Abstract
Background:
Several clinical trials of neoadjuvant chemoimmunotherapy (neoCIT) for patients with locally advanced oesophageal carcinoma have been reported recently, with promising anti-tumour efficacy. However, systematic evaluation of the treatment patterns and clinical outcomes in a real-world cohort is lacking.
Objectives:
This study aimed to describe the real-world treatment patterns and short- and long-term outcomes of neoCIT in patients with resected oesophageal squamous cell carcinoma (ESCC).
Design:
A multicentre retrospective cohort study.
Methods:
Data from patients with resected ESCC who received neoCIT in routine clinical practice between January 2020 and December 2022 were retrospectively collected from three tertiary centres in China.
Results:
A total of 324 consecutive patients were enrolled, and the majority (82.4%) received two cycles of neoCIT. In total, 225 patients achieved a radiographic partial response (PR), resulting in an objective response rate of 69.4%. The rates of minimally invasive oesophagectomy and radical resection were 71.9% and 98.5%, respectively. Fifty-nine patients (18.2%) experienced postoperative complications, and the most common complication was anastomotic leakage (10.2%). The pathological complete response and major pathological response rates were 27.2% and 37.0%, respectively. As of August 31, 2024, the median follow-up was 24.8 months. The estimated 3-year rates of disease-free survival and overall survival were 62.3% and 71.5%, respectively. Multivariate analyses revealed that tumour grade (p = 0.047), number of neoadjuvant therapy cycles (p = 0.042), resection status (p = 0.043), pathological N stage (p < 0.001) and adjuvant treatment (p < 0.001) were independent prognostic factors for disease-free survival. However, advanced pathological N stage was identified as the sole independent factor that correlated with worse overall survival (p < 0.001).
Conclusion:
This real-world study validates the clinical efficacy and safety of neoCIT for resected oesophageal squamous cell carcinoma while demonstrating encouraging survival outcomes.
Introduction
Two landmark phase III trials reported a consistent survival benefit of neoadjuvant chemoradiotherapy (neoCRT) compared with oesophagectomy alone in patients with locally advanced oesophageal carcinoma (LA-EC).1,2 However, the use of neoCRT, accompanied by increased perioperative complication rates and mortality, may limit its wide clinical application.3,4 Alternative neoadjuvant treatment regimens that balance the efficacy and toxic effects are still under continuous investigations in patients with EC. 4
Although advances in 2021 rewrote the guidelines for adjuvant nivolumab in resected EC, 5 the efficacy and safety of perioperative anti-programmed death (PD)-1 or PD-ligand 1 (PD-L1) therapy have yet to be definitively confirmed. 4 In recent years, an increasing number of phase Ib/II clinical trials have demonstrated promising pathological responses (major pathological response (MPR) rate: 37.5%–89.0%; and pathological complete response (pCR) rate: 16.7%–51.4%) with a manageable safety profile in locally advanced oesophageal squamous cell carcinoma (LA-ESCC) patients who received neoadjuvant PD-1 inhibitors combined with CRT or chemotherapy.4,6,7 However, almost all these trials were designed as single-arm studies, conducted at a single centre, and based on small samples of participants.4,6,8 Despite two open-label, randomised, phase III trials published in 2024, which confirmed the superiority of neoadjuvant PD-1 inhibitors combined with chemotherapy over chemotherapy alone for patients with resected ESCC in terms of improving pathological response rates, the short follow-up period leaves uncertainty about whether neoadjuvant chemoimmunotherapy (neoCIT) leads to better survival rates.9,10
Therefore, we attempted to systematically describe the real-world treatment patterns and short- and long-term outcomes of neoCIT in patients with resected ESCC across different clinical practice settings.
