Abstract
Objective
We performed this meta-analysis to compare the efficacy and toxicity of regorafenib and TAS-102.
Methods
Electronic databases were searched to identify studies comparing the efficacy and safety of regorafenib and TAS-102 in patients with chemotherapy-refractory metastatic colorectal cancer using pooled analyses.
Results
Three clinical trials were included in this analysis. Regarding the reasons for treatment discontinuation, regorafenib was significantly associated with disease progression (odds ratio [OR] = 0.33, 95% confidence interval [CI] = 0.21–0.50) and adverse events (OR = 4.38, 95% CI = 2.69–7.13). However, overall (OR = 0.97, 95% CI = 0.81–1.17) and progression-free survival (OR = 1.01, 95% CI = 0.86–1.18) did not significantly differ between the groups. The most common treatment-related adverse events in the regorafenib group were neutropenia (OR = 0.06, 95% CI = 0.03–0.11), hand–foot syndrome (OR = 50.34, 95% CI = 10.44–242.84), and liver dysfunction (OR = 34.51, 95% CI = 8.30–143.43). Conversely, the incidence of thrombocytopenia did not differ between the two groups.
Conclusions
Regorafenib and TAS-102 have similar efficacy but different adverse event profiles. Differences in the toxicity profiles of the two drugs will help guide treatment selection.
Keywords
Introduction
Globally, colorectal cancer (CRC) is the third most common cancer 1 and the second leading cause of cancer-associated death. 2 In the initial diagnosis, approximately 25% of patients with CRC present with synchronous metastatic disease, and more than half of patients are diagnosed with metastases.3,4
There has been substantial progress in the treatment of unresectable or metastatic CRC (mCRC), with combinations of chemotherapy drugs 5 and molecular targeted drugs,6–8 contributing to prolonged overall survival (OS) and improved quality of life (QOL). 9 However, the outcomes of these treatments are not satisfactory, and many patients experience disease progression after standard therapy. Thus, well-tolerated, effective treatment options for patients with chemotherapy-refractory mCRC are desired.
Regorafenib, an oral multi-molecular targeted drug that inhibits tumor angiogenesis and apoptosis, 10 was reported to prolong OS and progression-free survival (PFS) compared with the effects of placebo in patients with mCRC who are resistant to standard treatment in the CORRECT trial. 11 The drug has been approved by the US Food and Drug Administration and European Medicines Agency for the treatment of refractory mCRC. Meanwhile, the novel combination regimen trifluridine (FTD)/tipiracil (TPI), also known as TAS-102, has recently emerged as a treatment for mCRC.
FTD is the active antitumor component of TAS-102, and it is incorporated into DNA in tumor cells, resulting in double-strand DNA breaks. 12 TPI is a potent inhibitor of thymidine phosphorylase that serves to increase the efficacy of FTD by preventing its degradation.13,14 The RECOURSE trial indicated that TAS-102 improved OS and PFS compared with the effects of placebo in the refractory setting. 15
Based on these findings, regorafenib and TAS-102 are both considered new treatment options for salvage line therapy. Recent retrospective studies described the comparable efficacy of these drugs, although their toxicity profiles differed. 16 Thus, it remains unclear which drug should be administered as a standard therapy for refractory mCRC.
Our meta-analysis examined existing data from previous retrospective trials comparing regorafenib and TAS-102 regarding their benefits and drawbacks and provided an overview of the efficacy and safety of both drugs.
Methods and materials
Search strategy
Studies on the efficacy and safety of regorafenib and TAS-102 in the treatment of refractory mCRC published through December 2018 were retrieved. The searchable databases included PubMed, EMBASE, and Cochrane Library, and the following keywords were used: “metastatic colorectal cancer,” “TAS-102,” AND “regorafenib.” In addition, relevant Medical Subject Heading terms were utilized. The reference lists of all relevant articles were also manually searched to identify additional pertinent studies.
Eligibility criteria
Articles were selected for inclusion in this meta-analysis based on the following criteria: (1) inclusion of patients with treatment-refractory mCRC; (2) the studies reported data for efficacy (OS, PFS, and the reason for treatment discontinuation) and toxicity (incidence of severe adverse effects); and (3) the full texts were available. When studies included overlapped or duplicated data, those with the more complete information were included. In addition, only studies published in English were included.
Quality assessment
Two investigators independently assessed the quality of the included studies. We selected the risk of bias items recommended by The Cochrane Handbook for Systematic Reviews of Interventions.
Data extraction
Two authors extracted the relevant data from individual studies separately, and differences were settled through discussion. From each of the eligible studies, the main variables extracted were the authors’ names, year of publication, country of publication, number of patients, mean age, number of male patients, and endpoints of interest. We extracted the corresponding variables and risk estimates of mortality with 95% confidence intervals (CIs).
Statistical analysis
A sensitivity analysis was also performed to examine the impact of heterogeneity on the overall results. To assess the heterogeneity of the included studies and determine the model for analysis, the I2 statistic and chi-squared test were used. 17 A fixed-effects model was used if the heterogeneity was insignificant (I2 ≤ 50%). If the heterogeneity was significant (I2 > 50%) or uncertain, we used a random-effects model for further analysis. 18 P < 0.05 denoted statistical significance. Review Manager version 5.3 software (RevMan; The Cochrane Collaboration, Oxford, UK) was used to perform further statistical analyses.
Results
Overview of the literature search and study characteristics
In total, 156 articles were initially identified. Based on the eligibility criteria, 153 articles were excluded because of the absence of relevant outcome data. Therefore, three trials19–21 comparing regorafenib and TAS-102 were included in the analysis. The search process is described in Figure 1. All studies included in this study were based on moderate- to high-quality evidence. Table 1 provides a brief description of these three studies.

