Abstract
Background:
PSMA-negative but FDG-positive (PSMA−/FDG+) lesion in dual-tracer (68Ga-PSMA and 18F-FDG) positron emission tomography/computed tomography (PET/CT) is associated with an unfavorable response to Lutetium-177 (177Lu)-PSMA-617. This study sought to develop both radiomics and clinical models for the precise prediction of the presence of PSMA−/FDG+ lesions in patients with castration-resistant prostate cancer (CPRC).
Methods:
A cohort of 298 patients who underwent dual-tracer PET/CT with a less than 5-day interval was included. The evaluation of the prognostic performance of the radiomics model drew upon the survival data derived from 40 patients with CRPC treated with 177Lu-PSMA-617 in an external cohort. Two endpoints were evaluated: (a) prostate-specific antigen (PSA) response rate, defined as a reduction exceeding 50% from baseline and (b) overall survival (OS), measured from the initiation of 177Lu-PSMA-617 to death from any cause.
Results:
PSMA−/FDG+ lesions were identified in 56 (18.8%) CRPC patients. Both radiomics (area under the curve [AUC], 0.83) and clinical models (AUC, 0.78) demonstrated robust performance in PSMA−/FDG+ lesion prediction. Decision curve analysis revealed that the radiomics model yielded a net benefit over the ‘screen all’ strategy at a threshold probability of ⩾4%. At a 5% probability threshold, the radiomics model facilitated a 21% reduction in 18F-FDG PET/CT scans while only missing 2% of PSMA−/FDG+ cases. Patients with a low estimated score exhibited significantly prolonged OS (hazard ratio = 0.49, p = 0.029) and a higher PSA response rate (75% versus 35%, p = 0.011) compared to those with a high estimated score.
Conclusion:
This study successfully developed two models with accurate estimations of the risk associated with PSMA−/FDG+ lesions in CRPC patients. These models held potential utility in aiding the selection of candidates for 177Lu-PSMA-617 treatment and guiding 68Ga-PSMA PET/CT-directed radiotherapy.
Plain language summary
This study developed two models with accurate estimations of the risk associated with specific lesions in prostate cancer.
Introduction
Metastatic castration-resistant prostate cancer (mCRPC) remains incurable despite significant advancements in diverse drug regimens. The emergence of prostate-specific membrane antigen (PSMA) as a theragnostic agent with heightened expression in mCRPC lesions has revolutionized mCRPC management.1 –4 Lutetium-177 (177Lu) PSMA-617 is a radiolabeled small-molecule inhibitor that binds with high affinity to PSMA and delivers β particle radiation. The success of the VISION and TheraP trial has established the antitumor activity and favorable safety profile of 177Lu-PSMA-617 in men with mCRPC.1,2 A pivotal criterion for radioligand therapy (RLT) in both trials is the manifestation of high PSMA expression within the tumor, as determined by discernible tracer uptake on 68Ga-PSMA positron emission tomography/computed tomography (PET/CT) scans. 5 However, the substantial heterogeneity of metastases in prostate cancer (PCa), particularly in advanced mCRPC with de-differentiation, poses a challenge.6,7 Advanced disease exhibits reduced PSMA expression, leading to limited or absent uptake on 68Ga-PSMA PET/CT. This subset of patients experiences discordance between 68Ga-PSMA and 18F-fluorodeoxyglucose [FDG] PET/CT imaging, indicative of a more aggressive phenotype with suboptimal responses to PSMA-directed therapies.8 –11 The TheraP trial, in contrast to the VISION trial, implemented additional 18F-FDG PET/CT scanning to identify and exclude patients with FDG-positive but PSMA-negative (PSMA−/FDG+) lesions, resulting in the exclusion of 18% (52/291) of patients.1,2 Furthermore, the augmented assessment of tumor burden afforded by supplemental 18F-FDG PET/CT scans holds significant implications in the selection of candidates for metastases-directed stereotactic body radiotherapy (SBRT).12,13
The identification of patients at risk of discordant lesions in 68Ga-PSMA and 18F-FDG (dual-tracer) PET/CT represents a crucial advancement, contributing to enhanced precision in treatment strategies and a reduction in unnecessary costs.8,9,14 In light of this, our research endeavored to develop PSMA−/FDG+ lesion prediction nomograms, grounded in clinical data and 68Ga-PSMA-11 PET/CT tumor characteristics. The predictive efficacy of these nomograms will be systematically compared with the Renji model, the sole reported PSMA−/FDG+ lesion prediction model, employing dichotomized thresholding of Gleason score (GS; 8) and prostate-specific antigen (PSA; 7.9 ng/ml) to categorize patients into distinct risk groups. 15 In addition, we assessed the survival impact of the radiomics nomogram using an external cohort.
