Abstract
Background:
Trifluridine/tipiracil plus bevacizumab (FTD/TPI + BEV) has shown efficacy and tolerability in refractory metastatic colorectal cancer (mCRC). Because randomized controlled trial (RCT) data comparing FTD/TPI + BEV with FTD/TPI are lacking, this meta-analysis evaluated outcomes with both regimens.
Data Sources and Methods:
Electronic databases, congress proceedings (past 3 years), trial registries, systematic review bibliographies, gray literature, and guidelines through June 2021 were searched for RCTs, non-RCTs, and prospective observational studies involving >20 previously treated patients with mCRC receiving FTD/TPI + BEV or FTD/TPI. Absolute and relative disease control rate (DCR), progression-free survival (PFS), overall survival (OS), adverse event (AE) rates, and discontinuation rates due to AEs were evaluated using fixed-effects and random-effects models. Study quality, heterogeneity, and publication bias were assessed.
Results:
In all, 29 of 875 screened publications were selected (26 studies: 5 RCTs, 11 non-RCTs, and 10 prospective observational studies). One RCT compared FTD/TPI + BEV with FTD/TPI. FTD/TPI + BEV versus FTD/TPI had a higher absolute DCR [64% (6 studies; n = 289) versus 43% (10 studies; n = 2809)], median PFS [4.2 (5 studies; n = 244) versus 2.6 (6 studies; n = 1781) months], 12-month PFS [9% (5 studies; n = 244) versus 3% (6 studies; n = 1781)], median OS [9.8 (5 studies; n = 244) versus 8.1 (6 studies; n = 1814) months], and 12-month OS [38% (5 studies; n = 244) versus 32% (6 studies; n = 1814)]. Grade ⩾3 febrile neutropenia, asthenia/fatigue, diarrhea, nausea, and vomiting rates were similar (1%–7%). Grade ⩾3 neutropenia rate was higher with FTD/TPI + BEV than with FTD/TPI [43% (6 studies; n = 294) versus 29% (12 studies; n = 7139)]. Discontinuation rates due to AEs were similar [8% (5 studies; n = 244) and 7% (10 studies; n = 3724)]. Low study quality, heterogeneity, and/or publication bias were detected in certain instances.
Conclusion:
Despite fewer patients treated with the combination, this meta-analysis consistently suggested that FTD/TPI + BEV provides benefits over FTD/TPI in refractory mCRC and has similar safety, except for more frequent grade ⩾3 neutropenia.
Keywords
Highlights
This meta-analysis evaluated efficacy and safety outcomes with trifluridine/tipiracil plus bevacizumab (FTD/TPI + BEV) and FTD/TPI monotherapy in patients with refractory metastatic colorectal cancer (mCRC).
The results suggest that FTD/TPI + BEV provides benefits over FTD/TPI monotherapy in patients with refractory mCRC and has a similar safety profile, except for a higher rate of grade ⩾3 neutropenia.
Given the lack of data from large randomized controlled trials comparing FTD/TPI + BEV with FTD/TPI monotherapy in this population, this meta-analysis may help guide treatment selection.