Methods
The reporting of this study conforms to the STrengthening the Reporting of OBservational studies in Epidemiology (STROBE) cohort reporting guidelines (Table S1). 11
Study design
Patients who underwent surgical treatment for resected ESCC (T2-4aN0-3M0 per the American Joint Committee on Cancer eighth edition staging manual) between January 2020 and December 2022 from National Cancer Center/National Clinical Research Center for Cancer/Cancer Hospital & Shenzhen Hospital, Chinese Academy of Medical Sciences and Peking Union Medical College, Anyang Tumour Hospital, The Affiliated Anyang Tumour Hospital of Henan University of Science and Technology and Shaanxi Provincial People’s Hospital were retrospectively included. The detailed flow chart of patient selection is illustrated in Figure 1.

Flow diagram of this real-world cohort study.
Perioperative treatment
Neoadjuvant treatment regimens were formulated by the local multidisciplinary team (MDT) according to the National Comprehensive Cancer Network and Chinese Society of Clinical Oncology guidelines, Chinese expert consensus and latest clinical research findings.4,12,13 In addition to these abovementioned references, MDT decisions on adjuvant therapy were impacted primarily by postoperative complications, pathological remission status and patient consent. Radiologic response was evaluated according to the Response Evaluation Criteria in Solid Tumors (RECIST, version 1.1).
Oesophagectomy
Surgical incision and procedure were collaboratively determined by experienced thoracic surgeons in local departments on the basis of guidelines recommendation.12,13 McKeown and Ivor-Lewis oesophagectomy with two-yield lymphadenectomy were mandatory procedures. Patients who presented with neck lymph node metastasis were candidates for additional cervical lymph node dissection. The Sweet procedure was performed only in patients with oesophagogastric junction tumours, and who were not tolerable or suitable for right thoracic approach.
Pathological assessment
The resected oesophageal tumour and lymph nodes were evaluated by two local pathologists who had specialised in chest pathological diagnosis for more than 10 years. In case of any discrepancy, a joint review was performed using a double-headed microscope to reach a consensus diagnosis. In accordance with the widely accepted criteria,1,2,9,10 no intact tumour cells or carcinoma in situ found in the whole tumour bed or all resected lymph nodes were classified as pCR (tumour regression grade (TRG) 1), whereas MPR was defined as 10% or fewer residual tumour cells, including TRG 1 and 2.
Clinical outcomes
The key outcomes of this study included objective response (defined as a complete or PR according to the RECIST, version 1.1), MPR, pCR, disease-free survival (DFS; calculated from the date of surgery to the date of first recurrence, death or last follow-up) and overall survival (OS; defined as the date from the first cycle of neoCIT until death from any cause or last follow-up).
Statistical analysis
All data analyses were performed using MedCalc Statistical Software version 22.0 (MedCalc Software Ltd, Ostend, Belgium). If the continuous variables were normally distributed, the means (standard deviations, SDs) were presented, and Student’s t tests were used to analyse the difference; conversely, the medians (interquartile ranges, IQRs) and Mann–Whitney U test were used. Categorical variables were reported as absolute numbers and frequencies, and differences were assessed via the Chi-square test or Fisher’s exact test (when more than 20% of cells have expected frequencies <5). The Kaplan–Meier method was used to plot DFS and OS curves and assess the Cox proportional hazards assumption. The Cox multivariable proportional regression model was performed by including all covariables with p < 0.05 in the univariable model to identify factors associated with DFS/OS. A two-sided p value of less than 0.05 was considered statistically significant.
Results
Patient characteristics
Among the 324 consecutive patients enrolled in this study, a majority of patients had clinical T3 stage disease (94.1%) and clinical N-positive status (88.9%), respectively. Nearly two-thirds of the tumours were located at the middle of the thoracic oesophagus, while the mean (SD) tumour length was 5.5 (1.9) cm. The baseline clinicopathological details are outlined in Table 1.
Associations between clinicopathological characteristics and pathological response status among the whole cohort.

Using the Fisher’s exact test.
BMI, body mass index; ICIs, immune checkpoint inhibitors; kg, kilogram; MPR, major pathological response; SD, standard deviation.
