Abstract
Background:
Neoadjuvant therapy provides substantial clinical benefits for patients with locally advanced tongue squamous cell carcinoma (TSCC). It improves the rate of complete tumor resection, decreases recurrence risk, and extends survival. However, accurate post-therapy risk stratification depends on the identification of reliable prognostic biomarkers.
Objectives:
This retrospective study assessed several immune microenvironment biomarkers—tumor-associated tissue eosinophils (TATEs), neutrophils (TANs), lymphocytes (TILs), and tertiary lymphoid structures (TLSs)—in 108 patients with stage III or IV TSCC who received neoadjuvant chemotherapy (n = 44) or immunochemotherapy (n = 64) between 2013 and 2022.
Design:
Retrospective cohort study.
Methods:
Post-treatment hematoxylin and eosin (H&E)-stained specimens were evaluated to quantify biomarker infiltration. Associations between biomarker levels, pathological response, and survival outcomes were analyzed.
Results:
A low stromal TATE (S-TATE) density (⩽20/mm²) was significantly correlated with higher rates of pathological complete response (pCR) (p < 0.001). In contrast, elevated S-TATE levels were associated with lymph node metastasis (p = 0.011) and vascular or neural invasion (p = 0.004). Multivariate analysis identified S-TATE > 20/mm² as an independent predictor of reduced overall survival (HR = 3.52, 95% CI: 1.01–12.32; p = 0.049) and shorter progression-free survival.
Conclusion:
S-TATE serves as an independent prognostic indicator in patients with locally advanced TSCC receiving neoadjuvant therapy. Quantifying S-TATE in post-treatment specimens may help tailor adjuvant therapy intensity and refine surveillance strategies. Patients with elevated S-TATE levels should receive closer follow-up.
Keywords
Introduction
Oral cavity cancer remains a major public health concern in China, with tongue squamous cell carcinoma (TSCC) representing its most aggressive and prevalent subtype, accounting for over half of all oral cavity cancer cases.1–3 Owing to the tongue’s rich vascularity and extensive lymphatic drainage, TSCC is characterized by early lymph node metastasis and a high risk of local recurrence. Despite standard treatment with surgery followed by adjuvant chemoradiotherapy, 4 approximately 50% of patients experience recurrence or distant metastasis within 5 years.5–7 Given the tongue’s critical roles in speech and swallowing, treatment must balance oncologic control with functional preservation.
Neoadjuvant therapy has recently emerged as a promising approach for locally advanced TSCC, offering potential benefits in tumor downstaging, organ preservation, and postoperative recovery.8–10 Moreover, immune checkpoint inhibitors (ICIs) have improved survival in recurrent or metastatic head and neck squamous cell carcinoma (HNSCC). 11 Building on these findings, several trials have shown that neoadjuvant immunochemotherapy can enhance pathological response rates and improve survival in locally advanced TSCC.12–14 However, prognostic biomarkers that accurately predict recurrence or survival after neoadjuvant immunochemotherapy remain lacking.
Tumor-associated tissue eosinophilia (TATE) has been described across multiple malignancies and may reflect dynamic immune–tumor interactions. 15 Prior studies have reported inconsistent results regarding its prognostic role—some linking high TATE density to poor outcomes,16–19 while others suggesting favorable prognostic implications in patients treated with ICIs. 20 These discrepancies highlight the need to clarify the biological and clinical relevance of TATE in the neoadjuvant immunochemotherapy setting.
Therefore, we conducted a retrospective study to evaluate the prognostic significance of TATE in patients with locally advanced TSCC who underwent neoadjuvant therapy and to compare outcomes between immunochemotherapy and chemotherapy subgroups. Furthermore, we developed a simple, clinically applicable prognostic risk-scoring system integrating S-TATE and key clinicopathologic factors to improve individualized risk stratification.
Methods
Patients and treatment
Patients with primary stage III or IV locally advanced tongue squamous cell carcinoma (TSCC) who received neoadjuvant chemotherapy or immunochemotherapy at Sun Yat-sen University Cancer Center between 2013 and 2022 were retrospectively included. In the neoadjuvant chemotherapy group, patients were treated with a regimen containing taxanes, platinum, and fluorouracil administered every 2–3 weeks for 5–6 weeks before surgery. In the neoadjuvant immunochemotherapy group, patients received taxanes, platinum, and ICIs on the same schedule. After completing neoadjuvant therapy, all patients underwent curative-intent surgery with selective lymphadenectomy. Adjuvant chemoradiotherapy was administered when clinically indicated. Patients with a prior history of head and neck squamous cell carcinoma (HNSCC) were excluded (Supplemental Figure 1). Tumor staging followed the Eighth Edition of the American Joint Committee on Cancer (AJCC) tumor–node–metastasis (TNM) classification. This study was approved by the Institutional Review Board of Sun Yat-sen University Cancer Center, and the reporting complied with the STROBE guidelines for cohort studies (Supplemental Table 1). 21
Data collection
Clinical and pathological data were extracted from electronic medical records, including age, sex, body mass index (BMI), smoking status, alcohol consumption, areca nut use, vascular invasion, perineural invasion, and lymph node metastasis. BMI values were obtained from the most recent measurements before the initiation of neoadjuvant therapy.
Pathological response evaluation
All available hematoxylin and eosin (H&E)-stained sections were retrieved from the Department of Pathology and independently reviewed by two experienced pathologists blinded to clinical outcomes. Pathological response was evaluated according to published criteria22,23: (a) Identification of the tumor bed: The tumor bed included both residual viable tumor areas and treatment-related changes such as necrosis, keratinous debris, granulation tissue, fibrosis, and multinucleated giant cell reactions. (b) Quantification of residual viable tumor cells (RVT): The proportion of viable tumor cells relative to the total tumor bed area was estimated for each section, and the mean value was calculated across all sections. Patients were categorized as follows: pathological complete response (pCR): RVT = 0%; partial pathological response (PPR): 0% < RVT ⩽ 50%; and no pathological response (NPR): RVT > 50%.
