Abstract
Mitochondrial dysfunction is a well-established pathological event in Parkinson’s disease (PD). Proteins misfolding and its impaired cellular clearance due to altered autophagy/mitophagy/pexophagy contribute to PD progression. It has been shown that mitochondria have contact sites with endoplasmic reticulum (ER), peroxisomes and lysosomes that are involved in regulating various physiological processes. In pathological conditions, the crosstalk at the contact sites initiates alterations in intracellular vesicular transport, calcium homeostasis and causes activation of proteases, protein misfolding and impairment of autophagy. Apart from the well-reported molecular changes like mitochondrial dysfunction, impaired autophagy/mitophagy and oxidative stress in PD, here we have summarized the recent scientific reports to provide the mechanistic insights on the altered communications between ER, peroxisomes, and lysosomes at mitochondrial contact sites. Furthermore, the manuscript elaborates on the contributions of mitochondrial contact sites and organelles dysfunction to the pathogenesis of PD and suggests potential therapeutic targets.
Keywords
Loss of dopaminergic neurons in the substantia nigra pars compacta (SNpc) and striatum (STr) and intra-neuronal accumulation of alpha-synuclein (α-syn) are the major pathological hallmarks of PD (Elfarrash et al., 2019; Wegrzynowicz et al., 2019). Dopamine plays a significant role in the regulation of voluntary movements and its decrease not only affects motor functions but also a spectrum of non-motor functions. Thus, it serves as a biomarker in the premotor phase of PD (Mulcahy et al., 2020; Schneider et al., 2021). Several recent reports suggest that dopaminergic dysfunction is associated with the accumulation of α-syn in the mitochondrial structure (Danyu et al., 2019; Ganjam et al., 2019). Mitochondrial dysfunction plays a critical role in the progression of PD by impairing fusion-fission, transport and selective autophagic degradation of misfolded proteins (Yamaguchi et al., 2020; Ahmed et al., 2021; Mani et al., 2021). Mitochondria communicate bi-directionally with endoplasmic reticulum, peroxisomes and lysosomes at the mitochondrial contact sites and maintain cellular homeostasis (Xia et al., 2019; Picca et al., 2020). ER is the largest store of Ca2+ in a cell. The ER-mitochondria contact site is called Mitochondria-associated membrane (MAMs) (Cherubini et al., 2020). Transfer of Ca2+ and phospholipid precursors to mitochondria from endoplasmic reticulum (ER) is the basic function at this site (Gomez-Suaga et al., 2017; Kostic et al., 2018; Che et al., 2021). Neurotoxins like rotenone (used to induce PD in rodent models) damage MAMs by altering mitochondrial Ca2+ homeostasis (Lee et al., 2018; Wu et al., 2018). Mitochondria and lysosomes interact at a contact site which is distinct from the mitophagy process, and that help to regulate cellular homeostasis (Wong et al., 2019). Additionally, lysosome and mitochondria form contact sites with ER for the transfer of calcium, phospholipids and cholesterol to the respective organelles (Atakpa et al., 2018; Deus et al., 2020). Lysosome-Mitochondria-ER contact sites are further regulated by lipid-transfer proteins such as the vacuolar protein sorting-associated protein 13 (VPS13) family, wherein VPS13A has been linked to ER-mitochondria contacts and VPS13C is linked to ER-lysosome contacts (Kumar et al., 2018). Mutations of VPS13 family genes are linked to different neurodegenerative diseases (Vonk et al., 2017) and interestingly, mutations in VPS13C are linked to PD pathology (Lesage et al., 2016). Lloyd-Evans and Waller-Evans (2019) reviewed lysosomal Ca2+ homeostasis and its role in various human diseases (Lloyd-Evans and Waller-Evans, 2019). Apart from Ca2+ signalling, lysosomal dysfunction was reported to affect the chaperone-mediated autophagic clearance of α-syn, which contributes to PD (Arias et al., 2015; Sun et al., 2019).
Mitochondria and peroxisomes work closely to maintain the lipid balance via fatty acid β-oxidation pathway (Cohen et al., 2014; Hosoi et al., 2017; Valm et al., 2017). Peroxisomes regulate mitochondrial dynamics and mitochondria-mediated apoptosis (Tanaka et al., 2019). Plasmalogen, a peroxisome-derived glycerophospholipid is an important component of mitochondrial membrane (Kimura et al., 2019) and its decrease has been reported in PD (Fabelo et al., 2011; Miville-Godbout et al., 2016). Altered metabolism and oxidative stress have been shown to play a crucial role in various neurodegenerative diseases including PD (Kurian et al., 2017; Kumar et al., 2019; L. Zhang et al., 2020; Xie et al., 2020).
Growing evidence suggests that the dysfunction in the mitochondrial contact sites with ER, lysosome and peroxisome are associated with PD. The present review summarises data on the individual organelle dysfunction and alterations in the inter-organellar crosstalk involving mitochondrial contact sites in PD. It also provides information on the role of redox signalling, autophagy, and potential therapeutic targets in pathogenesis of PD.
Mitochondrial Dysfunction in PD
Mitochondria specific genes such as PINK1, PARK2 (PARKIN), DJ-1 (PARK7) and leucine-rich repeat kinase 2 (LRRK2) play crucial roles in PD by regulating ROS homeostasis (Valente et al., 2004; Nichols et al., 2005; Guzman et al., 2010). PINK1 accumulation in the mitochondria activates PARKIN's E3 ubiquitin ligase activity and recruits PARKIN into the impaired mitochondria. This triggers the selective autophagic clearance of damaged mitochondria (Lazarou et al., 2012; Xi et al., 2021). Mutations in PINK1, PARKIN, DJ-1 and LRRK2 are linked to PD (Puspita et al., 2017). The involvement of mitochondrial dysfunction in PD was more clearly understood from an incident where several drug abusers self-injected 1-methyl-4-phenyl-1,2,3,6-tetrabydropyridine (MPTP). The bioactive form of MPTP is 1-methyl-4-phenylpyridinium (MPP+) which inhibits Complex I of the Electron Transport Chain (ETC) in mitochondria, resulting in increased ROS generation (Javitch et al., 1985; Mizuno et al., 1987). This in turn caused selective destruction of dopaminergic neurons in the SNPc and STr, resembling clinical symptoms of PD (Langston et al., 1983). Unlike nuclear DNA, mitochondrial DNA (mtDNA) is more vulnerable to oxidative damage due to lack of histones (Gureev et al., 2017). mtDNA encodes 13 essential proteins of ETC (Gustafsson et al., 2016) and their mutation/deletions triggers oxidative stress which results in neuronal damage (Iannielli et al., 2018). Polymerase γ1 (POLG1) enzyme is a nuclear-encoded gene product that plays an important role in polymerase synthesis and mtDNA maintenance (Ropp and Copeland, 1996). Mutations in POLG1 are shown to be associated with PD, and this corroborates with mtDNA mutation (Gui et al., 2015; Hsieh et al., 2019).
Impaired Mitophagy
The selective removal of damaged mitochondria by autophagosomes and the subsequent catabolism by lysosomes is termed mitophagy. Alterations in fission and fusion process (a crucial part of mitophagy) play an important role in neurodegenerative diseases like AD, PD, stroke etc. (Chen et al., 2021; Trombetta-Lima et al., 2021). The regulation of fission and fusion is governed by several key proteins including the dynamin-related protein 1 (DRP1), the mitochondrial fusion proteins 1, 2 (MFN1, MFN2) and the optic atrophy 1 (OPA1) protein (Yu-Wai-Man et al., 2014; Xi et al., 2018). Stressful conditions such as nutritional deficiency or exposure to toxins impairs mitochondria driven -MFN1, MFN2 and OPA1 mediate fusion process (Archer, 2013). Severe mitochondrial damage promotes DRP1 and FIS1 facilitates fission (X. Zhang et al., 2020) and produces smaller fragments of mitochondria for clearance by mitophagy (Chen et al., 2021). Despite this data, it still remains unclear as to how the defective mitochondria decide between fusion and fission. Deletion of the MFN2 gene in mouse induces ETC impairment and causes dopaminergic degeneration (Lee et al., 2012; Pham et al., 2012). Further, Berthet et al. (2014) showing that DRP1 gene knock-out in mouse resultss in depletion of mitochondria and dopaminergic degeneration leading to PD (Berthet et al., 2014).
