Abstract
Objective
In 2012, the Commission on Human Medicines mandated lowering the acetaminophen toxicity nomogram treatment threshold in the UK to 100 µg/ml at 4 h post-ingestion. The present study aim was to evaluate biochemical and liver toxicity patterns in patients who presented with acetaminophen overdose and had low serum acetaminophen concentrations (<150 µg/ml).
Methods
Patients admitted to the emergency department with a clear history of acute acetaminophen overdose with or without other medication or ethanol were consecutively enrolled into this retrospective cohort study. Patients with serum acetaminophen concentration >150 µg/ml or an unknown ingestion time were excluded. Data were extracted from electronic medical records and are presented as mean ± SD or median (interquartile range).
Results
A total of 103 patients were included (median age, 17 [4–21] years) and 80 (78%) were female. The median ingested acetaminophen dose was 5000 (2850–7650) mg. At baseline, the median serum acetaminophen concentration was 42 (4.5–64.8) µg/ml, and median alanine aminotransferase and aspartate aminotransferase levels were 22 (17–28) and 27 (16–45) IU/L, respectively. Twenty patients were treated with acetylcysteine, with none developing adverse reactions. No patient developed hepatotoxicity, including patients with initial multiple product ingestion or other risk factors.
Conclusions
Patients presenting with an acute acetaminophen overdose with acetaminophen level <150 µg/ml, including patients with other risk factors, are at low risk of hepatotoxicity.
Introduction
Acetaminophen toxicity is a commonly cited reason for emergency department visits and is the leading cause of acute liver failure in developed countries. 1 The Rumack–Matthew nomogram is intended to guide clinicians in predicting a patient’s risk of hepatotoxicity based on a plasma acetaminophen concentration ascertained within 4–24 h post-ingestion without acetylcysteine treatment. 2 The Rumack–Matthew nomogram is considered the mainstay of risk assessment in acute acetaminophen poisoning.
Patients falling above the threshold concentration of 200 µg/ml at 4 h after acute ingestion are at risk of hepatotoxicity, with rates of severe hepatotoxicity and mortality in these patients reported to reach 60% and 5%, respectively. 3 The Rumack–Matthew nomogram treatment threshold was reduced by 25% (equal to 150 µg/ml at 4 h post-ingestion) in the USA, at the request of the United States Food and Drug Administration, with the same adjustment also carried out in Australia and Canada. 4 In 2012, the Commission on Human Medicines in the UK mandated lowering the nomogram treatment threshold to 100 µg/ml at 4 h post-ingestion. 5 Bateman et al. 5 investigated 1246 patients with acetaminophen toxicity in the year preceding, and 1251 patients in the year following, the change in treatment threshold recommendation. The study revealed a 13.2% increase in patients requiring admission for acetylcysteine therapy in the year following the change and the number of patients with adverse reactions to acetylcysteine significantly increased from 87 to 145 after treatment changes. Similarly, anaphylactoid reactions occurred in more patients, but the rate was not significantly different from the year preceding the threshold change (29 [9%] versus 55 [10.7%], P = 0.426). 5 The nomogram utilizes population-based data. Therefore, the risk of developing complications, even with serum concentrations below the threshold recommended for initiating treatment, may exist in certain patients. There are insufficient data to address the clinical presentations of individuals with initial levels below the treatment threshold of < 150 µg/ml at 4 h post-ingestion.5,6 According to the guidelines of Dart et al., 7 glutathione depletion (e.g., starvation), enzyme-inducing medication, and chronic alcoholism were identified as risk factors for hepatotoxicity. 7 A separate study concluded that an initially normal prothrombin time (PT) or international normalized ratio (INR) is associated with a lower risk of developing hepatotoxicity. 8
Given the potential inaccuracy of patient histories, particularly regarding ingestion time, it is unsurprising that some cases with initial plasma concentration below the treatment line progressed to develop hepatotoxicity. 2 The aim of the present study was to investigate patterns of biochemical parameters and liver toxicity markers among patients who presented with serum acetaminophen concentrations below the nomogram threshold (150 µg/ml) to determine whether a conservative strategy demonstrates an increase in rate of positive patient health outcome and not just an overtreatment with little added benefit.
