Abstract
Objective
To explore the relationship between immune scores and prognosis of patients with esophageal squamous cell carcinoma (ESCC) and construct a corresponding clinical prediction model.
Methods
The present research was a retrospective cohort study. We obtained the clinical information and immune scores of 137 patients with ESCC from The Cancer Genome Atlas database, and a Cox proportional risk model was used to construct the clinical prediction model. The concordance index, receiver operating characteristic curve, calibration curve, net reclassification improvement (NRI), and integrated discrimination improvement (IDI) were used to evaluate model performance and prediction accuracy.
Results
Patients with a high immune score (> −121.4) showed a worse prognosis than those with a low immune score (< −645.8; hazard ratio=3.743, 95% confidence interval [CI]=1.385–10.115, P=0.009). The concordance index of the predictive model was 0.733 (95% CI=0.655–0.812). The calibration curve showed that the 3- and 5-year overall survival rates predicted by the model were highly consistent with the observed values. The NRI and IDI for the 3-year overall survival indicated that the model with the immune scores was superior for classifying the risk probability and distinguishing cases.
Conclusion
Immune scores may be an independent predictor of prognosis in patients with ESCC.
Keywords
Introduction
Esophageal cancer is one of the most common malignant gastrointestinal tumors worldwide, and it is the eighth most common cancer overall and the sixth leading cause of cancer-related death. 1 In 2018, there were 572,000 new cases of esophageal cancer globally (representing 3.2% of all cancers) and 508,585 new deaths related to esophageal cancer (5.3% of all cancer-related deaths). 2 Esophageal carcinoma is divided into two subtypes: esophageal squamous cell carcinoma (ESCC) and esophageal adenocarcinoma. ESCC is the main histological type of esophageal cancer, accounting for 90% of cases worldwide. ESCC is particularly aggressive, and many patients have a poor prognosis, 3 with the 5-year survival rate ranging from 4% to 40%. 4 In the past decade, several new treatments for esophageal cancer have been developed, but the prognosis for patients has only slightly improved, and the global 5-year survival rate remains low.5–7 The introduction of new treatment methods and the accurate assessment of prognostic factors will help to improve the survival rate of patients with ESCC.
Immunotherapy has recently attracted the attention of oncologists and clinicians, with some reports of significant improvements in the prognosis of cancer patients receiving this treatment.8–10 This situation indicates that understanding the relationship between the immune system and tumor prognosis is necessary for future developments of tumor immunotherapy.
Tumor microenvironment studies have revealed that infiltrated immune cells are related to the growth, invasion, and metastasis of tumors.11,12 Additionally, immune infiltration is associated with the prognosis of patients with ESCC. 13 The immune score calculated based on gene expression data can indicate the immune characteristics and be used to estimate the degree of immune cell infiltration in tumor tissues. This method has previously been used to estimate the relationship between immune scores and the prognosis of breast cancer. 14 However, few studies have investigated the relationship between immune scores and the prognosis of ESCC. As a result, there is limited guidance for the clinical application of this metric.
The present situation prompted us to study the relationship between immune scores and the prognosis of ESCC. We used this information to construct an appropriate clinical predictive model and then produced a nomogram for guiding the clinical treatment of patients with ESCC.
Methods
Data collection and patient selection
The present research was a retrospective cohort study; the data of this study were obtained from The Cancer Genome Atlas (TCGA) database, and all participants were patients with ESCC. The TCGA project began in 2005 with the aim of using genome sequencing and bioinformatics to classify mutations associated with cancer. This project is supervised by the Cancer Genomics Center at the National Cancer Institute and the US government-funded National Human Genome Institute (https://www.cancer.gov/about-nci/organization/ccg/research/structural-genomics/tcga). The TCGA database currently contains more than 200 types of information on cancer and the clinical characteristics of patients, and it provides a large data set suitable for tumor genome analysis. 15
We used the cBioPortal (http: //www.cbioportal.org/) to download the clinical information of patients with ESCC, including their sex, age, height, weight, TNM (tumor, nodes, metastases) stage of cancer, histology information, overall survival (OS), and OS time. Height and weight information were used to calculate body mass index (BMI) values. The immune score was calculated using gene expression data to estimate the level of immune cell infiltration. This method involves using expression data to estimate immune cells in malignancies and the algorithm of single-sample gene set-enrichment enrichment analysis (SsGSEA), as described in detail in previous studies. 16 Detailed information is available elsewhere (https://bioinformatics.mdanderson.org/estimate/disease.html). We deleted records of duplicate cases and those with missing clinical information or data for calculating the immune score. Our retrospective cohort study followed the guideline from https://www.equator-network.org/ (STROBE Statement—Checklist of items that should be included in reports of cohort studies). Each recommendation on the STROBE checklist was carefully addressed.