Methods
Study design and participants
Between April 2019 and January 2022, patients with CRPC who underwent dual-tracer PET/CT at two academic hospitals (Fudan University Shanghai Cancer Center, Shanghai, and Qilu Hospital, Shandong) were retrospectively assessed for eligibility based on predefined criteria. Inclusion criteria encompassed histological confirmation of prostate adenocarcinoma and the availability of a comprehensive medical record. Exclusion criteria included the presence of active malignancies other than PCa, histologic features indicative of pure neuroendocrine or small-cell cancer, and prior 177Lu-PSMA-617 treatment. CRPC was defined according to the European Association of Urology guidelines. 16 Ethical approval was obtained from the institutional ethics committees of both institutes, aligning with the Declaration of Helsinki. Informed consent requirements were waived due to the retrospective nature of the study. The reporting of this study conformed to the reporting recommendations for tumor marker prognostic studies. 17
PET/CT imaging and image analysis
Radiotracers were administered on different days with an interval of fewer than 5 days. For 18F-FDG PET/CT, patients fasted for at least 6 h. The blood glucose levels before the injection of the tracer should be lower than 10 mmol/L. Routine scans commenced 60 min post-tracer administration (3.7 MBq/kg). 68Ga-PSMA-11 PET/CT did not necessitate fasting, with patients ingesting 500 mL of water during a 2-h period before acquisition (2.0 MBq/kg) without dietary preparation. A 60-min interval was also adopted for uptake time. PET/CT scans were performed using a Siemens mCT Flow PET/CT scanner (Siemens Healthcare, Knoxville, TN, USA). A non-contrast-enhanced CT scan was performed using the following parameters: slice thickness of 3 mm, increment of 2 mm, and soft tissue reconstruction kernel, 120 keV. Immediately after CT scanning, a whole-body PET (from the level of the skull base to the knee) was acquired in 3D (matrix 200 × 200). A multimodality computer platform (Syngo; Siemens Healthcare) was used for image review and manipulation. Transaxial, coronal, and sagittal reconstructions of CT, PET, and fusion PET/CT data for interpretation can be produced by this system.
All scans were visually evaluated independently by three blinded nuclear medicine specialists with at least 5-year experience in PET/CT reading. Any assessment results inconsistency was resolved by joint discussion. During visual interpretation, each reviewer marked regions of suspected disease based on a two-point scale: zero as negative and one as positive. The region was graded as one only when a visually positive lesion was found. Positive lymph nodes [LNs] were verified only if the 68Ga-PSMA-11 or 18F-FDG uptake was locally accumulated and higher than the blood pool (mediastinal blood pool). Positive visceral lesions were verified only if the 68Ga-PSMA-11 or 18F-FDG uptake was locally accumulated and higher than the background activity of the surrounding involved organ or region. Positive bone lesions were verified only if the 68Ga-PSMA-11 or 18F-FDG uptake was locally accumulated and higher than physiologic bone marrow. The uptake of a lesion was semi-quantitatively expressed as the maximum standardized uptake value (SUVmax). The PET/CT imaging interpretation at two academic hospitals followed these prespecified protocols.
Development of the prediction models
A clinically driven, evidence-based approach guided variable selection. First, a very recent literature review was used to identify significant predictors for the presence of PSMA−/FDG+ disease. 7 Second, three consensus meetings were organized with clinical experts including urologists (JP, TZ, SC, TB, JW, DY, and YZ), two expert uroradiologists (CL and SS), and two uropathologists (HG and QW). The principles of variable selection were usefulness, availability, and relevance. Finally, 17 variables were selected for model construction. Variables included clinical information [including baseline PSA value, GS, distant metastases at diagnosis, history of abiraterone/docetaxel treatment, albumin (ALB), alkaline phosphatase (ALP), alanine transaminase (ALT), aspartate aminotransferase (AST), total bilirubin (TBIL), and lactate dehydrogenase (LDH)] and 68Ga-PSMA-11 PET/CT tumor characteristics [including the number of metastases, LN status, bone status, visceral status, SUVmax, and average SUV (SUVmean)]. Clinical laboratory assessments were done no more than 7 days before dual-tracer PET/CT.
