Abstract
Spondyloarthritis is a group of chronic inflammatory diseases that includes axial spondyloarthritis (axSpA) and psoriatic arthritis (PsA). AxSpA primarily affects the axial skeleton, manifesting with hallmark features such as inflammatory back pain and a strong association with human leukocyte antigen-B27. On the other hand, axial involvement in PsA (axial PsA) poses unique challenges in the diagnosis, classification, and management. These challenges stem from a limited understanding of this condition and an absence of a specific definition for its diagnosis. Although shared genetic and environmental contributors are observed, the presence of differences suggests the possibility that axial PsA may, in fact, represent a distinct clinical entity rather than axSpA. The prevailing classification criteria, such as ClASsification criteria for Psoriatic ARthritis for PsA and the Assessment of SpondyloArthritis International Society criteria for axSpA, are insufficient in capturing the full scope of axial PsA. Moreover, treatment paradigms for axial PsA are primarily extrapolated from axSpA due to the lack of targeted trials in this specific population. Biologic disease-modifying anti-rheumatic drugs, encompassing tumor necrosis factor inhibitors, interleukin (IL)-17 inhibitors, and Janus kinase inhibitors, have demonstrated efficacy in axSpA and PsA. However, IL-23 inhibitors have not shown efficacy in axSpA, and currently, no results from randomized controlled trials in axial PsA are available. While axial PsA exhibits features that overlap with axSpA, emerging evidence underscores its distinct pathophysiology and clinical characteristics, highlighting the need for standardized definitions and tailored therapeutic approaches to optimize outcomes. Ongoing studies evaluating therapeutic efficacy and molecular characterization hold promises to enhance understanding and management of axial PsA, thus paving the way for personalized treatment strategies. This review aims to provide an overview of the similarities and differences between axial PsA and axSpA and seeks to disentangle the intersections between these two diseases.
Keywords
Introduction
Spondyloarthritis (SpA) is a group of chronic inflammatory diseases with shared clinical, genetic, and pathophysiological characteristics. The SpA spectrum includes axial spondyloarthritis (axSpA), peripheral SpA (perSpA), psoriatic arthritis (PsA), reactive arthritis (ReA), inflammatory bowel disease (IBD) associated SpA, and undifferentiated SpA. 1 These diseases typically present with peripheral musculoskeletal manifestations such as dactylitis, enthesitis, and arthritis, and can affect the axial skeleton, including the sacroiliac joints (SIJ) and spine. Additionally, SpA can be accompanied by extra-musculoskeletal manifestations (EMMs) like acute anterior uveitis, skin psoriasis (PsO), and IBD.
According to the Assessment of SpondyloArthritis International Society (ASAS), SpA can be categorized into axial SpA (axSpA) and perSpA. 2 AxSpA includes both non-radiographic (nr-axSpA) and radiographic axSpA (r-axSpA, also known as ankylosing spondylitis (AS)). 3 This distinction between nr-axSpA and r-axSpA is nowadays mainly for research and drug approval purposes, as disease severity and burden are similar between the two subgroups, except for function and mobility.4–6
PsA shares features with both perSpA, such as peripheral arthritis, enthesitis, and dactylitis, and axSpA, characterized by axial involvement. Axial involvement in PsA (axial PsA) is common, and despite its prevalence, our understanding of axial PsA still remains limited.7–10 Some studies suggest that axial PsA may be clinically similar to axSpA, indicating a shared pathophysiological mechanism, while recent research proposes that axial PsA might have a distinct pathogenesis.11–14
The increasing understanding of the similarities and differences between axSpA and axial psoriatic arthritis (axial PsA) has revealed that, besides their existing commonalities, there are significant pathophysiological discrepancies. These differences could influence the approaches taken in clinical management. Currently, the treatment paradigm for axial PsA, which includes recommendations and guidelines,15–17 is largely based on data extrapolated from studies and treatment guidelines for axSpA.18,19 This dependence is due to the absence of universally accepted definitions or classification criteria for axial PsA. For instance, interleukin (IL)-12/23 and IL-23 inhibitors, which are not recommended for managing axSpA, 20 are approved treatment options for (per)PsA. However, clear evidence supporting their efficacy in axial PsA remains limited, although some post hoc analyses suggest a potential benefit.21,22 These topics will be discussed in more detail later in the treatment section of this review.
There is an ongoing debate among SpA experts between “lumpers” and “splitters.” Since PsA was distinguished from rheumatoid arthritis and the concept of SpA was introduced as a group, there has been continuous discussion about whether conditions with similar pathogenesis should be grouped together or separated for research and clinical purposes. 23 Although PsA and axSpA are distinct entities within the SpA spectrum, they share overlapping features, particularly in terms of axial involvement. With the growing body of data highlighting the similarities and differences between axSpA and axial PsA, this review aims to compare and contrast the current understanding of these two conditions.
Search strategy
A literature search was conducted in PubMed up to July 15, 2024, using the following search terms: (“psoriatic arthritis” OR PsA) AND (“axial psoriatic arthritis” OR “axial PsA” OR “axPsA”) AND (“axial spondyloarthritis” OR “ankylosing spondylitis” OR “axSpA”). The publications were selected based on their relevance to the review’s key themes, including classification, pathogenesis, clinical features, imaging, and treatment of axial PsA and axSpA, as determined by the authors. In addition to this systematic search, other publications—including original studies, clinical trials, and narrative reviews—were included if they were deemed relevant by the authors.