Ethics statement
The present study was performed in compliance with the Declaration of Helsinki. Informed consent was not required as personal identifying information was not included. Ethical approval was not required because the present study used data from publicly available databases.
Statistical analysis
The outcome of the study was the OS. X-tile software (version 3.6.1, Yale University School of Medicine, New Haven, CT, USA) was used to obtain the optimal cutoffs of the immune score for dividing patients into three groups. 17 A Kaplan–Meier plot (K–M plot) of these three groups along with 3- and 5-year survival receiver operating characteristic (ROC) curves were mapped, and the area under the ROC curve (AUC) value was used to assess the prognostic value and power of the immune score.
A multivariate Cox proportional risk regression model was used to determine the independent predictors of OS. After simultaneously considering the effects of age, histology, sex, BMI, and TNM stage, adjusted hazard ratios (HRs) and 95% confidence intervals (CIs) were estimated. The nomogram was formulated based on the results of multivariate Cox regression analysis, and the results for the nomogram were resampled 1000 times using the bootstrap method for internal data verification. We used the calibration curve to calibrate the nomogram for the 3- and 5-year OS rates by comparing the observed survival rate and the survival rate predicted by the nomogram. 18
The concordance index (c-index) was first proposed in 1996 19 for determining the difference between the value predicted by the Cox model and the actual value and to evaluate the performance of the predictive model. We compared the predictive models with and without immune scores and calculated the integrated discrimination improvement (IDI) and the net reclassification improvement (NRI) to evaluate the improvement in the prediction accuracy of the new predictive model. 20
All statistical tests were two-sided, and
Results
Characteristics of patients
The final analysis was conducted with 137 patients aged 61.36±11.05 years (mean±standard deviation). These 137 patients included 117 (85.40%) men, 44 patients (32.12%) aged 56 to 65 years, 79 (57.66%) cases of ESCC, 68 (53.28%) with TNM stage T3, 65 (47.45%) with TNM stage N0, 128 (93.43%) with TNM stage M0, and 78 (56.93%) with BMIs of 18.5 to 24.9 kg/m2. The median OS time was 13.17 months (range=0–122.01 months). The optimal cutoffs of the immune score calculated using X-tile software were −645.8 and −121.4. These two cutoffs were used to divide the patients into the following three groups: 23 (16.79%) with low immune scores (< −645.8), 38 (27.74%) with moderate immune scores (−645.8 to −121.4), and 76 (55.47%) with high immune scores (−121.4).
Table 1 presents information on patients in the three immune-score groups. The patients in the groups with low, moderate, and high immune scores were aged 57.83±11.86, 61.61±10.23, and 62.30±11.13 years, respectively. Compared with patients in the moderate- and low-immune-score groups, patients with high immune scores were more likely to have a BMI of ≥25 kg/m2, TNM stage T3, and TNM stage N1. Table 2 shows the differences in survival between groups with different immune scores.
Characteristics of patients with ESCC according to the subgroups of different immune scores.

BMI, body mass index; ESCC, esophageal squamous cell carcinoma; M, metastases; N, nodes; T, tumor.
Survival information of patients with ESCC according to the subgroups of different immune scores.

ESCC, esophageal squamous cell carcinoma; SD, standard deviation.
Verifying the immune score model
The optimal threshold for the immune score was calculated using the X-tile software, and patients were divided into three groups. Figure 1 shows the difference in survival among the three groups. Patients with higher immune scores had a poor prognosis. The ROC curve was used to verify the power of immune scores to predict the OS of patients, and the AUCs for 3 and 5 years were 0.906 and 0.872, respectively. This result indicated that the immune score has a strong predictive value for the prognosis of patients with ESCC (Figure 2).