Histopathological biomarker evaluation
Assessment of TLS
TLSs were examined on all available H&E-stained sections and classified into three maturation stages24,25:(a) early TLS (E-TLS): loose lymphocytic aggregates; (b) primary follicle-like TLS (PFL-TLS): compact oval lymphoid clusters without germinal centers (GCs); and (c) secondary follicle-like TLS (SFL-TLS): mature structures containing GCs with follicular dendritic cell networks and proliferating centroblasts/centrocytes. Each case was categorized into one of three groups based on TLS presence and maturity 26 : (i) TLS-mature: at least one SFL-TLS in any section; (ii) TLS-immature: at least one E-TLS or PFL-TLS, but no SFL-TLS; and (iii) TLS-negative: no TLS of any stage observed.
Evaluation of tumor-associated immune cells (TATE, TAN, and TIL)
To minimize inter- and intra-observer variability, biomarker assessment followed a standardized protocol. Two pathologists independently evaluated TATEs, tumor-associated neutrophils (TANs), and tumor-infiltrating lymphocytes (TILs) on representative H&E-stained slides. For TATEs and TANs, stromal cell density was quantified at the invasive margin (IM) under 400× magnification (Olympus BX53 microscope). Areas with necrosis, ulceration, keratinous debris, or multinucleated giant cell reactions were excluded.27,28 Ten consecutive high-power fields with the maximal density of TATEs or TANs were examined, and results were recorded as cells per mm². 19 Eosinophils were defined as nucleated cells with dense, bright red cytoplasmic granules, excluding erythrocytes overlapped by inflammatory cells. Neutrophils were identified as cells with multilobulated nuclei and pale cytoplasmic granules, excluding nuclear debris or apoptotic remnants. For TILs, density was quantified as the percentage of stromal area occupied by mononuclear inflammatory cells (lymphocytes and plasma cells) at the tumor edge under 200× magnification (Olympus BX53). Necrotic areas, keratinous debris, and pre-existing lymphoid stroma were excluded. The IM was defined as a 1-mm zone centered on the tumor–host interface.29,30
Statistical analysis
The primary endpoint was overall survival (OS), defined as the time from diagnosis to death from any cause or last follow-up. The secondary endpoint was progression-free survival (PFS), defined as the time from surgery to disease progression, death, or last follow-up. Disease progression included newly diagnosed HNSCC, local recurrence, or nodal/distant metastasis confirmed by imaging or biopsy. Survival curves were estimated using the Kaplan–Meier method and compared with the log-rank test. Associations with OS or PFS were examined using Cox proportional hazards regression, and a risk score model was derived from multivariable analysis. Cutoff values for continuous variables were determined using maximally selected rank statistics. 31 Between-group comparisons used the Mann–Whitney U test or χ² test. 32 Associations between pathological response categories, S-TATE levels, and treatment types were analyzed using the χ² or Cochran–Mantel–Haenszel test as appropriate. The Kruskal–Wallis test evaluated differences in TATE density or risk scores among pathological response groups. Additional χ² tests explored correlations between S-TATE levels and pathological parameters (lymph node metastasis, vascular invasion, and perineural invasion). The Spearman rank correlation test assessed the relationship between prognostic risk scores and RVT percentages. All p values were two-sided, with p < 0.05 considered statistically significant. Hazard ratios (HRs) and 95% confidence intervals (CIs) were reported. All analyses were performed using R software (version 4.3.1).
Results
Patient characteristics
Clinical and pathological data were retrospectively collected from patients with locally advanced TSCC who received neoadjuvant chemotherapy (N = 44) or neoadjuvant immunochemotherapy (N = 64) (Supplemental Figure 1). Baseline characteristics are summarized in Table 1. Figures 1 and 2 provide group comparisons and representative histology. Figure 1(a)–(c) shows cutoffs for continuous variables in survival analyses derived from maximally selected statistics. Between-group differences were significant for BMI, smoking history, TILs, lymph node metastasis (LNM), vascular or perineural invasion, and adjuvant therapy. Figure 1(d)–(f) compares S-TATE (stromal TATE), S-TAN (stromal TAN), and TILs; only TILs differed significantly (Mann–Whitney U test, p = 0.018), whereas S-TATE (p = 0.292) and S-TAN (p = 0.237) were evenly distributed. Figure 2(a)–(c) displays H&E-stained images illustrating high/low S-TATE and S-TAN at the tumor edge and varying stromal TIL percentages. Figure 2(d) shows immature and mature TLSs. Figure 2(e)–(g) depicts representative features of pathological responses, including necrosis, histiocytic inflammation, giant cell reaction to keratinous debris, and fibrosis.
The clinicopathological characteristics of patients in the two different treatment groups.

BMI, body mass index; LNM, Lymph node metastasis; S-TAN, Tumor-associated neutrophils in stroma; S-TATE, Tumor-associated tissue eosinophils in stroma; TILs, tumor-infiltrating lymphocytes; TLS, tertiary lymphoid structures.