Epidemiology and classification
AxSpA is a chronic inflammatory disease that primarily affects the axial skeleton and has a prevalence between 0.2% and 1.4% in the general population, depending on the geographical region. 24 PsA is also an inflammatory rheumatic disease and is less common in comparison to axSpA, affecting 0.04% and 0.58% of adults in the general population.25–27 It is a heterogenetic disease, strongly associated with PsO, occurring in up to 30% of PsO patients,28–31 and therefore, sometimes is subsumed as psoriatic syndrome or psoriatic disease. 32 Although the concept of SpA encompasses both axSpA and PsA, some significant differences exist between these two conditions. PsA is particularly associated with skin PsO, psoriatic nail involvement, affection of small joints, and dactylitis, which are less common in axSpA. Furthermore, PsA patients are generally older and have a lower prevalence of the human leukocyte antigen-B27 (HLA-B27). PsA is traditionally associated with peripheral musculoskeletal manifestations, but a significant proportion of these patients have axial involvement, with a prevalence ranging from 4% to 70% described in the literature in different PsA cohorts.7,11,12,33–35 The variation in the reported prevalence of axial PsA primarily stems from the lack of a universally accepted definition of axial PsA. This discrepancy is often attributed to differences in diagnostic imaging techniques, such as radiographs or magnetic resonance imaging (MRI), and disease duration. The prevalence of axial PsA is significantly higher in PsA patients with longstanding disease.36,37 Of note, axial involvement without peripheral musculoskeletal manifestations is rare, described in less than 5% of PsA patients.12,36 In addition, a key clinical difference between axial PsA and axSpA is which part of the axial skeleton is affected first. While axSpA predominantly affects the SIJ and rarely involves the spine alone (spinal involvement without SIJ affection can be seen only in 1%–2% of axSpA patients), 38 axial PsA can affect all areas of the axial skeleton, with reported isolated spinal involvement of between 20% and 30%.35,39,40 Notably, cervical spine involvement is more common in axial PsA and may even be the sole manifestation, further distinguishing it from axSpA.7,8,11,12,41 These similarities and differences can blur the distinction between axial PsA and axSpA, making differential diagnosis challenging.
The second major difference is related to the classification criteria used for both diseases. The ClASsification for Psoriatic ARthritis (CASPAR) criteria are used to classify PsA and require, as a stem criterion, the presence of confirmed inflammatory articular disease involving the joints, axial skeleton, or entheses. Once this requirement is met, PsA can be classified based on features such as current, previous, or family history of PsO, psoriatic nail dystrophy, negative rheumatoid factor, dactylitis, and radiographic evidence of joint involvement. 42 Although axial symptoms are recognized, CASPAR criteria emphasize peripheral manifestations and specific psoriatic features. On the other hand, the ASAS classification criteria were specifically designed to define axSpA. 3 These criteria are aimed to detect early and subtle axial disease using advanced imaging techniques like MRI, which specifically highlights active inflammation in the SIJ, together with radiographs and the presence of the HLA-B27. Despite these differences, around a quarter of r-axSpA patients also fulfill the CASPAR criteria for PsA. 11 Noteworthy, PsO is also part of the SpA features in the ASAS classification criteria for axSpA. Overall, the CASPAR and ASAS criteria lack sufficient sensitivity and specificity for the classification of axial PsA, and thus cannot be employed in clinical practice for this purpose. Axial PsA is instead suspected in patients with PsA who present with back pain and is mainly evaluated using the clinical presentation, which can be supported by HLA-B27 status, and imaging of the axial skeleton. 43
Pathogenesis
The pathogenesis of axSpA and PsA, including axial PsA, remains incompletely understood. Along with immunological and genetic mechanisms, emerging data highlight the importance of the gut microbiome and other environmental factors, such as biomechanical loading. 44
The discovery of HLA-B27 in 1973 revealed a strong association between genetic predisposition and SpA, specifically axSpA.45,46 HLA-B27 is now recognized as the most significant genetic factor in the pathogenesis of axSpA, present in 70%–80% of patients. 47 Genome-wide association studies have identified numerous other genes linked to axSpA beyond HLA-B27, including major histocompatibility complex (MHC) variants and non-MHC genes such as endoplasmic reticulum aminopeptidase, the Tumor Necrosis Factor (TNF)-Receptor gene family, and NPEPPS (encoding puromycin-sensitive aminopeptidase). 48 Despite significant advancements in understanding the genetic background of axSpA, MHC, and non-MHC genes together explain less than 50% of the genetic predisposition, leaving the other half unexplained.49–51 In addition to these genetic associations, recent studies have highlighted the significant impact of gut microbiome alterations, or dysbiosis, on SpA pathogenesis.44,52 It is hypothesized that these gut alterations may be associated with HLA-B27, although the exact mechanism remains unclear. It is believed that HLA-B27 influences interactions between the host immune response and the gut microbiome, which may play a crucial role in disease pathogenesis. 53
PsA also results from a complex interaction of genetic and environmental factors that stimulate immune responses that affect skin, joints, and other tissues. 31 The strong genetic association with HLA, especially the presence of specific alleles of HLA-B (such as HLA-B27, HLA-B39, HLA-B38, and HLA-B08) and of HLA-C06 (*02), influences the development of PsA.54–56 Beyond HLA, other non-HLA genes (such as IL23R, TNFAIP, PTPN22) and cytokines (such as TNF, IL-17, and IL-23) also contribute to the pathogenesis by inducing immune activation.28,31,57 In addition to these genetic factors, environmental triggers such as biomechanical factors (also called “mechano-inflammation”), obesity, and the microbiome changes (gut and skin) may further contribute to disease onset and progression.31,54,58,59 However, while no specific studies have focused exclusively on axial PsA, it is highly likely that the pathogenesis of axial involvement in PsA shares similar factors with axSpA, including the aforementioned genetic, immunological, and environmental influences. Presently, a paucity of data exists regarding the microbiome, the immune cell pathways, and the epigenetic or environmental factors that are specifically associated with axial PsA. Consequently, further exploration into these non-genetic mechanisms is necessitated, particularly within well-defined axial PsA populations. Previous studies showed some genetic markers that may be associated with the presence of axial PsA; among these, HLA-B27, HLA-B08, and HLA-B38 should be highlighted. 60 Conversely, the HLA-C06 allele is primarily a risk factor for PsO, not for PsA. Although HLA-B27 is also associated with PsA, its prevalence in this patient population is significantly lower (15%–45%) compared to those with axSpA,9,28 but it should be noted that the reason for this low prevalence may be due to the collider bias, meaning that patients with HLA-B27 are tent to be diagnosed as axSpA rather than PsA (or axial PsA). However, this may still indicate shared genetic links between the two diseases, but the relationship between HLA-B27 and axial PsA remains controversial. 61