Introduction
Colorectal cancer (CRC) is the third most common cancer and the second leading cause of cancer-related mortality worldwide, accounting for more than 1.9 million new cases and 935,000 deaths in 2020. 1 Approximately 20% of newly diagnosed patients have metastatic CRC (mCRC), 2 and 70% of patients with CRC overall will ultimately experience metastatic relapse. 3 Patients with mCRC have a poor prognosis, with a median overall survival (OS) of approximately 30 months from initiation of first-line systemic therapy4,5 and a 5-year relative survival of less than 15%. 2 Although systemic treatments such as chemotherapy, targeted therapy, immunotherapy, and their combinations have improved OS in these patients over the last decade, more effective therapeutic approaches are needed.4–6
Trifluridine/tipiracil (FTD/TPI), an oral cytotoxic chemotherapy consisting of a thymidine-based nucleoside analog (trifluridine) and a thymidine phosphorylase inhibitor (tipiracil), has demonstrated improved clinical efficacy with a manageable safety profile in patients with heavily pretreated mCRC7,8 and is a standard-of-care treatment for refractory mCRC.4–6 FTD/TPI has a unique mechanism of action that involves incorporation of phosphorylated FTD into DNA (resulting in DNA dysfunction) and inhibition of FTD degradation by thymidine phosphorylase with coadministration of TPI (resulting in increased FTD bioavailability). 9 In the phase III RECOURSE trial, FTD/TPI administered beyond second-line treatment was associated with a significant improvement in OS compared with placebo [median OS, 7.1 versus 5.3 months; hazard ratio (HR), 0.68; 95% confidence interval (CI), 0.58–0.81; p < 0.001] in patients with refractory mCRC. 8 The most common grade 3–4 adverse events (AEs) with FTD/TPI in that study were neutropenia (38%) and leukopenia (21%). In a subgroup analysis of the RECOURSE trial data, FTD/TPI was shown to be effective regardless of age, geographic region, or KRAS mutation status. 10
Findings from preclinical and clinical studies suggest that the combination of FTD/TPI plus bevacizumab (FTD/TPI + BEV; a vascular endothelial growth factor inhibitor) is a feasible treatment option for patients with CRC.11,12 FTD/TPI + BEV has been shown to enhance antitumor activity in human CRC xenografts compared with either treatment alone; phosphorylated FTD levels increased when FTD/TPI was combined with BEV in these tumor models, suggesting that BEV might facilitate FTD accumulation in tumor cell DNA. 11 Although FTD/TDI is most commonly used as a single-agent treatment in clinical practice, data from a smaller randomized controlled trial (RCT), the phase II EudraCT trial (n = 93), showed benefits with FTD/TPI + BEV compared with FTD/TPI monotherapy in patients with refractory mCRC. 12 In that trial, median progression-free survival (PFS) was 2.6 months with FTD/TPI monotherapy and 4.6 months with FTD/TPI + BEV (HR, 0.45; 95% CI, 0.29–0.72; p = 0.0015), and median OS was 6.7 months with FTD/TPI monotherapy and 9.4 months with FTD/TPI + BEV (HR, 0.55; 95% CI, 0.32–0.94; p = 0.028). 12 FTD/TPI + BEV is recommended by the National Comprehensive Cancer Network Clinical Practice Guidelines on Oncology (NCCN Guidelines®) as an option for treating patients with refractory mCRC.13,14 Given the lack of data from larger RCTs comparing FTD/TPI + BEV with FTD/TPI monotherapy in patients with mCRC, we conducted this meta-analysis to evaluate efficacy and safety outcomes with the two regimens.
Methods
Data sources
A meta-analysis was conducted using studies identified in a systematic literature review. The systematic literature review involved searching electronic databases (MEDLINE®, Embase®, and Cochrane Library databases); congress proceedings (the American Society of Clinical Oncology, European Society of Medical Oncology, and American Association for Cancer Research) for the past 3 years; clinical trial registries (Clinicaltrials.gov and UMIN registry); systematic review bibliographies; gray literature; and clinical guidelines through June 2021 to identify studies involving patients with mCRC treated with FTD/TPI + BEV or FTD/TPI monotherapy. Specific inclusion and exclusion criteria were applied as a two-stage screening process to identify relevant publications (Supplemental Table 1); the electronic search strategy used for one of the databases, MEDLINE®, is shown in Supplemental Table 2. In the first stage, abstracts returned by the search strategy were examined independently by two researchers and screened based on the inclusion and exclusion criteria. In the event of any conflict, a third independent reviewer was consulted whose decision would be considered final. In the second stage, full texts of all the studies included in the first stage were obtained. Two reviewers examined these independently for inclusion or exclusion, and disagreements were resolved by a third independent reviewer, whose decision was considered final.
Quantitative synthesis
A feasibility analysis was carried out to evaluate the possibility of data synthesis for relevant outcomes based on study design, patient setting, and consistency in reporting outcomes across studies. Based on the composition of the identified studies, the quantitative synthesis was limited to previously treated patients with mCRC in RCTs, non-RCTs, and prospective observational studies. Only studies with a sample size of more than 20 patients were analyzed in the quantitative synthesis. In addition, only patients who were treated with FTD/TPI + BEV or FTD/TPI monotherapy were included in the meta-analysis; patients who received placebo were excluded. Data from the included publications were extracted by one reviewer. To identify and rectify any errors in data extraction, a second reviewer checked and validated all the data by conducting an independent internal data check.