Mutations in PINK1 and PARKIN, the major regulatory proteins in mitophagy are associated with PD pathogenesis (Vincow et al., 2013; Park and Koh, 2020). Furthermore, the involvement of other PD related genes like DJ-1, LRRK2 in mitophagy in PD pathogenesis is also well documented in various reports (Deas et al., 2011; J Liu et al., 2019; Wang et al., 2019).
Endoplasmic Reticulum Dysfunction in PD
In addition to its role in calcium storage and protein synthesis and regulation of mitochondrial energy metabolism, ER performs protein folding and trafficking (Field et al., 2007; Choi et al., 2017; Sowers et al., 2018; Lima et al., 2019). ER dysfunction or an unhealthy cellular environment produces misfolded proteins (Chen et al., 2019). The aggregation of misfolded proteins activates unfolded protein response (UPR) via ER stress sensors such as ATF6 (activating transcription factor 6), PERK (protein kinase RNA-like endoplasmic reticulum kinase) and IRE1 (inositol-requiring enzyme 1) (IRE1). This helps in protein-folding homeostasis. Activated sensors induce expression of genes that code for the proteins involved in the cellular clearance of misfolded proteins (Adams et al., 2019). ER stress-induced by glucose starvation, hypoxia, disruption of calcium homeostasis, or oxidative stress causes the accumulation of misfolded proteins. Prolonged ER stress and consistent accumulation of misfolded proteins stimulate the proapoptotic pathways in cells through activation of the transcription factor C/EBP homologous protein (CHOP) and cysteine proteases caspase-4/12 (Nakagawa et al., 2000; Ferri and Kroemer, 2001; Hitomi et al., 2004; Marciniak et al., 2004) leading to cell death. This was clearly demonstrated in 6-OHDA mouse model of PD, where the upregulated CHOP causes neuronal death through apoptosis (Sun et al., 2013; Aimé et al., 2020). Similarly, knockdown of ATF6, a UPR protein, is shown to aggravate MPTP induced neurotoxicity in a mouse model (Egawa et al., 2011). Consistent with the experimental reports on PD models (Bellucci et al., 2011), the post mortem examination of PD patients’ brains revealed the activation of UPR protein with α-syn co-localization (Hoozemans et al., 2012). Further, accumulation of misfolded α-syn is reported to activate IRE1-induced autophagy-dependent neuronal death in a drosophila model of PD. Thus inhibition of IRE1 is found to be neuroprotective (Yan et al., 2019). Knockdown of Herp (ER stress protein) and mutation in LRRK2 exacerbated the ER-stress mediated neuronal death and α-syn aggregation in PD (Belal et al., 2012; Lee et al., 2019). These data indicate that ER dysfunction has a crucial impact on PD pathogenesis.
ER–Mitochondria Contact Sites
Bernhard and Rouiller (1956), initially explained the inter-organellar contact sites between mitochondria and ER (Bernhard and Rouiller, 1956). ER contact sites with mitochondria are termed as “Mitochondria-Associated Membranes (MAMs)”. About 5 to 20% of the mitochondrial surface is physically in contact with ER and varies depending on the cell type (Rizzuto et al., 1998). A symbiotic relationship between ER and mitochondria is crucial for the maintenance of cellular Ca2+ homeostasis (Calvo-Rodríguez et al., 2016; Wu and Kao, 2016; Butler et al., 2017). Calcium concentration in ER regulates several enzymes involved in the synthesis of secretory proteins such as N-glycosylation and oxidative protein folding like calnexin or calreticulin (Guo et al., 2020; Kweon et al., 2020). Cytosolic chaperone GRP75 is the first complex identified in mammalian cells that bridges the mitochondrial voltage-dependent anion channel (VDAC) of the outer mitochondrial membrane (OMM) with inositol 1,4,5-triphosphate receptor (IP3R) of ER and forms MAMs (Szabadkai et al., 2006; Yeo et al., 2021). MAMs allow the exchange of lipids (Shiao et al., 1995), calcium (Ca2+) (Rizzuto et al., 1998; Csordás et al., 1999) and reactive oxygen species (Verfaillie et al., 2012; Eisner et al., 2013) between both the organelles. This allows for = their adaptations in cellular stress. Knockdown of GRP75 is reported to decrease the transfer of Ca2+ from the ER to mitochondria (Csordás et al., 2006). MFN1 and MFN2, the two key mitochondrial fusion proteins are reported to strengthen the MAMs and facilitate mitochondrial Ca2+ uptake (Filadi et al., 2016; Che et al., 2021). MFN2 is localized at both ER and mitochondrial membranes. At the MAMs MFN1 and MFN2 form homo-and heterotypic interactions, the latter with MFN1 (Szabadkai et al., 2006; de Brito and Scorrano, 2008). In addition, knockout of MFN2 is reported to decrease ER-mitochondria contact and crosstalk (Krols et al., 2016). Calcium homeostasis is also assisted by cytosolic Ca2+ reuptake into ER through the sarco/endoplasmic reticulum (SR/ER) Ca2+ATPase pump (SERCA) (Chemaly et al., 2018). Alterations in SERCA due the mutations, toxins or DNA damage increases the Ca2+ transfer from ER to mitochondria. This causes mitochondrial Ca2+ overload leading to apoptosis through the opening of mPTP (mitochondrial permeabilization transition pore), cytochrome c release and other pro-apoptotic factors (Bittremieux et al., 2016).
With respect to ER-Mitochondria contacts, pathogenic mutations of α-syn are reported to be localized in MAMs (Guardia-Laguarta et al., 2014; Paillusson et al., 2017). Dysfunction in MAMs alters the IP3R-GRP75-VDAC complex, mitochondrial Ca2+ homeostasis and leads to mitochondrial dysfunction and causes dopaminergic death in PD (Calì et al., 2012; Guardia-Laguarta et al., 2015). Furthermore, knockout of DJ-1 disrupts the IP3R-GRP75-VDAC complex and forms a causative factor for PD (Y. Liu et al., 2019). Another PD associated protein PINK1 is involved in mitophagy process.LRRK2 is also reported to be localized in MAMs and its dysfunction causes PD (Park and Koh, 2020; Toyofuku et al., 2020; Zhao et al., 2021). PARKIN, an E3 ubiquitin ligase is associated with MFN2 at MAMs and supports Ca2+ homeostasis. Hence, mutations in PARKIN are shown to decrease the tethering between ER and mitochondria, which is strongly linked to PD (Palacino et al., 2004; Calì et al., 2013; Basso et al., 2018). Hailey et al. (2010) demonstrated the recruitment of autophagy-related proteins (ATG14, ATG5) at MAMs during starvation in cells (Hailey et al., 2010; Hamasaki et al., 2013) and also reported that the knockdown of MFN2 gene inhibits the formation of autophagosomes. Here, it is important to mention that dysregulation in autophagy is one of the major pathological hallmark in PD (Filadi et al., 2018; Janikiewicz et al., 2018). PINK1 and Beclin1 (autophagy marker) localize in the MAMs. Silencing of PINK1 impairs Beclin1 enrichment and triggers cell death in SH-SY5Y cells (Gelmetti et al., 2017). These leads to disruption in the crosstalk between mitochondria and ER (MAMs) and plays a crucial role in PD (Figure 1 and Table 1).