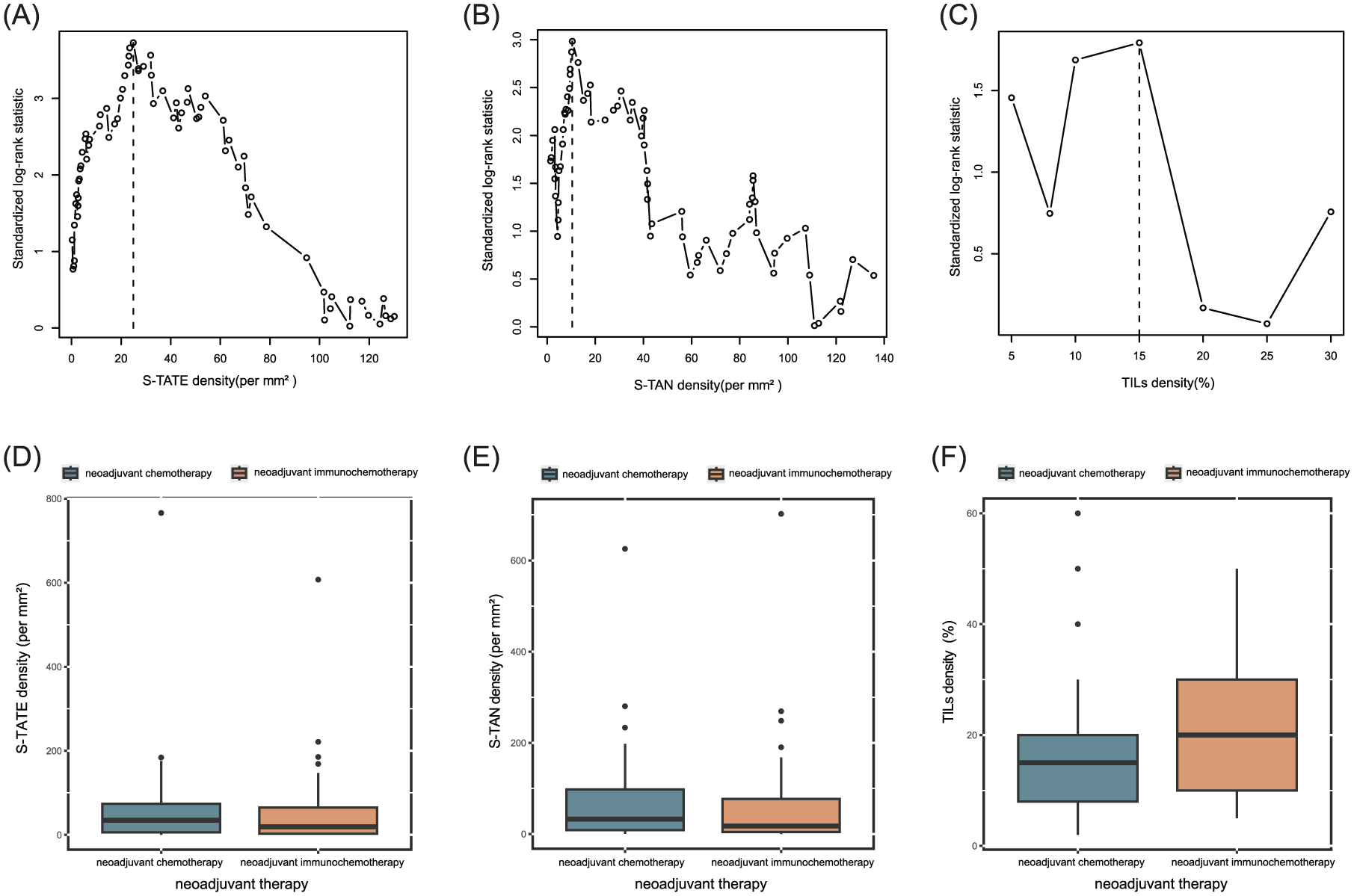
(A-C) Maximally selected log-rank test for OS shows that the optimal stratum for density of S-TATE, S-TAN and TILs. (D-F) the differences of S-TATE, S-TAN and TILs density between neoadjuvant chemotherapy group and neoadjuvant immunochemotherapy group. Only TILs shows the statistical differences (Mann-Whitney U test, P = 0.018 < 0.05), S-TATE (Mann-Whitney U Test: P = 0.292) and S-TAN (Mann-Whitney U Test: P = 0.237) don’t show the statistical differences.

(A) Representative images of different levels of S-TATE (200 × original magnification) in H&E sections of TSCC. (B) Representative images of different levels of S-TAN (200 × original magnification) in H&E sections of TSCC. (C) Representative images of different levels of TILs (200 × original magnification) in H&E sections of TSCC. (D) The respective H &E-stained images of immature TLS group and mature TLS group. (E) Representative H&E pictures of pathological response, which defined as tumor necrosis with associated histiocytic inflammation and/or giant cell reaction to keratinaceous debris. (100 × original magnification) (F) giant cell reaction to keratinaceous debris. (100 × original magnification) (G) Fibrosis, inflammatory cells infiltration, and residual viable tumor. (40 × original magnification).
Low S-TATE predicts higher pCR, and neoadjuvant immunochemotherapy further improves response
Figure 3(a) shows a graded association between S-TATE and pathological response: patients with pCR had lower S-TATE densities than those with PPR or NPR (Kruskal–Wallis, p < 0.001). Figure 3(b) confirms this trend categorically (χ², df = 2, n = 108 = 43.518; p < 0.001). As shown in Figure 3(c), the pCR proportion was higher with neoadjuvant immunochemotherapy than with chemotherapy alone (χ² = 10.34; df = 2; n = 108; p = 0.006). To disentangle treatment and S-TATE effects, a Cochran–Mantel–Haenszel test demonstrated that S-TATE remained an independent factor for response (χ² = 42.239; df = 2; p < 0.001).

(A) BOX plot of pathological response grades by density of S-TATE (Kruskal-Wallis test: P < 0.0001). (B) Proportion of patients with NPR, PPR or pCR under the stratification of S-TATE level (χ² (df = 2, n = 108) = 43.518, P < 0.001). (C) Proportion of patients with NPR, PPR or pCR under the stratification of neoadjuvant therapy (χ² (df = 2, n = 108) = 10.34, P = 0.006 < 0.05).
High S-TATE is linked to LNM and vascular/perineural invasion
We next examined whether S-TATE correlated with LNM. As shown in Figure 4(a), LNM was more frequent in tumors with high S-TATE (χ2, df = 1, n = 108 = 6.531; p = 0.011). Figure 4(b) shows that high S-TATE is also associated with vascular or perineural invasion (χ2 = 8.359; df = 1; n = 108; p = 0.004). These findings suggest that elevated S-TATE may promote nodal spread and invasive behavior.