Neoadjuvant therapy
All 324 patients were treated with neoadjuvant PD-1 inhibitors. The majority (98.8%) received neoCIT alone, and only 4 patients (1.2%) were treated with neoCIT combined with radiotherapy. No deaths related to neoadjuvant treatment occurred. According to the RECIST (version 1.1) criteria, 225 patients (69.4%) achieved a PR, 3 patients (1.0%) had progressive disease (PD) and no patient achieved a complete response.
Perioperative outcomes
Altogether, 223 patients (68.8%) achieved T downstaging, with 96 (29.6%) reaching pathological T0; and 177 (61.5%) achieved N downstaging (Figure 2). The pCR and MPR rates were 27.2% (95% confidence interval (CI), 22.6%–32.3%) and 37.0% (95% CI, 32.0%–42.4%), respectively (Table 2). Notably, the MPR status was only significantly related to radiographic response status (p < 0.001), whereas the MPR rates were comparable among patients receiving different neoadjuvant PD-1 inhibitors, chemotherapy regimens and cycles (all p > 0.05).

Sankey diagrams demonstrating the corresponding relationships between clinical and pathological T stage (a) and between clinical and pathological N stage (b).
Surgical, pathological and postoperative outcomes among the whole cohort.
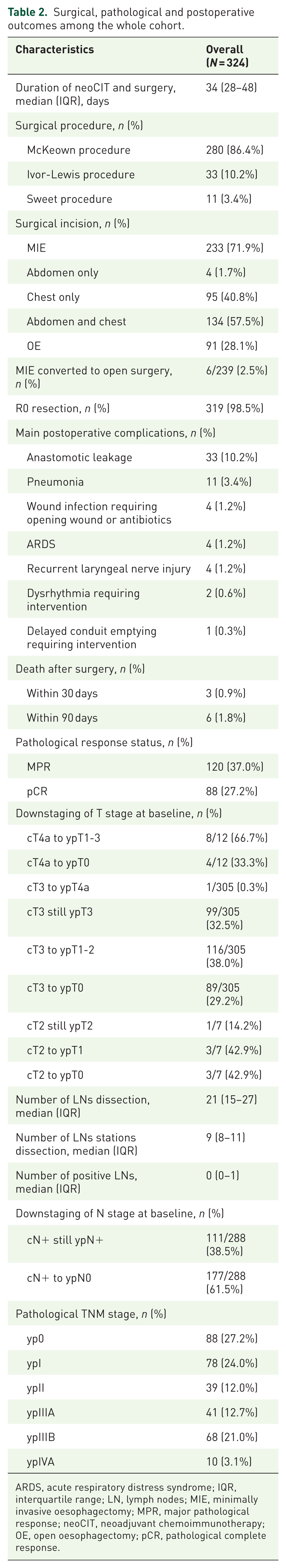
ARDS, acute respiratory distress syndrome; IQR, interquartile range; LN, lymph nodes; MIE, minimally invasive oesophagectomy; MPR, major pathological response; neoCIT, neoadjuvant chemoimmunotherapy; OE, open oesophagectomy; pCR, pathological complete response.
The main postoperative complications are summarised in Table 2. Anastomotic leakage occurred in 33 patients (10.2%). Additionally, six patients (1.8%) experienced nononcologic death within 90 days postoperatively.
Adjuvant therapy
Among the 318 patients who did not die during the first 90 days after oesophagectomy, 140 patients (44.0%) underwent adjuvant therapy (Table 3). The results showed that tumours located at the proximal third oesophagus, <3 cycles of neoCIT, McKeown procedure, R0 resection, early pathological T and N stage, MPR and pCR were significantly associated with a decreased likelihood of receiving adjuvant therapy (all p < 0.05. Table S2).
Adjuvant therapy and follow-up outcomes among patients who did not die within 90 days after oesophagectomy.
