Abstract
Background:
Postoperative early recurrence (PER) remains a major challenge to long-term survival after successful conversion therapy and curative resection for initially unresectable colorectal liver metastases (CRLM). Existing prediction models rely heavily on clinicopathological parameters and lack molecular biomarkers, limiting their predictive accuracy.
Objectives:
To define the optimal recurrence-free survival (RFS) cutoff for PER and develop a comprehensive predictive nomogram incorporating molecular and clinical variables to predict PER in patients with initially unresectable CRLM who undergo curative resection following conversion therapy.
Design:
Retrospective cohort study.
Methods:
Clinicopathological and molecular data from 411 patients with initially unresectable CRLM undergoing curative resection after conversion therapy were analyzed. The minimum p value approach determined the optimal RFS cutoff for PER. Least absolute shrinkage and selection operator regression identified significant predictors, followed by multivariate logistic regression to build a nomogram. Model performance was assessed using the area under the curve (AUC), calibration curves, and decision curve analysis (DCA).
Results:
PER was defined as recurrence within 4 months postoperatively. Independent predictors included dual preoperative positivity for CEA and CA19-9 (odds ratio (OR) = 2.437, p < 0.001), number of liver metastases (OR = 1.061, p < 0.001), tumor progression during the chemotherapy-to-surgery interval (OR = 2.837, p = 0.003), KRAS exon 2 mutations (OR = 1.927, p = 0.006), and BRAF V600E mutations (OR = 2.410, p = 0.002). An AUC of 0.703 (95% confidence intervals (CI): 0.650–0.756) was achieved, with an internal validation AUC of 0.697 (95% CI: 0.670–0.723). Calibration curves showed good agreement (p > 0.05), and DCA indicated clinical benefit at recurrence risk thresholds above 30%.
Conclusion:
We identified 4 months as the optimal RFS threshold for PER and proposed a novel nomogram integrating molecular and clinical factors for perioperative decision-making in patients with initially unresectable CRLM.
Plain language summary
Simple Summary: Predicting Early Cancer Recurrence After Liver Surgery
The Problem: Patients with advanced colorectal cancer that has spread to the liver (called “initially unresectable CRLM”) sometimes respond well to chemotherapy (“conversion therapy”) enough to allow surgery. However, cancer often returns quickly after surgery (“postoperative early recurrence” or PER), reducing long-term survival. Existing prediction tools don’t include genetic information and are not accurate enough.
What This Study Did? Researchers studied 411 patients who had liver surgery after successful chemotherapy. They aimed to:
1. Define what “early recurrence” means (how soon after surgery is “early”?).
2. Build a new prediction tool that combines both clinical factors and genetic biomarkers.
Key Findings
1. Timing of recurrence: Cancer returning within 4 months after surgery significantly lowers survival. This was defined as PER.
2. Top 5 predictors of early recurrence:
High levels of both tumor markers (CEA & CA19-9) before surgery
More tumors in the liver
Liver cancer growing during the wait for surgery after chemotherapy
KRAS gene mutation (exon 2)
BRAF gene mutation (V600E type)
3. Prediction tool performance:
The new model (a scoring system called a nomogram) was 70% accurate.
It works best for patients with >30% predicted recurrence risk, helping doctors decide if stronger treatments are needed.
Why This Matters? This is the first tool to combine genetic markers (KRAS/BRAF) with clinical factors to identify high-risk patients before surgery. Doctors can use it to:
Recommend closer monitoring after surgery
Offer personalized treatments (e.g., stronger post-surgery therapy)
Help patients understand their individual risks
Limitations: Based on past patient data (not real-time testing). Genetic testing may not be available everywhere. Further validation is needed.
Introduction
Recent advances in conversion therapy for colorectal liver metastases (CRLM) have expanded surgical opportunities for patients initially deemed unresectable. Through multimodal approaches combining systemic therapies such as chemotherapy and targeted agents with local interventions, including hepatic arterial infusion chemotherapy and radiofrequency ablation, a growing proportion of patients become eligible for curative-intent resection. Nevertheless, postoperative recurrence persists as a major clinical challenge, with reported recurrence rates ranging from 75% to 79% even after successful conversion therapy.1,2 Notably, patients converted from unresectable to resectable status experience significantly higher recurrence rates than those with initially resectable disease.3–5 Although postoperative early recurrence (PER) is strongly linked with worse clinical outcomes, there is no consensus on its temporal definition. Existing studies propose thresholds ranging from 6 months to 2 years for PER,2,6–9 reflecting both clinical heterogeneity and underlying differences in tumor biology.
