Abstract
Background:
Age is a known prognostic factor for various cancers. However, few studies explored the association between age and prognosis of esophageal cancer (EC) comprehensively, especially from a nonlinear perspective.
Design:
Retrospective cohort study.
Objectives:
Our study aims to explore the possible nonlinear associations between age and prognosis in EC patients receiving curative surgery and radiotherapy, respectively.
Methods:
Cox regression models with restricted cubic splines were used to model the possible nonlinear relationship between age and prognosis in surgical and radiotherapy groups, respectively. Surveillance, Epidemiology, and End Results database was used to validate the age-prognosis patterns found in Jing-Jin-Ji Esophageal and Esophagogastric Cancer Radiotherapy Oncology Group database. Age-prognosis patterns were further validated by survival comparisons between different age subgroups and in subsequent sensitivity and subgroup analyses. Primary endpoint is overall survival. Secondary endpoints are cancer-specific survival and progression-free survival.
Results:
A total of 56,457 patients from two large cancer databases were included. Patients receiving surgery and radiotherapy showed two distinct nonlinear age-prognosis patterns. Age showed a U-/J-shaped association with prognosis in the radiotherapy group, with a nadir at approximately 65- to 70-years-old. As for surgical cohort, relative risk for all-cause mortality and cancer-specific mortality increased with age with p for nonlinearity <0.05. The above age-prognosis relationships were validated by sensitivity, subgroup, and comparative survival analyses. Youngest and middle-aged patients showed better survival results compared to that of other age subgroups in surgical and radiotherapy cohorts, respectively [Radiotherapy, youngest/middle: hazard ratio (HR) = 1.06, 95% confidence interval (CI): 1.02–1.10, p = 0.001; Radiotherapy, oldest/middle: HR = 1.21, 95% CI: 1.18–1.24, p < 0.001; Surgical, middle/youngest: HR = 1.19, 95% CI: 1.14–1.25, p < 0.001; surgical, oldest/youngest: HR = 1.85, 95% CI: 1.75–1.97, p < 0.001].
Conclusion:
Patients receiving surgery and radiotherapy showed two distinct age-prognosis patterns. Younger and middle-aged patients were associated with better survival in surgical and radiotherapy groups, respectively. Additional studies are warranted to explore the underlying mechanisms and clinical implications of this phenomenon.
Introduction
Age is a known prognostic factor of various cancers, and the associations between age and prognosis may vary depending on the type of cancer.1,2 Age has been found to have a nonlinear relationship with prognosis in many cancers. In metastatic colorectal cancer, it was found to have a U-shaped relationship with prognosis, meaning that youngest and oldest patients have poorer prognosis compared to those in the middle age group. 1 In patients with diffuse large B-cell lymphoma, the relative hazard for death did not change significantly during the first to fifth decades, but began to increase exponentially in patients aged over 62 years. 2 However, the association between age and the prognosis of esophageal cancer (EC) has been rarely studied, especially from a nonlinear perspective. Most previous studies either assumed the linearity of age–prognosis relationship in EC, or used experiential prespecified cutting points to define younger versus older patients,3,4 omitting the possibility that age, as a continuous variable, may exhibit a nonlinear effect on prognosis. Moreover, previous studies mainly focused on subgroup of patients receiving certain treatments, either surgical or nonsurgical.5–7 Few studies have attempted to comprehensively demonstrate the full picture of age–prognosis relationship in EC, and even fewer have attempted to explore the similarities and differences of the age–prognosis patterns between different treatments groups.
As surgery and radiotherapy were the two most common curative treatment options for EC, we explored the possible nonlinear association between age and prognosis in patients receiving radical surgery and radiotherapy, respectively. Two distinct age–prognosis association patterns were found and illustrated, the universality of which were confirmed in two large-scale databases. One database came from a hospital-based collaborative group called Jing-Jin-Ji Esophageal and Esophagogastric Cancer Radiotherapy Oncology Group (3JECROG) of China, an East-Asia country with highest EC incidence, mortality, and dominating histological type of squamous cell carcinoma (SCC). The other is the Surveillance, Epidemiology, and End Results (SEER) database, a population-based cancer registry covering approximately 48.0% population of the USA, a country with relatively low incidence and mortality of EC but high incidence of esophageal adenocarcinoma (AC). Additionally, we also tried to explore the underlying reasons accounting for the age–prognosis association patterns by analyzing and integrating the demographic and clinicopathological information from two datasets.