In summary, while HLA-B27 is present in 70%–80% of axSpA patients and is a well-known genetic marker for the condition, its prevalence in PsA and axial PsA is significantly lower, ranging from 13% to 45%.9,13,28,35,62–64 However, when HLA-B27 is present in axial PsA, it may be associated with more severe axial and peripheral manifestations. 65 In addition, other alleles such as HLA-B*08 have been associated with asymmetric sacroiliitis in axial PsA, whereas HLA-B27 tends to correlate with symmetric sacroiliitis.66–68 These different associations may help explain some of the heterogeneity observed in axial PsA and help distinguish it from axSpA.
Furthermore, in a recent perspective, McGonagle et al. 69 suggest that axial PsA may be characterized predominantly by ligament-centric soft-tissue involvement, contrasting with the bone-centric pathology seen in r-axSpA. While HLA-B27-associated bone marrow inflammation and osteitis are hallmarks of r-axSpA, axial PsA primarily involves inflammation of the spinal ligaments and entheses, which can be termed as “ligamentitis.” In addition, a “tree trunk and root” model is proposed to describe how axial PsA may present with prominent soft-tissue ossification and ligamentous involvement (tree trunk), as distinct from the early entheseal osteitis (tree root) seen in axSpA. 70 This distinction may, therefore, lead to different clinical and radiographic presentations, with axial PsA showing less bone marrow edema and more para-syndesmophyte formations compared to r-axSpA. Additionally, the immune cell composition and responses in the entheses and ligaments of axial PsA differ from the bone marrow-centric inflammation in r-axSpA, likely contributing to varied responses to therapies such as IL-12/23 and IL-23 inhibitors. This viewpoint emphasizes the importance of recognizing axial PsA as a distinct entity with unique therapeutic needs, diverging from simply applying the treatment approaches used for r-axSpA. However, further studies are needed to confirm this theory and better understand the underlying mechanisms.
Clinical characteristics and outcome measures
Axial PsA and axSpA manifest distinctive characteristics with regard to back pain and inflammatory back pain (IBP). The latter is a hallmark of axSpA and is, therefore, explicitly defined by the ASAS criteria, which include onset before 40 years of age, insidious onset, improvement with exercise, no improvement with rest, and nocturnal pain. 71 In axial PsA, IBP is less prevalent than in axSpA, and different IBP criteria do not perform well in established disease. 72 Studies indicate that neither rheumatologist-assessed IBP nor IBP criteria reliably assess axial involvement in PsA. 73 Although the Calin and Berlin criteria showed greater sensitivity in axial PsA patients compared to the ASAS criteria, many patients—especially women—still did not meet these criteria. 74
Demographically, PsA patients with axial involvement tend to be older at diagnosis and include a higher proportion of females, leading to a more balanced sex distribution compared to axSpA.10,11,63,75,76 As anticipated, peripheral musculoskeletal manifestations are more prevalent in axial PsA than in axSpA. In addition, enthesitis and dactylitis are more prevalent in axial PsA, significantly contributing to the overall disease burden.62,75 It is also noteworthy that the presence of radiographic damage in peripheral joints may be associated with axial involvement in PsA. 36 Conversely, the presence of peripheral arthritis can be associated with a delayed radiographic progression in r-axSpA. 77 AxSpA patients frequently present with EMMs, uveitis, and IBD, whereas these conditions are significantly less prevalent in axial PsA.11–13,62,63 As patients with EMMs are more likely to be diagnosed with axSpA, caution should be taken to avoid potential classification bias at the entry of patients into a group. In addition, PsO is more prevalent in axial PsA due to its strong association with PsA, and psoriatic nail changes are associated with axial PsA, 36 which is not the case in axSpA. A recent latent class analysis of a large SpA cohort supports this overlap and heterogeneity and reinforces the need for refined clinical subtyping within the SpA spectrum by identifying mixed phenotypes with both axial and peripheral features, particularly in patients with PsO and related features (dactylitis and nail disease). 78
Elevated inflammatory markers, such as erythrocyte sedimentation rate (ESR) and C-reactive protein (CRP), are commonly observed in both axial Psoriatic Arthritis (PsA) and axial Spondyloarthritis (axSpA), and it is well established that high CRP levels correlate with more severe radiographic damage in axSpA.79,80 These studies demonstrated that patients with axial PsA exhibited comparable ESR and CRP levels to those observed in axSpA patients, indicating a similar degree of systemic inflammation.13,62,63,76 However, recent studies have suggested a reduction in the standard CRP threshold (e.g., from ⩾0.5 to around 0.25 mg/dL) to enhance sensitivity in detecting active disease, particularly in established PsA.81,82 Outcome measures such as the Bath Ankylosing Spondylitis Disease Activity Index (BASDAI) and the Axial Spondyloarthritis Disease Activity Score (ASDAS) are frequently utilized instruments for the assessment of disease activity in axSpA. Baraliakos et al. 83 demonstrated that both the BASDAI and ASDAS are effective in assessing disease activity of axial symptoms in axial PsA, despite these tools being originally designed for axSpA. The utility of BASDAI in axial PsA was supported in another publication, noting comparable levels between axial PsA and AS patients. 84 While these tools may provide valuable insights into the management and monitoring of disease activity in axial PsA, it is important to consider that they might not fully capture the entire disease activity. On the other hand, serum CRP and responses to BASDAI questionnaires may be largely dependent on peripheral manifestations (and even skin disease), which are highly prevalent in PsA. 16 In conclusion, the development of specific disease activity assessment tools for axial PsA is desirable. Finally, comorbidities play a significant role in both axial PsA and axSpA, with depression being more prevalent in axial PsA, highlighting the psychological burden associated with psoriatic diseases. 85 However, it must be noted that many factors, including the prevalence and progression of comorbidities, have not been adequately addressed and prospectively investigated specifically in axial PsA.