Outcomes
Outcomes evaluated in this meta-analysis included objective response rate (ORR), disease control rate (DCR), PFS, OS, AE rates, and discontinuation rates due to AEs. Grade ⩾3 neutropenia, febrile neutropenia, asthenia/fatigue, diarrhea, nausea, and vomiting were reported in most studies; therefore, these AEs were used in the safety analysis. Because neutropenia is frequently observed during the 2-week rest period after initiation of FTD/TPI treatment 15 and granulocyte colony-stimulating factor (G-CSF) is often used to treat neutropenia, data were collected for AE monitoring schedule and G-CSF use to determine if cases of neutropenia were accurately captured and appropriately treated.
Statistical analysis
The meta-analysis assessed absolute (pooled) and relative outcomes using fixed- and random-effects models, which were premised on an inverse-variance weighting approach. 16 Dichotomous outcomes (ORR, DCR, AE rates, and discontinuation rates due to AEs) were analyzed as proportions. Time-to-event outcomes (OS and PFS) were analyzed as rates with corresponding 95% CIs at specific landmark time points (e.g. 12 and 24 months) and were determined by pooling the Kaplan–Meier curves using Guyot’s algorithm. 17 For sparse dichotomous outcomes, relative treatment effects were estimated as risk ratios (RRs) or risk differences (RDs). For time-to-event outcomes, relative treatment effects were analyzed as HRs with corresponding 95% CIs.
Quality assessment of the included studies was carried out using the Cochrane risk-of-bias tool for RCTs, 18 the Downs and Black checklist for non-RCTs, 19 and the Newcastle–Ottawa scale for observational studies. 20 Heterogeneity (i.e. differences in patient characteristics) between the studies was assessed using I2 statistics21,22 and τ2 statistics. 23 Publication bias (i.e. the tendency to publish studies with beneficial outcomes or statistically significant findings) was evaluated using funnel plots (i.e. plots of effect estimates against sample sizes) 24 and Egger’s weighted regression p values. 23 An asymmetrical funnel plot and an Egger’s weighted regression p < 0.1 suggested significant publication bias.23,25 Due to low testing power, funnel plots and Egger’s weighted regression p value were not considered when the meta-analysis included fewer than 10 studies.
The statistical analysis was carried out using R software. All comparative assessments were performed by way of a pairwise meta-analysis; indirect treatment comparisons were not carried out. Missing parameters (e.g. standard deviations and standard errors) required for the meta-analysis were estimated based on Cochrane guidelines. 18
Results
Study selection
Among 875 screened publications, 29 publications were selected, which reported on 26 studies: 5 RCTs,7,8,12,26–30 11 non-RCTs,31–41 and 10 prospective observational studies15,42–50 (Figure 1; Supplemental Table 3). Six studies involved FTD/TPI + BEV as an intervention. Only one RCT was identified comparing FTD/TPI + BEV with FTD/TPI monotherapy in pretreated patients with mCRC, the phase II EudraCT trial (n = 93). 12 Pooled across all 26 studies, FTD/TPI monotherapy and FTD/TPI + BEV were used in 9383 and 289 patients, respectively.