Patients and methods
Study design and setting
This retrospective cohort study was conducted at King Khalid University Hospital (KKUH), King Saud University Medical City (KSUMC), a tertiary-academic hospital with 1200 beds in Riyadh, Saudi Arabia. The study was conducted following the Helsinki Declaration of 1975 (as revised in 2013) and was approved in 2019 by the Institutional Review Board at King Saud University in Riyadh, Saudi Arabia (E-19-4040). This study is reported as per the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines. 9 All patient details have been de-identified to ensure confidentiality and privacy. The requirement of informed consent was waived by the Institutional Review Board due to the retrospective nature of the study.
Study population
Patients who had presented to the emergency department between June 2015 and June 2019 with acute acetaminophen ingestion, with or without other medications or ethanol administration, provided that the time of acetaminophen ingestion had been reported, were consecutively recruited. Patients who were positive for acetaminophen in the urine on comprehensive drug testing via gas chromatography-mass spectrometry, despite a negative quantitative serum acetaminophen concentration, or patients with initial serum acetaminophen concentration <150 µg/ml at 4 h post-ingestion, were included. Patients with initial acetaminophen concentration obtained at 4–24 h post-ingestion were also included if the concentration was below the 150 µg/ml line. Patients with serum acetaminophen concentration >150 µg/ml, or unknown ingestion time, were excluded. Likewise, patients with no proven acetaminophen toxicity by laboratory testing, even if clinically suspected, were excluded.
Data collection
Data regarding age, sex, weight, ingested acetaminophen dose, ingestion time, serum acetaminophen concentration(s), alcohol use, nutrition status, history of liver diseases, co-ingestion of other medications (e.g., opioids or antihistamines), and acetylcysteine use and dose were collected from electronic hospital records. Baseline and daily data for serum creatinine, blood urea nitrogen, alanine aminotransferase (ALT), aspartate aminotransferase (AST), bilirubin, PT, and INR were obtained for the first 48 h post-ingestion. Lastly, information on hepatic encephalopathy, pH value, and lactate were collected.
Statistical analyses
Categorical variables are presented as frequencies and percentages, while continuous variables are reported as mean ± SD and median with interquartile range (IQR) to highlight that data did not show normal distribution. Categorical variables were compared using χ2-test or Fisher’s exact test and continuous variables were compared using Student’s independent t-test. Continuous variables without normal distribution were analyzed with Mann–Whitney U-test. Data were analyzed using STATA software, version 14 (StataCorp LP, College Station, TX, USA) and a P-value <0.05 was considered statistically significant.
Results
A total of 778 patients were screened during the study period. Of these, 675 patients were excluded from the study (645 who proved to have no acetaminophen toxicity, 19 patients with unknown acetaminophen ingestion time, and 11 patients with acetaminophen serum concentration > 150 µg/ml). Consequently, 103 patients were included in the study. The median (IQR) age of the study population was 17 (4–21) years, and the median weight was 55 (17.4–65) kg. Female patients represented 78% of participants (80/103). Only 2% of patients had a history of alcohol use, and none had a history of liver disease upon admission. About half of the patients had co-ingested other medications with acetaminophen (antihistamines, opioids, and non-steroidal anti-inflammatory drugs), and 13 patients (12.6%) required hospital admission for further management. More detailed study population demographic and clinical characteristics are summarized in Table 1.
Study population demographic and clinical characteristics at baseline, 24 h and 48 h of hospital presentation in patients with acute acetaminophen ingestion.

Data presented as median (interquartile range), mean ± SD or n (%) prevalence.
Upon presenting to the emergency department, serum acetaminophen concentration was tested in all patients and the median concentration was 42 (4.5–64.8) µg/ml. The median total ingested acetaminophen dose was 5000 (2850–7650) mg, equating to 98.2 (59.5–153.8) mg/kg. The median time between patients’ arrival at the emergency department and the first determination of serum acetaminophen concentration was 62 (25–138) min. These results are summarized in Table 2.
Acetaminophen and acetylcysteine data from patients with acute acetaminophen ingestion.