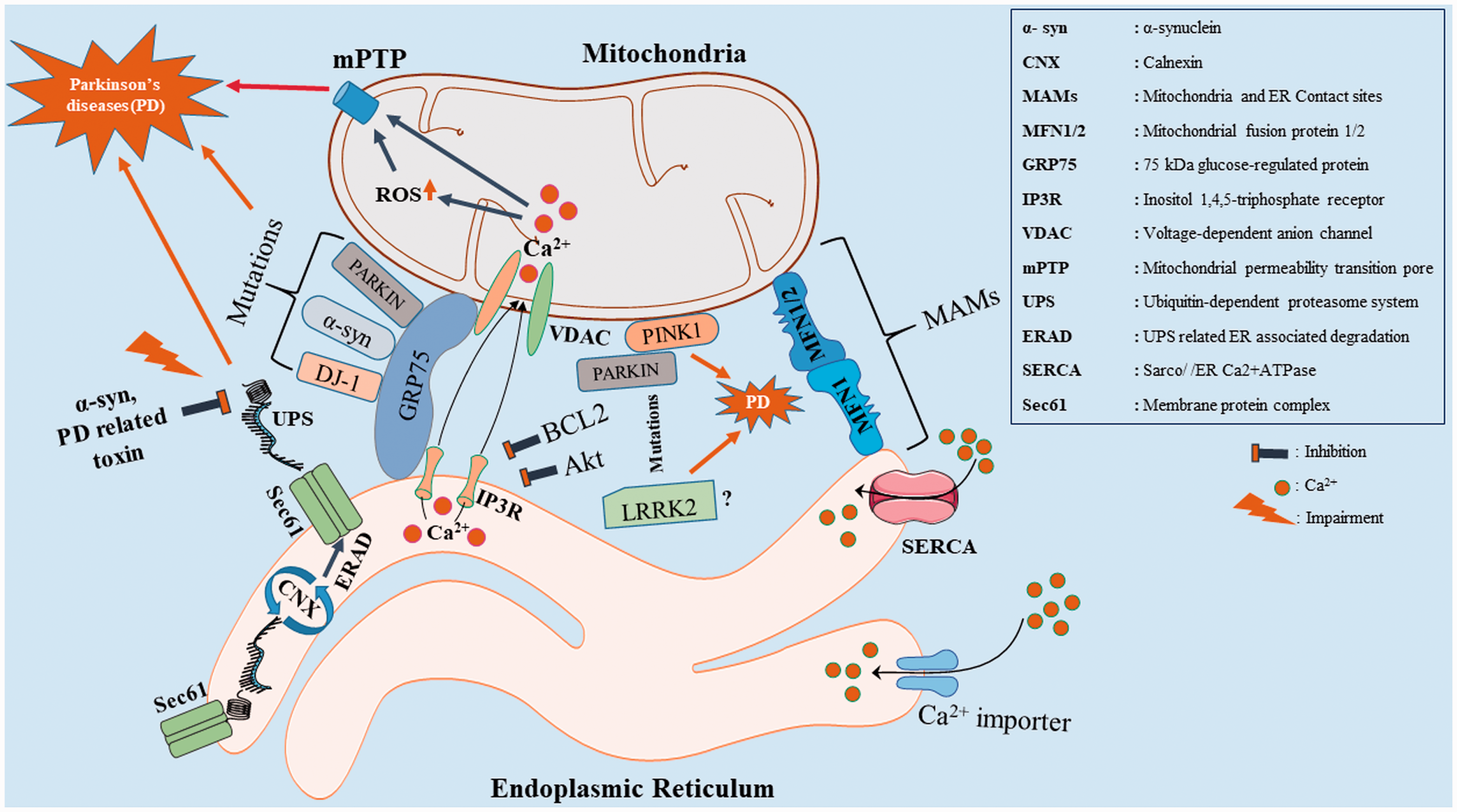
Diagram Shows the Physical Interactions Between the ER and Mitochondria (MAMs) From the Perspective of PD Associated Proteins (α-syn, PINK1, PARKIN, DJ-1, LRRK2). MAMs comprise of molecules like IP3R-GRP75-VDAC, MFN1-MFN1/2. IP3R-Grp75-VDAC complexes which are essential for Ca2+ flux between the ER and mitochondria. MFN 1&2 are involved in mitochondrial fission and fusion and mitophagy. Alterations in DJ-1, α-syn and PARKIN impair IP3R-GRP75-VDAC complex (weakening the MAMs and trigger the Ca2+ induced mitochondrial permeability transition pore (mPTP) dependent apoptosis) and lead to dopaminergic cell death. PINK1 recruits PARKIN in mitophagy process and their mutations alter the mitochondrial fission - fusion. LRRK2 (? – denotes that the exact function at MAMs is unclear), a mitochondrial specific protein associated with PD is also reported to be localized in MAMs. Furthermore, the ubiquitin-dependent proteasome system (UPS) is involved in the clearance of unfolded protein (proper folding of protein is confirmed through calnexin (CNX) folding cycle) degradation. Alterations in the UPS related ER associated degradation (ERAD) pathway cause α-syn accumulation which in implicated in PD.
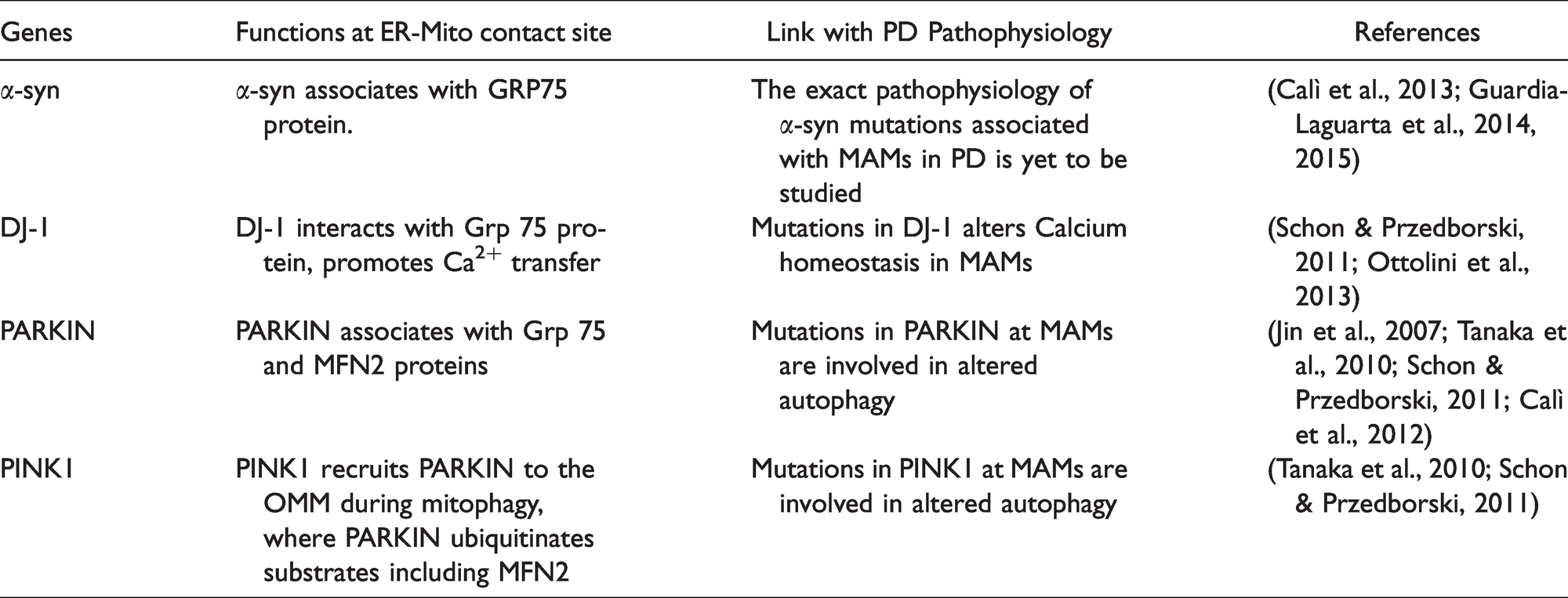
List of PD Related Genes Associated With MAMs.
Lysosomal Dysfunction in PD
The mitochondria-lysosomal contact sites through autophagosomes play a crucial role in the autophagy, biogenesis of both the organelles and exchanges of lipid, amino acids etc. Autophagy is essential for neuronal survival and serves as an adaptive response to nutrient deprivation (Clarke and Mearow, 2016; Fan et al., 2020). Lysosomes play an important role in clearing the damaged cellular components through autophagy (Li et al., 2019; Bi et al., 2021). Lysosomes contain more than 60 hydrolases which facilitate the removal of damaged cellular components and aggregated proteins (Eriksson et al., 2020). Interestingly, lysosomal proteolytic enzymes decline with ageing (Mazzulli et al., 2016). Mutations in lysosomal proteins - lysosomal type 5 P-type ATPase (ATP13A2) are reported to cause PD (Dehay et al., 2012; Mangone et al., 2020) while glucocerebrosidase (GBA), a lysosomal glycoside hydrolase is shown to be associated with pathology PD. Decrease in another chaperone-mediated autophagy protein and lysosomal-associated membrane protein 2 A (LAMP2) is linked to α-syn accumulations (Alvarez-Erviti et al., 2013). Thus, lysosomal dysfunction and associated impaired autophagy contribute to PD (He et al., 2018). The process of autophagy is regulated by mammalian target of the Rapamycin (mTOR) pathway in eukaryotes (Nicklin et al., 2009; Xi et al., 2019). The importance of attenuation of PI3K-Akt-mTOR activation has been well established in experimental models of PD (Inoki et al., 2005; Elstner et al., 2011; Heras-Sandoval et al., 2014; Xu et al., 2014). Rodríguez-Blanco et al. (2012) reported that MPP+ induces inhibition of the Akt/mTOR pathway due to increase in accumulation of ROS (Rodríguez-Blanco et al., 2012). Ablation of PTEN, a negative regulator of Akt (upstream) activates mTOR signalling and is neuroprotective in mouse models of PD (Domanskyi et al., 2011). Although the exact mechanisms involved in the pathogenesis and etiology of PD remain unknown, accumulating evidence suggests a major role of mTOR signalling (Bockaert and Marin, 2015; Dijkstra et al., 2015).