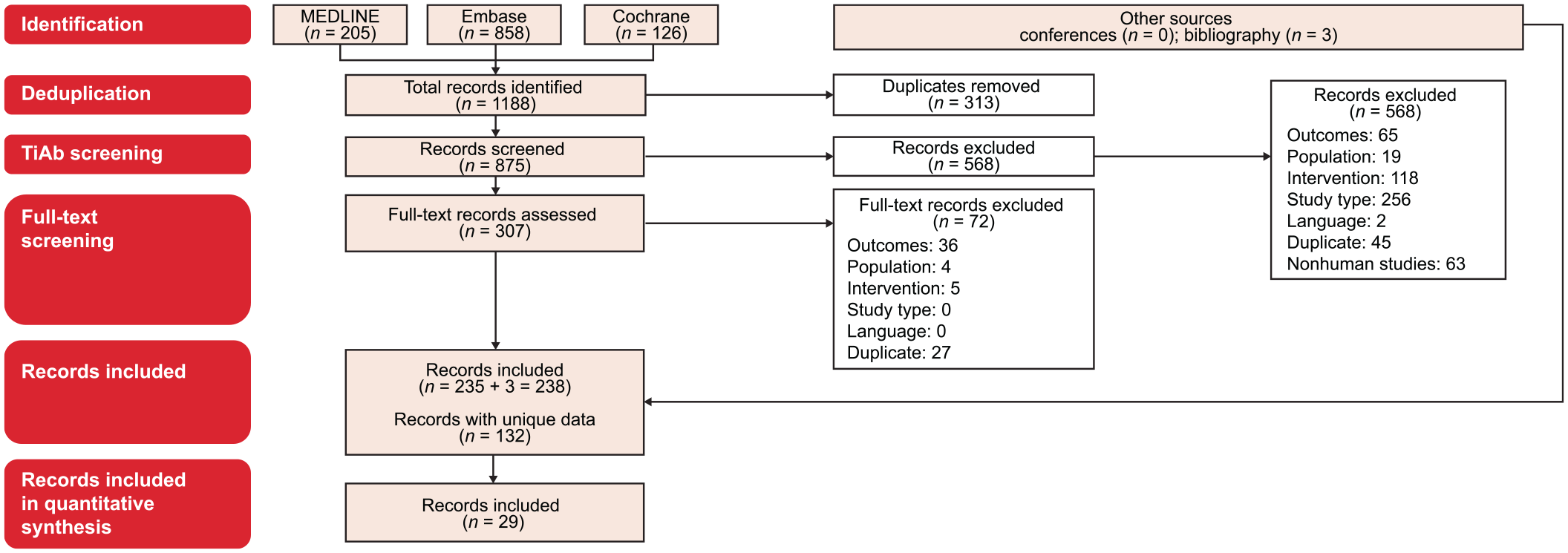
PRISMA diagram. Only records for RCTs, non-RCTs, and prospective observational studies with a sample size of more than 20 patients were included in the quantitative synthesis.
Quality assessment
Applying the Cochrane risk-of-bias tool 18 to the five RCTs, three studies were judged low risk in all seven categories; one study was judged low risk in four categories and high risk in three categories; and one study was judged low risk in three categories, high risk in two categories, and unclear risk in two categories (Supplemental Table 4). Applying the Downs and Black checklist 19 to the 11 non-RCTs, one study was good quality, five studies were fair quality, and five studies were poor quality (Supplemental Table 5). Applying the Newcastle–Ottawa scale 20 to the 10 observational studies, one study was high quality, six studies were medium quality, and three studies were low quality (Supplemental Table 6).
Absolute (pooled) efficacy
Absolute ORR was 4% with FTD/TPI + BEV (6 studies; n = 289) and 2% with FTD/TPI monotherapy (9 studies; n = 2784) (Supplemental Figure 1). Absolute DCR was 64% with FTD/TPI + BEV (6 studies; n = 289) and 43% with FTD/TPI monotherapy (10 studies; n = 2809) (Figure 2).

Absolute (pooled) DCRs for (a) FTD/TPI + BEV and (b) FTD/TPI monotherapy.
Absolute median PFS was 4.2 months with FTD/TPI + BEV (5 studies; n = 244) and 2.6 months with FTD/TPI monotherapy (6 studies; n = 1781); absolute 12-month PFS was 9% (95% CI, 6%–14%) and 3% (95% CI, 2%–4%), respectively (Figure 3; Supplemental Table 7). Absolute median OS was 9.8 months with FTD/TPI + BEV (5 studies; n = 244) and 8.1 months with FTD/TPI monotherapy (6 studies; n = 1814); absolute 12-month OS was 38% (95% CI, 32%–45%) and 32% (95% CI, 30%–34%), respectively (Figure 3; Supplemental Table 8).