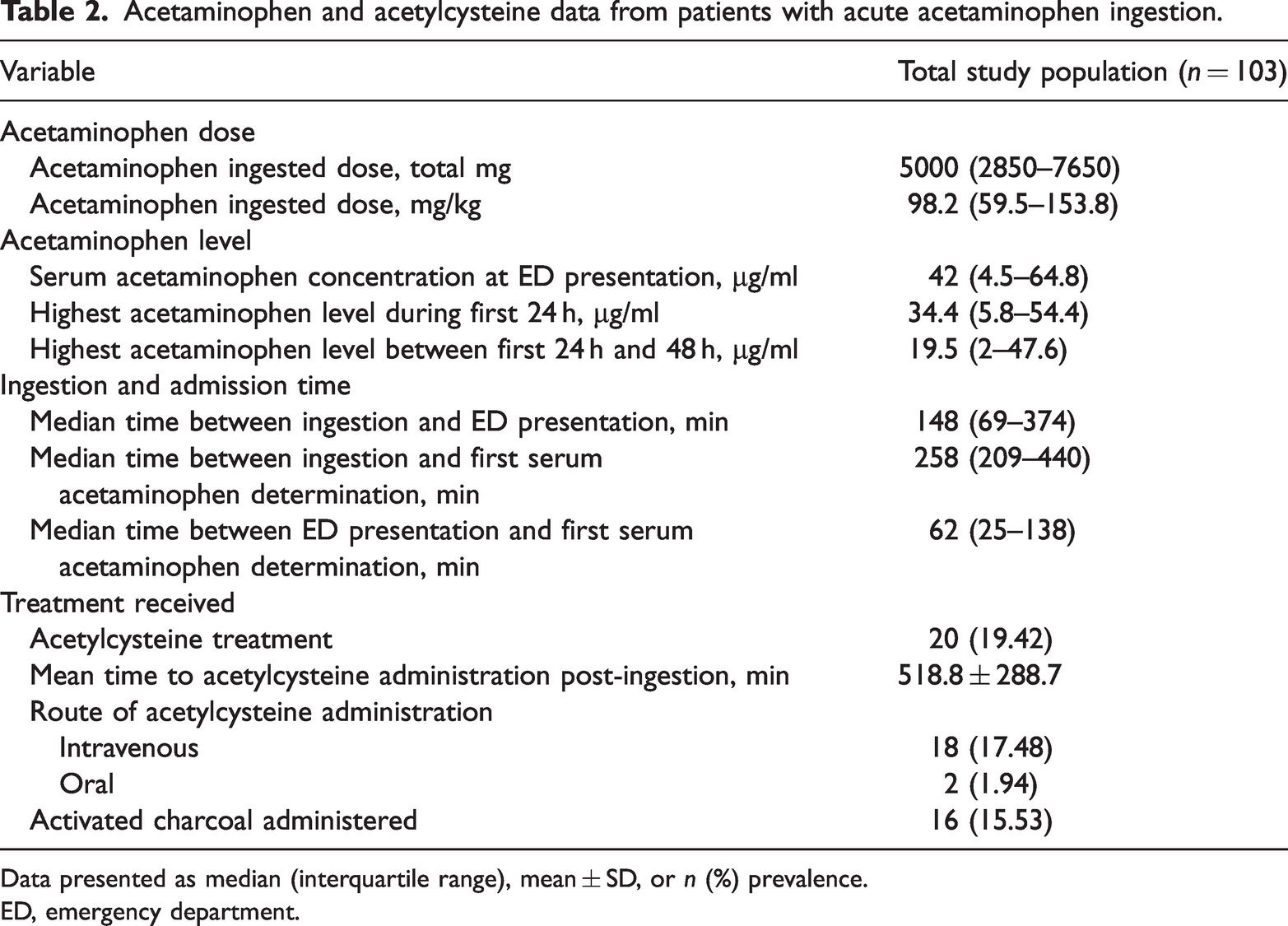
Data presented as median (interquartile range), mean ± SD, or n (%) prevalence.
ED, emergency department.
Relevant laboratory tests showed normal readings, where the median ALT, AST, and total bilirubin at baseline were 22 (17–28) IU/L, 27 (16–45) IU/L, and 6 (4–9) μmol/L, respectively. These values were slightly elevated but remained within normal range among patients followed for 24 or 48 h post-ingestion. Regarding the coagulation profile, the median PT was 14 (13.7–14.8) s, and INR readings were also normal. Additional applicable laboratory values are shown in Table 1.
A total of 16 patients (15.53%) were managed with charcoal only and 20 (19.42%) received acetylcysteine without any recorded adverse drug reaction for either treatment, or development of any hepatotoxicity, including in patients with initial co-ingestion or risk factors of hepatotoxicity.