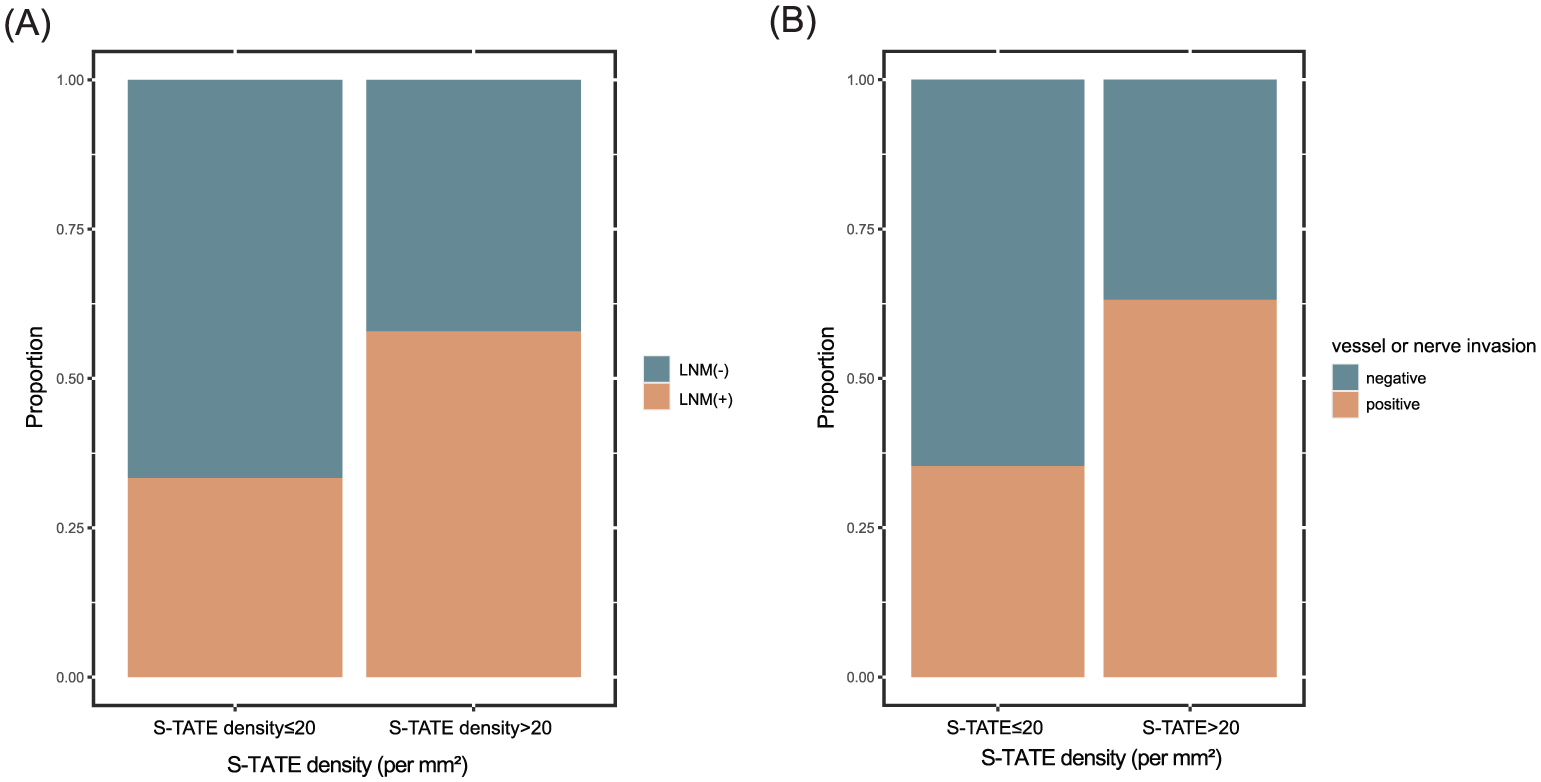
(A) Proportion of patients with or without LNM under the stratification of S-TATE levels (χ² (df = 1, n = 108) = 6.531, P = 0.011 < 0.05). (B) Proportion of patients with or without vessel or nerve invasion under the stratification of S-TATE level (χ² (1, n = 108) = 8.359, P = 0.004 < 0.05).
High S-TATE predicts worse survival and higher risk of progression after surgery
Kaplan–Meier analyses (Figure 5(a) and (b)) show that patients with low S-TATE had better OS and PFS than those with high S-TATE (log-rank p < 0.05). The survival advantage of low S-TATE persisted within both treatment strata (Figure 5(c) and (d); log-rank p < 0.05). On univariable Cox analysis, predictors of OS included BMI (HR 0.084; 95% CI 0.011–0.62; p = 0.015), S-TATE (HR 4.10; 95% CI 1.50–11.0; p = 0.005), S-TAN (HR 3.90; 95% CI 1.40–11.0; p = 0.012), differentiation (moderate grade: HR 2.317; 95% CI 0.526–10.214; p = 0.267; high grade: HR 7.558; 95% CI 1.635–34.944; p = 0.010), LNM (HR 5.50; 95% CI 2.10–15.0; p < 0.001), and neoadjuvant therapy (HR 0.32; 95% CI 0.13–0.78; p = 0.012; Table 2). Multivariable Cox analysis (including variables meeting proportional hazards assumptions) identified neoadjuvant therapy (HR 0.3323; 95% CI 0.134–0.822; p = 0.017), tumor differentiation (HR 2.6867; 95% CI 1.119–6.450; p = 0.027), S-TATE (HR 3.5219; 95% CI 1.006–12.324; p = 0.049), and BMI (HR 0.1138; 95% CI 0.015–0.880; p = 0.037) as independent predictors of death. Figure 5(e) summarizes the multivariable results.