These aforementioned distinct clinical and demographic characteristics in axial PsA suggest underlying pathophysiological differences from axSpA, underscoring the need for tailored diagnostic and comprehensive management strategies that address both axial and peripheral symptoms. Although both conditions may present with elevated inflammatory markers, many patients exhibit normal acute-phase reactants. This underscores the need for developing specific assessment tools for axial PsA that are not primarily dependent on these reactants, allowing for more accurate monitoring of disease activity and treatment responses.
Imaging
Imaging plays a pivotal role in the diagnosis and management of SpA, particularly in the presence of axial symptoms. Imaging techniques to show inflammatory changes with MRI and structural changes with conventional radiography or computed tomography in the SIJ and spine are vital for diagnosing, disease activity assessment, and monitoring. The modified New York criteria (mNYc), primarily crafted for axSpA, have been widely used in the evaluation of SIJ radiographs and for the definition of axial PsA in numerous studies. 86 Furthermore, MRI is of great value for identifying active inflammatory changes, particularly bone marrow edema, in the spine and SIJ, and the latter constitutes an important element in the axSpA classification criteria. 3 Recently, the ASAS MRI working group updated the definition of positive MRI changes in the SIJ to include both active and structural lesions, thereby enhancing the diagnostic accuracy for axSpA. 87 The active inflammatory findings on MRI are crucial for early diagnosis, particularly in nr-axSpA, where radiographic changes have not yet occurred. Nevertheless, this comprehensive definition also incorporates structural lesions for the accurate diagnosis, given that it is not always possible to detect active lesions, particularly considering longer symptom duration before diagnosis. Similarly, the group provided updated definitions for spinal MRI lesions in axSpA patients as for SIJ-MRI. 88 In the context of axSpA, the primary focus of MRI is on the SIJ, given the significance of sacroiliitis as a diagnostic feature. But as a recent study has demonstrated that in axial PsA, spinal MRI has the capacity to detect typical active or structural changes in more than one-third of the cases, even in the presence of normal SIJ imaging. 40 Consequently, a different approach is recommended for suspected axial PsA. Given the fact that axial PsA can manifest with isolated spinal involvement, it should be recommended that MRI of the entire spine—or, at the very least, the region corresponding to the patient’s pain—be considered for a comprehensive assessment. These imaging modalities facilitate not only the initial diagnosis but also the monitoring of disease progression and the guidance of treatment decisions. Given the lack of a widely accepted formal definition of axial PsA and the overlap in clinical presentations between the two conditions, the imaging insights that were gained from axSpA are currently the only valuable tools for defining axial PsA. Furthermore, imaging enables clinicians to identify numerous patients with axial PsA who may exhibit silent axial involvement, characterized by imaging changes in the axial skeleton without any clinical symptoms.11,89
Radiographs are a standard diagnostic tool for assessing structural damage in the SIJ for axSpA. In r-axSpA, bilateral and symmetrical sacroiliitis is common, characterized by erosions, sclerosis, joint-space irregularities, and eventual ankylosis. These features are hallmark signs of chronic inflammation in the past and are typically more pronounced in axSpA compared to axial PsA, where up to 30% of patients may show mNY-positive SIJ changes.11,90 In axial PsA, however, SI joint involvement is often described as asymmetrical, 91 or unilateral, making radiographic diagnosis more challenging.11,40,92,93 Additionally, in their recent publication, de Hooge et al. 90 found that radiographic sacroiliitis in axial PsA is associated with a higher likelihood of developing syndesmophytes over a 2-year period, particularly when combined with elevated CRP levels, underscoring the importance of early and accurate detection of imaging changes. On the other hand, it should be noted that sacroiliitis on imaging is not always necessarily associated with the diagnosis of axial PsA. 13
