Abstract
Background:
Serum C-reactive protein (CRP) level is frequently considered a prognostic predictor of immunotherapy. However, the prognostic role of the interim serum CRP levels before the third course of immunotherapy (CRP-C3) in patients with metastatic colorectal cancer (mCRC) receiving immunotherapy remains unclear.
Objectives:
This study aimed to determine the predictive value of CRP-C3 for immunotherapy response and progression-free survival (PFS) and to compare its performance with other biomarkers, such as tumor mutational burden (TMB).
Design:
We conducted a retrospective cohort study of 205 patients with mCRC who received immunotherapy.
Methods:
Baseline CRP (CRP-B) and CRP-C3 levels were measured in this retrospective cohort study. Clinicopathological data were retrospectively analyzed using Cox regression analyses to identify independent predictive variables for immunotherapy response and PFS, then summarized in nomograms to predict prognosis.
Results:
The median PFS periods were 8.47 months in the CRP-C3-low subgroup and 2.47 months in the CRP-C3-high subgroup (p < 0.001). The objective response rate (ORR) was 40.6% and 8.9%, respectively (p < 0.001). Although CRP-B failed in the multivariate Cox analysis, CRP-C3 performed exceptionally well, surpassing TMB levels (both p < 0.001). Moreover, a prognostic model combining CRP-C3 and TMB levels could significantly stratify patients with mCRC receiving immunotherapy into low-, intermediate-, and high-risk subgroups (p < 0.001). Patients from different risk subgroups exhibited significantly different ORR (low- vs intermediate- vs high-risk groups: 59.2% vs 32.5% vs 0%, respectively, p < 0.001) and Kaplan–Meier survival curves (p < 0.001).
Conclusion:
This retrospective study indicates a potential association between decreased CRP-C3 levels and improved PFS in patients with mCRC receiving immunotherapy. The combination of CRP-C3 and TMB appeared to enhance predictive performance, suggesting their possible utility as prognostic indicators.
Keywords
Introduction
Colorectal cancer (CRC) is the second leading cause of cancer-related death worldwide, with approximately 900,000 deaths annually. 1 The prognosis of metastatic CRC (mCRC) remains poor, as evidenced by a discouraging 5-year survival rate of approximately 14%. 2 Immunotherapy has offered promising results in CRC cases exhibiting genomic instability characterized by high tumor mutational burden (TMB-H), deficient mismatch repair, or microsatellite instability-high (MSI-H).3,4 Additionally, a combined approach involving immunotherapy and antiangiogenic agents has demonstrated remarkable outcomes in patients with CRC with genomic stability typified by low TMB (TMB-L), proficient mismatch repair (pMMR), microsatellite instability-low (MSI-L), and microsatellite stable (MSS) status.5,6
However, the efficacy of immunotherapy in patients with mCRC exhibits significant heterogeneity, necessitating the exploration of effective predictors. The systemic inflammatory response is associated with prognostic outcomes in various malignancies. 7 The level of C-reactive protein (CRP), a marker of acute inflammation, is often moderately elevated in chronic inflammatory conditions. 8 In cancer, the tumor microenvironment serves as a site for CRP-mediated modulation of the inflammatory milieu. 9 Previous studies have emphasized the correlation between serum CRP levels and the incidence and behavior of diverse tumors, such as lung cancer 10 and CRC. 11 Furthermore, baseline serum CRP concentration potentially serves as a predictive marker for assessing the efficacy of immunotherapy in solid tumors, such as non-small cell lung cancer, 12 hepatocellular carcinoma, 13 and urothelial carcinoma. 14 However, the prognostic significance of interim serum CRP levels before the third course of immunotherapy (hereafter referred to as CRP-C3) in patients with mCRC undergoing immunotherapy remains unclear.
Therefore, this retrospective study aimed to investigate the potential role of baseline and interim serum CRP levels in predicting the efficacy of immunotherapy in patients with mCRC, similar to TMB status.