PRISMA flowchart of study selection.
The primary characteristics of the eligible studies.

Clinical and methodological heterogeneity
Pooled analysis comparing PFS between regorafenib and TAS-102
Pooling the PFS data from all studies revealed that the two drugs had similar effects on PFS (OR = 1.01, 95% CI = 0.86–1.18, P = 0.93, Figure 2).
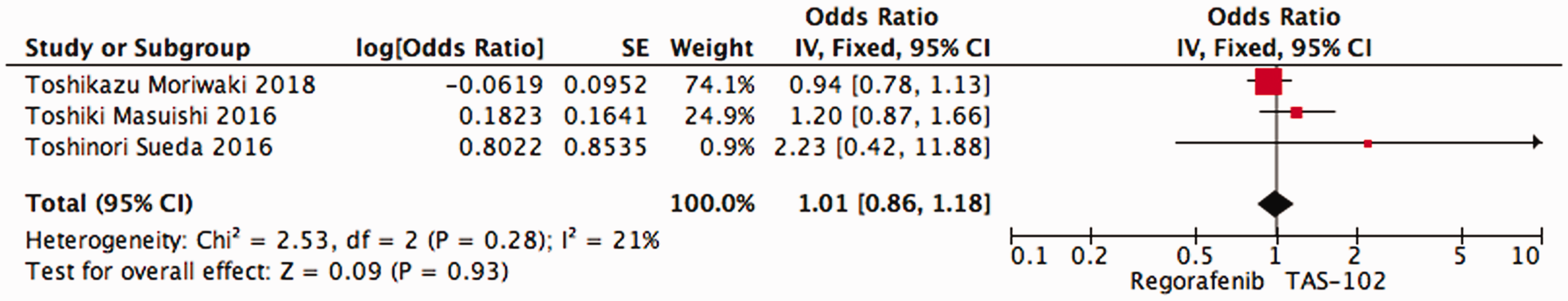
Pooled analysis of progression-free survival in the regorafenib and TAS-102 groups.
Pooled analysis comparing OS between regorafenib and TAS-102
The pooled data revealed no difference in OS between the two drugs (OR = 0.97, 95% CI = 0.81–1.17, P = 0.77, Figure 3).

Pooled analysis of overall survival in the regorafenib and TAS-102 groups.
Pooled analysis comparing the reasons for treatment discontinuation between regorafenib and TAS-102
Concerning the reasons for treatment discontinuation, regorafenib was associated with significantly rates of disease progression (OR = 0.33, 95% CI = 0.21–0.50, P < 0.00001, Figure 4) and adverse events (OR = 4.38, 95% CI = 2.69–7.13, P < 0.00001, Figure 5) than TAS-102.

Pooled analysis of the reasons for treatment discontinuation due to disease progression in the regorafenib and TAS-102 groups.

Pooled analysis of the reasons for treatment discontinuation due to adverse events in the regorafenib and TAS-102 groups.
Pooled analysis comparing adverse events between regorafenib and TAS-102
The results of systematic evaluations of adverse events are presented in Figures 6–9. The incidence of the most common toxicities was higher in the regorafenib group than in the TAS-102 group. The most common treatment-related adverse events in the regorafenib group were neutropenia (OR = 0.06, 95% CI = 0.03–0.11, P < 0.00001, Figure 6), hand–foot syndrome (OR = 50.34, 95% CI = 10.44–242.84, P < 0.00001, Figure 7), and liver dysfunction (OR = 34.51, 95% CI = 8.30–143.43, P < 0.00001, Figure 8). Conversely, the incidence of thrombocytopenia (OR = 1.97; 95% CI = 0.98–3.97; P = 0.06, Figure 9) did not differ between the two groups.