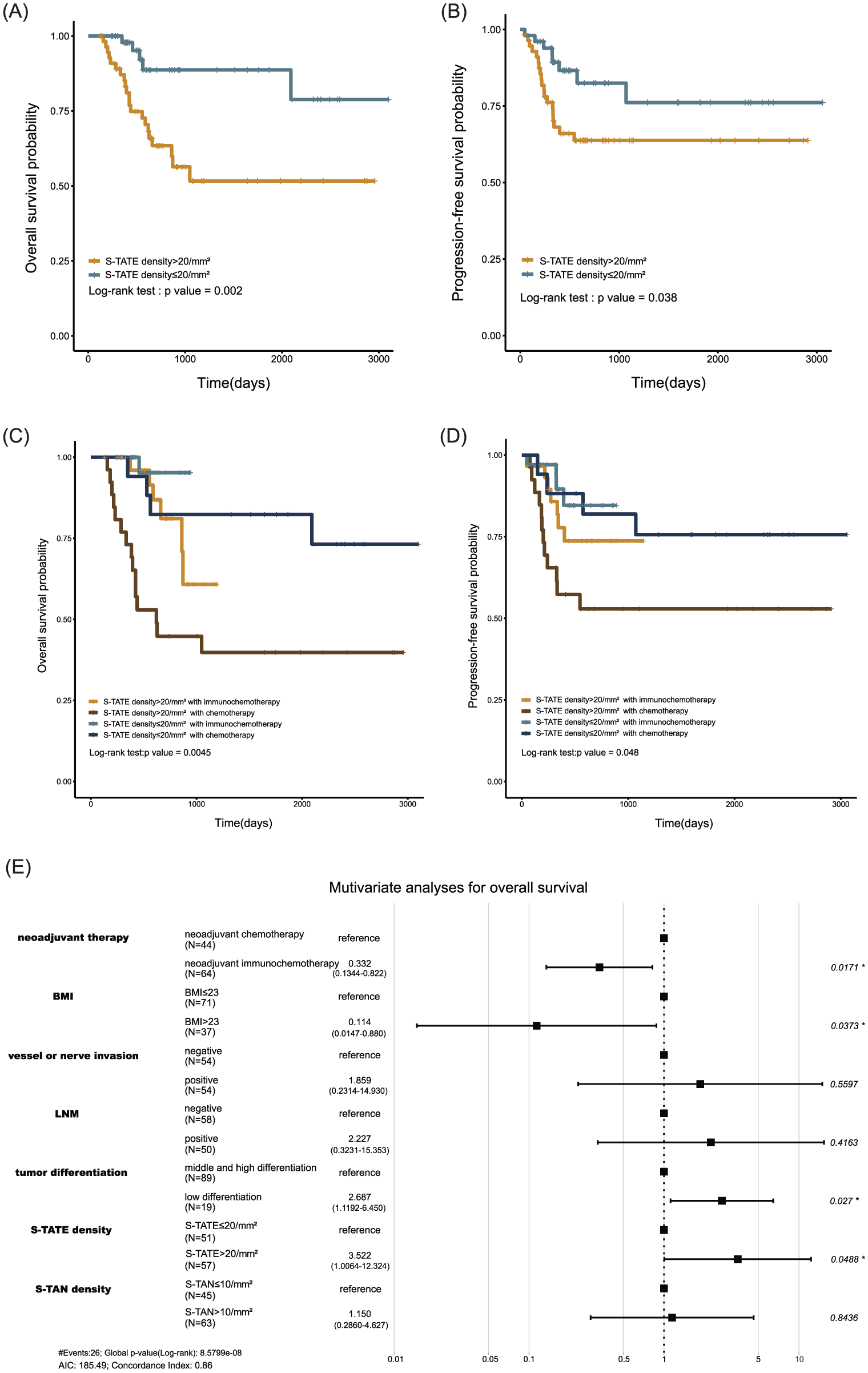
(A,B) Kaplan-Meier curves by S-TATE for OS and PFS. (C,D) Kaplan-Meier curves by S-TATE for OS and PFS under the stratification of neoadjuvant therapy. (E) Forest plot of Multivariate Cox analysis, showing the neoadjuvant therapy, tumor differentiation, S-TATE, and BMI can be predictors for death in the patients with local-regionally advanced TSCC.
Univariate analyses for overall survival.
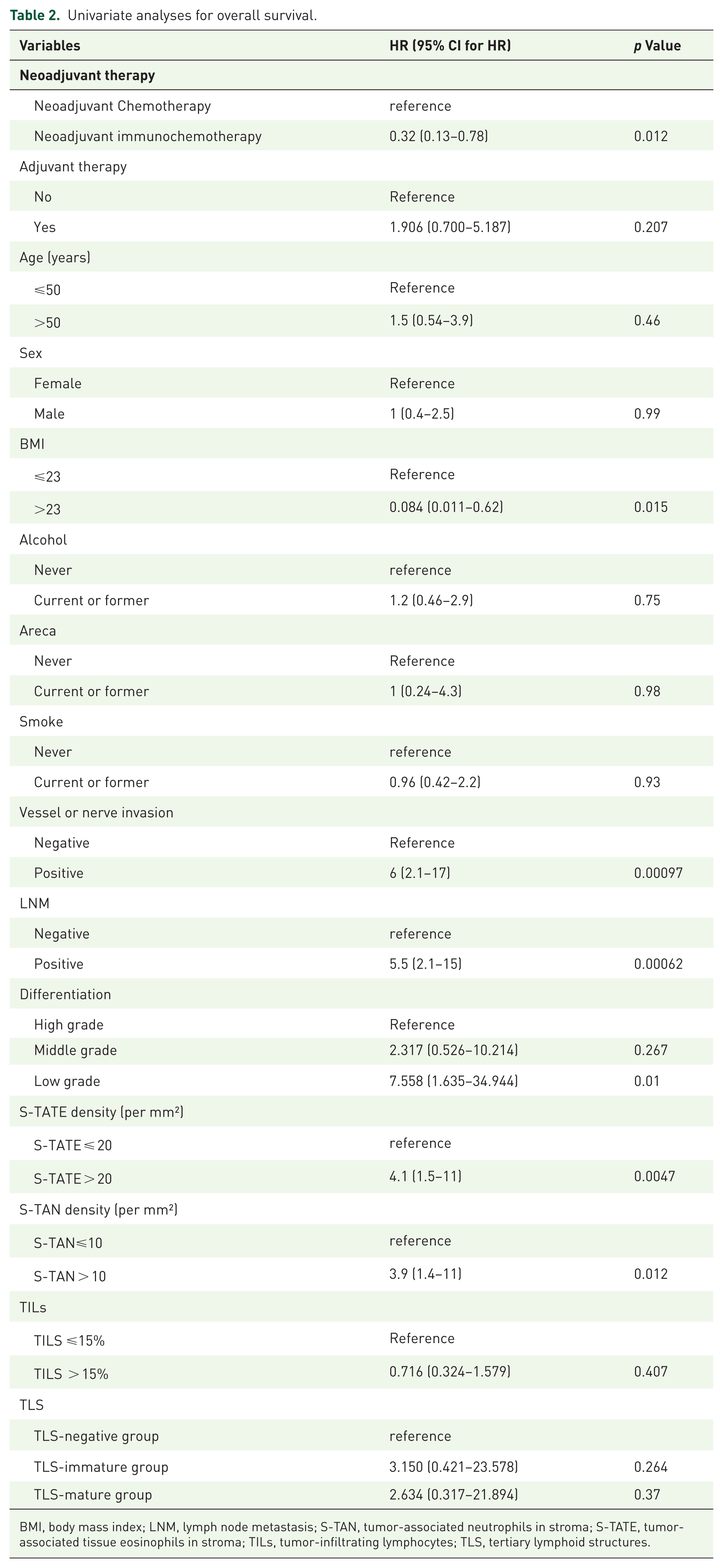
BMI, body mass index; LNM, lymph node metastasis; S-TAN, tumor-associated neutrophils in stroma; S-TATE, tumor-associated tissue eosinophils in stroma; TILs, tumor-infiltrating lymphocytes; TLS, tertiary lymphoid structures.