Materials and methods
Patients
Patients were considered eligible for inclusion in the study if they met all of the following criteria: (1) histologically confirmed diagnosis of mCRC, (2) received immunotherapy (as monotherapy or in combination with other therapies) at Sun Yat-sen University Cancer Center between January 2019 and December 2023. Patients were excluded if they met any of the following conditions: (1) absence of baseline or on-treatment CRP measurements (n = 48), (2) presence of other malignancies or severe diseases affecting the immune system (n = 5), (3) absence of baseline and post-treatment radiological assessments (n = 8), (4) receipt of immunotherapy prior to the defined study period (n = 25), or (5) receipt of immunotherapy in the neoadjuvant or adjuvant setting (n = 17; Supplemental Figure 1). After applying these criteria, a total of 205 eligible patients were included in the retrospective cohort study and were subsequently categorized into CRP-C3-high and CRP-C3-low groups. This study involved a comprehensive review of patients’ medical histories, laboratory findings, and radiological results. Drug dosages and treatment intervals were in accordance with the recommended guidelines, with appropriate adjustments based on individual patient characteristics. Genetic testing was performed on the biopsy tissues or surgical specimens obtained from these patients. Radiological response to treatment was evaluated using computed tomography (CT) or magnetic resonance imaging (MRI) initially at baseline, then 6–12 weeks after treatment initiation, and approximately every 2 months thereafter, following the Response Evaluation Criteria in Solid Tumors (RECIST) version 1.1 (EORTC, Brussels, Belgium; NCI, Bethesda, MD, USA; and CCTG, Kingston, ON, Canada).
Data collection
Demographic and clinical characteristics included age, sex, body mass index (BMI), smoking status, CRP level (mg/L), neutrophil-to-lymphocyte ratio (NLR), Eastern Cooperative Oncology Group (ECOG) performance status (PS), tumor size (cm), primary tumor location, microsatellite status, TMB, and prior treatments. Smoking status was classified according to established criteria commonly used in lung cancer research 15 : never smokers (fewer than 100 cigarettes smoked over a lifetime), former smokers (ceased smoking ⩾1 year before diagnosis or treatment), and current smokers (active smokers or those who quit <1 year before diagnosis or treatment). In this study, smoking status was dichotomized for analysis, with No representing never smokers and Yes representing both former and current smokers.
Serum CRP levels and NLR values were assessed within 5 days prior to immunotherapy initiation, and serum CRP level was reassessed before CRP-C3. All reagents used for CRP and NLR detection adhered to the World Health Organization standards. Imaging modalities, such as CT, MRI, and/or ultrasonography, were utilized to detect tumor size, number, lymph nodes, metastatic locations, and tumor recurrence. Progression-free survival (PFS) was defined as the duration from the initiation of immunotherapy to CRC relapse, progression, or death. Tumor responses to immunotherapy were categorized as complete response (CR), partial response (PR), stable disease (SD), and progressive disease (PD) based on RECIST version 1.1. The reporting of this study conforms to the STROBE statement 16 (Supplemental Table 1).
Statistical analyses
Categorical variables were analyzed using Pearson’s chi-squared test. The cutoff value for CRP-C3 (best cutoff value = 27.1 mg/L) was determined using the X-tile application (Figure 1). 17 Based on the cutoff value, the patients were divided into different groups for further analysis. Independent predictive variables for PFS were determined using univariate and multivariate Cox regression analyses for PFS with the Forward-Looking Linear Regression approach. The risk stratification survival curves were represented by Kaplan–Meier curves and analyzed using the log-rank test. Statistical analyses were performed using the IBM SPSS statistics version 19.0 (IBM, Armonk, NY, USA). A nomogram for predicting the PFS or overall survival (OS) of patients with mCRC, incorporating CRP-C3 and TMB, was constructed using R version 3.5.1. All tests were two-sided, and statistical significance was set at p < 0.05.