Lysosome–Mitochondria Contact Sites and PD
Mitochondria-lysosome contact sites can regulate mitochondrial dynamics by promoting mitochondrial fission and inter-mitochondrial contact site untethering (Wong et al., 2018, 2019). In yeast, mitochondria and vacuole (yeast version of lysosomes) develop a physical communication called “vacuole and mitochondrial patch” (vCLAMP) consisting of YPT7, LAM6, VPS39 proteins and an unknown mitochondrial component (Elbaz-Alon et al., 2014; Hönscher et al., 2014; Soto-Heredero et al., 2017). YPT7-VPS39 binds with a subunit of the translocase of outer membrane (TOM) of mitochondria (González Montoro et al., 2018). In mammalian cells, mitochondria-lysosome contact sites form with ∼ 15% of lysosomes in contact with mitochondria at any given time point (Wong et al., 2018). In addition, release of Ca2+ from lysosomes to mitochondria at these contact sites also play an important role in regulating mitochondrial functions (W. Peng et al., 2020). Efflux of Ca2+ from the lysosomes is triggered through TRPML1, transient receptor potential mucolipin (TRPML) subfamily, which is activated by an increase in ROS levels (Zhang et al., 2016). A decline in TRPML1 causes accumulation of ROS, which results in fragmented mitochondria and loss of mitochondrial membrane potential (Coblentz et al., 2014). Ca2+ release from the lysosome activates calcineurin, a Ca2+ dependent phosphatase which stimulates the transcription factor EB (TFEB) and initiates the transcription of PGC1α (Peroxisome proliferator-activated receptor gamma coactivator 1-alpha) and PPARα (peroxisome proliferator-activated receptor α) genes. Thus, Ca2+ release from lysosomes controls mitochondrial biogenesis (Mansueto et al., 2017; Todkar et al., 2017). However, excessive ROS levels may cause lysosomal dysfunction and autophagic failure and cell death (Li et al., 2015; Zhang et al., 2016).
MCOLN1calcineurin-dependent TFEB activation clears α-syn through initiation of autophagy (Ivankovic et al., 2016; Zhuang et al., 2020). Dephosphorylated form of TFEB translocates to the nucleus and regulates gene expressions for various genes (Napolitano and Ballabio, 2016). Under starvation, mTORC1 dephosphorylates and translocate TFEB to nucleus and facilitates the autophagy process (Martina et al., 2012).
In stress conditions, PARKIN and PINK1 facilitate the formation of mitochondrial-derived vesicles (MDVs) and these vesicles eliminate defective proteins and lipids from mitochondria via autophagy (McLelland et al., 2014; Matheoud et al., 2016; Jian et al., 2018). However, there is no evidence for these occurring at mitochondria-lysosome contact sites. Interestingly, a small fraction of MDVs is transported to peroxisomes (Sugiura et al., 2014), while the majority is transferred to lysosomes for degradation. Mitochondrial dysfunction causes a decrease in the formation of MDVs resulting in the accumulation of misfolded proteins. Mutations in PINK1 and PARKIN are directly linked to decrease in the formation of MDVs (Vincow et al., 2013; McLelland et al., 2014). In addition, vacuolar sorting protein 35 (VPS35) has a role in MDVs formation.
Acute mitochondrial stress induces lysosomal biogenesis whilst chronic mito-stress inhibits lysosomal biogenesis (Fernández-Mosquera et al., 2017). Defects in lysosomes disrupt the autophagy-lysosomal pathway (Plotegher and Duchen, 2017; Todkar et al., 2017) and impairs the autophagic clearance of misfolded proteins. This contributes to lysosomal dysfunction(Koike et al., 2008; Xie et al., 2015). Dysfunction of the lysosomal and mitochondrial contact sites impairs autophagy and triggers the accumulation of α-syn leading to PD (Decressac et al., 2013; Moors et al., 2017). Silencing of a mitochondrial localized (Junn et al., 2009) PD related gene, DJ-1 is found to be associated with the accumulation of autophagy markers in M17 neuroblastoma cell lines (Thomas et al., 2011). In support of the above-mentioned data, mutations in PARKIN gene (PARK2) are also reported to cause mitochondrial (Pacelli et al., 2011) and lysosomal (Guerra et al., 2019) dysfunction. To add, alterations in lysosomal (TFEB) and mitochondrial (PGC1α) transcription factors i.e impairment of inter-organelle communication influences PD progression advertently (Smolders and Van Broeckhoven, 2020). Mutations in VPS35 is shown to impair vacuolar transport. This is observed in PD (Vilariño-Güell et al., 2011; Zhou et al., 2017). Another protein, VPS13C (a human homolog of VPS13) encodes a member of the vacuolar protein sorting-associated 13 gene family. Mutations in VPS13 in yeast are associated with vCLAMP tethering between lysosome-mitochondria (Lang et al., 2015; Park et al., 2016; Peter et al., 2017) and triggers the mitochondrial dysfunction associated with PD (Lesage et al., 2016). RAB7 is a GTPase and is the key modulator of lysosomal maturation, positioning and network dynamics (Zhen and Stenmark, 2015). RAB7 also regulates the mitochondrial–lysosome tethering and untethering (Stroupe, 2018; Wong et al., 2018). An important PD related gene, LRRK2 knockout has been reported to deplete RAB7 (Dodson et al., 2012; Esteves et al., 2015), and cause aggregation of α‐syn (Dinter et al., 2016). Interestingly, increased levels of LRRK2 are also observed in larger granular structures along with RAB7 in the post-mortem brains of patients with Lewy body disease (Higashi et al., 2009). Deficiency of the mitochondrial specific protein PINK1 causes extension of vacuolar chamber and lysosomal dysfunction which impairs mitophagy and is a risk factor in PD (Demers-Lamarche et al., 2016). Altered organelle proteins at mitochondria-lysosomal contacts sites lead to impairment of autophagy/mitophagy and redox signalling, which are potential targets in PD drug discovery (Figure 2 and Table 2).
