the traditional view of PsA as a disease characterized by auto-immunity targeting synovial tissues underwent substantial reconsideration. This was triggered by advanced imaging data63,64 highlighting the occurrence of prominent inflammatory processes located in close proximity, yet outside the synovium, in PsA. These observations provided seminal evidence that the enthesis is likely to be the earliest site of inflammation being primed in the musculoskeletal system. This line of thought led to a theoretical model of PsA pathogenesis, focused on the entheses,65,66 later reinformed by experiments in animal models of SpA and human tissues,67,68 and ultimately leading to the concept of the synovio-entheseal complex. 69
Crucially, plenty of evidence – stemming from imaging-based research projects – documented the occurrence of enthesopathic and synovitic findings in people with PsO but with no clinical symptoms and signs of PsA. 70 However, no consensus exists on how to define – let alone diagnose – ‘pre-clinical’ PsA in clinical practice. This is a significant area of unmet need as the transition from ‘pre-clinical’ to (early) PsA would mark the genuine onset of the early clinical stage of PsA. There are however many challenges when researching this transition phase including the incomplete understanding of the bio-pathologic processes underpinning the development of PsA and the lack of specificity of available imaging methods when assessing the synovial joints, and surrounding enthesis and soft tissues in PsA, with findings of inflammatory features in synovial joints such as effusion, synovial hypertrophy and power-Doppler signal also reported in healthy individuals (up to 52%) in the absence of joint pain. 71 Furthermore, musculoskeletal symptoms occurring during the pre-clinical stages of PsA development are non-specific. 72 These limitations can clearly contribute to the diagnostic delay experienced by people who develop PsA. 4 The lack of longitudinal data exploring the transition from ‘pre-clinical’ PsA to early PsA is currently, the main obstacle to consensus on a definition of early PsA.
To facilitate research in this area, an EULAR task force has proposed that three distinct stages are relevant to the prevention of PsA: (1) people with PsO at higher risk of PsA; (2) subclinical PsA and (3) clinical PsA. 73 Risk factors include arthralgia as a short-term predictor and the severity of PsO, nail involvement, family history of PsA and obesity considered to be long-term predictors for the development of PsA. 73 An operational definition of ‘sub-clinical’ has also been proposed for use in clinical trials aimed at exploring the transition from PsO to PsA. EULAR defines ‘sub-clinical PsA’ as arthralgia (pain affecting the joints) and/or imaging evidence of synovial/entheseal inflammation, but without clinically detected synovitis, in patients affected by PsO. However, these proposals have to be considered only as a first step, mostly intended to facilitate future research, with more data needed to shape the definitions of both ‘sub-clinical’ or ‘early’ PsA. Reflecting the relevant interest in this field of research, other organizations, like the Psoriasis and Psoriatic Arthritis Clinics Multicenter Advancement Network, proposed their consensus terminology for preclinical phases of PsA for use in research studies. 74
Interestingly, register data point towards a ‘window of opportunity’ of 2 years between onset of musculoskeletal inflammatory symptoms (typically, articular early morning stiffness) and clinical diagnosis of PsA. 75 However, current evidence shows that a quarter of patients already have radiographic erosions 76 at the time of diagnosis, even when presenting with less than 2 years of symptom duration, suggesting that disease is already well established with a limited chance of drug-free remission at diagnosis, prompting the question of ‘how early is early’ when aiming to intervene to induce full remission of arthritis or even prevention. Yet, exciting insights are coming from observational studies in PsO with indirect evidence suggesting the potential for the ‘interception’ of PsA before its clinical development. Indeed, two recent retrospective studies77,78 reported a lower risk of PsA development in PsO patients treated with a bDMARD compared with those treated with narrow-band ultraviolet light B phototherapy or those treated with topicals or no treatment, with similar incidence rates found (i.e. 1.2 and 1.6 cases per 100 patients/year, respectively) in both cohorts. Furthermore, IL-23/IL-17 axis drug blockers have excellent efficacy on skin outcomes leading to clearance in up to 50% of cases. 79 Although overall responses in signs and symptoms of PsA are more modest, a rapid improvement of dactylitis, a marker of disease severity in PsA, has been reported, suggesting the potential for disease modification if used in early disease. 79 Furthermore, another retrospective study of patients with PsO and joint pain found no evidence of current or previous dactylitis in patients given biologic therapy for their skin PsO. 80 These observations suggest that biological therapy could potentially change or modulate the course of PsA, contributing to a milder or less severe outcome. 81
Conclusion
Response to treatment in PsA may be enhanced when initiated in the early stages of the disease course; however, data are needed to establish the optimal therapeutic strategies and to allow for treatment personalization. These will improve the understanding of the true impact of early treatment and intervention timings on the long-term disease outcome including the avoidance of structural progression and disability prevention. However, early intervention in PsA will only be achieved through the successful shortening of the time to diagnosis. This together with an increased understanding of the factors leading to symptom onset remains one of the main unmet needs to improve the lives of people with PsA.
Footnotes
Acknowledgements
The authors are supported by the National Institute for Health Research (NIHR) Leeds Biomedical Research Centre (LBRC). The views expressed are those of the authors and not necessarily those of the (UK) National Health Service (NHS), the NIHR or the (UK) Department of Health.