Evaluation of the predictive performance of the models
The Akaike information criteria (AIC) determined the model with optimal discrimination, while the receiver operating characteristic curve (ROC) and area under the curve (AUC) gauged discrimination ability. Calibration curves were plotted via bootstrapping with 1000 resamples to assess the calibration of the developed models. Decision curve analysis (DCA) was conducted to estimate a net benefit for prediction models defined as the proportion of true positives minus the proportion of false positives.18,19 The net benefit of the model at each threshold probability was estimated.
In addition, survival data of 177Lu-PSMA-617-treated mCRPC patients with complete medical records from an external cohort (Nanjing cohort) 20 were utilized to evaluate the prognostic value of the radiomics model. Two endpoints were evaluated: (a) PSA response rate, defined as a reduction exceeding 50% from baseline 1 and (b) overall survival (OS), measured from the initiation of 177Lu-PSMA-617 to death from any cause.
Statistical analyses
Image findings and baseline characteristics were collected, with continuous data presented as medians and interquartile range (IQR), and categorical data as frequencies and percentages. Categorical variables were compared using the chi-square test or Fisher exact test. Continuous variables were compared using Student’s t-test or Mann–Whitney U test, when appropriate. Predictors of PSMA−/FDG+ lesion were identified through univariate logistic regression analysis, with variables (p < 0.05) included in stepwise multivariable logistic regression analysis. In addition, another two continuous parameters, SUVmax and SUVmean, were important reference indicators for selecting candidates for the 177Lu-PSMA-617 treatment.1,2,21 Thus, these two variables were included in the stepwise multivariable logistic regression analysis irrespective of the univariate analysis results. The Kaplan–Meier method was used to estimate OS outcomes. Hazard ratio (HR) and 95% confidence intervals (95% CI) were calculated. Statistical analyses were performed with R software (version 4.2.0; R Core Team, Vienna, Austraia).
Results
Metastases and heterogeneity detected by dual-tracer PET/CT
The workflow of our study design is outlined in Figure 1. A cohort of 298 patients with CRPC was incorporated into the study. The demographic and clinical characteristics of these patients are listed in Table 1 and Supplemental Table S1. The median age at the time of PET/CT was 68 (IQR, 63–73), with a corresponding median PSA level at enrollment was 2.5 (IQR, 1.1–8.0). Notably, 79.5% (237/298) presented with GS ⩾8 disease and 78.9% (235/298) exhibited PSMA-positive metastases.

Study flowchart.
Characteristics of the 298 patients in the study cohort.

FDG, fluorodeoxyglucose; LN, lymph node; PSA, prostate-specific antigen; PSMA, prostate-specific membrane antigen.
A total of 117 PSMA−/FDG+ lesions were identified in 56 patients (Supplemental Table S2/S3). These lesions were distributed across node [29 (24.8%)], bone [83 (70.9%)], and visceral [5 (4.3%)]. Of the 56 patients, 15 (36.6%) had multiple PSMA−/FDG+ lesions. The validation of PSMA−/FDG+ lesions involved several procedures: histological analysis after salvage LN dissection or dual-tracer PET/CT-guided target biopsy (n = 3), follow-up 18F-FDG PET/CT or conventional imaging confirmation (n = 108), or target radiation therapy with consecutive PSA declines of ⩽0.02 ng/ml (n = 6). Only one patient experienced interpretation discrepancy among three readers, attributed to a supraclavicular LN lesion with intense FDG uptake. A subsequent false-positive result was identified in this lesion following a tumor-free histopathological report post 18F-FDG PET/CT-guided target biopsy.