Given these biological and clinical challenges, the present study focuses specifically on PER in patients with initially unresectable CRLM. This distinct subgroup is characterized by inherently aggressive tumor biology, highlighting the need for critical reevaluation of surgical decision-making frameworks. Current prognostic models10–13 are primarily derived from resectable CRLM data or heterogeneous populations, limiting their precision in assessing recurrence risks in candidates who underwent conversion therapy. To that end, we conducted a comprehensive assessment of PER risk in this subgroup of patients, incorporating baseline and preoperative clinical characteristics, serum biomarker levels, and molecular profiles. Ultimately, we aim to establish a robust predictive model that accurately identifies high-risk patient subgroups and supports the development of individualized treatment strategies.
Methods
Study population
This study retrospectively analyzed 411 CRLM patients who received curative resection at Sun Yat-sen University Cancer Center (SYSUCC) from 2006 to 2022 and subsequently developed recurrence. The flowchart for patient selection is presented in Figure 1. At first, a total of 686 patients with CRLM who experienced postoperative recurrence after initial liver metastasis resection were initially considered. Among them, 275 patients were excluded for the following reasons: 167 were deemed initially resectable by the multidisciplinary team (MDT), 20 had other malignant neoplasms, 51 lacked follow-up data, 11 were diagnosed with other malignancies on postoperative pathology, and 26 underwent palliative resection of liver metastases. Finally, 411 patients with initially unresectable CRLM who underwent R0/R1 resection following conversion therapy (primarily first-line regimens like FOLFOX/FOLFIRI/FOLFOXIRI plus bevacizumab or anti-EGFR antibodies), with or without adjunctive ablation or radiotherapy.

Flowchart for the selection of initially unresectable CRLM patients with recurrence following liver metastasis resection and overview of the training and validation procedure for the predicting models.
This study was developed and reported in accordance with the TRIPOD (Transparent Reporting of a multivariable prediction model for Individual Prognosis Or Diagnosis) statement 14 (Supplemental File 1).
Patients’ management
Definitions
In the present study, initially unresectable CRLM was defined based on the criteria used in the CELIM trial. 15 Specifically, CRLM was considered initially unresectable if there were five or more liver metastases or if the disease was deemed technically unresectable by an experienced MDT, based on the following criteria: tumor involvement of all hepatic veins, bilateral hepatic arteries, or bilateral portal vein branches, or an insufficient postoperative functional liver remnant volume. Synchronous metastasis is defined as intrahepatic metastatic lesions detected within 12 months of primary colorectal cancer diagnosis. 1 The formula for calculating tumor burden score (TBS) 16 was as follows: TBS² = (maximum tumor diameter)² + (number of tumors)². According to the standard operating procedures of the clinical laboratory at SYSUCC, preoperative tumor marker positivity was defined as CEA >5 ng/mL and CA19-9 >35 U/mL. The number of liver metastases was based on the definitive count obtained during surgery and pathology. Overall survival (OS) was defined as the time from hepatectomy to either death or final follow-up. Recurrence-free survival (RFS) was calculated from the date of liver metastasectomy to the date of recurrence. Post-recurrence survival (PRS) was defined as the time from the first relapse to either death or last follow-up.