When comparing the characteristics between patients who received acetylcysteine and those who did not, the mean baseline prothrombin time (15.7 versus 14 s, respectively) and mean total bilirubin at baseline (11.35 versus 6.85 mmol/L, respectively) were significantly higher among patients who received acetylcysteine than those who did not receive acetylcysteine (all P < 0.05). The mean bilirubin levels continued to be elevated among the acetylcysteine group, even at 24 h of admission (13.66 versus 3.10 mmol/L, respectively; P = 0.01). However, there were no statistically significant between-group differences in liver enzyme levels at baseline, or at 24 h or 48 h post-admission. The mean acetaminophen levels at presentation to the emergency department were higher among patients treated with acetylcysteine compared with those not treated with acetylcysteine (71.63 versus 38.46 µg/ml, respectively; P < 0.001), with no statistically significant between-group difference at 24 h; while at 48 h, patients in the acetylcysteine treatment group exhibited lower acetaminophen levels (10.30 versus 52.80 µg/ml, respectively; P = 0.01). Of note, patients in the acetylcysteine treatment group had ingested higher doses of acetaminophen versus those who did not receive acetylcysteine treatment (8759.71 versus 5617.25 mg, respectively; P = 0.01), but had significantly lower acetaminophen levels at 48 h. Further data comparing the two treatment groups are summarized in Table 3.
Comparison between patients with acute acetaminophen ingestion who received acetylcysteine versus those who did not.
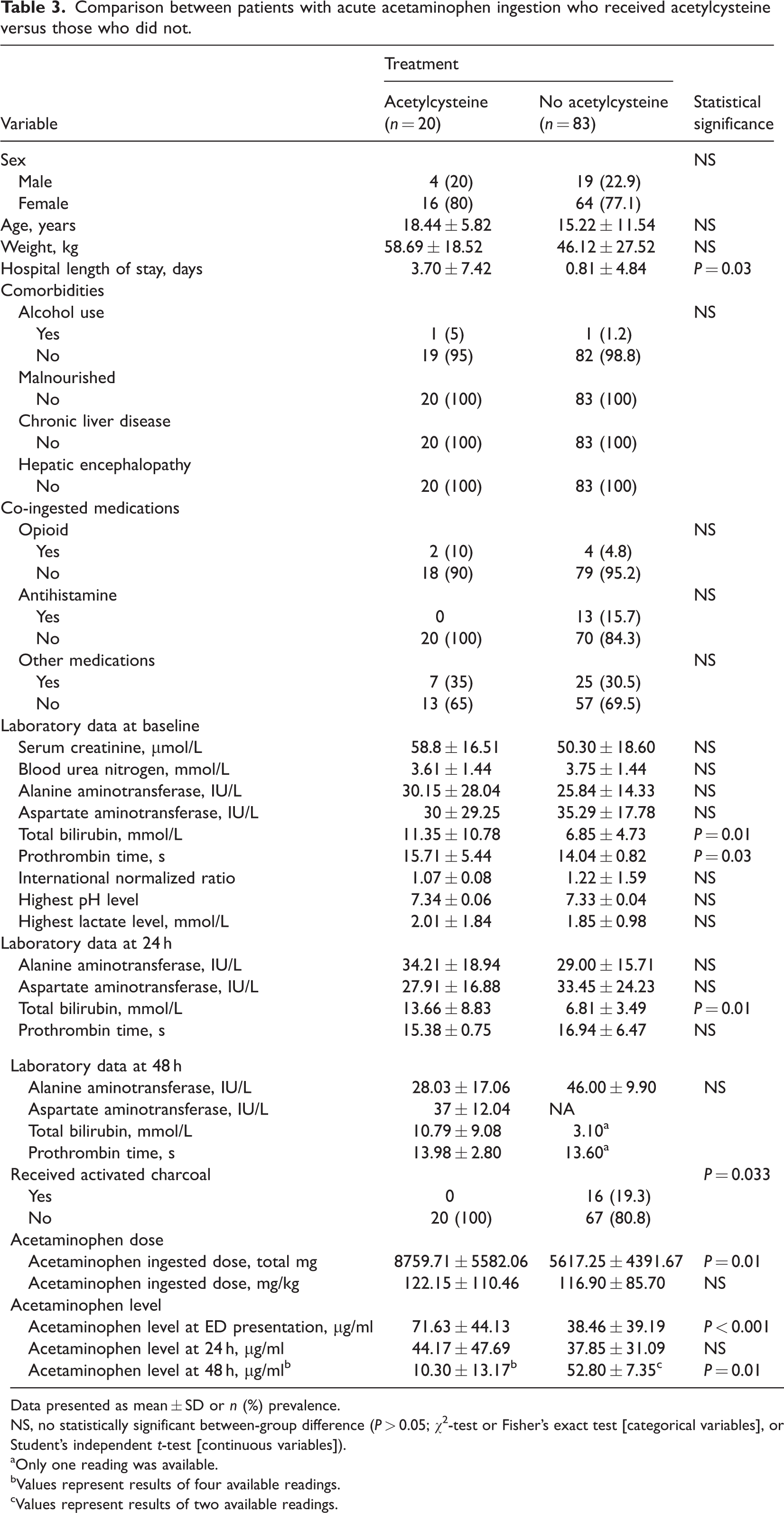
Data presented as mean ± SD or n (%) prevalence.