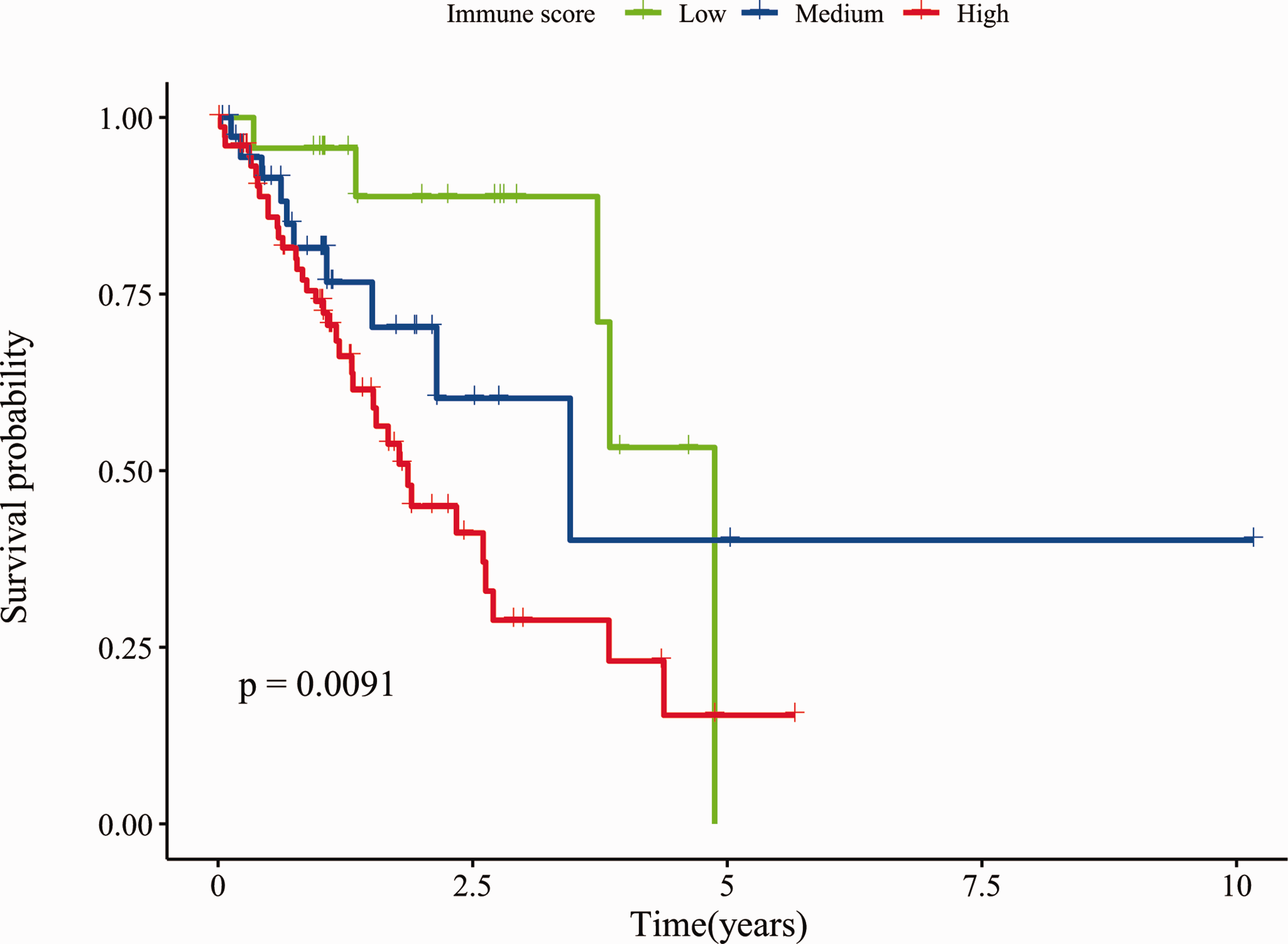
Kaplan–Meier curves depicting the associations of immune score subgroups with the overall survival (OS) for 137 patients with esophageal squamous cell carcinoma. Comparison of OS among patients with immune scores ≤ −645.8 (group 1, Low), immune scores between −645.8 and −121.4 (group 2, Medium), and immune scores > −121.4 (group 3, High).