Statistical analysis
All statistical analyses in this study were conducted using IBM SPSS Statistics 27 (IBM, NY, USA) or R (version 4.2.0). Missing data were excluded on a complete case basis. Differences of categorical variables in the distribution between groups were assessed using chi-square or Fisher’s exact test, as appropriate. p Values less than 0.05 were considered statistically significant. The survival curve was plotted using the Kaplan–Meier method, and the differences in survival distributions between subgroups were compared by the log-rank test. A minimum p value approach17,18 was employed to determine the optimal cut-off value for RFS, allowing stratification of patients into early or late recurrence groups based on differences in postoperative survival. Least Absolute Shrinkage and Selection Operator (LASSO) regression analysis was utilized to screen predictors, and the selected variables were incorporated into a multivariate logistic proportional hazards model using the survival package in R 4.2 to identify independent risk factors associated with RFS. Based on the results of multivariate analysis, a risk predictive model for PER in patients with initially unresectable CRLM was constructed using the rms package in R. Internal validation was conducted using the Bootstrap method with 1000 resamples. 19 The model’s discriminatory ability was assessed by calculating the area under the receiver operating characteristic curve (AUC), calibration was evaluated using calibration curves, and goodness-of-fit was assessed using the Hosmer–Lemeshow test. The clinical applicability of the prediction model was evaluated using decision curve analysis (DCA). 20
Results
Determination of the optimal RFS cut-off
The present study randomized 411 patients with initially unresectable CRLM into early and late recurrence groups. Multiple RFS cutoff values were systematically evaluated by calculating corresponding p values through log-rank tests to identify the optimal threshold that best distinguished PRS outcomes. As shown in Figure 2(a), the minimum p value (p = 4E−04) was observed when the RFS cutoff was set at 4 months, which was subsequently used to define early versus late recurrence.

Identification of RFS threshold for early and late recurrence. (a) Different cutoff thresholds (x-axis), with the corresponding p values (y-axis), show that the optimal threshold for defining early and late recurrence based on the difference in post-OS is 4 months. (b, c) Kaplan–Meier curves showing the difference between OS (b) and PRS (c) between the early and late recurrence groups.
Among patients with PER (RFS ⩽4 months; N = 162, 39.4%), the median RFS and PRS were 1.5 and 29 months, respectively. By contrast, patients with late recurrence (RFS >4 months; N = 249, 60.6%) had a median RFS of 8.5 months and a median PRS of 51 months. Of note, median OS was significantly longer in the late recurrence group compared with the early recurrence group (62 vs 32 months, p < 0.0001; Figure 2(b)). Similarly, the PRS was also significantly longer in the late recurrence group (p = 0.00012; Figure 2(c)).
Clinicopathological characteristics of the patients
Table 1 summarizes and compares the demographic, perioperative clinicopathological, conversion therapy, and molecular profiles between the PER and postoperative late recurrence (PLR) groups. Interestingly, patients in the ER group exhibited greater tumor burden, reflected by elevated CEA and CA19-9 levels, a higher number of liver metastases, and more aggressive molecular profiles, including KRAS exon 2 and BRAF mutations. Moreover, the PER group showed worse responses to conversion therapy, a lower incidence of left-sided primary tumors, and more frequent development of extrahepatic metastases. Notably, a higher proportion of patients experienced interval progression (IP) of liver lesions compared with the PLR group. No significant differences were found between the groups for other evaluated parameters.
Clinicopathological characteristics of the patients with recurrence.

Non-normally distributed data were presented as median and compared using the non-parametric Mann–Whitney U test. Values are presented as N (%) unless stated otherwise. p Values indicate statistical significance between groups (bolded for
IQR, interquartile range; OR, odds ratio; OS, overall survival; PER, postoperative early recurrence; PLR, postoperative late recurrence; PRS, post-recurrence survival; RFS, recurrence-free survival; SD, standard deviation.
ER occurrence and predictive factors
To develop a predictive model for ER, 9 of 21 candidate variables were identified through LASSO regression analysis (Figure 2) based on their non-zero coefficients. Key predictors included clinical factors such as IP of liver metastases between chemotherapy completion and surgery, number of liver metastases, and preoperative concurrent positivity for serum CEA and CA19-9, alongside molecular features encompassing KRAS exon 2 and BRAF V600E mutations.
A subsequent multivariate logistic regression analysis was conducted using the nine LASSO-selected variables to identify independent predictors of ER. As shown in Table 2, multivariable analysis identified the following independent predictors: KRAS exon 2 mutation (odds ratio (OR) = 1.927, 95% confidence interval (CI): 1.203–3.086; p = 0.006); preoperative dual positivity of CEA and CA19-9 (OR = 2.437, 95% CI: 1.458–4.072; p < 0.001); the number of liver metastases (per additional lesion: OR = 1.061, 95% CI: 1.029–1.095; p < 0.001); IP post-chemotherapy (OR = 2.837, 95% CI: 1.439–5.593; p = 0.003); BRAF V600E mutation (OR = 6.738, 95% CI: 2.045–22.200; p = 0.002). All variables demonstrated statistical significance (p < 0.05) and were confirmed as independent predictors of ER.