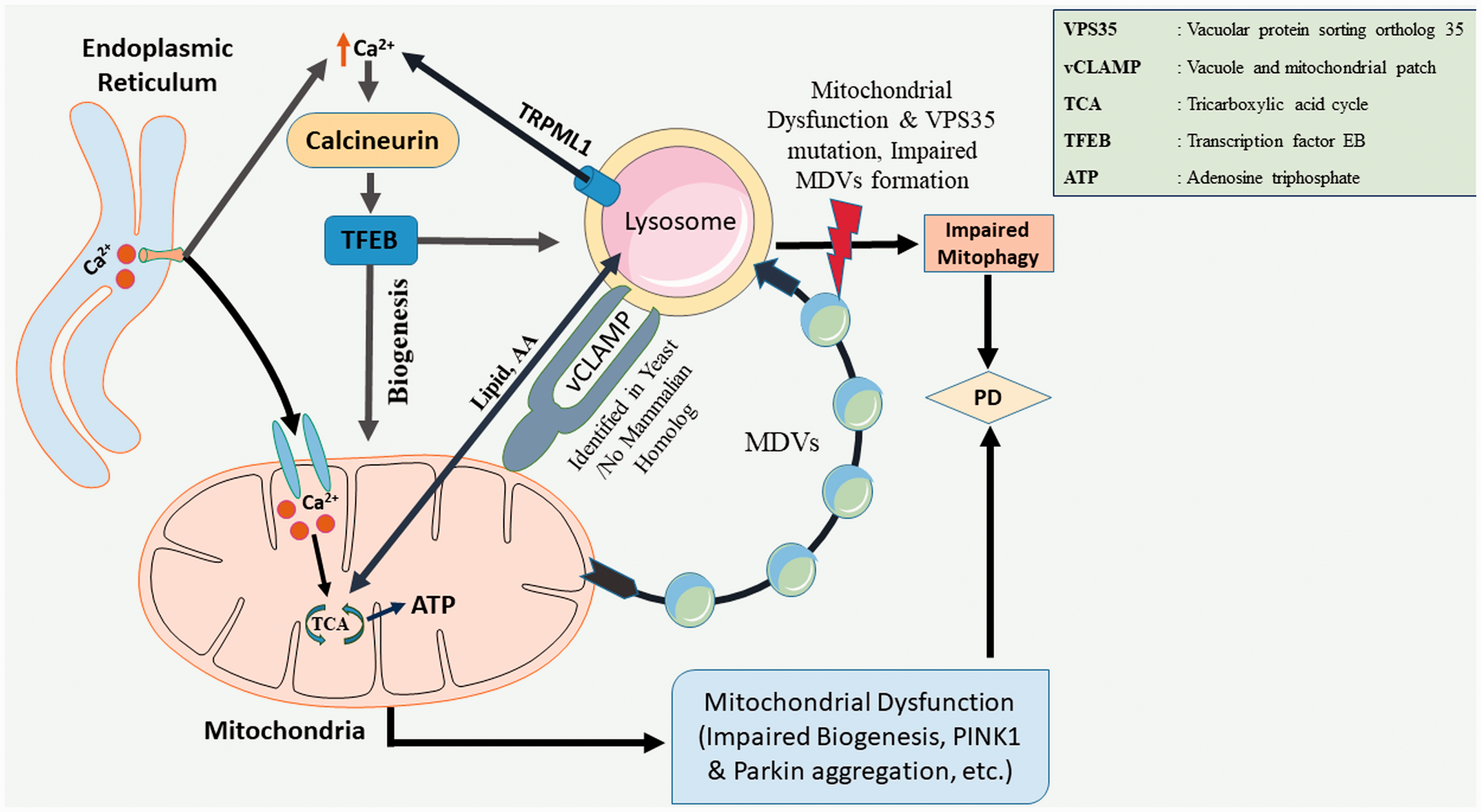
Lysosome-Mitochondria Connection in PD. In yeast, mitochondria and lysosome establish a physical connection through a vacuole and mitochondria patch (vCLAMP), no mammalian homolog has been identified yet. This connection facilitates the exchange of lipids and nutrients between the organelles. TRPML1, a lysosomal calcium channel, acts as a ROS sensor releases Ca2+ in cytosol and activates Calcineurin/TFEB signalling cascade, and initiates autophagy. Mitochondrial-derived vesicles (MDV) remove damaged proteins and lipids from the intact mitochondria. Vacuolar sorting protein 35 (VPS35) plays a key role in MDVs formation. Impaired autophagy and mutations in VPS35 are associated with PD pathogenesis.
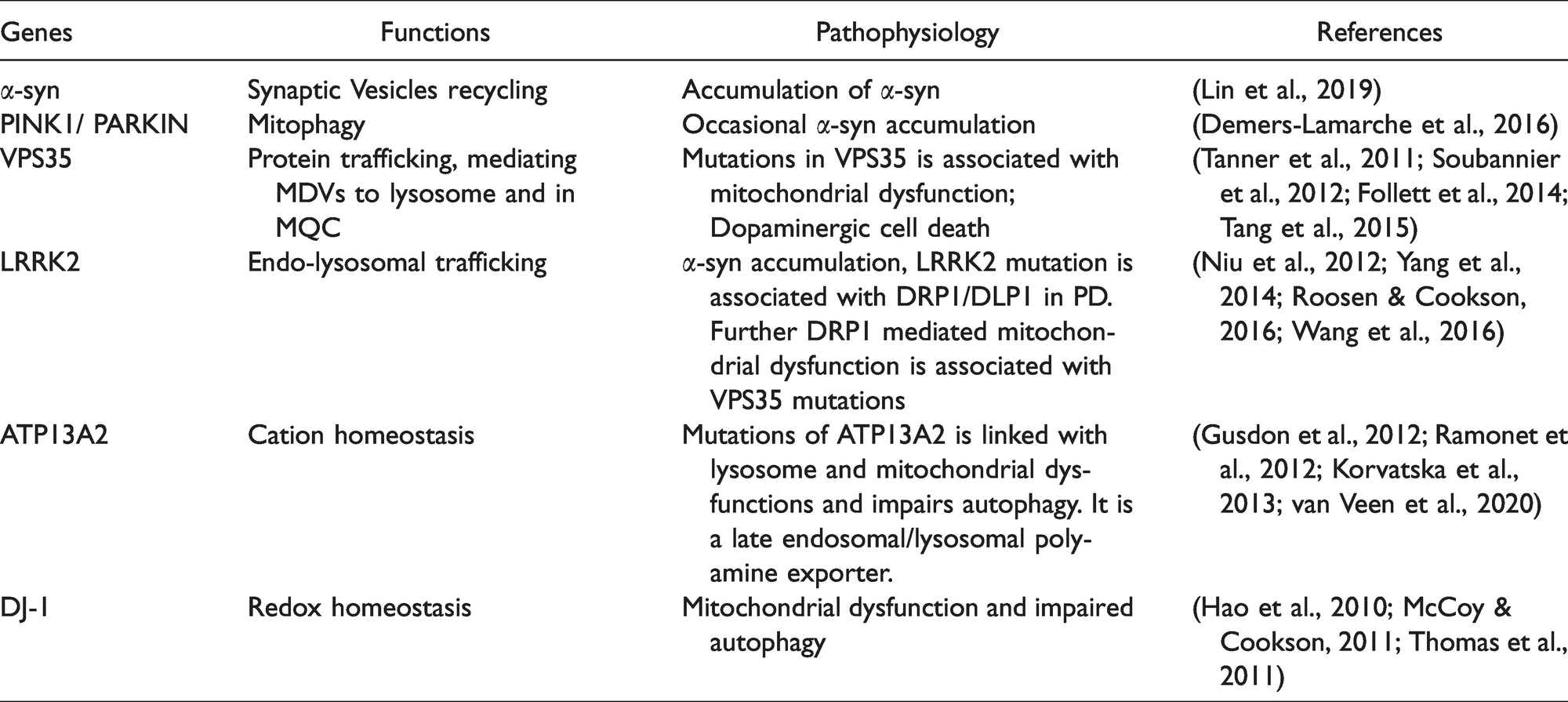
Mitochondria and Endo-Lysosomal Genes Associated PD.
Peroxisome Dysfunction in PD
Peroxisomes play important role in several biochemical pathways like fatty acid (FA) oxidation, glyoxylate metabolism, bile acid and docosahexaenoic acid synthesis, catabolism of amino acids, polyamines and purines, and synthesis of plasmalogens and cholesterol (Fransen et al., 2012; Waterham et al., 2016; Cipolla and Lodhi, 2017). Peroxisomes consume about 20% of the total cellular oxygen (Legakis et al., 2002) and account for 35% of total H2O2 generation in mammalian tissues (Boveris et al., 1972). Thus they were originally named based on their role in hydrogen peroxide production and catabolism (Fransen et al., 2012).
Peroxisomal biogenesis factor- 5 (PEX5), a redox-sensitive protein imports the ataxia-telangiectasia mutated (ATM), a phosphatidylinositol-3 kinase-like protein kinase which inhibits mTORC1, thereby peroxisomes participates in autophagy process (Zhang et al., 2013, 2015). Null mutations in either Peroxisomal Biogenesis Factor 2, 5 or 13 (PEX2, PEX5 or PEX13) genes are shown to increase the accumulation of α-syn, which in turn impairs pexophagy (Yakunin et al., 2010). Plasmalogens, a class of glycerophospholipids are an integral part of the cellular membrane (Lessig and Fuchs, 2009). Peroxisomes are involved in critical steps of plasmalogen biosynthesis. Decreased plasmalogens levels are recorded in the blood and brains of PD patients (Miville-Godbout et al., 2016, 2017). However, the pathogenic mechanism behind the decrease in plasmalogens levels in PD is still not clear. Furthermore, dysfunction of peroxisomes is a risk factor in PD (Yakunin et al., 2010). None-the-less, it is evident that peroxisomes play a crucial role in redox homeostasis and pexophagy.