Nomogram predicting 3- and 5-year survival. The nomogram was used by scoring each variable. The scores for all variables were then combined to obtain total scores, and a vertical line was drawn from the total score line to estimate 3-year and 5-year survival rates.
Univariate and multivariate analyses of OS
The univariate regression analysis showed that the risk factors for the OS rate were a high immune score (HR=3.626 vs low immune score, 95% CI=1.413–9.302,
Univariate analyses of OS among patients with ESCC according to clinical pathological characteristics and immune scores.

BMI, Body mass index; CI, confidence interval; ESCC, esophageal squamous cell carcinoma; HR, hazard ratio; M, metastases; N, nodes; OS, overall survival; T, tumor.
The multivariate Cox regression model showed that that the risk factors for the OS rate were an age of 66 to 75 years (HR=2.707 vs <50 years, 95% CI=1.125–6.514,
Multivariate analyses of OS among patients with ESCC according to clinical pathological characteristics and immune scores.
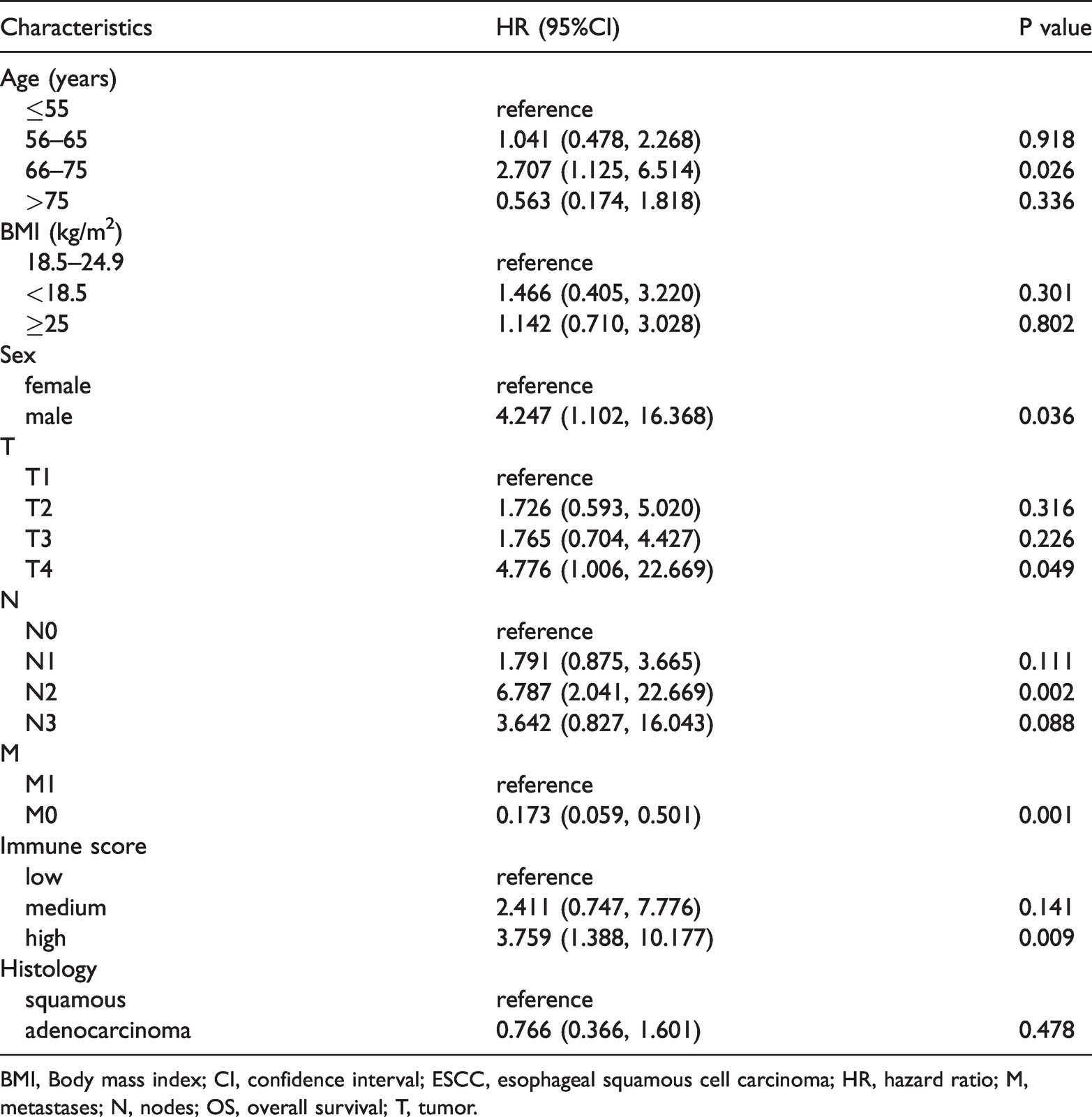
BMI, Body mass index; CI, confidence interval; ESCC, esophageal squamous cell carcinoma; HR, hazard ratio; M, metastases; N, nodes; OS, overall survival; T, tumor.