NS, no statistically significant between-group difference (P > 0.05; χ2-test or Fisher’s exact test [categorical variables], or Student’s independent t-test [continuous variables]).
Only one reading was available.
Values represent results of four available readings.
Values represent results of two available readings.
Discussion
The present study highlighted the clinical characteristics and presentations of patients considered to be at low risk of hepatotoxicity based on the Rumack–Matthew nomogram. The Rumack–Matthew nomogram provides a guide to the severity of acetaminophen overdose to determine the initiation of N-acetylcysteine (NAC) therapy. However, the nomogram cannot always be utilized, as patients may present late or acetaminophen ingestion may not have occurred in a one-time toxic ingestion. 10 Although the median serum acetaminophen concentration at presentation to the emergency department was 42 (4.5–64.8) µg/ml in the present study, which is far below the Rumack–Matthew threshold and values reported previously by Levine et al., 8 of 100 (44–179) µg/ml, it concurs with values reported in two previous studies (30 [0–660] µg/ml and 48 [23.1–114.4] µg/ml).5,10 In the present study, selected patients were admitted for further observation and/or NAC treatment. The use of biomarkers in patients with acetaminophen toxicity should be part of the management approach, 11 as acetaminophen concentration upon admission is not a cornerstone of either diagnosis or initiating NAC in cases of acetaminophen poisoning. Furthermore, it was reported recently that a significant percentage of patients with acetaminophen toxicity showed undetectable acetaminophen levels despite remarkable liver injury. 10
In the present study, markers of hepatic injury showed levels comparable to those of Levine et al. 8 However, other studies have revealed a marked rise in hepatic injury markers that may necessitate liver transplantation despite NAC therapy.10,12 These discrepancies may be attributed to many variables, including ingestion of massive doses, or concomitant medications, alcohol intake, starvation, older age, comorbid liver disease, and late presentation. In the present case, none of the patients developed hepatic encephalopathy, were scheduled for liver transplantation, or died.
Changes in AST and ALT activity have also been questioned for their ability to predict outcomes, as the development of hepatotoxicity is not consistently predicted by abnormal ALT or AST activity at presentation. In individuals receiving NAC treatment, initially normal ALT or AST activity was found to have a negative predictive value for the later development of hepatotoxicity. 13 In contrast, when combined in a predictive model, arterial lactate and serum creatinine were shown to be independent predictors of poor outcome and may be helpful in identifying individuals at risk. 14
At therapeutic doses, most acetaminophen is metabolized in the liver through glucuronidation and sulfation pathways into non-toxic metabolites. A small fraction undergoes metabolism by CYP450 enzymes, primarily CYP2E1, forming N-acetyl-p-benzoquinone imine (NAPQI), a toxic metabolite. A small amount is excreted unchanged in urine. In cases of overdose, the glucuronidation and sulfation pathways become saturated, leading to increased metabolism of acetaminophen by CYP450 enzymes to NAPQI. This metabolite is responsible for hepatocyte necrosis and, in severe cases, liver failure. Plasma acetaminophen concentration typically correlates with dose-dependent toxicity. The pathogenesis of acetaminophen-induced acute liver injury may involve not only the direct hepatotoxic effects of NAPQI, but also interactions with immune cells, including macrophages, neutrophils, and various lymphoid cell types. These cells represent an inflammatory response to acetaminophen-induced liver injury.15–18
Regarding the management of acetaminophen toxicity in the present study, most included patients (78.4%) were managed conservatively, and the remaining received either NAC (19.42%) or activated charcoal (15.53%). A study into the impact of lowering the acetaminophen overdose treatment threshold to 100 mg/L concluded that the number of patients admitted and treated with acetylcysteine increased without reducing adverse reactions. 6 This supports the present findings, indicating that intentional non-adherence to guidelines might be a conscious choice by physicians and is not influenced by external factors. However, adherence places a significant cost burden on the healthcare system by overwhelming the financial resources that may be allocated for other interventions.