Development of nomogram
Among the 17 evaluated variables, 11 met the criteria for variable selection, qualifying for multivariable analysis (Table 2). Two models emerged: the radiomics model [Figure 2(a)] integrating clinical data and 68Ga-PSMA-11 PET/CT tumor characteristics, and the clinical model [Figure 2(b)] based solely on clinical variables. In the stepwise logistic regression analysis, the radiomics model, constructed by SUVmax, PSA, number of lesions, bone metastases, prior docetaxel therapy, and ALP, exhibited the lowest AIC value (224.13). Consequently, these six variables were selected for radiomics model construction and the subsequent generation of online calculator (https://peterjianfuscc.shinyapps.io/DynNomapp/). The radiomics model displayed an AUC of 0.83 [95% CI: 0.77–0.89; Figure 3(a)], with calibration curves illustrating a commendable alignment between predicted and actual PSMA−/FDG+ lesion positivity (Supplemental Figure S1). The clinical model, constructed by PSA, prior docetaxel therapy, and ALP, yielded the lowest AIC value (233.34) and an AUC of 0.78 (95% CI: 0.70–0.86).
Logistic regression model to predict the presence of PSMA−/FDG+ disease (n = 298).

CI, confidence interval; FDG, fluorodeoxyglucose; LDH, lactate dehydrogenase; LN, lymph node; PSA, prostate-specific antigen; PSMA, prostate-specific membrane antigen; OR, odds ratio; SUV, standardized uptake value; TBIL, total bilirubin.
A significant difference (p ⩽ 0.05).
Included in stepwise multivariable logistic regression analysis regardless of univariate analysis results.

Radiomics model and clinical model. (a) A radiomics model with clinical variables and 68Ga-PSMA PET/CT tumor characteristics incorporated. (b) Clinical model constructed based on clinical variables to predict the presence of PSMA−/FDG+ lesion.

Performance of radiomics model and clinical model in PSMA−/FDG+ lesion predicting. (A) Comparison of ROC between the radiomics model, clinical model, and Renji model for the prediction of the presence of PSMA−/FDG+ lesion. (B) DCA of the radiomics model, clinical model, and Renji model for the prediction of PSMA−/FDG+ lesion.
Model predictive performance analysis
Subsequently, the predictive capabilities of the developed nomograms were systematically evaluated in comparison with the established Renji model. 15 In our cohort, the Renji model exhibited a higher AIC value (269.36) compared to both the radiomics and clinical models, indicative of its inferior fit. Furthermore, the AUC value for the Renji model (0.67; 95% CI: 0.60–0.74) in our cohort was observed to be lower than that of both the radiomics and clinical models, underscoring its diminished discriminatory power. In DCA, both the radiomics and clinical models demonstrated a net benefit over either the ‘screen all’ strategy or the Renji model at threshold probabilities of ⩾4% and ⩾8%, respectively [Figure 3(b)]. The reduction in 18F-FDG PET/CT and the rate of missing PSMA−/FDG+ lesions were detailed across various threshold probabilities derived from the radiomics and clinical models (Table 3). Encouragingly, with a 5% cutoff, the radiomics model avoided 21% (62/298) of unnecessary 18F-FDG PET/CT scans while only missing 2% (1/56) of PSMA−/FDG+ cases. At a 10% probability threshold, the radiomics model averted 49% (145/298) of 18F-FDG PET/CT scans while missing 16% (9/56) of PSMA−/FDG+ lesions. Conversely, using a 10% cutoff from the clinical model resulted in a 27% (15/56) omission of men with PSMA−/FDG+ lesions but spared 67% (201/298) of additional 18F-FDG PET/CT scans.
Reduction in 18F-FDG PET/CT and number of PSMA−/FDG+ lesions missing according to the threshold probabilities of 5%, 10%, 15%, 20%, and 25% derived from the radiomics model and clinical model in included patients with CRPC.

CRPC, castration-resistant prostate cancer; FDG, fluorodeoxyglucose; PSMA, prostate-specific membrane antigen.
Association between PSMA−/FDG+ lesion risk and survival outcome post 177Lu-PSMA-617 treatment
The prognostic performance of the radiomics model was elucidated using a cohort of 40 eligible mCRPC patients from the Nanjing cohort (Table 4). 20 At baseline, the median age was 68 (IQR, 61–75), and the median PSA level at enrollment was 17.7 (IQR, 76.6–383.5). Remarkably, 92.5% (37/40) of patients presented with >5 PSMA-positive lesions, with 32.5% (13/40) having received prior docetaxel treatment.