Nomogram validation
The independent prognostic factors associated with the OS rate identified in the multivariate analysis were used in the internal validation of the nomogram. The nomogram was used by drawing a vertical line to determine the score for each variable. The scores for all of the nomogram variables were then summed to obtain the total score, and a vertical line was drawn from the total score to obtain the OS rates for 3 years and 5 years (Figure 3).

Area under the curves (AUCs) for 3- and 5-year survival rates. The receiver operating characteristic curve was used to verify the power of immune scores to predict the overall survival of 137 patients with esophageal squamous cell carcinoma.
Performance of the nomogram
The c-index of the predictive model was 0.733 (95% CI=0.653–0.813), indicating that the predictive model had good recognition ability. Figure 4 shows the calibration curve of the model, which demonstrates that the 3- and 5-year OS rates predicted by the model were highly consistent with the observed results. The NRI and IDI for the 3-year OS were 0.682 (95% CI=0.133–1.332) and 0.04 (
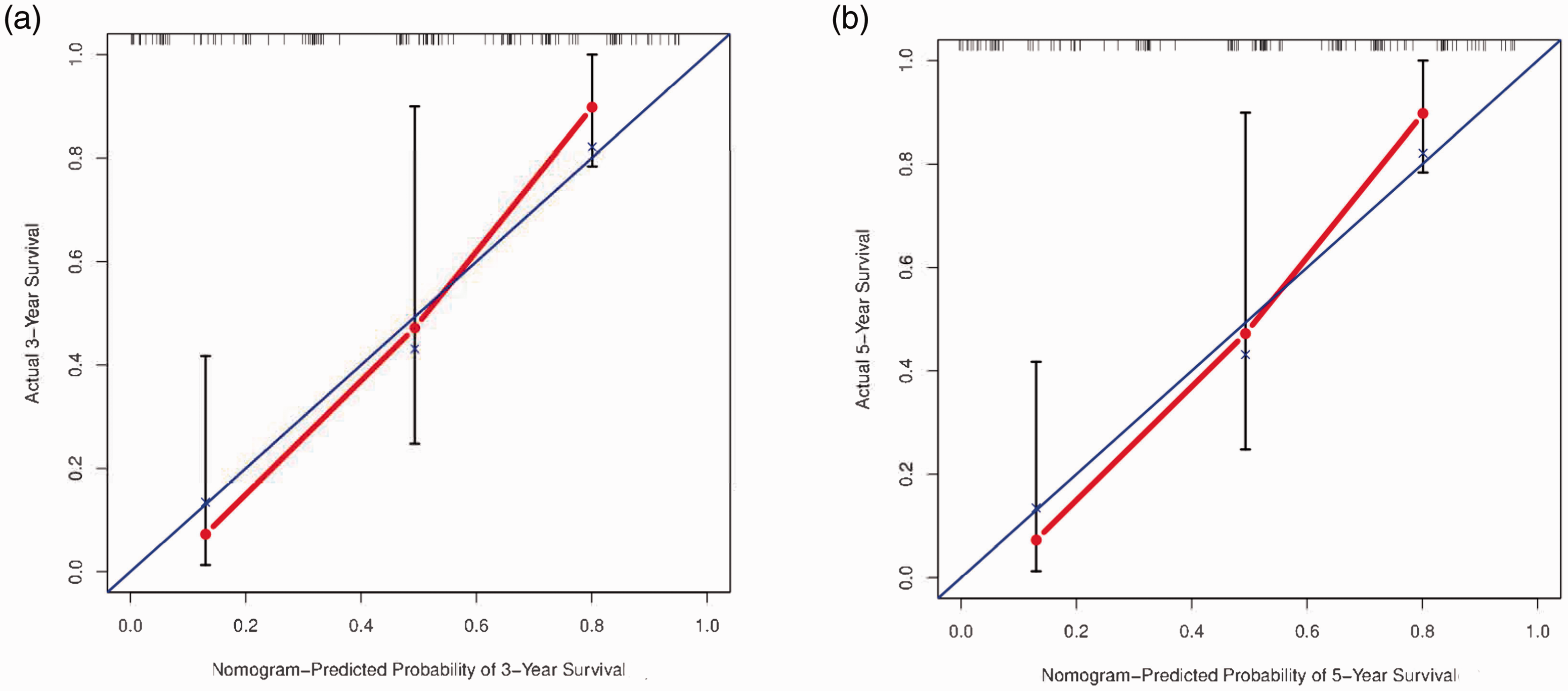
Calibration curves for the nomogram. (a) Calibration plot of the 3-year overall survival (OS). (b) Calibration plot of the 5-year OS. The x-axis is the predicted survival calculated by the nomogram, and the y-axis is the actual survival estimated by the Kaplan–Meier method. The blue line is the reference line, representing the actual observed values, and the red line represents the predicted values of the model.
Discussion
The relationships between the tumor microenvironment and cancer prognoses have received increasing attention.10,21,22 Although treatments for esophageal cancer have improved, 5-year survival rates remain unsatisfactory worldwide. Attempting to understand the relationship between ESCC and the host immune system is a very promising approach. 23 The present study evaluated the relationship between the immune score and clinical prognosis of patients with ESCC using their gene expression data. The obtained information was used to construct a clinical predictive model and a nomogram.
Previous studies have found that certain immune-related parameters—especially tumor-infiltrating lymphocytes—are closely related to the prognosis of patients with ESCC, suggesting that different immune states significantly impact the prognosis of these patients.24–26 Numerous high-quality clinical predictive models of ESCC have been constructed, but few studies have used the immune score in an independent predictive model.27,28 Therefore, we explored the relationship between immune scores and ESCC prognosis based on data obtained from the TCGA database. Moreover, a nomogram was constructed to provide a convenient approach for clinical staff to predict the survival status of individual patients.
The multivariate Cox regression model showed that male sex was a risk factor for ESCC, which is consistent with previous findings. 28 Because lifestyle habits such as smoking and drinking alcohol are risk factors for the onset and progression of ESCC, we speculate that the poor prognosis in male patients with ESCC is associated with poor lifestyle habits. 29 Age is a predictor of the prognosis of tumors. More specifically, previous studies have shown that older patients with esophageal cancer generally have a worse prognosis, which may be related to several factors such as the higher rate of complications when treating the elderly and those with poor autotrophic levels. 