Results of multivariate logistic regression analysis for ER.

p < 0.05 indicates a statistically significant difference.
CI, confidence interval.
A nomogram was constructed to visually represent the predictive model, providing a practical tool for estimating individualized probabilities of ER (Figure 3). Of note, a total score exceeding 65 points corresponds to an estimated ER probability exceeding 90%.

Predictor selection using the LASSO regression with tenfold cross-validation. (a) Lambda selection for LASSO regression using minimum (left dashed line) and 1 − SE criteria (right dashed line). (b) Coefficient trajectories plotted against log(lambda), with five predictors retained under the 1 − SE criterion.
To apply the nomogram, point values corresponding to each patient-specific variable were first identified on the topmost scale. These individual points were then summed to yield a total score, which was subsequently used to estimate the predicted probability of early recurrence by referencing the corresponding value on the bottommost scale. For high/very-high-risk patients, score > ~60%–70% should be managed with intensified adjuvant chemotherapy, enhanced surveillance, and consideration for clinical trials.
The predictive model demonstrated moderate discriminative ability, achieving an AUC of 0.703 (95% CI: 0.650–0.756) in the primary analysis. Internal validation via bootstrap resampling (1000 iterations) produced a comparable AUC of 0.697 (Figure 4). Calibration curve analysis revealed good agreement between predicted and observed recurrence rates (Figure 5), supported by a nonsignificant Hosmer–Lemeshow test result (p = 0.661), indicating no substantial model overfitting.

Nomogram for predicting ER risk and its algorithm.
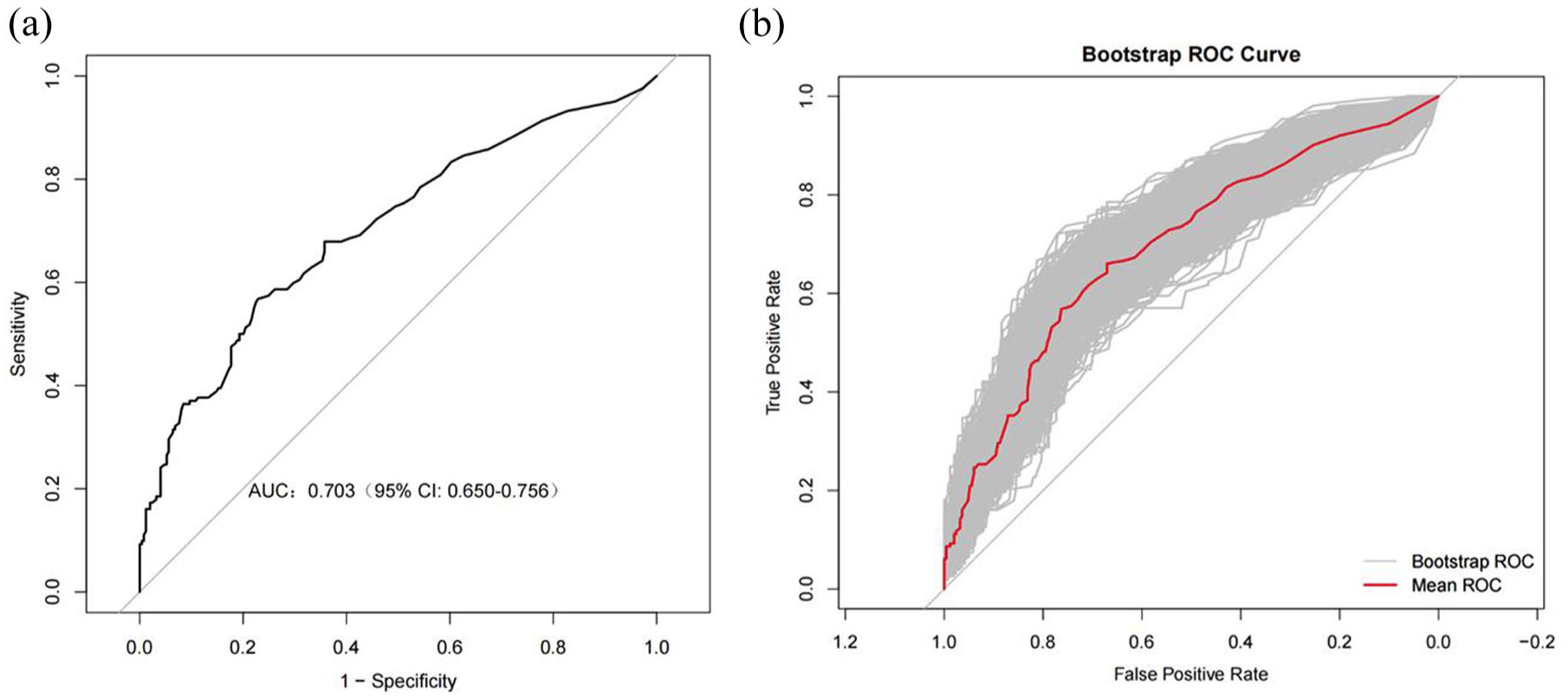
Discriminatory performance of the predictive model and internal validation. (a) The receiver operating characteristic curve and the AUC of the predictive model. (b) AUC of the model after internal validation using bootstrap resampling (n = 1000). The shaded gray area represents the 95% confidence interval.