Pooled analysis of the incidence of neutropenia in the regorafenib and TAS-102 groups.

Pooled analysis of the incidence of hand–foot syndrome in the regorafenib and TAS-102 groups.

Pooled analysis of the incidence of liver dysfunction in the regorafenib and TAS-102 groups.

Pooled analysis of the incidence of thrombocytopenia in the regorafenib and TAS-102 groups.
Discussion
Despite advances in treatment and diagnosis, many patients with mCRC experience disease progression despite the use of standard therapies, resulting in high mortality rates. 22 Recently, there has been interest in clinical research for the treatment of chemotherapy-resistant mCRC to further improve outcomes in this specific setting. Thus, the need for efficacious and less toxic treatment options has been recognized.
Recent trials have reported survival benefits with tolerable toxicity profiles for regorafenib and TAS-102 versus placebo after the failure of standard chemotherapies in patients with mCRC.11,15,23,24 Both drugs have been accepted for use in the US, Europe, and Japan. 21 However, criteria for the appropriate selection of regorafenib or TAS-102 have not yet been established. Thus, we performed a meta-analysis of the efficacy and toxicity of both drugs in patients with mCRC who were intolerant to standard chemotherapies.
In terms of efficacy, no difference in PFS or OS was observed between the two drugs. These results indicate that regorafenib or TAS-102 confer similar improvements in survival among patients with mCRC who are refractory to standard treatments. In a study by Moriwaki and colleagues, 21 regorafenib provided an OS benefit in patients aged <65 years, whereas TFD tended to be more beneficial in elderly patients. The reason for this difference is unclear. It is possible that tolerance to regorafenib decreases with age, whereas tolerance to TFD was similar between the age groups. Additionally, Sueda et al. 19 reported that patients who received crossover treatment with both drugs exhibited better OS than patients who received only one of the drugs. This result indicates that strategies that maximize the availability of both drugs can prolong OS in patients with refractory disease. Moreover, potential predictive or prognostic biomarkers of regorafenib efficacy were evaluated in a retrospective study. 25 The results illustrated that the KRAS and PIK3CA mutational status may be predictive of clinical benefit for regorafenib treatment. In studies of TAS-102, no correlation was observed between the efficacy of TAS-102 and two potential biomarkers: thymidine kinase 1 and thymidine phosphorylase. 26 To our disappointment, no predictive biomarkers for the efficacy and safety of TAS-102 have been found. Therefore, it is relevant that selection criteria, including patients’ clinical factors and a history of previous therapy, are associated with efficacy profiles. However, the results emphasize the need for further investigation.
Several factors may affect patient adherence to treatment, with perhaps the most important being the management of symptoms and/or side effects of treatments.27,28 The adverse events associated with regorafenib and TAS-102 treatment were reversible and not life-threatening. 29 In our analysis, the most frequently reported grade ≥3 adverse events in the regorafenib group were neutropenia, hand–foot syndrome, and liver dysfunction. Despite their frequency, these adverse events were mostly manageable, but they can cause significant suffering and require dose adjustment and treatment discontinuation. Because regorafenib-related toxicities might worsen patient QOL, it is possible that regorafenib-related adverse events affected disease progression during treatment and prompted treatment discontinuation. Therefore, it is highly important to consider toxicity-driven dosing. Currently, an initial reduction of the regorafenib dose is being investigated by some ongoing studies to clarify the efficacy and safety of this treatment (NCT02368886, UMIN000014661). 20
This study had some limitations. First, all included studies were retrospective nonrandomized analyses; thus, the possibility of bias cannot be eliminated, which may have affected the results. Second, the efficacy of regorafenib and TAS-102 may differ among different patient subgroups. Further investigations of these regimens in a large-scale study with greater statistical power are needed.
Conclusion
Our meta-analysis found that regorafenib and TAS-102 have similar efficacy but different adverse event profiles in patients with mCRC who are refractory to standard chemotherapy. To our knowledge, patient comorbidities and the safety profiles of the drugs are the major drivers of the selection of regorafenib or TAS-102. Further analyses of biomarkers, assessments of initial dose reduction, or the use of combination therapy may aid in further tailoring the treatment for chemotherapy-resistant mCRC to obtain maximal clinical benefits.