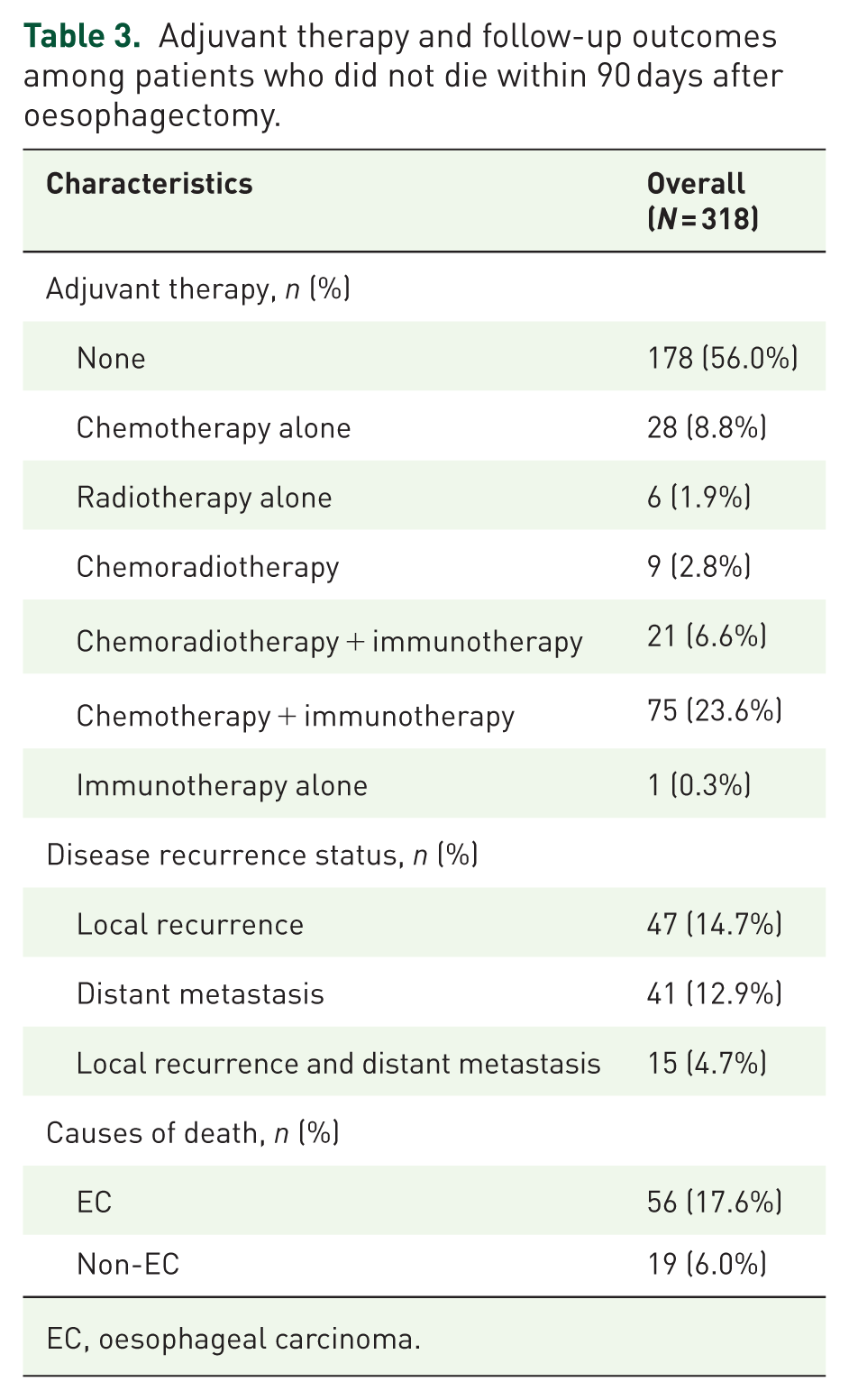
EC, oesophageal carcinoma.