Characteristics of the 177Lu-PSMA-617-treated patients from the Nanjing cohort, n = 40.

IQR, interquartile range; LN, lymph node; PSA, prostate-specific antigen; PSMA, prostate-specific membrane antigen; ALP, alkaline phosphatase.
The radiomics score was computed using the formula: radiomics score = 100 − 0.833 × SUVmax of 68Ga-PSMA-11 PET/CT (continuous variable) + 0.055 × PSA value (continuous variable) + 21.659 × number of lesions (categorical variable) + 19.244 × bone metastases (categorical variable) + 28.520 × prior docetaxel treatment (categorical variable) + 37.145 × ALP value (categorical variable). Patients with >5 PSMA-positive lesions, bone metastases, prior docetaxel treatment, or ⩾ULK ALP value were assigned a value of 1, the rest of the patients were assigned a value of 0. With a median follow-up of 10.9 (IQR 7.6–16.2) months, 87.5% (35/40) of patients died and PSA response was achieved in 55.0% (22/40) of patients [Figure 4(a)]. In addition, in comparison to patients with ⩾median predicted scores, those with <median predicted score exhibited a higher PSA response rate [75% versus 35%, p = 0.011; Figure 4(b)] and a significant prolonged OS [median 12.3 months versus 9.4 months; HR = 0.49, 95% CI = 0.25–0.97, p = 0.029; Figure 4(c)].
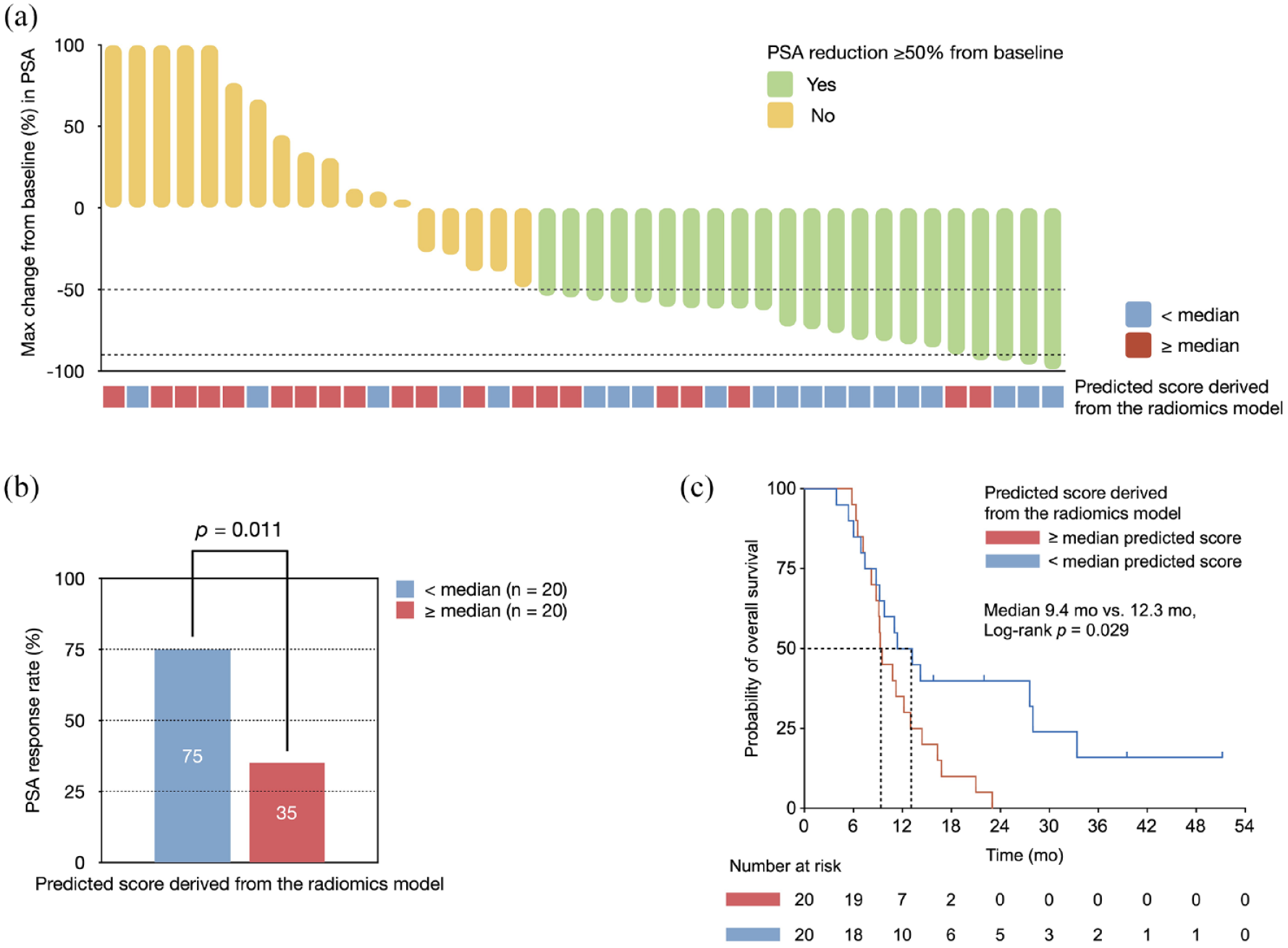
Survival data of the 177Lu-PSMA-617-treated patients with mCRPC from the Nanjing cohort (n = 40) stratified by the PSMA−/FDG+ risk according to the radiomics model. (a) Maximal percentage change from baseline in PSA in patients receiving 177Lu-PSMA-617. (b) Impact of predicted score on the PSA response rate. (c) Kaplan–Meier curves for OS according to the predicted score.
Discussion
In the context of a retrospective multicenter cohort, we crafted two nomograms to assess the risk of PSMA−/FDG+ disease in CRPC patients undergoing dual-tracer PET/CT. Notably, our radiomic model (AUC, 0.83) and clinical model (AUC, 0.78) showcased commendable efficacy, surpassing the antecedent model (AUC, 0.67). 15 Intriguingly, the probability of PSMA−/FDG+ disease exhibited a robust correlation with the PSA response rate and overall survival outcomes in patients with mCRPC subjected to 177Lu-PSMA-617 treatment. 20
Tumor heterogeneity emerges as a pivotal factor in treatment resistance and failure. 4 Patients scheduled for PSMA-directed therapy, including RLT and imaging-guided metastases radiation, often exhibit heterogeneity in tumor biology and prior treatments.1,2,22,23 In a retrospective study, Kerstin et al. stratified mCRPC patients treated with 177Lu-PSMA-617 according to the presence of PSMA−/FDG+ disease, revealing significantly lower OS rates in those with PSMA−/FDG+ disease despite comparable clinical characteristics. 24 TheraP trial’s exploratory analysis also highlighted the adverse impact of a high volume of 18F-FDG avid tumors on survival, irrespective of treatment assignment. 