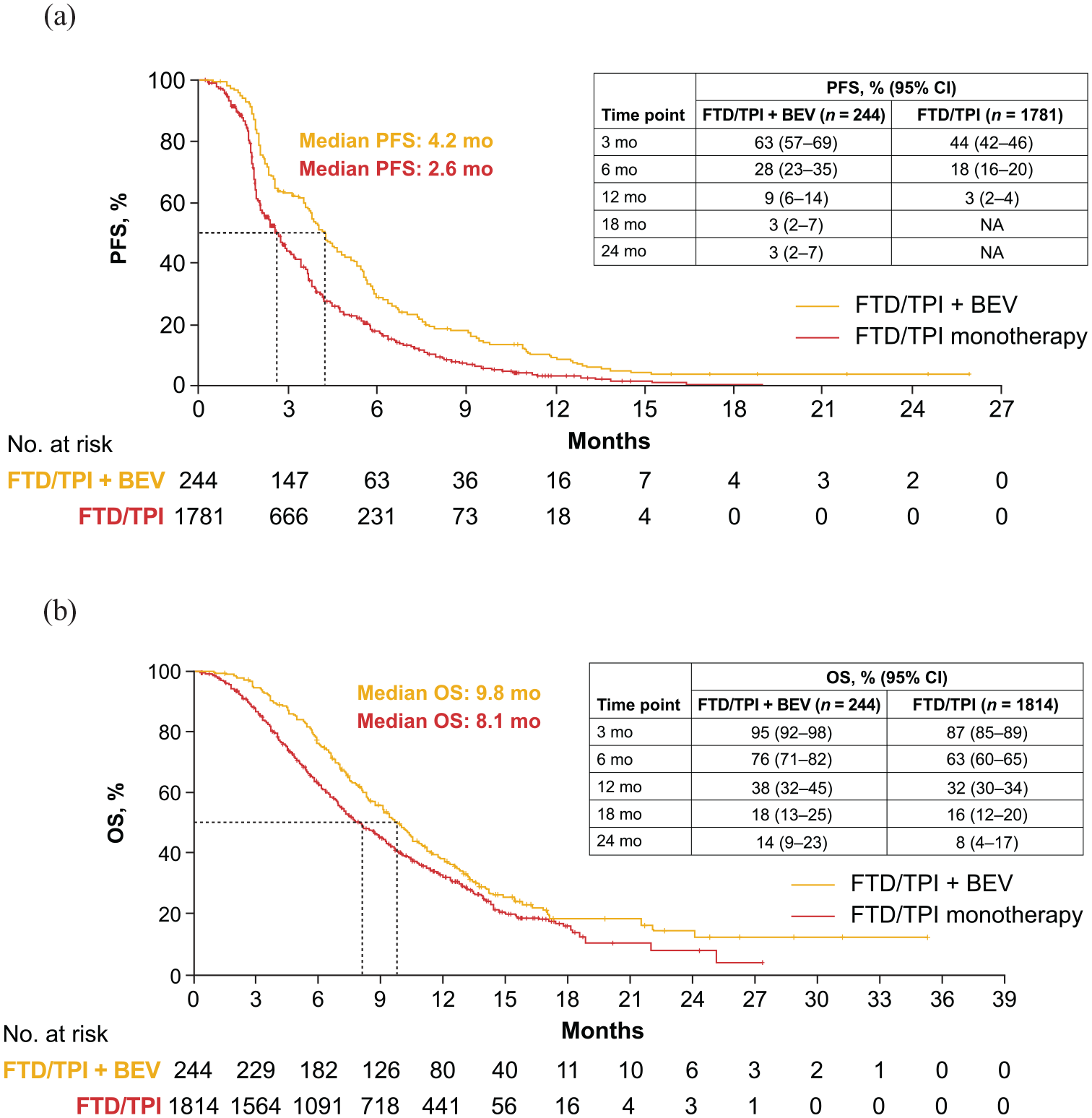
Pooled absolute (a) PFS and (b) OS for FTD/TPI + BEV and FTD/TPI monotherapy.
For the absolute DCR analysis for FTD/TPI monotherapy, which included 10 studies, the funnel plot and Egger’s weighted regression p value suggested the presence of publication bias. Funnel plots and Egger’s weighted regression p values were not considered for the other absolute efficacy analyses because fewer than 10 studies were included.
Relative efficacy
The relative efficacy analysis only included the EudraCT trial. 12 In that trial, FTD/TPI + BEV, compared with FTD/TPI monotherapy, was associated with numerically (although not statistically significantly) higher ORR (RD, 0.02; 95% CI, −0.02 to 0.07; p = 0.35) and DCR (RR, 1.32; 95% CI, 0.94–1.86; p = 0.11). 12 FTD/TPI + BEV, compared with FTD/TPI monotherapy, was associated with a 55% significant reduction in the risk for progression (HR, 0.45; 95% CI, 0.29–0.72; p = 0.0015) and a 45% significant reduction in the risk for death (HR, 0.55; 95% CI, 0.32–0.94; p = 0.03). 12 Funnel plots and Egger’s weighted regression p values were not considered for the relative efficacy analyses because only one study was included.