Survival outcomes
As of 31 August 2024, the median follow-up time was 24.8 months, with an IQR of 19.9–32.2 months. The estimated DFS rate at 3 years was 62.3%, with a median DFS of not reached (Figure 3(a)). In the univariate survival analysis, sex, tumour location, histologic grade, number of cycles of neoadjuvant therapy, radiographic response status, resection status, pathological response status, pathological T stage, pathological N stage and adjuvant therapy were independently associated with DFS (all p < 0.05). After the above factors were incorporated into a Cox multivariate proportional hazards model, well-differentiated tumour, <3 cycles of neoadjuvant therapy, R0 resection, a lower pathological N stage and not receiving adjuvant therapy remained independent predictors of better DFS (all p < 0.05. Table S3).

Survival outcomes of the real-world cohort, with Kaplan–Meier curves showing DFS (a) and OS (b).
The median OS was not reached, and the estimated 3-year OS rate was 71.5% (Figure 3(b)). Similarly, through univariate and multivariate survival analyses, advanced pathological N stage (p < 0.001) was identified as the sole independent factor that correlated with worse OS (Table S4).
Discussion
Like the demographic features and treatment patterns reported in prospective clinical trials (Table S5), this real-world cohort across different centres revealed that patients who received two cycles (82.4%) of neoadjuvant PD-1 inhibitors (100%) combined with nab-paclitaxel (92.9%) and carboplatin (94.8%) were affected mainly by locally advanced (cT2-4aN1-3M0, 88.9%) and thoracic (99.1%) ESCC. The pCR outcome was achieved in 88 of the 324 patients (27.2%; 95% CI, 22.6%–32.3%), which was markedly higher than the pooled estimate of 9.0% (95% CI, 6.0%–14.0%) for neoadjuvant chemotherapy, 14 and numerically comparable to the pooled proportion of 32.0% (95% CI, 26.0%–39.0%) for neoCRT. 14
Over the past 4 years, numerous clinical trials have explored the efficacy and safety of conventional preoperative treatment in addition to anti-PD-1 antibodies followed by oesophagectomy for LA-ESCC.7,10 Although the various combination therapies all indicated favourable anti-tumour efficacy, the rates of MPR and pCR exhibited greater numerical differences. This presents new challenges for clinicians and patients making clinical decisions, as we do not know whether the differences in efficacy rates are due to variations in study designs. First, the present study and a meta-analysis reported no difference in the rate of MPR or pCR between different neoadjuvant PD-1 inhibitors. 15 An inconsistent finding from another meta-analysis was that patients who received pembrolizumab- and tislelizumab-based (vs toripalimab- and sinilimab-based) neoCIT had better rates of MPR and pCR. 8 However, a deeper review of the related protocols revealed that a greater proportion of studies designed ⩾3 cycles of pembrolizumab- or tislelizumab-based neoCIT for resectable ESCC, where a plausible interpretation is that the above statistically significant difference may be attributed to this confounding bias. 8 Second, the randomised phase III trial revealed that compared with neoadjuvant camrelizumab, nab-paclitaxel and cisplatin treatment could bring markedly improved rates of MPR and pCR compared to neoadjuvant camrelizumab, paclitaxel and cisplatin treatment. 9 However, as reported in basic research studies, dexamethasone can induce immunosuppression by blocking naïve T-cell proliferation and differentiation and abrogating anti-PD-1/PD-L1-mediated T-cell activation. 8 Thus, we should note that dexamethasone administration prior to paclitaxel infusion for allergic prophylaxis might compromise the clinical efficacy of preoperative camrelizumab treatment. 9 According to the current study and a recent meta-analysis, no difference was found in the efficacy outcomes between preoperative paclitaxel-based and nab-paclitaxel-based CIT. 8 Third, the optimal number of neoCIT cycles for LA-ESCC patients remains another contentious issue. Two meta-analyses align with our findings, demonstrating that, while patients with LA-ESCC who received ⩾3 cycles of neoCIT presented higher MPR and pCR rates than those treated with 2 cycles did, these differences did not reach statistical significance. 16 This real-world cohort study found that patients who received ⩾3 cycles (vs ⩽2 cycles) of neoCIT had worse DFS. This is primarily attributed to their poor response (stable disease and PD) to neoCIT and the subsequent increase in the number of neoCIT cycles, which may lead them to undergo salvage oesophagectomy. Taken together, future studies should comprehensively integrate molecular subtypes, the immune microenvironment and minimal residual disease to precisely inform the selection of neoadjuvant immunotherapy agents, chemotherapy drugs and the number of treatment cycles.8,17