Peroxisome–Mitochondria Contact Sites and PD
Mitochondria and peroxisomes share similarities in common biochemical pathways such as fatty acid β-oxidation, ROS homeostasis and clearance of defective organelles(Pascual-Ahuir et al., 2017). The functional and physical interactions of peroxisomes with mitochondria was first reported in yeast (Ušaj et al., 2015). In yeast, peroxisomes localize near sites where mitochondria and ER contact each other (Costello et al., 2017; Ušaj et al., 2015). A recent report demonstrated the potential physical tether between peroxisomes and mitochondria. This tethering involves contact between PEX11 protein (which involve in peroxisomes biogenesis) and the mitochondrial MDM34 protein (Ušaj et al., 2015). Subsequently, MDM34 is a crucial structural constituent of the mitochondria-ER tether of ERMES (Kornmann et al., 2009). Fan et al. (2016) demonstrated the existence of peroxisomal-mitochondrial contact sites in mammalian cells (Fan et al., 2016). The contact sites facilitate the passage of ions and lipids (Cohen et al., 2014). Shai et al. (2018) reported that the mitochondria and peroxisomal contact sites are strengthened with two tethering proteins, FZO1 (the yeast mitofusin protein and a homolog of the human MFN1 and MFN2) (De Vecchis et al., 2017) and PEX34 (a peroxisomal membrane protein, whose molecular function is unclear) (Shai et al., 2018). Mitochondria and peroxisomes share the same fission proteins DRP1 and FIS1 (Schrader, 2006; Camões et al., 2009; Delille et al., 2009). Supporting the mitochondria and peroxisomal cross-talk, it was observed that ablation of peroxisomal biogenesis genes PEX3 or PEX5 triggers mitochondria-dependent apoptosis (Tanaka et al., 2019). Mao et al. (2014) reported that mitochondria and peroxisomes also share the same autophagy proteins Atg11 scaffold and the DRP1/DNM1-containing fission complex in selective autophagy (mitophagy and pexophagy). It is also reported that pexophagy process is associated with mitochondria-peroxisome contact sites (Mao et al., 2014; Zhong et al., 2016). Alterations in peroxisome biogenesis (Yakunin et al., 2010), pexophagy (Yakunin et al., 2010; Eun et al., 2018; Wang et al., 2020) and mitophagy(Vives-Bauza and Przedborski, 2011) are linked to the abnormal accumulations of α-syn in brain. Furthermore, recent investigations have demonstrated the importance of peroxisome in cancer, ageing and various other neurodegenerative diseases (Fransen et al., 2013; Islinger et al., 2018).
In yeast, the ERMES complex strengthens the contact site and facilitates the exchange of ions/proteins between mitochondria and ER in yeast. On the other hand, peroxin-11 on binding to the ERMES complex at MDM34 facilitates the transfer of information transfer between mitochondria and peroxisomes (Ušaj et al., 2015). The vCLAMP has also been reported to support phospholipid, iron and Ca2+ exchange at the ERMES (Elbaz-Alon et al., 2014). Dysfunctions in the communications or the contact sites of the involved organelles are reported in various neurodegenerative diseases including PD and involve several pathological pathways. Connecting the evidence, peroxisomal impairment in lipid and ROS metabolism impacts the mitochondrial redox homeostasis (Lismont et al., 2015). It is reported that excessive ROS production due to peroxisomal impairment leads to mitochondria-mediated cell death (López-Erauskin et al., 2012). Ganglioside-induced differentiation-associated protein 1 (GDAP1) regulates the mitochondrial network. While the mutation of GDAP1 impairs the mitochondrial fragmentations in neuroblastoma N1E-115 cells(Niemann et al., 2005), the absence of GDAP1 causes the elongation of peroxisomes(Huber et al., 2013). Mitochondrial and peroxisomal dysfunctions are linked to PD pathogenesis (Subramaniam and Chesselet, 2013). In addition to the MDVs transport to lysosome, vesicular transport is also reported in peroxisomes (Sugiura et al., 2014). However, the reason for the MDVs delivery to peroxisomes is unclear (Mohanty and McBride, 2013). At present, a membrane-anchored protein ligase called MUL1/MAPL (mitochondria-anchored protein ligase) is reported to be delivered to the peroxisomes (Neuspiel et al., 2008; Braschi et al., 2009). The retromer complex containing VPS35, VPS26 and VPS29 is identified as a MAPL binding partner using affinity chromatography approach (Braschi et al., 2010). It was reported that VPS35 participates in various cellular transport pathways and its defect have been linked to mitochondrial dysfunction and PD (Sugiura et al., 2014). Deletion of VPS35 gene in SH-SY5Y and NLT cell lines causes intracellular accumulation of α-syn (Tang et al., 2015). A separate report, in a Chinese cohort study, showed that the gene MUL1 is a risk factor for PD (Taximaimaiti and Li, 2019). Another protein, MIRO1, a Rho GTPase gets localized both in the peroxisomes and mitochondria in mammals (Castro et al., 2018; Okumoto et al., 2018; Farré et al., 2019). MIRO1 modulates the dynamics of both the organelles (Covill-Cooke et al., 2020). Here it is noteworthy that Gem1, a yeast orthologue of MIRO is an important regulator of mitochondrial and peroxisomal contact sites (Shai et al., 2018). In PD patients, α-syn accumulation in the brain is correlated with the upregulation of MIRO (Shaltouki et al., 2018). Very recently, Guillén-Samander et al. (2021) reported that VPS13D, an essential gene involved in autophagy/mitophagy process (Anding et al., 2018) establishes a bridge connecting ER, Mitochondria and peroxisome by MIRO, which is a PARKIN substrate directly associated with PD (Guillén-Samander et al., 2021). Although, indirectly there is substantial literature that provides evidence on the disturbances in redox homeostasis and autophagy processes and their link to PD, to the best of our knowledge, as of today, there is no data available that directly links the mitochondrial and peroxisomal contact sites dysfunctions to PD pathogenesis. Thus, mitochondria-peroxisome contact sites (Figures 3 and 4) provide novel research opportunities in relation to PD pathogenesis and therapeutics.
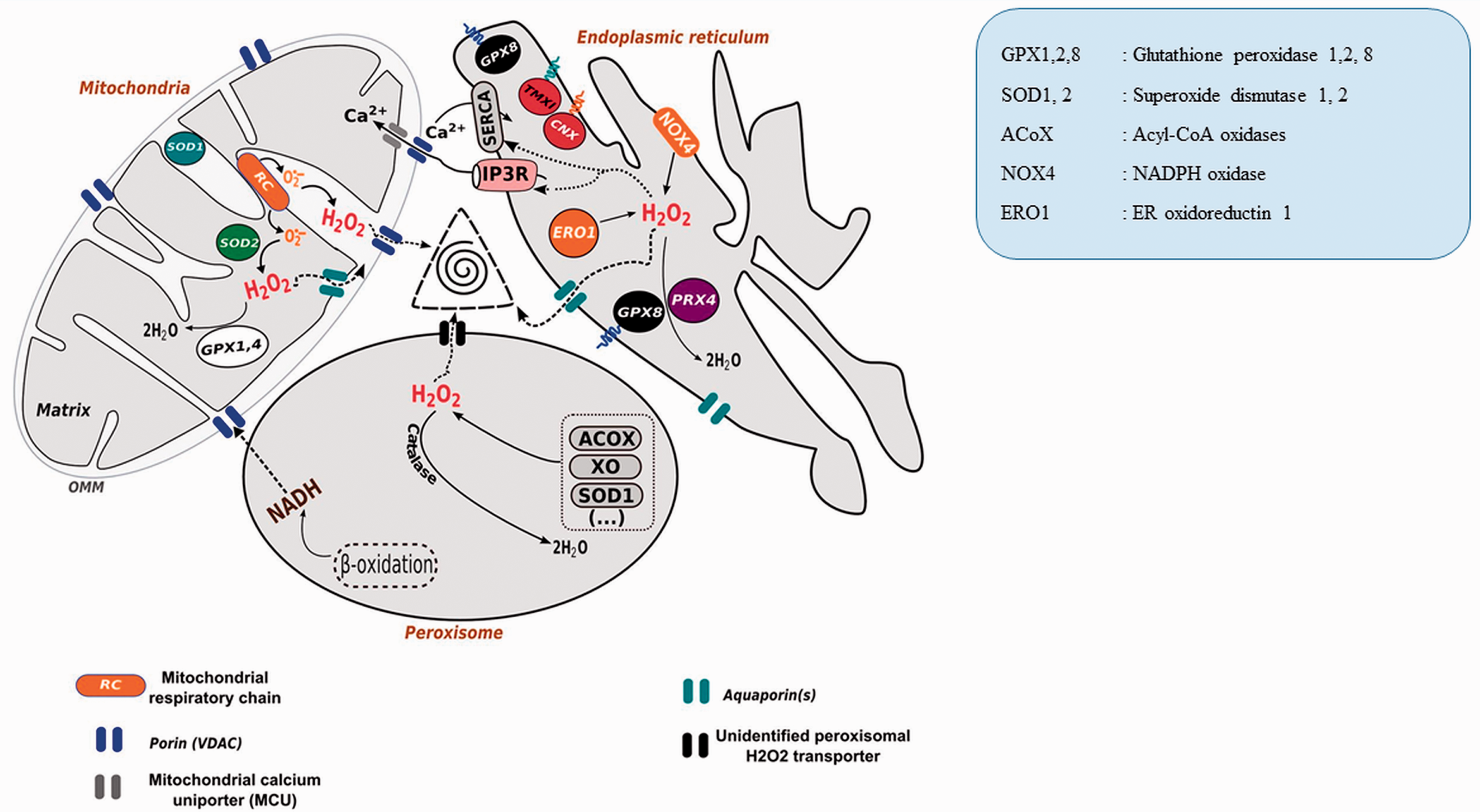
Redox Triangle (ER-Mitochondria-Peroxisome). ER, mitochondria, and peroxisome, generate ROS in their biochemical reactions. All the three organelles releases ROS through porin (ER), aquaporins (lysosome), unknown channel (Peroxisome) and form redox triangle causing oxidative damage of the associated organelles (Yoboue et al., 2018) (Open access article, reused the image as per terms of the Creative Commons CC BY license).