Spinal radiographs serve as an important tool in visualizing structural changes along the vertebral column in SpA. In axSpA, particularly r-axSpA, spinal radiographs often show syndesmophytes, which are bony growths originating inside the ligaments of the spine. These changes are typically marginal, originating at the edges of the vertebral bodies, and reflect the chronic inflammatory process associated with axSpA. In axial PsA, however, spinal involvement can present differently. Spinal radiographs in axial PsA patients may reveal nonmarginal syndesmophytes, 94 which morphologically differ from the marginal syndesmophytes typical of axSpA, have a greater volume, therefore they can be called “para-marginal,” and axial PsA might show increased paravertebral ossifications, which are less commonly seen in axSpA.91,95 Moreover, degenerative changes should also be considered through the evaluation process of axial PsA, since mechanically associated changes, such as diffuse idiopathic skeletal hyperostosis, can be regularly observed in these patients when taking into account their older age, and comorbidities such as obesity.69,96 In regards to scoring methods for research purposes to assess damage and radiographic progression, there is a need for the development of radiographic scoring tools that are specifically designed for axial PsA. The current tools, including the Bath Ankylosing Spondylitis Radiology Index for the spine, the modified Stoke Ankylosing Spondylitis Spine Score, and the Radiographic Ankylosing Spondylitis Spinal Score, are reliable; however, they may not fully capture the unique spinal involvement patterns observed in axial PsA. 97 Nevertheless, it was shown that the Psoriatic Arthritis Spondylitis Radiology Index may exhibit superior efficacy for this objective. 98 The differential pattern of spinal involvement between axSpA and axial PsA is significant for both diagnosis and management. While axSpA presents with a more predictable and severe spinal pathology, 99 the variability seen in axial PsA necessitates a broader diagnostic approach. This includes considering additional imaging modalities and recognizing the diverse presentation of axial involvement in PsA, which can also impact treatment decisions and prognostic assessments. However, it currently remains unclear whether this is due to true differences in the pathogenesis. 37
Advanced imaging techniques such as MRI can offer additional insights into early inflammatory (bone marrow edema, enthesitis, etc.) and structural changes, which are not yet visible on radiographs, and may provide a more nuanced understanding of the disease process in axial PsA. Currently, definitions depicted from axSpA studies are used for the interpretation/definition of MRI lesions for axial PsA. 87 It is expected that the presence of entheseal involvement on MRI is more common in axial PsA, reflecting the broader range of inflammatory sites characteristic of PsA, but no data confirmed this expectation. However, there is an ongoing debate about whether current MRI protocols can fully capture the inflammatory processes in axial PsA. This potential limitation may be attributed to the previously mentioned “ligamentitis” theory. 69 A recent MRI study of the spine in patients with PsA reported low rates of axial inflammation defined by MRI and poor alignment with IBP determined by clinical evaluation. 100 Supporting this complexity, a recent study described a subgroup (35%) of patients with axial PsA with negative X-rays but positive MRI findings—so-called non-radiographic axial PsA—emphasizing the importance of MRI in complementing conventional imaging. 35