Next, to assess its clinical usefulness, DCA was performed. As shown in Figure 6, the nomogram demonstrated meaningful net benefit when the predicted risk threshold exceeded 0.6 (60% probability), indicating strong clinical discriminative ability in identifying true high-risk patients. Together, our findings support the model’s potential to inform perioperative decision-making and optimize individualized management strategies (Figure 7).

Calibration curve of the predictive model illustrating the degree of consistency between the predicted and observed probabilities. The solid line represents the theoretically perfect calibration (bias-corrected ideal), while the dashed line reflects the model’s apparent predictive performance. The Hosmer–Lemeshow test yielded a p value >0.05, indicating a good model fit and no significant deviation between predicted and actual outcomes.

DCA of the nomogram. The red solid line represents the net benefit of the nomogram across a range of threshold probabilities for predicting early recurrence. The curve indicates that when the predicted risk of ER exceeds 0.6, the model provides a positive net clinical benefit, supporting its utility in identifying high-risk patients.
Discussion
PER, as a predictor of poor prognosis, has been consistently linked to worse OS in patients with various malignant tumors. Despite advances in CRLM management, there remains no consensus on the precise definition of ER. Existing definitions predominantly rely on arbitrary time cutoffs, (commonly between 6 and 12 months postoperatively), largely based on clinical experience rather than robust biological rationale or statistical validation that considers the heterogeneity in tumor recurrence dynamics. In this study, using a minimum p value approach applied to postoperative survival data, we identified an optimal RFS cutoff of 4 months to distinguish early from late recurrence in patients with initially unresectable CRLM. This threshold aligns closely with the widely accepted 6-month ER definition reported in previous studies.6,7 Subsequent survival analyses demonstrated significant differences in both postoperative OS and PRS between the early and late recurrence groups, highlighting that PER is a major determinant of long-term prognosis following radical resection.