A risk score system featuring stromal TATE
Given that neoadjuvant therapy, S-TATE, BMI, and tumor differentiation independently predicted OS, we constructed a TATE-based prognostic score from the multivariable model (Table 3). Figure 6(a) shows that risk points differed by pathological response (Kruskal–Wallis, p < 0.001). Risk points correlated with RVT percentage (Figure 6(b); Spearman’s ρ = 0.533; p < 0.001).
TATE prognostic scoring system.
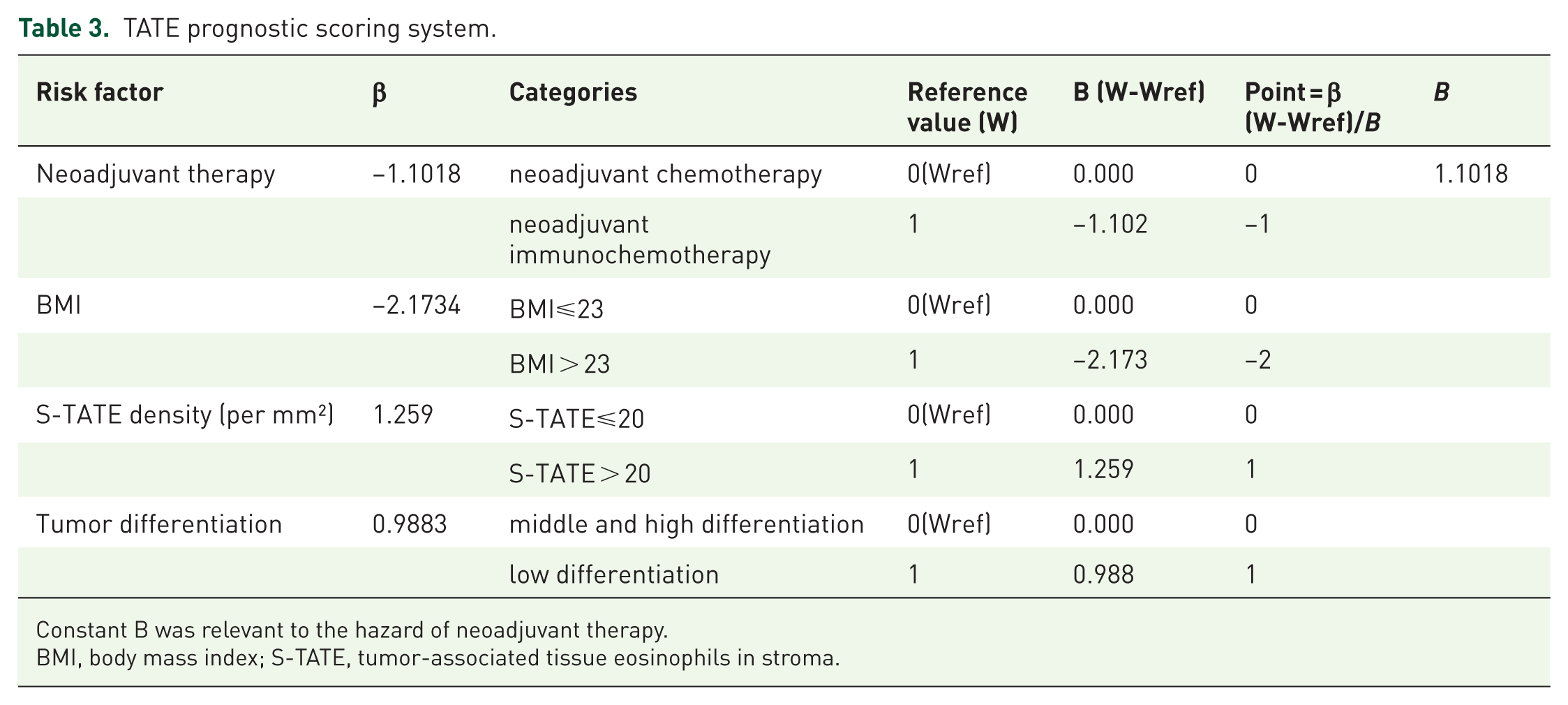
Constant B was relevant to the hazard of neoadjuvant therapy.
BMI, body mass index; S-TATE, tumor-associated tissue eosinophils in stroma.