These differences in MRI findings underscore the importance of MRI in distinguishing between axSpA and axial PsA. The ability to detect subtle variations in inflammation patterns can aid in accurate diagnosis and inform more targeted therapeutic strategies. Furthermore, MRI’s potential role for the management of disease activity, which is still under debate, highlights its value in the ongoing management of both axSpA and axial PsA. Future studies with novel techniques, including artificial intelligence, will also be important to interpret the imaging findings and to better understand the process of axial involvement.
Treatment
Since the recognition of axial PsA as a distinct clinical entity, 95 no tailored treatment strategy has yet been developed for this patient population. Given the clinical and imaging overlap with axSpA, current treatment recommendations for axial PsA are largely derived from axSpA guidelines, relying on evidence from studies conducted in axSpA populations.16–19 However, recent studies suggest a need for specific treatment recommendations that consider the unique features of axial disease in PsA.
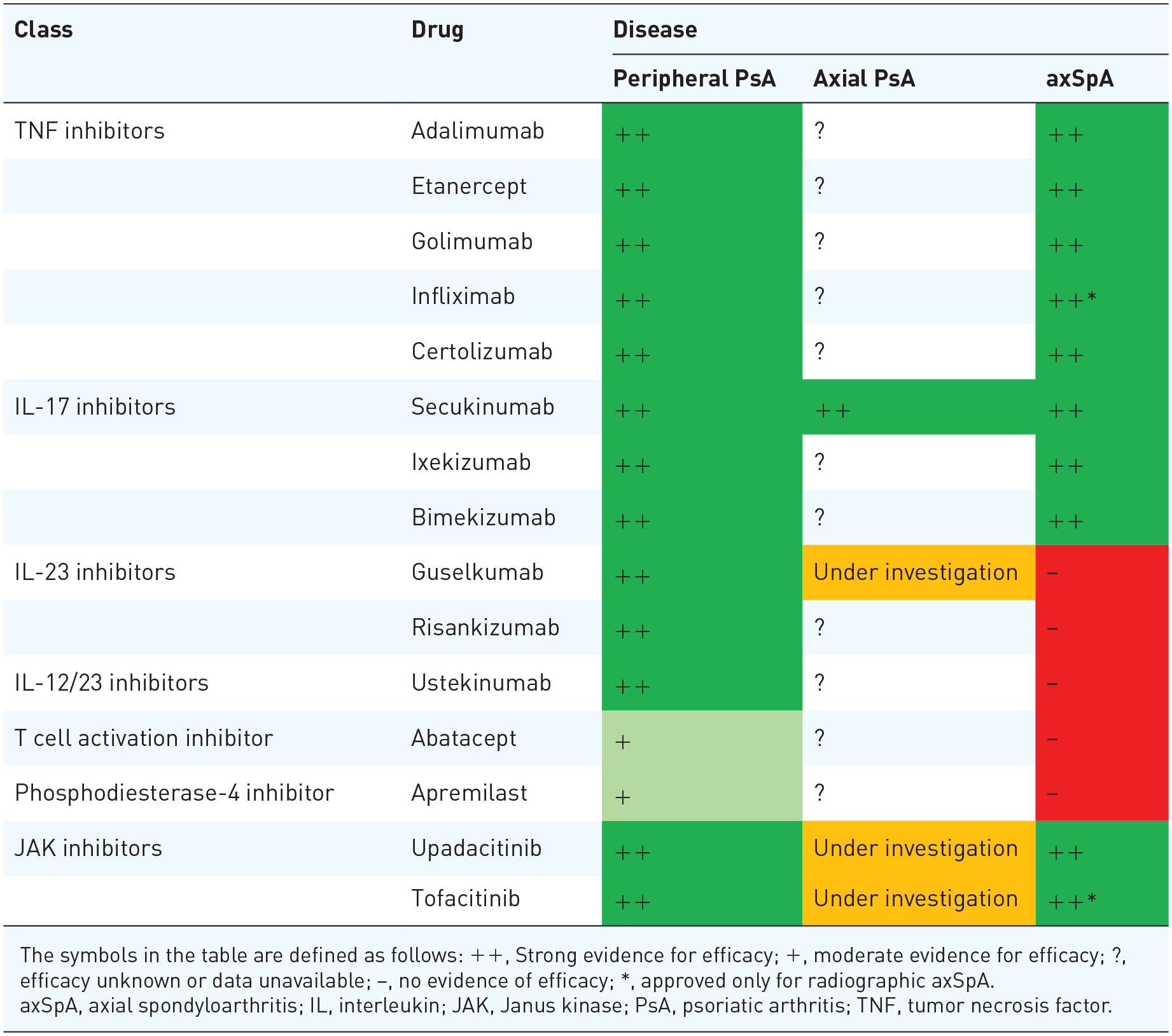
Over the past few decades, significant advancements have been made in the treatment of axSpA and PsA. 101 Nonpharmacological management—such as physiotherapy, regular exercise, and topical therapies for PsO—forms the foundation of management for both axSpA and PsA. Many biological disease-modifying anti-rheumatic drugs (bDMARDs) as well as targeted-synthetic DMARDs (tsDMARDs) are applicable for both axSpA and PsA, though there are important differences reflected in the treatment guidelines of GRAPPA and ASAS.16,17 Other manifestations, including enthesitis, dactylitis, arthritis, or EMMs, present treatment challenges in both diseases and must also be addressed. 102 The table provides an overview of the various b- and ts-DMARDs and their indications for peripheral PsA, axial PsA, and axSpA (Table 1).
Comparison of drug efficacy in peripheral PsA, axial PsA, and AxSpA.

The symbols in the table are defined as follows: ++, Strong evidence for efficacy; +, moderate evidence for efficacy; ?, efficacy unknown or data unavailable; −, no evidence of efficacy; *, approved only for radiographic axSpA.
axSpA, axial spondyloarthritis; IL, interleukin; JAK, Janus kinase; PsA, psoriatic arthritis; TNF, tumor necrosis factor.
Conventional synthetic DMARDs (csDMARDs), mainly methotrexate, but also sulfasalazine (SSZ), and leflunomide, remain the recommended first-line therapies for peripheral PsA.16,17 SSZ may also be considered for treating peripheral involvement in SpA; however, SSZ only plays a minimal role in axSpA and axial PsA, yet is not recommended as a first-line therapy. 19
Oral phosphodiesterase (PDE4) inhibitors as tsDMARDs, such as apremilast, are primarily used for the treatment of peripheral PsA. 102 A placebo-controlled phase III study failed to demonstrate efficacy for apremilast in axSpA. 103 However, the MOSAIC study, the results of which were recently presented as a congress abstract, indicated that apremilast can reduce axial inflammation as assessed by MRI in oligoarticular PsA with axial involvement.104,105 Abatacept, a T cell activation inhibitor, has demonstrated efficacy in treating peripheral PsA, achieving an ACR20 response in a phase III study, 106 but has not shown efficacy in axSpA. 107
bDMARDs have been widely used for the management of axSpA, axial PsA, and peripheral PsA, often employed when patients do not adequately respond to first-line treatments (nonsteroidal anti-inflammatory drugs (NSAIDs) or csDMARDS). TNF inhibitors (TNFi) such as adalimumab, etanercept, certolizumab pegol, golimumab, and infliximab are approved therapies for axSpA and PsA.16–18,101 The efficacy of TNFi has been validated in numerous randomized controlled trials (RCTs); however, current recommendations for axial PsA are based solely on data from axSpA studies, as there are no RCTs specifically targeting axial PsA patients.8,102 Recent evidence indicates that remission and minimal disease activity (MDA) are achievable in axial PsA patients treated with anti-TNF agents, with approximately 31.2% achieving BASDAI 50 and 50% achieving MDA after 12 months. 108 Predictors of MDA include male sex, younger age, and shorter disease duration, highlighting specific patient factors that can guide treatment expectations.