When examining risk factors for ER in patients with initially unresectable CRLM, the number of liver metastases emerges as a critical determinant, in line with numerous studies. For instance, Abdalla et al. 21 reported that patients presenting with 10 or more liver metastases had a postoperative recurrence rate as high as 74.6% and a 5-year OS rate of approximately 30%. More recently, Viganò et al. 22 identified that the presence of over 15 liver metastases significantly increased the risk of very early recurrence (defined as within 3 months after liver resection). Our study mainly focuses on initially unresectable CRLM patients, who typically have a relatively large number of liver metastases, reflected by a median number of six liver metastases here. Consistent with prior findings, we observed that the risk of ER escalates with increasing numbers of metastases. 23 Clinically, patients with numerous small metastases are particularly susceptible to recurrence. This phenomenon may be explained by the higher likelihood of micrometastatic disease dissemination in such patients, with micrometastases often falling below the detection threshold of imaging modalities. Consequently, these lesions may lead to incomplete surgical clearance due to insufficient resection margins, thereby increasing postoperative recurrence risk. 24 These findings underscore the need for MDTs to optimize perioperative conversion therapy strategies, closely monitor imaging changes, and consider wider surgical excision or adjunctive ablation techniques during hepatectomy to improve oncologic outcomes. The number of liver metastases in this study was based on the final intraoperative and pathological counts, which is more objective. As these counts were largely consistent with preoperative imaging assessments, it can not only provide prognostic information but also hold potential significance for evaluating the benefit of surgical intervention, though this warrants further investigation. Fortunately, relevant clinical trials, an ongoing phase III RCT study (NCT06208371) evaluating localized interventions plus chemotherapy versus palliative chemotherapy alone in patients with 10 or more CRLM may determine which subgroups derive survival benefit from surgical resection.
Moreover, our study identified IP between chemotherapy completion and surgery as a novel independent risk factor for early postoperative recurrence, observed in approximately 12% of patients. This finding is consistent with the multicenter study by Viganò et al., 25 which demonstrated IP as an independent negative prognostic factor for both OS (hazard ratio (HR) = 3.144) and RFS (HR = 2.350). Previous research has established chemotherapy response as the strongest predictor of postoperative outcomes,26–28 with disease progression during systemic therapy often serving as a contraindication to surgical intervention. 26 Biologically, IP represents an aggressive tumor phenotype characterized by rapid proliferation and chemoresistance acquisition through epigenetic reprogramming or clonal evolution during treatment intervals. This dynamic tumor behavior contributes significantly to early recurrence and poor prognosis.7,29 While traditional static tumor staging and morphological assessments maintain prognostic value, they insufficiently capture the risks associated with evolving tumor biology. Although prospective validation in larger cohorts is warranted, our findings support the routine evaluation of IP in clinical practice. Patients demonstrating significant progression during the chemotherapy-to-surgery interval should undergo thorough reassessment of surgical candidacy to better tailor treatment strategies and improve clinical outcomes.
Serum tumor markers CEA and CA19-9 offer molecular-level insights into tumor biology and are routinely employed for diagnosis, surveillance, and prognostic stratification in colorectal cancer. Although current studies primarily recommend CEA for prognostic assessment,10,30–32 recent studies highlight that preoperative CA19-9 levels are also strongly associated with clinical outcomes.33–35 Herein, we found that preoperative CA19-9 positivity significantly correlated with ER (p < 0.001), suggesting that CA19-9 could serve as an independent predictive marker. Importantly, further analysis demonstrated that combining CEA and CA19-9 improved predictive accuracy; patients with dual positivity for both markers exhibited an early recurrence rate of 58%, significantly higher than the 33% observed in those with either single-marker positivity or dual negativity. Notably, our multivariate analysis confirmed dual positivity as an independent risk factor for ER (OR = 3.2, 95% CI: 1.8–5.6). These findings support the incorporation of combined preoperative CEA and CA19-9 assessment into standard clinical evaluation algorithms to improve individualized recurrence risk prediction.
In the context of molecular aggressiveness, our findings highlight KRAS and BRAF mutations as independent predictors of early recurrence, emphasizing the importance of routine multigene profiling in patients with initially unresectable CRLM. In the present cohort, KRAS mutations were detected in 39.7% of patients, consistent with previously reported incidences of 35%–45%.36–38 Of note, 34.2% of these mutations were localized to exon 2, specifically at codons G12 and G13. Compared to their wild-type counterparts, patients harboring KRAS mutations have been shown to exhibit a significantly greater incidence and burden of micrometastases (p < 0.001), 39 which, in turn, is associated with increased rates of R1 resection (OR = 2.41, 95% CI: 1.33–4.19) and occult residual disease, ultimately contributing to ER.39,40 Interestingly, KRAS-mutated subtypes demonstrated heterogeneous prognostic impacts. High-risk variants such as p.G12A and p.G13D were associated with a 40% reduction in post-recurrence median survival (HR = 1.40, p < 0.001) and lower resectability rates compared to wild-type patients (48.1% vs 70.1%), 41 while p.G12C and p.G12D mutations independently predicted worse post-recurrence outcomes (HR = 1.72, p = 0.003). 42 Although direct evidence linking KRAS exon 2 mutations to ER remains limited, a previous study revealed that postoperative detection of KRAS exon 2 mutations in serum correlated dynamically with minimal residual disease evolution, supporting its utility as a molecular surveillance marker. 43 This suggests that dynamic monitoring of circulating tumor DNA (ctDNA) could be integrated into recurrence risk assessment frameworks.44,45 By non-invasively tracking postoperative ctDNA clearance rates, clinicians may better predict recurrence risk and make more informed decisions regarding individualized interventions, such as intensifying adjuvant chemotherapy or considering secondary surgical resection.