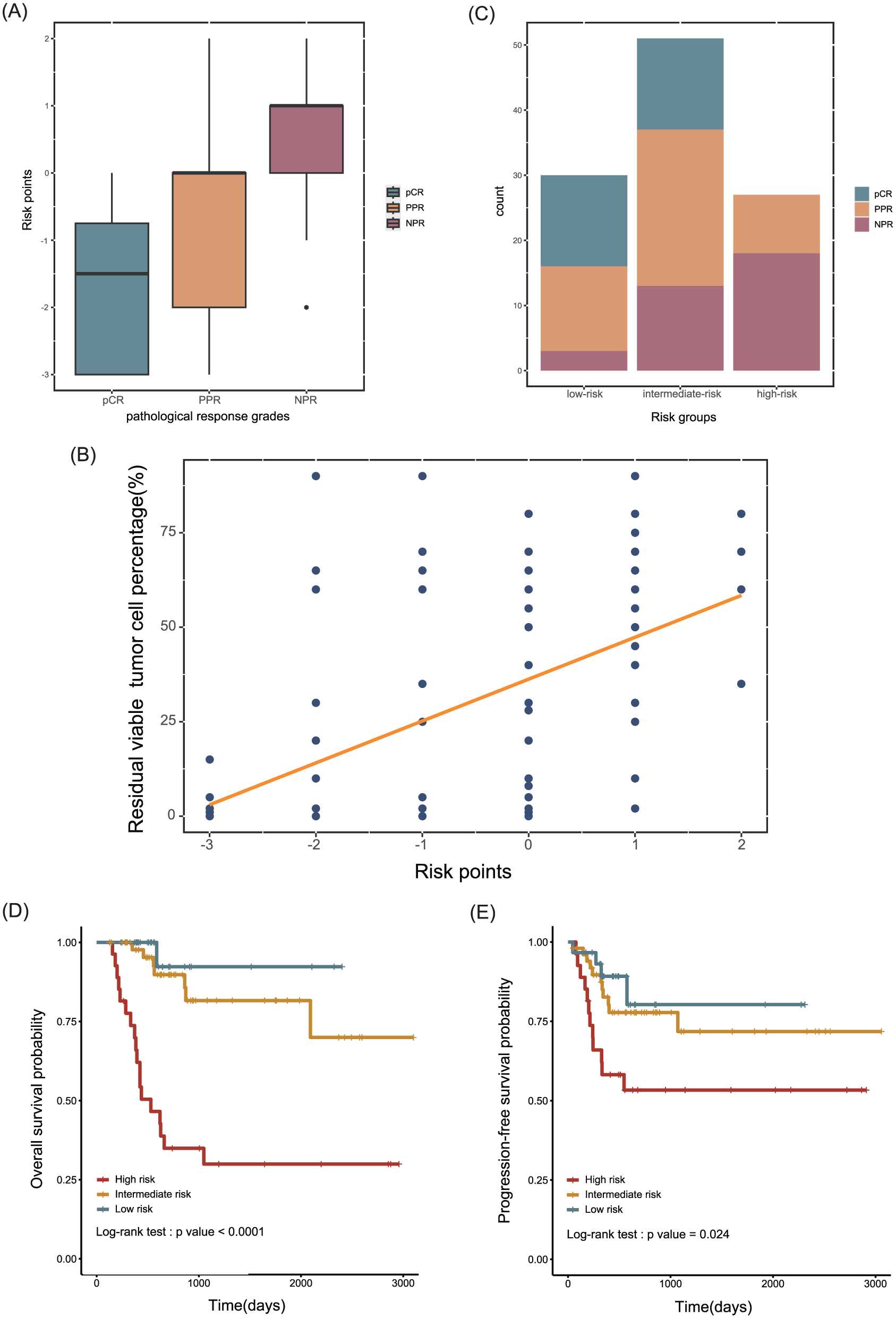
(A) BOX plot of pathological response grades by risk points (Kruskal-Wallis test, p-value < 0.001). (B) Scatter plots of risk points by RVT, showing a trend of RVT following by risk points. (Spearman’s rank correlation test, ρ = 0.533, p-value < 0.001) (C) Proportion of patients with NPR, PPR or pCR under the stratification of risk groups. (χ² (df = 4, n = 108) = 28.406, P < 0.001) (D,E) Kaplan-Meier curves of risk groups for OS and PFS.
Based on total points, patients were stratified into low- (scores −3 to −2), intermediate- (−1 to 0), and high-risk (1–2) groups. Figure 6(c) shows the distribution of response categories across risk groups (χ² = 28.406; df = 4; n = 108; p < 0.001). Survival curves demonstrated stepwise separation for both OS (Figure 6(d); log-rank p < 0.001) and PFS (Figure 6(e); log-rank p = 0.024).
Discussion
Neoadjuvant immunochemotherapy provides a promising therapeutic option for patients with locally advanced TSCC, improving both OS and pathological response. Nevertheless, predicting tumor progression and survival after such treatment remains a major challenge. Programmed death-ligand 1 (PD-L1) expression assessed by the combined positive score (CPS) is an established biomarker for response to ICIs in advanced HNSCC,33,34 but several neoadjuvant trials have shown no clear correlation between PD-L1 expression and pathological response.13,14 Therefore, reliable prognostic biomarkers are urgently needed. Joris L. Vos et al. reported that microvessel density was significantly higher in MPR samples (p = 0.001). 13 Wu-tong Ju et al. observed that the MPR group exhibited higher levels of CD4+ T-cell infiltration in resected specimens (p = 0.02). 33 Ravindra Uppaluri et al. found that inflammatory gene signatures were enriched in non-responders. 34 Moreover, N. Honoré et al. demonstrated that tumor-agnostic circulating tumor DNA (ctDNA) assays could predict disease progression and survival. 35
TATE, a major component of the tumor microenvironment (TME), has been reported in multiple tumor types, including lymphomas, breast, ovarian, uterine, bladder, lung, pancreatic, gastric, esophageal, and colorectal cancers, as well as HNSCC. 15 In our study, a higher level of stromal TATE (S-TATE) was significantly associated with a lower pCR rate in both neoadjuvant immunochemotherapy and neoadjuvant chemotherapy groups, suggesting that TATE may exert general pro-tumorigenic effects rather than mediating treatment resistance. Several in vivo studies have examined eosinophil responses to tumor cells engineered to secrete cytokines and/or chemokines, aiming to clarify the pro-tumorigenic role of eosinophils. Evidence indicates that eosinophils promote tumor progression in the TME by modulating immune cell composition and function and by secreting growth factors and matrix metalloproteinases.