The IL-17 inhibitors have also gained attention for their utility in treating peripheral PsA, axial PsA, and axSpA. IL-17 inhibitors are preferred over TNFi inhibitors in patients with significant PsO skin involvement.17,102 The only published phase III study yet that specifically addressed axial PsA was the MAXIMISE study, which demonstrated that secukinumab significantly improved axial symptoms compared to placebo in patients with PsA inadequately managed with NSAIDs. Specifically, 63% and 66% of patients treated with secukinumab 300 and 150 mg, respectively, achieved ASAS20 responses at week 12, compared to 31% with placebo. However, it is important to note that patients were chosen based on symptoms in the spine without needing to confirm this with imaging. This should be kept in mind when interpreting the results. Furthermore, MRI findings indicated a reduction in inflammation at the spine and SIJs, thereby supporting the study’s conclusion regarding the efficacy of secukinumab in reducing axial disease symptoms.109,110
IL-23 inhibitors have demonstrated efficacy in treating both PsO and PsA. Ustekinumab, an IL-23p40 inhibitor, initially showed promise in RCTs involving TNFi-naïve patients with axSpA 111 ; however, it failed to meet the ASAS40 response endpoint, leading to the early termination of subsequent phase III studies. 20 Similarly, risankizumab failed to meet the ASAS40 response in axSpA, suggesting a limited role for IL-23 inhibition in axSpA treatment. 112 In post hoc analyses of the DISCOVER 1/2 studies, guselkumab showed better BASDAI and ASDAS responses compared to placebo, 22 but the results of these studies should be interpreted cautiously since the power was not calculated for this objective and the efficacy assessment of guselkumab on the axial involvement of PsA was solely based on clinical assessment. The ongoing STAR trial is specifically investigating guselkumab’s efficacy in axial PsA and active MRI at baseline, with results highly anticipated. 113 Interestingly, a recent mechanistic data from ex vivo study has shown that axial peri-entheseal bone—particularly in the spine and hip—produces markedly higher levels of IL-23 compared to peripheral sites, which may help explain the paradoxical lack of efficacy of IL-23 blockade in axial disease and raise the question of whether higher dosing might be needed for axial involvement. 114
Janus kinase (JAK) inhibitors, which are also classified as tsDMARDs, represent a recent advancement in treating axSpA and PsA, and are recommended for these conditions alongside the above-mentioned bDMARDs. In phase III RCTs, tofacitinib and upadacitinib demonstrated significantly superior ASAS-40 responses in axSpA patients compared to placebo.115,116 The SELECT-PsA-1 study demonstrated that upadacitinib was not inferior to adalimumab in treating peripheral PsA. 117 A post hoc analysis of the SELECT-PsA 1/2 study indicated upadacitinib’s efficacy in axial PsA compared to placebo. Here, it must be noted as a limitation that patient selection was solely at the discretion of the local investigators. 118 The ongoing investigator-initiated PASTOR and UP-SPOUT studies, in which patients are recruited based on confirmed active inflammation either in SIJ or spine, aim to specifically evaluate tofacitinib’s and upadacitinib’s efficacy in axial PsA (ClinicalTrials.gov identifier: NCT06454188). 119
Recent evidence suggests that IBP in axial PsA may be significantly more responsive to corticosteroid treatment compared to IBP in r-axSpA. 120 This differential response may be attributed to the unique inflammation profile in PsA, which is more enthesis-driven and characterized by a greater predominance of macrophages and IL-23/IL-17 axis involvement. These findings imply distinct underlying mechanisms for IBP in axial PsA versus r-axSpA, supporting the need for condition-specific therapeutic approaches.
Overall, the treatment strategies for axSpA and (axial) PsA are largely similar, particularly with the use of TNF, IL-17, and JAK inhibitors demonstrating efficacy in both conditions. However, a disparity appears to exist in the efficacy of IL-12/23 inhibitors, which have demonstrated efficacy in PsA but failed to show benefit in axSpA, while post hoc analyses suggest promising results in patients with axial PsA. Given the differences between these conditions, caution is warranted when extrapolating treatment outcomes from axSpA studies to axial PsA. Data from dedicated studies specifically addressing treatment efficacy in axial PsA are critically needed. To advance understanding, well-designed clinical trials targeting axial PsA populations, using standardized definitions, are urgently required.
Summary
Axial PsA and axSpA are distinct yet overlapping entities within the broader spectrum of spondyloarthritis, posing unique challenges in diagnosis, classification, and management. This conceptual ambiguity can be illustrated by Rubin’s vase (Figure 1), a visual illusion where perception shifts between two distinct images—either a vase or two faces—depending on the observer’s focus. Similarly, the classification of axial PsA and axSpA has long been influenced by perspective: some researchers view them as part of a shared disease spectrum, while others recognize fundamental differences. However, as our understanding evolves, the definition of axial PsA is becoming clearer, akin to how focusing on one image in Rubin’s vase eventually stabilizes perception. In Figure 2 similarities and differences between axSpA and axial PsA are illustrated (Figure 2). Despite recent advances in the understanding of SpA through genetic insights, novel imaging techniques, and innovative treatment approaches, the distinctions and overlaps between these conditions remain insufficiently understood and require further clarification.

Rubin’s Vase: highlighting the impact of perspective on evaluations.