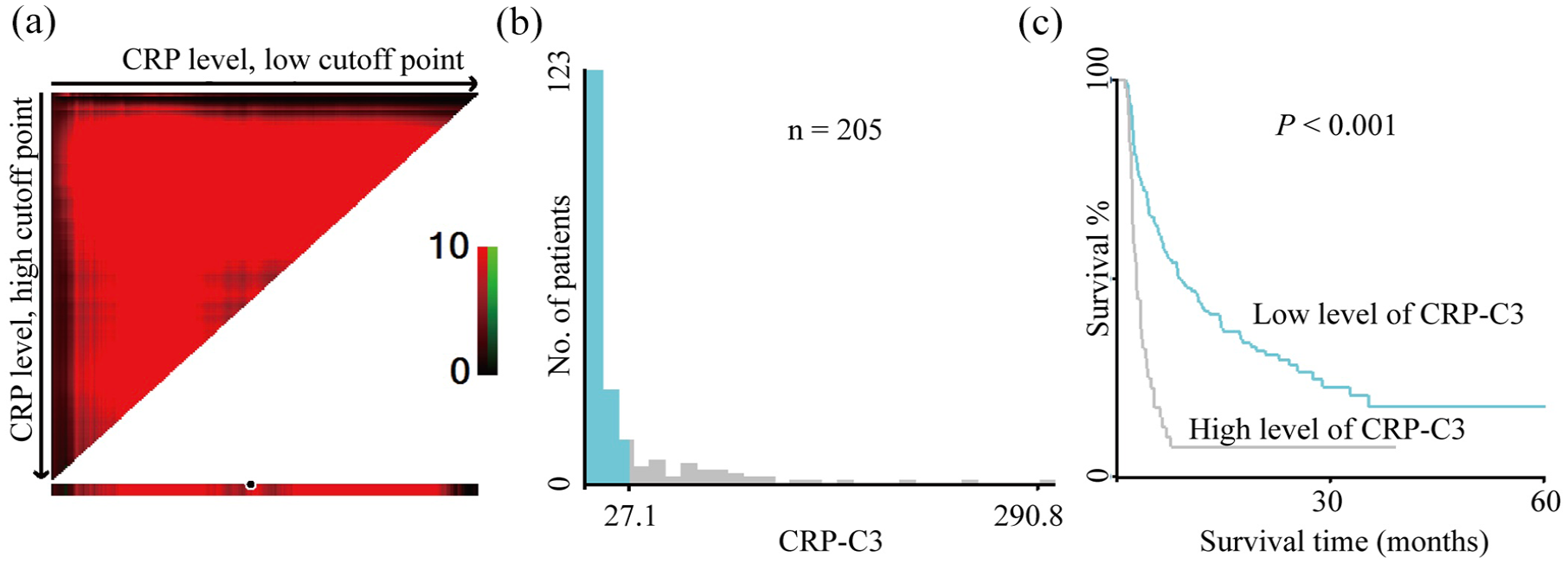
X-tile plots of the prognostic marker CRP-C3 in patients with mCRC receiving immunotherapy. Xtile plot showing the χ2 log-rank values created when the cohort was divided into two populations. The cutoff point highlighted by the black/white circle (a) is demonstrated on a histogram of the entire cohort (b) and a Kaplan–Meier plot (c). CRP levels were divided at the optimal cutoff point, as defined by the most significant point on the plot (⩽27.1 and >27.1 mg/L for patients with mCRC receiving immunotherapy, p < 0.001).
Results
Patient characteristics
A total of 205 patients with mCRC received immunotherapy at the Sun Yat-sen University Cancer Center. In this cohort, 130 (63.5%) patients were male and 75 (36.6%) were female, with an average age of 50.5 years. Regarding microsatellite status, 164 (80.0%) patients exhibited the MSS or MSI-L status, whereas 41 (20.0%) patients exhibited the MSI-H status. In terms of treatment regimens, 30 patients (14.6%) received immunotherapy alone, 15 (7.3%) received immunotherapy combined with chemotherapy, 120 (58.5%) received immunotherapy combined with targeted therapy, and 40 (19.5%) received a combination of immunotherapy, chemotherapy, and targeted therapy. Table 1 provides a comprehensive summary of patients’ baseline clinical characteristics.
Clinical characteristics of patients with mCRC categorized based on CRP-C3 levels.

Variables are expressed as n (%).
BMI, body mass index; CRP-C3, C-reactive protein levels before the third cycle of immunotherapy; ECOG PS, Eastern Cooperative Oncology Group Performance Status; mCRC, metastatic colorectal cancer; MSI-H, microsatellite instability-high; MSI-L, microsatellite instability-low; MSS, microsatellite stable; NLR, neutrophil-to-lymphocyte ratio.