Current clinical trials investigating neoadjuvant therapy for LA-ESCC do not routinely mandate adjuvant therapy.15,16 Although adjuvant therapy was administered in our clinical practice primarily to patients with nonradical resection, suboptimal pathological response or advanced postoperative stage to mitigate the risk of recurrence, the DFS was significantly worse in the group that received adjuvant therapy than in the group that did not. We postulate that this discrepancy may be driven by the fact that 99.7% of patients in the adjuvant therapy cohort received traditional chemotherapy and/or radiotherapy associated with substantial treatment-related toxicities and uncertain clinical efficacy. In the CheckMate 577 trial regarding patients with resected, LA-EC or gastroesophageal junction cancer who had received neoCRT and had residual pathological disease, adjuvant nivolumab therapy could reduce the risk of disease recurrence or death by 71% over placebo. 5 However, owing to differences in preoperative therapy strategies and pathological subtypes, whether postoperative immunotherapy could similarly improve survival in ESCC patients receiving neoCIT requires further validation through prospective randomised controlled trials. 18
The retrospective nature of this study necessitates careful consideration of the following limitations. First, the radiographic and pathological response statuses were assessed at the participating hospitals rather than through an independent review committee, potentially introducing selection bias or assessment discrepancies. Second, this study included a subset of patients who were unable to tolerate neoCIT-related adverse events or who exhibited suboptimal radiographic responses, leading to salvage oesophagectomy being performed, which subsequently impacted the objective evaluation of neoCIT efficacy and postoperative safety. Third, although the median follow-up duration of our cohort population exceeded 2 years, the analyses of long-term survival rates in ESCC patients remain methodologically premature. Fourth, this study was a single-arm investigation regarding neoCIT and lacked a control group receiving other neoadjuvant treatments. Fifth, adjuvant therapy had an adverse effect on prognosis, which was likely influenced by confounding factors, and therefore required extreme caution in interpretation.
Conclusion
The real-world study demonstrated that neoCIT achieved remarkable radiographic and pathological response rates in patients with resectable thoracic LA-ESCC. This multimodal therapy enables radical resection with manageable postoperative morbidity in the majority of cases while demonstrating promising survival outcomes.
Supplemental Material
sj-docx-1-tam-10.1177_17588359261443147 – Supplemental material for Treatment patterns and clinical outcomes of neoadjuvant chemoimmunotherapy for patients with resected oesophageal squamous cell carcinoma: a Chinese multicentre retrospective study
Supplemental material, sj-docx-1-tam-10.1177_17588359261443147 for Treatment patterns and clinical outcomes of neoadjuvant chemoimmunotherapy for patients with resected oesophageal squamous cell carcinoma: a Chinese multicentre retrospective study by Xiangyang Yu, Jianfei Zhu, Feng Wang, Kai Ma, Xiaotong Guo, Baihua Zhang, Lixu Wang, Xin Yu, Shengcheng Lin, Calvin S. H. Ng, Ran Yang, Jie He and Zhentao Yu in Therapeutic Advances in Medical Oncology
Footnotes
Acknowledgements
We would like to thank Dr Lei Liu, from Chengde Central Hospital, The Second Clinical College of Chengde Medical University, for supporting the testing of specimens from some of the enrolled patients and the exploration of future research direction through his research grant (No. 202204A017).
Authors’ note
Presented at the 33rd European Conference on General Thoracic Surgery of the European Society of Thoracic Surgeons, Budapest, Hungary, 25–27 May 2025.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