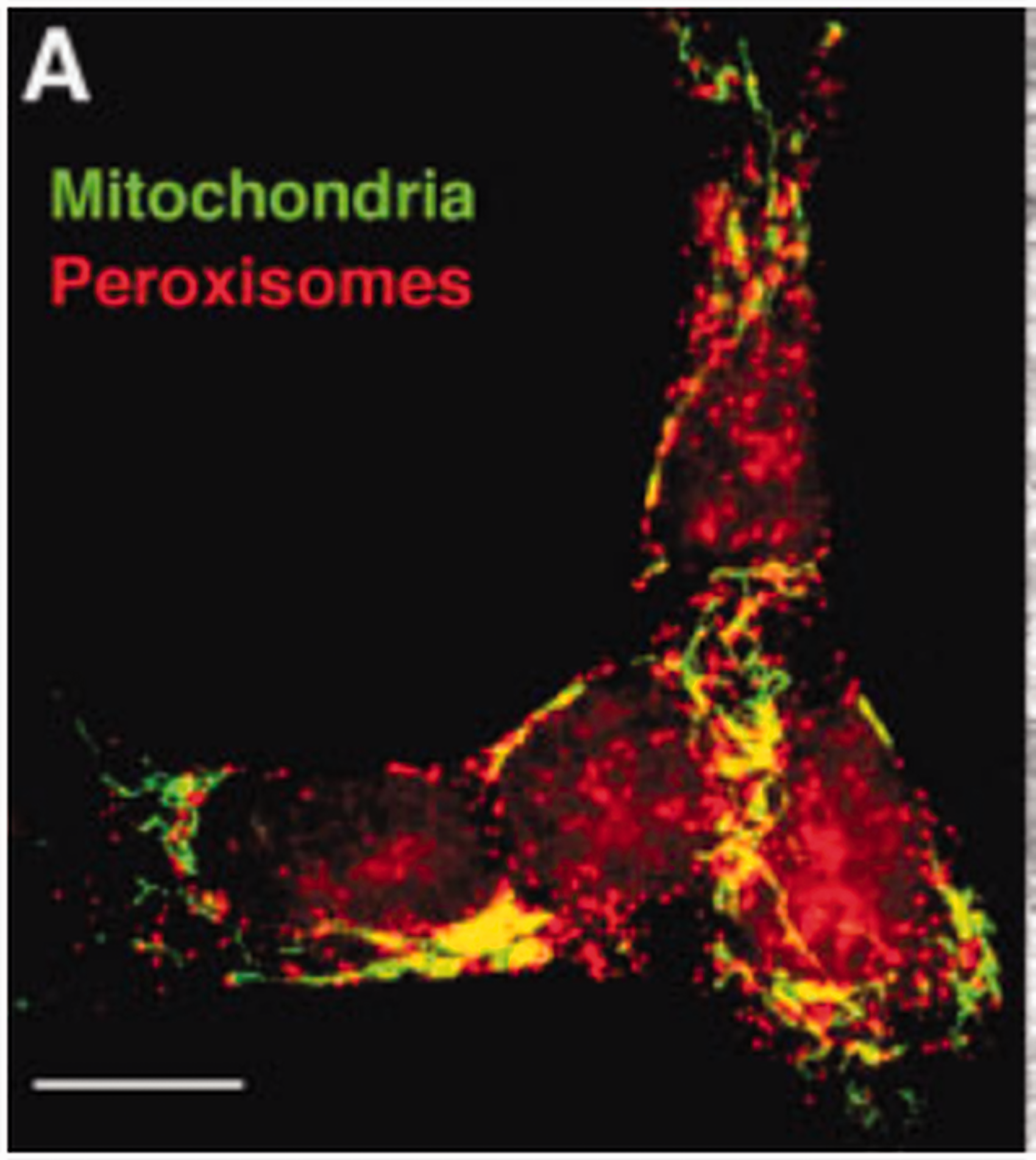
Mitochondria and Peroxisomal Contact Sites. A. Fluorescence microscope image showing close contact between peroxisome and mitochondrial in neuroblastoma cell line. The cells were stained with peroxisomal membrane protein (PMP70) that emitted red fluorescence and mitochondrial MnSOD, a mitochondrial antioxidant, emitted green fluorescence, that established the existence of contact sites in neuroblastoma cells captured by Fluorescence microscopy (10 μm). (Reused as per the BioEssays journal's copyright permission policy) (Schrader and Yoon, 2007).
PD and Organelle Specific Targeted Drug Delivery Strategies
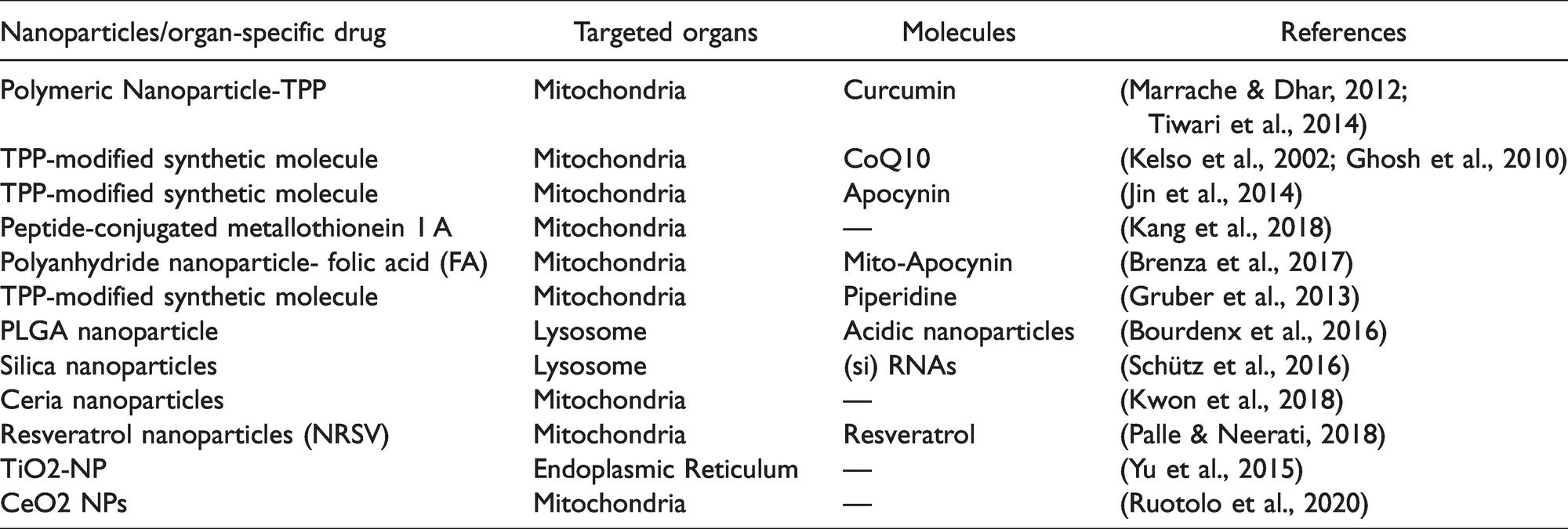
Current treatments in PD are predominantly symptomatic and palliative care approaches. It is unfortunate that none of the therapeutic approaches have proven to be completely effective in preventing or slowing down the progression of PD. The prime obstacle that confronts the brain-targeted therapies is crossing the blood-brain barrier (Torres-Ortega et al., 2019). Towards this end, nano-drug delivery systems are being considered as potential options as they provide high bioavailability, have reduced adverse reactions and specifically target the desired tissues or organelles ( Chidambaram et al., 2020; Patra et al., 2018; Y. Peng et al., 2020 ). Functional groups can be attached to the surface of the nanoparticles (NPs) and this can help in reaching the desired target organelle. For example, mitochondria-specific delivery can be achieved by surface modification with triphenyl phosphonium (TPP) cations (Boddapati et al., 2005). Furthermore, targeted delivery approaches have been explored to target specific organelles like mitochondria, lysosome etc. Wittrup et al. (2010) studied antibody-conjugated magnetic nanoparticles to understand the role of GRP75, an important protein of MAMs, in endocytosis of macromolecules (Wittrup et al., 2010). Likewise, Au-functionalized siRNA particles were used to knock down GRP75 in PC-3 cells (Shah et al., 2020). Few other studies have been discussed in (Table 3). Future research in this area may lead to the development of specific organelle and mitochondrial membrane contact site delivery, especially for neurodegenerative diseases.