At the final follow-up on March 15, 2024, 73 (35.6%) patients died, 39 were lost to follow-up, and 93 remained alive. The mean follow-up duration after immunotherapy was 17.7 months. The median PFS time was 6.0 (95% confidence interval, 4.4–7.6) months.
Except for the location of the primary tumor, there were no significant differences in demographic and clinical characteristics between the CRP-C3-high and CRP-C3-low subgroups. Among these patients, 14 (6.8%) achieved CR, 55 (26.8%) achieved PR, 68 (33.2%) maintained SD, and 68 (33.2%) experienced PD. The objective response rate (ORR) was 33.7% (69 of 205 patients), and the disease control rate (DCR) was 66.8% (Table 2).
The association between tumor response and levels of CRP-C3 or TMB in patients with mCRC receiving immunotherapy.

Variables are expressed as numbers of patients (%).
CR, complete response; CRP-C3, C-reactive protein levels before the third cycle of immunotherapy; DCR, disease control rate; mCRC, metastatic colorectal cancer; ORR, objective response rate; PD, progressive disease; PR, partial response; SD, stable disease; TMB, tumor mutational burden.
Independent risk factors for PFS and OS of patients with mCRC
To evaluate the influence of clinicopathological variables on PFS in patients with mCRC receiving immunotherapy, we initially used univariate Cox proportional models. These models identified several potential prognostic indicators for PFS: age >45 years, BMI >22.67 kg/m2, ECOG PS ⩾ 1, CRP-C3 level >27.1 mg/L, NLR ⩽3.2, use of combination immunotherapy, MSS/MSI-L status, and TMB <10 Mb (determined via tumor tissue next-generation sequencing). The optimal baseline CRP and CRP-C3 cutoff values were 21.81 and 27.10 mg/L, respectively. These cutoff values were determined using X-tile (Figure 1 and Supplemental Figure 2).
However, in the multivariate Cox proportional analysis, only CRP-C3, MSS/MSI-L status, and TMB levels emerged as independent predictors of PFS, whereas the other variables failed to demonstrate a significant independent prognostic value (Table 3). Regarding OS, we observed that an elevated CRP-C3 level served as an independent prognostic factor (Supplemental Table 2). Moreover, Kaplan–Meier curves based on serum CRP-C3 and TMB levels were generated, as depicted in Figure 2. Kaplan–Meier curves unequivocally revealed significant associations, suggesting that lower CRP-C3 levels or elevated TMB levels correlated with more favorable PFS or OS (Figure 2, all p values ⩽0.0001). Furthermore, CRP-C3 levels could further stratify the risk of patients in the TMB-low or TMB-high subgroups, predicting their PFS and OS (Supplemental Figure 3).
Univariate and multivariate Cox regression analyses of risk factors for progression-free survival.

BMI, body mass index; CRP, C-reactive protein; CRP-C3, serum CRP levels before the third cycle of immunotherapy; ECOG PS, Eastern Cooperative Oncology Group Performance Status; HR, hazard ratio; MSI-H, microsatellite instability-high; MSI-L, microsatellite instability-low; MSS, microsatellite stable; NLR, neutrophil-to-lymphocyte ratio.

Kaplan–Meier plot of PFS and OS for CRP-C3 and TMB. (a, b) Patients in the CRP-C3-low group had more prolonged PFS and OS than those of patients in the high group (log-rank test). (c, d) Patients in the TMB-high group reported significantly better PFS and OS.
Association between tumor response for immunotherapy and CRP-TMB model
Given the substantial heterogeneity in outcomes among patients with mCRC receiving immunotherapy, we devised a novel prognostic model that incorporated serum CRP-C3 and TMB levels to enhance the precision of predicting patient outcomes. This model categorized patients into high-, intermediate-, and low-risk subgroups, based on their combined serum CRP-C3 and baseline TMB levels. The PFS and OS of patients with mCRC displayed significant variations across the different risk subgroups (Figure 3, all p < 0.01). Nomograms further validated the predictive value of CRP-C3 and TMB for patient PFS and OS (Figure 4).

Association between the CRPC3 and TMB and PFS or OS in patients with mCRC receiving immunotherapy. (a) All patients stratified into markedly different risk subgroups for PFS by the CRP-C3 and TMB model. (b) All patients stratified into markedly different risk subgroups for OS by the CRP-C3 and TMB model.