30 The present study found that compared with patients younger than 55 years, only patients aged 66 to 75 years had a significantly worse prognosis, and no statistically significant difference for patients aged ≥75 years was observed. We thought that this result might be because of the small number of older patients in our sample. For example, there were only 22 patients who were aged 75 or older. Based on the overall trend, older patients continue to have a worse prognosis because older patients tend to have multiple metabolic diseases. A particularly interesting finding was that after adjusting for confounders, we found that a lower immune score was significantly associated with a better prognosis. Cancer patients with a high immune score are thought to typically have a stronger immune system, which helps to eliminate their tumors.14,31 However, our results suggested that immune regulation may contribute to tumor escape and survival in ESCC. The occurrence of esophageal cancer is closely related to chronic inflammation. 32 Myeloid-derived suppressor cells (MdSCs) often play an immunosuppressive role, but inflammation can activate MdSCs, 33 leading to the direct inhibition of T cell activation and natural killer cell toxicity.34,35 Furthermore, previous studies have found that increased MDSCs in patients with esophageal cancer are associated with advanced disease, poor prognosis, and treatment resistance. 36 The expansion of regulatory T cells (Tregs) has been noted in both the peripheral blood and esophageal mucosa of patients with esophageal cancer. 37 Tregs are involved in regulating the proliferation and activation of B cells and T cells in normal physiological processes, but in cancer, Tregs are involved in inhibiting tumor immune responses by secreting immunosuppressor factors and interfering with the expression of tumor-associated antigens.38,39 These findings support our conclusion and suggest that some immune components in the tumor microenvironment may have a dual role. In other words, they mainly play an immunosuppressive role in normal physiological processes but may be associated with tumor immune escape and the poor prognosis of patients during the course of ESCC. 32
After establishing the predictive model and the nomogram, we used several methods to evaluate the performance and discrimination of the model. The c-index of the model and the 3- and 5-year OS AUC values showed that the model exhibited a good discriminatory ability. The calibration curve demonstrated the consistency between the predicted and observed values. Because previous studies have rarely used the immune score as an independent predictor, we compared our model with the model that did not include the immune score. The NRI and IDI values for the 3-year OS were 0.703 and 0.008, respectively, which showed that the prediction performance of the model was greatly improved by adding the immune score as an independent predictor. However, we found that the NRI and IDI values for the 5-year OS were not statistically significant, which we attribute to the small sample size used to construct the model. Therefore, the usefulness of the immune score in predicting the 5-year OS remains to be further elucidated in studies involving larger samples.