Administration of NAC at acetaminophen concentration levels below 150 µg/ml may be justified for certain high-risk groups, such as patients with chronic alcohol use disorders, those receiving enzyme-inducing medications, and those with malnutrition. There is no specific ‘cutoff value’ for NAC treatment below the Rumack–Matthew line. Initiating NAC when the acetaminophen concentration is below the line remains at the physician's discretion, and the closer the level is to the treatment line, the more likely the patient will receive NAC. There is occasional concern about the accuracy of the provided history; for instance, the time of ingestion may be inaccurate (the patient may provide different times upon repeated questioning), or unknown, prompting the physician to commence NAC therapy for safety reasons.5,19
In the present study, an increase in acetaminophen levels was observed at 48 h compared with 24 h in patients who did not receive NAC treatment. It is important to note that data for 48-h levels were only available for a small number of patients (four in the NAC-treated group and two in the group with no NAC treatment). Nevertheless, potential explanations include the possibility of faster and more efficient metabolism in those receiving NAC, however, acetaminophen levels would be expected to decrease over time. The elevation in levels at 48 h may be attributed to delayed gastric emptying and absorption of acetaminophen resulting from concurrent ingestion of medications that slow gut motility (such as opioids, antihistamines/anticholinergic agents) or the formation of bezoars. Another possible explanation is accidental acetaminophen ingestion or administration during hospital admission. 20
The present study investigated the demographic and clinical characteristics of 103 patients with serum acetaminophen concentrations below the Rumack–Matthew nomogram threshold (150 µg/ml). However, the study results may be limited by several factors. Firstly, the single-center, retrospective study design limits the broader applicability of the findings. Secondly, the small sample size precluded adjustments for potential confounding variables. Moreover, the study did not consider the costs associated with NAC use, which is crucial for monitoring resource allocation within the healthcare system. Further research is warranted to address these limitations. Furthermore, the reliability of patient-reported ingestion times poses a potential limitation. Lastly, the absence of data on the type of acetaminophen formulation consumed in attempted overdose further limits the results. For instance, the absorption of extended-release acetaminophen is slower than that of immediate-release acetaminophen, which may lead to lower acetaminophen levels at presentation but subsequent elevation with continued absorption.
Conclusions
In the absence of consensus regarding risk assessment and treatment initiation for patients not meeting the criteria of the Rumack–Matthew nomogram, various clinical scenarios may arise, prompting questions about NAC treatment initiation. Despite the limitations of the present single-center, retrospective study design and the modest sample size, the study offers insights into the demographic and clinical profiles of patients presenting with acetaminophen toxicity below the nomogram threshold. The present findings suggest that initiating NAC treatment in this population may not be necessary unless there are concerns about the accuracy of the history provided, or the presence of other compelling factors, which may be justified for certain high-risk groups, including patients with chronic alcohol use disorders, those receiving enzyme-inducing medications, and those with malnutrition. Practitioners may consider establishing an estimated cutoff below the treatment line in the Rumack–Matthew nomogram, considering clinical characteristics and how far the serum acetaminophen concentration is situated below the nomogram threshold. Future studies should aim to elucidate the role of clinical characteristics and the degree to which serum acetaminophen concentration falls below the treatment line in guiding treatment decisions. Additionally, investigating the efficacy and potential risks associated with establishing an estimated cutoff below the treatment line in the nomogram may provide valuable insights for clinical practice.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605241260362 - Supplemental material for Patterns of acetaminophen toxicity among patients with low-risk serum concentrations
Supplemental material, sj-pdf-1-imr-10.1177_03000605241260362 for Patterns of acetaminophen toxicity among patients with low-risk serum concentrations by Abdullah M. Alhammad, Ghada Alajmi, Aljawharah Alkhodair, Wael H. Mansy, Mohammad H. Aljawadi, Rania Aljadeed, Reem Alshammari, Raghad Alshehri, Mohammed N. Alarifi and Badr Alyahya in Journal of International Medical Research
Footnotes
Acknowledgments
The authors thank the Researchers Supporting Project number (RSPD2024R919), King Saud University, Riyadh, Saudi Arabia, for their support.
Author contributions
All authors made substantial contributions to the study conception and design, and the acquisition, analysis and interpretation of data, and/or took part in drafting the article or revising it critically for important intellectual content. All authors agreed to submit the article to the current journal, gave final approval to the version to be published, and agreed to be accountable for all aspects of the work.
Data availability
The dataset supporting the conclusions of this article is available by request from the corresponding author.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