(a, c) Nomograms for predicting the PFS and OS of patients with mCRC with different CRP-C3 and TMB levels. (b, d) The calibration curve for predicting patients’ PFS and OS at 1 year.
Table 4 summarizes the association between our CRP-C3 and TMB model and the response to immunotherapy. According to RECIST version 1.1, the low-risk group demonstrated CR in 14.3% (7/49), PR in 44.9% (22/49), SD in 24.5% (12/49), and PD in 16.3% (8/49) of the patients. In the intermediate-risk group, 5.7% (7/123) of the patients reported CR, 26.8% (33/123) reported PR, 39.0% (48/123) reported SD, and 28.5% (35/123) reported PD. Remarkably, in the high-risk group, none of the patients achieved CR or PR, 24.2% (8/22) exhibited SD, and 75.8% (25/33) reported PD. The ORRs were significantly higher at 59.2% (29/49) in the low-risk group, 32.5% (40/123) in the intermediate-risk group, and 0% in the high-risk group (p < 0.001). Similarly, the DCRs were 83.7% (41/49) in the low-risk group, 71.5% (88/123) in the intermediate-risk group, and 24.2% (8/33) in the high-risk group (p < 0.001).
The association between tumor response and subgroups of CRP-C3 and TMB model in patients with mCRC receiving immunotherapy.

Variables are expressed as numbers of patients (%).
CR, complete response; CRP-C3, C-reactive protein levels before the third cycle of immunotherapy; DCR, disease control rate; mCRC, metastatic colorectal cancer; ORR, objective response rate; PD, progressive disease; PR, partial response; SD, stable disease; TMB, tumor mutational burden.
Internal validation
An independent internal validation cohort consisting of 104 patients with mCRC who received immunotherapy between January and December 2024 was retrospectively collected using the same inclusion and exclusion criteria as the primary cohort. Several potential prognostic factors for PFS were identified in this cohort, including CRP-C3 level >27.1 mg/L, combination immunotherapy, MSS/MSI-L status, and TMB <10 Mb.
In multivariate Cox proportional analysis, only elevated CRP-C3 levels, combination immunotherapy, and MSS/MSI-L status were confirmed as independent predictors of PFS, whereas the other variables did not demonstrate statistically significant prognostic value (Supplemental Table 3).
Additionally, Kaplan–Meier survival curves stratified by serum CRP-C3 levels further supported its prognostic relevance. As shown in Supplemental Figure 4, lower CRP-C3 levels were significantly associated with improved PFS and OS (p = 0.0004 and p < 0.0001, respectively).
Discussion
In recent years, the landscape of mCRC treatment has been revolutionized by the advent of immunotherapy, which specifically targets immune-regulatory proteins, such as programmed cell death protein 1 (PD-1)/programmed death ligand 1 (PD-L1) or cytotoxic T-lymphocyte-associated protein 4 (CTLA-4) via antibodies. Despite these advancements, the number of patients achieving long-term benefits from immunotherapy remains limited. In addition, patients with similar mCRC molecular subtypes often exhibit varying treatment responses. Therefore, identification of biomarkers that can aid in patient categorization and enhance the efficacy of immunotherapy for mCRC is crucial. Although previous studies have explored serum CRP as a potential biomarker, its precise utility in predicting outcomes for patients with mCRC receiving immunotherapy remains unclear. This study revealed a significant correlation between serum CRP-C3 levels and PFS in patients with mCRC receiving immunotherapy. Furthermore, when combined with TMB levels in tumor tissues, serum CRP-C3 levels effectively facilitated risk stratification and assessed treatment efficacy.
The currently used clinical indicators for predicting the efficacy of immune-checkpoint inhibitor treatment, including PD-L1 expression level and TMB, MMR, and MSI status, have proven to be less satisfactory in practice.18,19 Although TMB has emerged as a useful predictor of patient survival after immunotherapy for various cancers, including CRC, the optimal cutoff value for TMB in relation to immunotherapy remains controversial, and not all mutations associated with high TMB result in robust antitumor immune responses. 20 Moreover, assessing TMB requires sequencing of the patient’s tumor, which is complex and costly. Thus, CRP-C3 serves as a simple and efficient biomarker with the potential to reclassify established patient subgroups (such as patients with mCRC with TMB-H or TMB-L) and effectively guide immunotherapy strategies. Moreover, antitumor immune infiltration after immunotherapy may lead to temporary enlargement of tumor lesions, known as pseudoprogression. 21 The accuracy of differentiating pseudoprogression can potentially be enhanced with the assistance of CRP-C3 levels.