BRAF mutations, particularly the V600E subtype, are well-established predictors of poor survival and recurrence in Colorectal Cancer (CRC). 46 Nonetheless, a critical clinical question remains: whether surgical resection offers a survival benefit over systemic therapy alone for patients harboring BRAF mutations. Evidence suggests that in cases with liver- or lung-limited metastases (LLM), surgical intervention may confer a significant advantage. In a cohort of 93 patients, those in the LLM subgroup who underwent local therapy achieved a 2.7-fold longer median OS compared to those receiving systemic treatment alone (HR = 0.16). 47 This finding is further supported by global data showing a 61% reduction in recurrence among patients with liver-limited BRAF V600E-mutant disease who underwent surgery. 48 In our cohort, 31.5% (6/19) of BRAF-mutant patients presented with extrahepatic metastases and experienced postoperative recurrence within 5 months. To conclude, oligometastatic CRLM (limited to a single organ) with favorable tumor biology and lower burden shows superior response to local therapy, emphasizing the necessity of stringent patient selection.
Herein, tumor progression during the interval between chemotherapy completion and surgery, the number of liver metastases, dual positivity of serum tumor markers before surgery, KRAS exon 2 mutations, and BRAF V600E mutation were identified as independent predictors for early postoperative recurrence. Collectively, our predictive model may assist in guiding clinical decision-making for patients undergoing liver metastasectomy after conversion therapy. Nevertheless, these findings do not support a definitive conclusion that surgery is futile in initially unresectable CRLM patients who experience ER, as a subset of patients has demonstrated prolonged survival through repeat hepatectomy after initial recurrence. 49
This nomogram provides an objective tool for postoperative recurrence risk stratification, designed to identify high-risk patients for intensified management and thus facilitate personalized adjuvant care.
Despite the novelties in this study, there are several limitations that should be taken into account. First, a key limitation of our model is that it requires external validation in independent cohorts before widespread clinical application, which will be the focus of our subsequent research. Second, the inclusion of patients with initially unresectable disease may limit the generalizability of our findings to the overall population of CRLM patients. Second, our study lacked specific information with regard to dynamic clinical factors like serum biomarkers or ctDNA, besides perioperative parameters. Lastly, as a single-center retrospective study, inherent limitations include selection bias and its extended timeframe.
Conclusion
The present study found that PER in patients with initially unresectable CRLM is associated with both high tumor burden and aggressive tumor biology. Significant predictors include the number of metastases and IP, KRAS/BRAF mutations, and dual CEA/CA19-9 positivity. Integration of these parameters into a biology-driven nomogram model could improve risk stratification and may guide postoperative surveillance and therapeutic decision-making.
Supplemental Material
sj-docx-1-tam-10.1177_17588359251411660 – Supplemental material for A clinical-molecular nomogram for predicting early recurrence following resection of initially unresectable colorectal liver metastases
Supplemental material, sj-docx-1-tam-10.1177_17588359251411660 for A clinical-molecular nomogram for predicting early recurrence following resection of initially unresectable colorectal liver metastases by Yu-Ting Lu, Xiao-Xia Huang, Wen-Chuan Chen, Qi-Yun Xiao, Jian-Li Duan, Zi-Jing Li, William Pat Fong, Feng-Hua Wang, Yu-Hong Li and De-Shen Wang in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