To the best of our knowledge, this is the first study to predict the OS in patients with ESCC based on the immune score and clinical information. Previous studies of prognostic risk factors for ESCC have mostly been based on tissue samples or radiological analyses that assess the primary tumor, such as tumor size, stage, histological integrity, degree of invasion, and regional lymph node metastasis.40,41 Although these factors have been shown to be valuable in evaluating the prognosis of patients with ESCC, this analytical approach is also flawed because it assumes that tumor progression is a cell-autonomic process, focusing only on the cancer cells and ignoring the immune response of the host. Many studies have reported the hypothesis that cancer development is influenced by the host immune system, and researchers have demonstrated the value of immunophenotypes in predicting the prognosis of breast, colorectal, and prostate cancers.42–44 Several cohort studies have also reported that the number, type, and location of tumor immune infiltrates in primary tumors are strongly associated with the survival of cancer patients. 45 We found that the immune score was closely related to the prognosis of patients with ESCC and thus used it as a predictor of tumor prognosis. An advantage of this approach is that the immune score quantifies the level of immunity, making it universal and providing a reference for other tumor prognostic studies in addition to ESCC. 11 Furthermore, considering that genomic, transcriptional, and proteomic analyses of clinical tumor tissues are already available in clinical practice, our study provides a simple tool for evaluating outcomes in patients with ESCC and useful information for the development of immunotherapy and other novel therapeutic approaches.46,47 Our predictive model and its associated nomogram allow both clinicians and patients to easily and quickly make individualized survival predictions, which is an essential function in the era of individualized treatment for tumors.
There is no denying that our study was also subject to some limitations. First, because only patients with complete information were included in the analysis, selection bias might have been present. The present study was a retrospective study, and it was not possible to verify a causal association between immune scores and OS. The current association could be due to confounding factors, or there could be a reverse causality. Second, the prognosis of patients with ESCC is related to the treatments they receive and their lifestyle habits, such as smoking and drinking alcohol after admission, but the TCGA database does not contain these data. At the same time, the number of samples with disease-free survival records does not currently meet the test efficiency, and we only focused on the OS of patients, which limited our research. Third, the ability to externally validate our predictive model was restricted by the small number of datasets available to calculate the immune score. The results of internal verification are based on the data of 137 patients, and a conclusion based on a larger sample size would be more helpful to verify this conclusion. Although a larger sample size is not available at present, we plan to address this in future work. Finally, considering the differences in medical resources, the use of immune scores to assess the survival of patients is still limited by technical, financial, and other factors, but it is undeniable that this will be a promising direction for the future.
Conclusions
This study developed and internally verified a nomogram based on immune scores for predicting the 3- and 5-year OS rates of patients with ESCC. This nomogram predicted the survival rates of individuals with a high c-index and was well-calibrated. The results of this study may help clinicians and patients to achieve personalized survival prediction.
Footnotes
Acknowledgments
The authors thank The Cancer Genome Atlas (TCGA) program staff for providing open access to the database.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This study was supported by the National Social Science Foundation of China (No. 16BGL183).