Absolute (pooled) safety
AEs were not consistently reported across the studies, and AE grading followed the National Cancer Institute Common Terminology Criteria for Adverse Events v3.0, v4.0, or v4.03, depending on when the respective studies were conducted. Data for the following six most commonly reported AEs were pooled across the studies: neutropenia, febrile neutropenia, asthenia/fatigue, diarrhea, nausea, and vomiting. Absolute rates for grade ⩾3 neutropenia were 43% with FTD/TPI + BEV (6 studies; n = 294) and 29% with FTD/TPI monotherapy (12 studies; n = 7139) (Figure 4(a) and (b)). Absolute rates for grade ⩾3 febrile neutropenia were 7% with FTD/TPI + BEV (4 studies; n = 217) and 3% with FTD/TPI monotherapy (6 studies; n = 4963) (Figure 4(c) and (d)). Absolute rates for grade ⩾3 asthenia/fatigue were 4% with FTD/TPI + BEV (6 studies; n = 294) and 4% with FTD/TPI monotherapy (11 studies; n = 7094) (Figure 4(e) and (f)). Absolute rates for grade ⩾3 diarrhea were 6% with FTD/TPI + BEV (5 studies; n = 249) and 2% with FTD/TPI monotherapy (12 studies; n = 7144) (Figure 4(g) and (h)). Absolute rates for grade ⩾3 nausea were 5% with FTD/TPI + BEV (6 studies; n = 294) and 1% with FTD/TPI monotherapy (10 studies; n = 6917) (Figure 4(i) and (j)). Absolute rates for grade ⩾3 vomiting were 3% with FTD/TPI + BEV (4 studies; n = 147) and 1% with FTD/TPI monotherapy (10 studies; n = 6608) (Figure 4(k) and (l)). Absolute discontinuation rates due to AEs were 8% with FTD/TPI + BEV (5 studies; n = 244) and 7% with FTD/TPI monotherapy (10 studies; n = 3724) (Supplemental Figure 2).

Absolute (pooled) rates for grade ⩾3 AEs for FTD/TPI + BEV and FTD/TPI monotherapy. (a) FTD/TPI + BEV: Grade ⩾3 neutropenia. (b) FTD/TPI monotherapy: Grade ⩾3 neutropenia. (c) FTD/TPI + BEV: Grade ⩾3 febrile neutropenia. (d) FTD/TPl monotherapy: Grade ⩾3 febrile neutropenia. (e) FTD/TPI + BEV: Grade ⩾3 asthenia/fatigue. (f). FTD/TPI monotherapy: Grade ⩾3 asthenia/fatigue. (g) FTD/TPI + BEV: Grade ⩾3 diarrhea. (h) FTD/TPI monotherapy: Grade ⩾3 diarrhea. (i) FTD/TPI + BEV: Grade ⩾3 nausea. (j) FTD/TPI monotherapy: Grade ⩾3 nausea. (k) FTD/TPI + BEV: Grade ⩾3 vomiting. (l) FTD/TPI monotherapy: Grade ⩾3 vomiting.
For absolute safety analyses for FTD/TPI monotherapy that included 10 or more studies, funnel plots and Egger’s weighted regression p values suggested no publication bias for neutropenia, asthenia/fatigue, diarrhea, nausea, and vomiting, but publication bias was suggested for discontinuation rates due to AEs. Funnel plots and Egger’s weighted regression p values were not considered for the other absolute safety analyses because fewer than 10 studies were included.