Organelle Specific Nano Drug Delivery in PD.
Conclusion
The communication between mitochondria and ER, lysosomes and peroxisomes is important for physiological homeostasis. However, not much research has been done in this area specifically with reference to PD pathology. Mitochondria are connected to ER, lysosomes, and peroxisomes bi-directionally at mitochondria contact site (MCS). In pathological conditions, alterations in Ca2+ homeostasis, impaired autophagy, redox imbalance, MDVs transport are observed at MCS. Interestingly, data suggests that the effects of structural and biochemical alterations at MAMs (mitochondria-ER contact site) are more prominent in PD pathology in comparison to alterations at mitochondria-lysosome and mitochondria-peroxisome sites. Derangements at MAMs impairs Ca2+ signalling which causes dopaminergic cell death. Also, mutations in PD associated genes such as α-syn, DJ-1, PARKIN are linked to IP3R-GRP75-VDAC complex dysfunction at MAMs. In addition, mutation in PINK and PARKIN impairs MFN1-MFN1/2 tethering at MAMs, consequently affecting autophagy and mitochondrial fission-fusion processes. Similarly, mitochondria-lysosomal contact sites regulate mitochondrial biogenesis and autophagy through Calcineurin/TFEB signalling and are crucial for clearing the damaged mitochondrial proteins by the MDVs transport mechanism. Dysfunction of this contact site and mutation in VPS35 are associated with PD pathogenesis. Although there is no direct evidence to show the involvement of derangement at mitochondria - peroxisome site in PD, alterations in proteins like VPS35, Mortalin/HSPA9, and sharing autophagy protein, Atg11 scaffold and DRP1/DNM1 impose deleterious effects and affect cellular homeostasis negatively. There are few investigations that have focused on the organ-specific nano delivery of molecule/genes to treat or understand the diseased condition. This review gives a glimpse of the role of mitochondrial contact sites in PD and establishes the role of mitochondria and organellar crosstalk in Parkinson’s disease. Further research may help to understand the role of mitochondrial contact sites in PD (Figure 5).
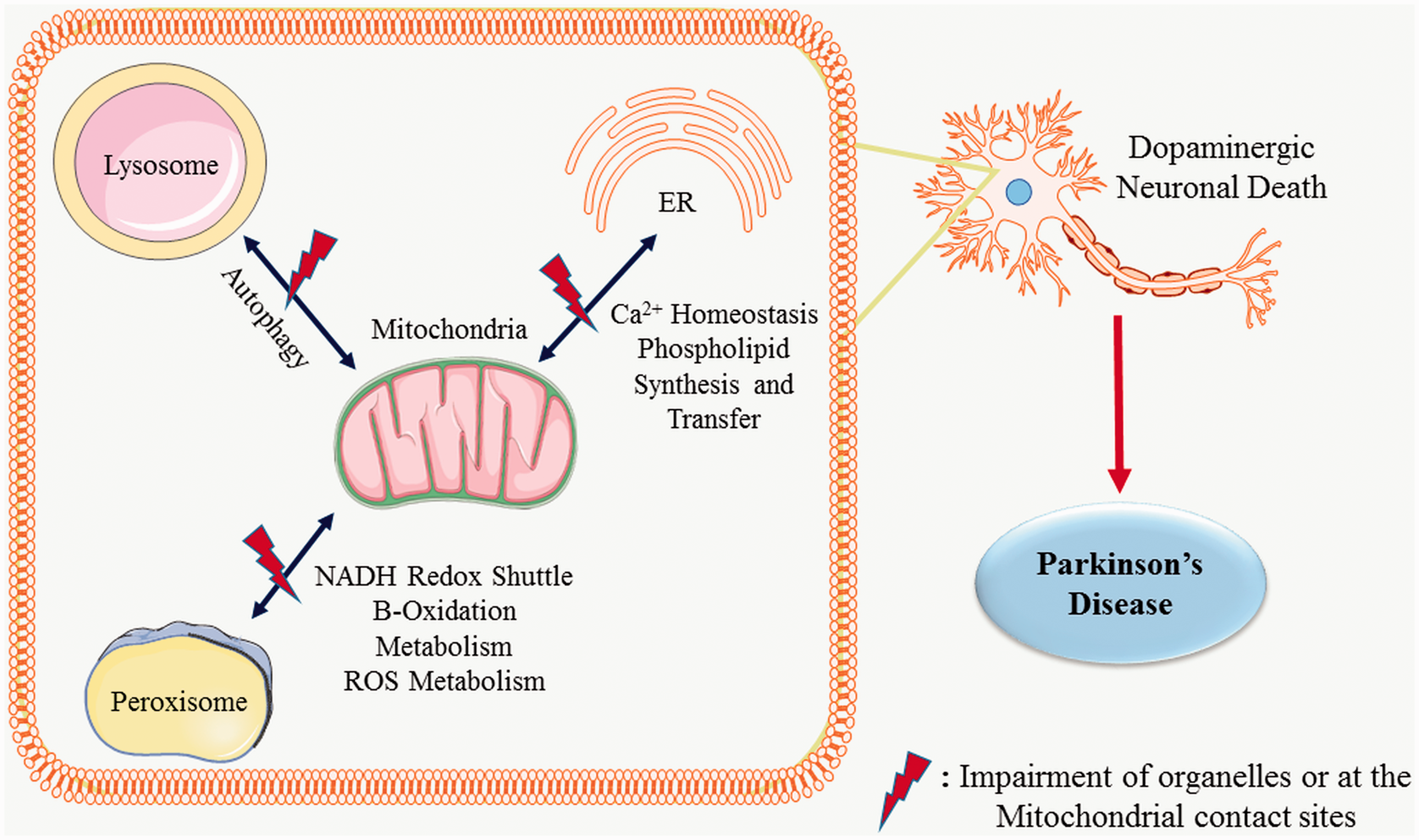
Individual Organelle Dysfunction and Altered Communication at Mitochondrial Contact Sites Between Lysosome, ER, and Peroxisome in Terms of Autophagy and Redox Homeostasis Lead to Dopaminergic Cell Death in PD.
One key “chicken-and-egg problem” question is PD begins with the dysfunction of these organelles and/or MCS, or vice-versa? Detailed investigations on the mitochondrial crosstalk with these organelles and further studies on the regulation of these events will provide additional knowledge on PD pathogenesis and potential therapeutics for PD.
Footnotes
Acknowledgment
The authors also thank “Edit n Stat,” Chennai, for assistance in English proof reading.
Author Contributions
B. R.: Literature collection, compilation, analysis of data and manuscript drafting, images drawing; A. B.: Literature collection, images drawing; S. T.: Literature collection and compilation; A. M. M.: Literature collection and compilation; M. B.: Literature collection and compilation; S. K. M.: Manuscript draft reading; V. P. V.: Manuscript draft reading; R. C.: Manuscript draft reading; M. M. E.: Manuscript draft reading; S. B. C.: Designing, manuscript draft reading, copy right permission request for images and manuscript correction; M. K. S.: Manuscript draft reading and manuscript correction.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: B. R. acknowledges the Indian Council of Medical Research (ICMR), New Delhi, Govt. of India, for the Senior Research Fellowship. M.K.S acknowledges Discovery Grant (grant number: 417652) from the Natural Sciences and Engineering Research Council of Canada.