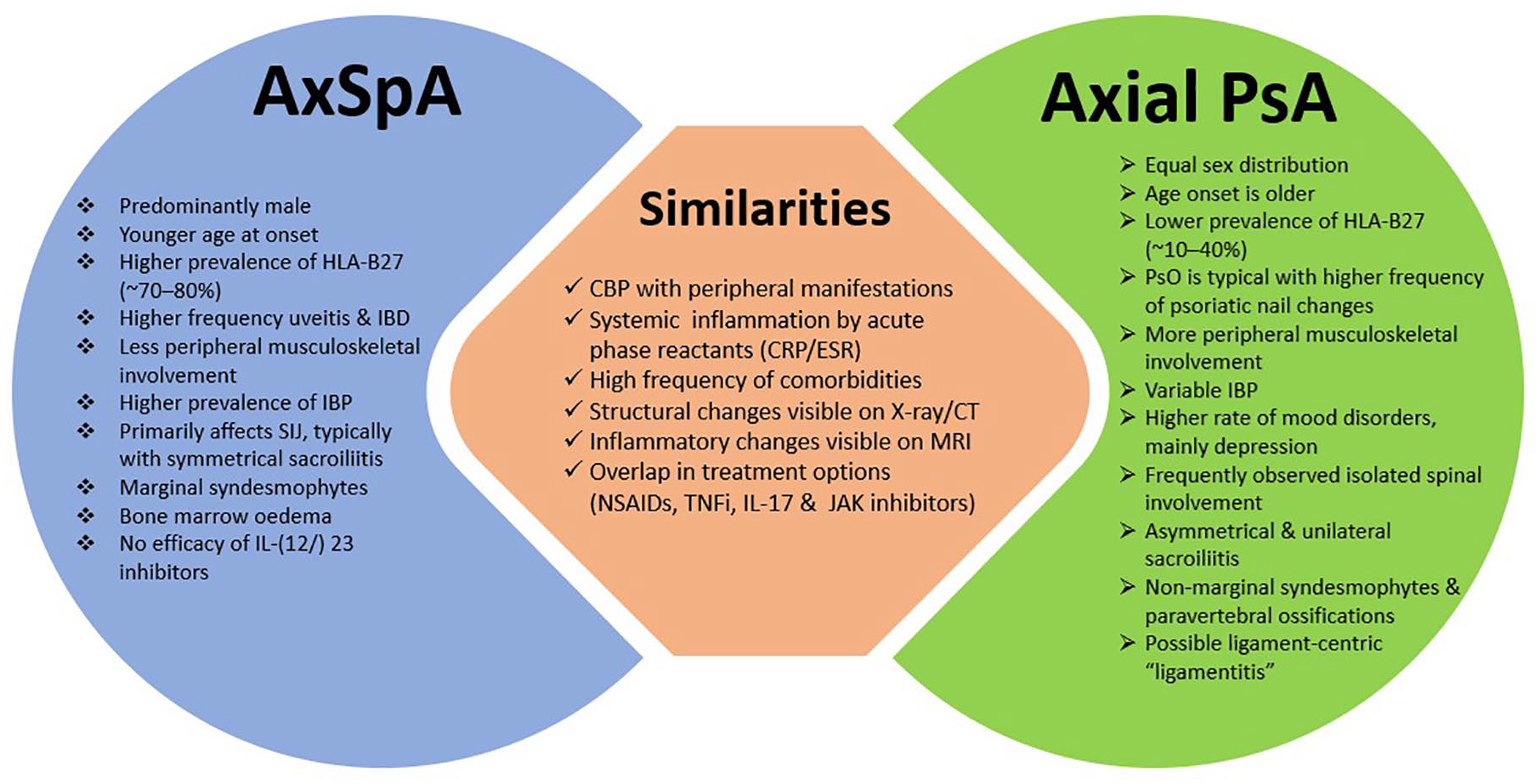
Similarities and differences between AxSpA and axial PsA.
Axial PsA is typically diagnosed based on clinical assessment and imaging findings. The ongoing AXIS study, an ASAS-GRAPPA collaboration, aims to establish classification criteria for axial PsA, marking a crucial step toward standardized diagnosis. 64 Furthermore, the upcoming STAR, PASTOR, and UP-SPOUT trials will evaluate the efficacy of guselkumab, tofacitinib, and upadacitinib in axial PsA patients, who have to have MRI-confirmed active inflammation in SIJ or spine as part of the inclusion criteria. Beyond these efforts, a GRAPPA Collaborative Research Network-led study (MONOLITH) has already started recruitment and aims to conduct a molecular characterization of axial PsA versus peripheral PsA, providing insights into the distinct biological underpinnings of these disease manifestations (ClinicalTrials.gov identifier: NCT05925842).
Despite significant overlaps in clinical features, axial PsA and axSpA differ substantially in their genetic, clinical, and radiographic presentations, which necessitate distinct diagnostic and therapeutic strategies. Imaging modalities such as radiography and MRI are indispensable for diagnosing axial PsA, particularly given its tendency for asymmetrical involvement and the frequent absence of back pain, which may lead to underdiagnosis. Studies have shown that longstanding PsA is often associated with axial involvement, suggesting that an inception cohort with shorter disease duration would be ideal for predicting long-term axial PsA outcomes.
Conclusion
In conclusion, although axial PsA and axSpA manifest similar characteristics, their distinguishing genetic elements, clinical manifestations, and therapeutic responses support their classification as discrete conditions rather than as two sides of a unified entity. At this point, the Rubin’s vase analogy serves as a reminder that, although historical classifications blurred these boundaries, emerging data now provide a clearer focus, reinforcing the need for specific classification criteria. Future research should prioritize the identification of biomarkers to facilitate early diagnosis and personalized treatment pathways. The AXIS study will be instrumental for a standard definition of axial PsA and will open the door for more consistent and reliable research outcomes. Future trials, alongside molecular characterization initiatives, will further refine disease-specific treatment guidelines and help identify new therapeutic targets, paving the way for improved patient care and outcomes in axial PsA.