Relative safety
As in the relative efficacy analysis, the relative safety analysis only included the EudraCT trial. 12 In that trial, the risk for grade ⩾3 neutropenia was significantly higher with FTD/TPI + BEV than with FTD/TPI monotherapy (RD, 0.29; 95% CI, 0.10–0.49; p = 0.0033). Risks were similar between the two regimens for grade ⩾3 febrile neutropenia (RD, 0.04; 95% CI, −0.04 to 0.13; p = 0.2961), asthenia/fatigue (RD, −0.04; 95% CI, −0.15 to 0.07; p = 0.48), diarrhea (RD, 0.09; 95% CI, 0.00–0.17; p = 0.04), nausea (RD, −0.04; 95% CI, −0.12 to 0.04; p = 0.31), and vomiting (RD, 0.02; 95% CI, −0.05 to 0.09; p = 0.55). The risk of discontinuation due to AEs in the EudraCT trial was similar with FTD/TPI + BEV and FTD/TPI monotherapy (RD, −0.02; 95% CI, −0.09 to 0.05; p = 0.57) despite a longer treatment duration with combination therapy than with monotherapy (median, 4.9 versus 2.4 months). Funnel plots and Egger’s weighted regression p values were not considered for the relative safety analyses because only one study was included.
AE monitoring schedule and G-CSF use
To determine if cases of neutropenia were accurately captured and appropriately treated, data were collected for the AE monitoring schedule and G-CSF use. The AE monitoring schedule, which was reported in 10 of the 26 studies (38%), was approximately every week or every 2 weeks (Supplemental Table 9). G-CSF was used in five studies (19%), not allowed in two studies (8%), and not reported in 19 studies (73%) (Supplemental Table 9).
Discussion
The results of this meta-analysis, which pooled data across 26 studies, suggest that the addition of BEV to FTD/TPI is a feasible treatment for patients with refractory mCRC. Given the lack of data from large RCTs comparing FTD/TPI + BEV with FTD/TPI monotherapy, this meta-analysis provides further important insights into the use of FTD/TPI + BEV that may help guide treatment decisions in this patient population.
Efficacy results were consistently superior with FTD/TPI + BEV than with FTD/TPI monotherapy in this meta-analysis. Absolute median PFS was 4.2 and 2.6 months with FTD/TPI + BEV and FTD/TPI monotherapy, respectively, and absolute median OS was 9.8 and 8.1 months, respectively. Efficacy findings of our meta-analysis were consistent with those of a recent meta-analysis with mCRC (pooled across 25 studies) in which median PFS was 4.35 and 2.53 months with FTD/TPI + BEV and FTD/TPI monotherapy, respectively, and median OS was 10.41 and 6.95 months, respectively. 51 Furthermore, efficacy results with FTD/TPI + BEV in our meta-analysis appeared to be superior to those with any later-line regimen reported in a systematic literature review of phase II and phase III trials (67 studies; 7556 patients) in refractory mCRC (median PFS, 3.2 months; median OS, 8.8 months), 52 although it is difficult to make comparisons between different analyses.
FTD/TPI + BEV and FTD/TPI monotherapy were shown to be safe and well tolerated in this meta-analysis. Absolute rates for grade ⩾3 febrile neutropenia, asthenia/fatigue, diarrhea, nausea, and vomiting were low with FTD/TPI + BEV and FTD/TPI monotherapy (1–7%). However, grade ⩾3 neutropenia was reported more frequently with FTD/TPI + BEV than with FTD/TPI monotherapy (43% and 29%, respectively). These safety results are in line with those of the previous meta-analysis with mCRC patients in which FTD/TPI + BEV, compared with FTD/TPI monotherapy, was associated with a significantly higher incidence of grade ⩾3 AEs (odds ratio, 2.19; 95% CI, 1.40–3.44). 51 In another meta-analysis, the addition of BEV to other cancer therapies increased the risk of serious neutropenic events in patients with various tumor types. 53 Because neutropenia is an overlapping AE with FTD/TPI and BEV, the benefits and risks of adding BEV to FTD/TPI should be considered carefully in patients with mCRC. However, the increased incidence of grade ⩾3 neutropenia with FTD/TPI + BEV may not be clinically significant as absolute discontinuation rates due to AEs were low with both the combination therapy and monotherapy (8% and 7%, respectively), suggesting that both regimens were well tolerated and that toxicity was generally manageable.
Data were collected for AE monitoring schedule and G-CSF use to determine if cases of neutropenia with FTD/TPI, which are frequently observed during the 2-week rest period after treatment initiation, 15 were accurately captured and appropriately treated. In the studies that reported the AE monitoring schedule, AEs were monitored approximately every week or every 2 weeks; thus, the monitoring schedules were sufficient to capture cases of neutropenia. G-CSF use was not consistently reported, so a relationship between the occurrence of neutropenia and use of G-CSF could not be determined.