25 The driver role of FDG-positive tumors in CRPC was in accordance with our survival analyses. In addition, radio-ablation of PSMA−/FDG+ lesions would improve the outcomes of the CRPC patients. 13 In a prospective cohort, Pan et al. reported that 23.0% of CRPC patients had PSMA−/FDG+ disease, and radio-ablation of all PSMA−/FDG+ lesions resulted in a high PSA response rate (86.2% of patients had a PSA decline ⩾90%) compared to historical cohorts. 13 Therefore, an accurate estimate of the risk of PSMA−/FDG+ lesions may improve the selection for additional 18F-FDG PET/CT staging and subsequently optimize precision treatment with affordable cost. 26
The investigation by Chen et al. elucidated that 23.2% of patients exhibiting PSA progression during androgen deprivation therapy harbored at least one PSMA−/FDG+ lesion, and these patients demonstrated higher PSA and GS levels according to the Renji model. 15 Our model exhibited commendable performance across a spectrum of threshold probabilities compared with Renji model. In addition, we discerned that docetaxel-refractory CRPC, prognostically linked to poor outcomes in 177Lu-PSMA-617 treatment, was associated with a higher incidence of PSMA−/FDG+ disease. 21 The predictive value of some laboratory variables was evaluated. ALP, as a more bone-related parameter, demonstrated a robust correlation with the presence of PSMA−/FDG+ lesions. Interestingly, variable bone metastases also contributed to an augmented positivity for PSMA−/FDG+ lesions.21,27 Hence, we posited that a deeper exploration of bone-related parameters was merited, given their potential as metrics for worsening disease under 177Lu-PSMA-617 treatment. Furthermore, it was rational to consider SUVmax in 68Ga-PSMA PET/CT as another imaging parameter associated with PSMA−/FDG+ lesions. This association was plausible as tumor PSMA expression may decrease or be lost during various lines of treatment, potentially leading to more conspicuous positive 18F-FDG PET/CT findings. 7 These findings harmonized with the results in the multivariable survival analysis of 177Lu-PSMA-617 treatment, where these three variables retained their status as independent prognostic factors for PSA progression-free survival. 21 This consistency underscored the potential translation of predictive imaging phenotypes in CRPC into judicious applications of PSMA-targeted RLT. For enhanced clinical applicability, we meticulously devised both radiomic and clinical nomograms, adaptable to both community and academic practice, serving as valuable screening tools for patient referral and strategic treatment sequence arrangements. For example, patients with a low probability of PSMA−/FDG+ disease may be more inclined to benefit from RLT, while those with a high probability may discuss clinical trials if available.