CRP has gained recognition as a promising indicator of the progression of numerous malignant tumors. Previous studies have not explored the prognostic importance of interim CRP levels in patients with mCRC. To bridge this gap, our multivariate Cox analysis revealed that both elevated serum CRP-C3 and low TMB levels served as significant independent prognostic indicators of poor PFS. Additionally, we formulated a prognostic model incorporating CRP-C3 and TMB, which effectively categorized patients into low-, intermediate-, and high-risk groups and exhibited significantly distinct DCR values (83.7%, 71.5%, and 24.2%, respectively; p < 0.001). In our study, serum CRP-C3 and TMB levels outperformed other factors, such as MSI status and baseline serum CRP levels, effectively predicting both PFS and OS in patients with mCRC receiving immunotherapy. Based on these results, we hypothesize that CRP could serve as an indicator of tumor immune escape. Baseline CRP level may reflect the extent of tumor immune evasion; however, it is not anticipated to directly affect the efficacy of immune checkpoint inhibitors. Recent advancements suggest that the expression of immune-inhibitory checkpoints, such as PD-1/PD-L1 and CTLA-4, serves as a potent mediator during the balance and escape phases of cancer immune editing. Immunotherapy can efficiently enhance the body’s innate ability to clear tumors and reduce the level of tumor immune escape. 22 CRP-C3 level may directly indicate the efficacy of immunotherapy. We can refer to this phenomenon in CRP as “bBuoys of immunotherapy effectiveness.” Owing to the pressing requirement for a predictive biomarker for immunotherapy response, we suggest integrating CRP-C3 into clinical practice as it offers insights into immunotherapy prognosis.
Both primary and acquired resistance to immunotherapy can arise from diverse tumor alterations, such as the upregulation of immune inhibitory molecules, loss of neoantigens, deficiencies in antigen presentation, interferon signaling, and exclusion of T cells. 23 These alterations significantly affect the efficacy of immunotherapy. In CRC and pancreatic cancer, 24 high levels of transforming growth factor-beta are frequently observed. This factor promotes the development of dense stromal components, limiting immune cell infiltration into the tumor,24,25 thereby contributing to the suboptimal response of mCRC to immunotherapy.
CRP is elicited by interleukin-6 (IL-6) in hepatic cells in response to infection or tissue damage. It can stimulate the complement system and augment phagocytosis, thereby assuming an opsonic function. 8 Recent findings suggest an association between CRP and tumor immunosuppression. CRP consistently upregulates the expression of the interleukin-1 beta (IL-1β) gene in expanded melanoma antigen recognized by T cells from patients with melanoma, and, IL-1β is a known stimulator of the production of CRP by hepatocytes. 26 A previous study has demonstrated that IL-1β (+) macrophages cause inflammation in pancreatic cancer. 27 Jiang J et al. 28 demonstrated that CRP inhibited the function of T cells by activating the p38 Mitogen-Activated Protein Kinase pathway in multiple myeloma, which can be activated by a wide range of cellular stresses, such as reactive oxygen species and inflammatory cytokines. CRP also modulates various cytokines, such as the upregulation of perforin, interferon-γ, and CD107a. By exerting these immunosuppressive effects, CRP may be involved as one component of the immunosuppressive regulatory pathway, partially reflecting the extent of immunosuppression. In patients diagnosed with non-small cell lung cancer, changes in CRP levels over a 4-week period have shown predictive ability in determining the response to immunotherapy. 12 Similar findings have been reported in metastatic renal cell carcinoma and metastatic melanoma.29,30 Additionally, IL-6, a chronic inflammatory protein that triggers CRP expression, facilitates the proliferation, survival, invasiveness, and metastasis of tumor cells via IL-6/JAK/STAT3 signaling, while strongly suppressing the antitumor immune response. 31 Thus, elevated IL-6 and CRP levels may play important roles in the poor outcomes observed in patients with mCRC treated with immunotherapy.