RCTs are needed to provide definitive evidence on the use of FTD/TPI + BEV in patients with refractory mCRC. The only RCT comparing FTD/TPI + BEV and FTD/TPI monotherapy in this population was the phase II EudraCT trial, which enrolled 93 patients. 12 The international, open-label, phase III SUNLIGHT trial (NCT04737187) compared the efficacy and safety of FTD/TPI + BEV and FTD/TPI monotherapy in patients with unresectable mCRC in the third-line setting.54,55 The SUNLIGHT trial had an estimated enrollment of 490 patients and a predicted primary completion date of December 2022. 55 The study sponsor announced positive results in a press release (September 12, 2022). 56 Upcoming SUNLIGHT trial results are expected to be consistent with the findings of this meta-analysis.
There are limitations in the review process and evidence that should be considered when interpreting the results of this analysis. As with any meta-analysis, these findings may have been confounded by study heterogeneity and publication bias. Specifically, the results of this meta-analysis may have been influenced by differences in prognostic characteristics (e.g. age, histology, number of metastases, mutational status, and performance status) between patients treated with FTD/TPI + BEV and patients treated with FTD/TPI monotherapy who were pooled across the various studies. In particular, study heterogeneity may have had a significant impact on pooled Kaplan–Meier curves, impeding interpretation of those results. In addition, a disparity existed in the number of patients treated with FTD/TPI monotherapy (n = 9383) and FTD/TPI + BEV (n = 289) in this analysis. However, the evaluation of efficacy included absolute outcomes, which were independently determined. Thus, the difference in the number of pooled patients for the two regimens was not expected to materially affect the analysis. The relative outcomes analysis was limited by the inclusion of only one RCT (EudraCT trial 12 ) that compared FTD/TPI + BEV with FTD/TPI monotherapy in patients with mCRC who were pretreated, in which sample sizes for the two regimens were similar. Another limitation was that publication bias was detected in certain instances, and some studies were deemed to be of low quality. Furthermore, safety could not be fully assessed because the overall incidences of grade ⩾3 AEs were not consistently reported in the studies. Consequently, six commonly reported AEs (neutropenia, febrile neutropenia, asthenia/fatigue, diarrhea, nausea, and vomiting) were examined. Despite these limitations, the precision of the calculated estimates may have increased by the inclusion of large sample sizes and the use of two models (fixed and random effects).
Conclusion
In summary, the results of this meta-analysis suggest that FTD/TPI + BEV provides benefits over FTD/TPI monotherapy in patients with refractory mCRC, with a higher DCR and longer PFS and OS, and has a similar safety profile. However, the combination therapy is also associated with a higher rate of grade ⩾3 neutropenia, although this finding is of uncertain clinical significance as absolute discontinuation rates due to AEs were low with both regimens. Given the lack of data from large RCTs comparing FTD/TPI + BEV with FTD/TPI monotherapy in this population, these findings may help guide treatment decisions. Results of the phase III SUNLIGHT trial54–56 will more directly delineate the role of FTD/TPI + BEV in mCRC.
Supplemental Material
sj-docx-1-tam-10.1177_17588359221146137 – Supplemental material for Trifluridine/tipiracil with or without bevacizumab in metastatic colorectal cancer: results of a systematic review and meta-analysis
Supplemental material, sj-docx-1-tam-10.1177_17588359221146137 for Trifluridine/tipiracil with or without bevacizumab in metastatic colorectal cancer: results of a systematic review and meta-analysis by Takayuki Yoshino, Julien Taieb, Yasutoshi Kuboki, Per Pfeiffer, Amit Kumar and Howard S. Hochster in Therapeutic Advances in Medical Oncology
Footnotes
Acknowledgements
These results were presented, in part, at the 2022 American Society of Clinical Oncology (ASCO) Annual Meeting; June 3–7, 2022; Chicago, IL, USA. Medical writing and editorial support for the development of this manuscript, under the direction of the authors, were provided by Mark Palangio and Jennifer Robertson of Ashfield MedComms, an Inizio company, and funded by Taiho Oncology, Inc.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