A study on cervical cancer revealed that hypoxia increased thymic stromal lymphopoietin (TSLP) secretion by tumor cells, which in turn enhanced CCL17 production and recruited eosinophils to the tumor site. 36 TSLP also upregulated anti-inflammatory cytokines (IL-10, IL-4, IL-5, and IL-13) and downregulated CD80/CD86 expression on eosinophils, 37 thereby promoting cervical cancer cell proliferation and inhibiting apoptosis through the upregulation of Ki-67, PCNA, and Bcl-2 and the downregulation of Fas and FasL.36,37 Furthermore, TSLP-induced eosinophils increased angiogenic factor production in co-culture with cervical cancer cells, mediated by IL-8 and VEGF. 36 IL-5 is essential for eosinophil differentiation, trafficking, and survival. 38 Host-derived IL-5 has been shown to promote malignant pleural effusion formation by enhancing eosinophil and myeloid suppressor cell recruitment. 39 Another study demonstrated that IL-5-driven pulmonary eosinophils expressed CCL22, thereby recruiting Tregs to support metastatic niche formation, while Tregs suppressed protective immune responses by NK cells and macrophages. 40 Additionally, eosinophils express indoleamine 2,3-dioxygenase (IDO), which degrades L-tryptophan via the kynurenine pathway, leading to suppression of effector T-cell responses and induction of immune tolerance. 41
Clinically, elevated S-TATE levels have been associated with LNM, as well as vascular and perineural invasion, suggesting that TATE contributes to tumor invasiveness and migratory potential. Consistent findings have been reported across multiple malignancies: Spiegel et al. in cervical carcinoma, Mahmoud Said et al. in laryngeal squamous carcinoma, and Dante De Paz et al. in TSCC all demonstrated a positive correlation between TATE and tumor invasion.16,17,19 Furthermore, Lee Tsung-Lun et al. showed that TATE promotes metastasis and angiogenesis in HNSCC, 18 while Fei Li et al. identified C-C chemokine ligand 6 (CCL6) as a key mediator of eosinophil-driven tumor migration and metastasis in vivo. 42 Consistent with these findings, our results demonstrated that higher S-TATE was associated with poorer OS and PFS, likely due to its ability to enhance tumor cell migration and invasion. Although the underlying molecular mechanisms remain unclear, multiple studies support the pro-tumorigenic role of TATE. Therefore, greater attention should be paid to evaluating TATE in post-treatment specimens of locally advanced TSCC. For patients with high TATE levels, closer surveillance and more intensive postoperative management should be considered to reduce the risk of recurrence or progression.
We acknowledge several limitations in our study. First, its retrospective design and the relatively small number of events from a single center may limit the robustness and generalizability of the survival analyses. In addition, the absence of data on PD-L1 status, HPV infection, and baseline TATE levels introduces potential bias. Second, our risk-scoring system lacks both internal and external validation in independent prospective datasets, raising concerns about its accuracy and consistency. Finally, cytokines and chemokines known to influence eosinophil migration and effector function were not investigated in this study.
Despite these limitations, our work provides novel insights into the prognostic relevance of eosinophil infiltration in TSCC. To our knowledge, this is the first study to evaluate the prognostic value of infiltrating eosinophils within the TME in patients with locally advanced TSCC treated with neoadjuvant therapy, particularly neoadjuvant immunochemotherapy. To integrate the effects of coexisting prognostic factors on survival outcomes, we established a risk-scoring system incorporating BMI, S-TATE, and tumor differentiation, which was significantly associated with OS in multivariate Cox analysis. Importantly, these factors are practical to assess and readily applicable in routine clinical practice.
Conclusion
In conclusion, S-TATE serves as an independent prognostic biomarker in patients with locally advanced TSCC treated with neoadjuvant therapy. Elevated S-TATE levels are associated with reduced pathological complete response rates, poorer survival, and greater risks of progression, LNM, and vascular or perineural invasion. Routine assessment of S-TATE in resected specimens may facilitate postoperative risk stratification and inform adjuvant therapy decisions. Further investigations are warranted to clarify the molecular mechanisms driving intrastromal TATE formation and its potential as a therapeutic target.
Supplemental Material
sj-docx-1-tam-10.1177_17588359261424663 – Supplemental material for Stromal tumor-associated eosinophils predict therapeutic resistance and survival in locally advanced tongue squamous cell carcinoma after neoadjuvant therapy
Supplemental material, sj-docx-1-tam-10.1177_17588359261424663 for Stromal tumor-associated eosinophils predict therapeutic resistance and survival in locally advanced tongue squamous cell carcinoma after neoadjuvant therapy by Jiaming Huang, Yanfeng Chen, Lei Yuan, Xinyi Xian, Xiujiao Shen, Wenjian Qin, Jing Zeng and Wanming Hu in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-2-tam-10.1177_17588359261424663 – Supplemental material for Stromal tumor-associated eosinophils predict therapeutic resistance and survival in locally advanced tongue squamous cell carcinoma after neoadjuvant therapy
Supplemental material, sj-docx-2-tam-10.1177_17588359261424663 for Stromal tumor-associated eosinophils predict therapeutic resistance and survival in locally advanced tongue squamous cell carcinoma after neoadjuvant therapy by Jiaming Huang, Yanfeng Chen, Lei Yuan, Xinyi Xian, Xiujiao Shen, Wenjian Qin, Jing Zeng and Wanming Hu in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