This study has some limitations. The measurement of serum CRP levels can be influenced by various factors, such as inflammation, infections, injuries, and stress. To minimize these potential sources of variation, we assessed the CRP levels when the disease status was stable. Moreover, our study did not detect serum IL-6 levels. Additionally, this study had a retrospective design, which limited the cohort size and restricted the research to a single institution. Future studies should prioritize prospective investigations to validate and expand the findings presented here.
Conclusion
Our study suggests that serum CRP-C3 levels may serve as a promising biomarker for assessing the efficacy of immunotherapy and predicting clinical outcomes in patients with mCRC receiving such treatment. Furthermore, the integration of CRP-C3 with TMB shows potential as a stratification tool for identifying patients who are more likely to benefit from immunotherapy. This preliminary evidence supports the potential utility of a more personalized approach, which could help reduce unnecessary treatments and improve patient care through more tailored and cost-effective therapeutic strategies. However, further validation in larger, prospective cohorts is warranted to confirm these findings and fully establish their clinical applicability.
Supplemental Material
sj-docx-1-tam-10.1177_17588359251356581 – Supplemental material for Interim C-reactive protein levels predict the prognosis of patients with metastatic colorectal cancer receiving immunotherapy: a retrospective cohort study
Supplemental material, sj-docx-1-tam-10.1177_17588359251356581 for Interim C-reactive protein levels predict the prognosis of patients with metastatic colorectal cancer receiving immunotherapy: a retrospective cohort study by Hai-Yan Fu, Yi-Yang Zhang, Jin-Shan Huang, Jin-Xin Hu, Kun-Hao Bai and Zhi-Qiang Wang in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-jpg-2-tam-10.1177_17588359251356581 – Supplemental material for Interim C-reactive protein levels predict the prognosis of patients with metastatic colorectal cancer receiving immunotherapy: a retrospective cohort study
Supplemental material, sj-jpg-2-tam-10.1177_17588359251356581 for Interim C-reactive protein levels predict the prognosis of patients with metastatic colorectal cancer receiving immunotherapy: a retrospective cohort study by Hai-Yan Fu, Yi-Yang Zhang, Jin-Shan Huang, Jin-Xin Hu, Kun-Hao Bai and Zhi-Qiang Wang in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-jpg-3-tam-10.1177_17588359251356581 – Supplemental material for Interim C-reactive protein levels predict the prognosis of patients with metastatic colorectal cancer receiving immunotherapy: a retrospective cohort study
Supplemental material, sj-jpg-3-tam-10.1177_17588359251356581 for Interim C-reactive protein levels predict the prognosis of patients with metastatic colorectal cancer receiving immunotherapy: a retrospective cohort study by Hai-Yan Fu, Yi-Yang Zhang, Jin-Shan Huang, Jin-Xin Hu, Kun-Hao Bai and Zhi-Qiang Wang in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-jpg-4-tam-10.1177_17588359251356581 – Supplemental material for Interim C-reactive protein levels predict the prognosis of patients with metastatic colorectal cancer receiving immunotherapy: a retrospective cohort study
Supplemental material, sj-jpg-4-tam-10.1177_17588359251356581 for Interim C-reactive protein levels predict the prognosis of patients with metastatic colorectal cancer receiving immunotherapy: a retrospective cohort study by Hai-Yan Fu, Yi-Yang Zhang, Jin-Shan Huang, Jin-Xin Hu, Kun-Hao Bai and Zhi-Qiang Wang in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-jpg-5-tam-10.1177_17588359251356581 – Supplemental material for Interim C-reactive protein levels predict the prognosis of patients with metastatic colorectal cancer receiving immunotherapy: a retrospective cohort study
Supplemental material, sj-jpg-5-tam-10.1177_17588359251356581 for Interim C-reactive protein levels predict the prognosis of patients with metastatic colorectal cancer receiving immunotherapy: a retrospective cohort study by Hai-Yan Fu, Yi-Yang Zhang, Jin-Shan Huang, Jin-Xin Hu, Kun-Hao Bai and Zhi-Qiang Wang in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