Our radiomics nomogram, intricately tailored to predict PSMA-/FDG+ disease, found validation in an external cohort where the risk derived from the radiomics model correlated with the PSA response rate and OS after 177Lu-PSMA-617 treatment. These results substantiated the burgeoning body of evidence underscoring the unfavorable outcomes linked to PSMA−/FDG+ lesions. 28 Consequently, it is judicious to contemplate intensifying treatment strategies for patients with a heightened risk of PSMA−/FDG+ lesions, necessitating prospective evidence and precise interventions such as FDG+ tumor ablation or combination treatments.
To the best of our knowledge, this study represents the pioneering effort in generating PSMA−/FDG+ lesions prediction nomograms. Despite the commendable predictive performances of our models, several limitations warrant consideration. First, the retrospective nature of the study introduces the potential for selection bias. Second, the moderate sample size in the development cohort underscores the imperative for prospective validation with a more substantial patient population. However, with the increasing number of ongoing clinical trials (Supplemental Table S4) exploring the integration of 177Lu-PSMA-617 in the frontline setting of mCRPC and oligo-metastatic CRPC, our models may assume a pivotal role in further optimizing individualized care. Lastly, we could not estimate the predictive value of the DNA repair defects 14,29 and the activation of the PI3K-Akt-mTOR pathway,30,31 factors that may exert influence on the expression of PSMA.
In conclusion, our study culminates in the development of radiomic and clinical nomograms exhibiting promising predictive performances for PSMA−/FDG+ lesions in CRPC. Patients identified with a heightened risk of PSMA−/FDG+ lesions had a poor prognosis post 177Lu-PSMA-617 treatment. Our models, complemented by online risk calculators, stand poised to contribute significantly to individual medical decision-making and the meticulous design of clinical trials.
Supplemental Material
sj-docx-1-tam-10.1177_17588359231220506 – Supplemental material for Nomogram to predict the presence of PSMA-negative but FDG-positive lesion in castration-resistant prostate cancer: a multicenter cohort study
Supplemental material, sj-docx-1-tam-10.1177_17588359231220506 for Nomogram to predict the presence of PSMA-negative but FDG-positive lesion in castration-resistant prostate cancer: a multicenter cohort study by Jian Pan, Tingwei Zhang, Shouzhen Chen, Ting Bu, Jinou Zhao, Xudong Ni, Benkang Shi, Hualei Gan, Yu Wei, Qifeng Wang, Beihe Wang, Junlong Wu, Shaoli Song, Feng Wang, Chang Liu, Dingwei Ye and Yao Zhu in Therapeutic Advances in Medical Oncology
Footnotes
Acknowledgements
We thank the patients who participated in the studies. And we thank Hongru Shen, Yunhai Pan, Chunxiang Zhang, Runzhi Pan, Qiqi Pan, Rui Shi, Yiyao Huang, Jinghui Wang, Huiru Sun, and Zhijun Zeng, who gave strong support to the present study.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