Materials and methods
Data source and elements
Established in 2015, 3JECROG was a collaborative group of several major cancer institutions, aiming to fill the gap in collecting and registering data for patients with EC in China. Initial participant institutions were mainly from Beijing (Jing), Tianjin (Jin), Hebei (Ji), and other provinces/municipalities of North China, but 3JECROG gradually developed into a national organization involving more than 10 hospitals localized in different provinces/municipalities of North, Southwest, Central, and East China. In addition, 3JECROG initially focused on patients receiving definitive radiotherapy, but some hospitals later supplemented the data of surgical patients, making 3JECROG become the largest hospital-based registry of EC in China. The final 3JECROG database contained 6619 EC patients that received radiotherapy from 2001 to 2020 and 4027 patients that received esophagectomy from 1993 to 2019. SEER program, on the other hand, is the largest authoritative source of information on cancer of the USA, collecting data on patients’ demographics, primary tumor, histological type of primary tumor, stage at diagnosis, treatment modalities and sequence, and follow-up for vital status. The latest SEER database released in November 2021 collecting data of 17 registries from 2000 to 2019 was used for analysis. EC patients diagnosed with the International Classification of Diseases for Oncology (third edition, ICD-O-3) codes C150–C159 receiving surgery and/or radiotherapy from SEER dataset were included in the analysis. Patients who only received chemotherapy or did not receive any treatment were excluded. Patients with missing data on age, treatment methods, or follow-ups were excluded. Patients with missing data on other items were retained without further processing. Items included in both datasets were age, sex, year of diagnosis, histological type, TNM stage according to the American Joint Committee on Cancer (AJCC)/Union of International Cancer Control (UICC) manual, treatment received, sequence of treatment, vital status, survival time, and cause of death. 3JECROG database also included facility type and location, radiotherapy dose, date, and status of recurrence/distant metastasis, etc. SEER database also included race, marital status, income (median household income with inflation adjusted to 2019), the region of residence, tumor grade, combined summary stage (localized, regional, distant), etc.
Study design and statistical analysis
Patients in both 3JECROG and SEER databases were divided into two cohorts (surgery and radiotherapy) according to the treatment they received, respectively. The primary endpoint of the study was overall survival (OS) in different treatment cohorts. The secondary endpoints include exploring cancer-specific survival (CSS) and progression-free survival (PFS) in different treatment groups. The above study endpoints were achieved through the following steps. First, we explored the age–prognosis associations of the radiotherapy group in 3JECROG. The relationship between age and OS, CSS, and PFS were all evaluated. OS was defined as the time interval between diagnosis to death from any cause. CSS was defined as the time interval between diagnosis to death from EC. PFS was defined as the time interval between diagnosis to disease progression or death from any cause. We used Cox proportional regression models with restricted cubic splines (RCS), a smoothly joined sum of polynomial functions, to analyze the potential nonlinear association between age and log-relative hazard ratios (HRs) of all-cause mortality (ACM), EC-specific mortality (ECSM), and disease progression and/or ACM (DP ± ACM).8–10 RCS model with four knots at the 5th, 35th, 65th, and 95th centiles was utilized as it yielded the smallest Akaike information criterion, and nonlinearity was tested by comparing the model with linear ones via Wald test. Next, we identified the nadir point and concave region of age–prognosis curves and divided patients into three age groups (i.e. younger, middle, older). Then we listed and compared baseline clinicopathological and demographic characteristics of the three age groups via Chi-square tests to explore intergroup imbalances possible to explain survival differences. The above procedures were repeated in the radiotherapy cohort of SEER database to validate the age–prognosis associations found in 3JECROG dataset. To further validate the age–prognosis association patterns, we combined the two radiotherapy cohorts from 3JECROG and SEER databases to conduct further sensitivity and subgroup analyses. First, we categorized the combined group into fifths evenly by age and compared the HRs of ACM and ECSM of each fifth with the youngest fifth via Cox analysis. Covariates included and adjusted in the Cox model were sex, TNM stage, tumor site, and pathological type. Sensitivity analyses were done by excluding patients with rare histological type, extremely short follow-up or survival time (<3 months), or extreme age (>100 years) to address potential bias. Moreover, to further validate the survival difference between different age groups, we compared OS and CSS of the three age groups via Kaplan–Meier method and log-rank tests. Subgroup analyses were conducted to investigate effect modification after stratifying the whole population by sex, race, histological type, year of diagnosis, tumor stage, treatment details, etc. Interaction between age and subgroup stratification factors was evaluated in multivariate Cox models, with interaction associated with p < 0.01 deemed significant.
The above procedures were repeated in surgical cohorts from SEER and 3JECROG datasets to explore the age–prognosis associations in surgical patients. All statistical analyses were conducted via SPSS Statistics (version 29.0; IBM Corp., Armonk, NY, USA) and R software (version: 4.3.2). The reporting of this study conforms to the STROBE statement (Supplemental Table 7). 11
Results
Study population
A total of 56,457 patients were enrolled in the final analysis, 34,952 of them received radiotherapy as curative treatment, while 21,505 received esophagectomy. Baseline characteristics of radiotherapy and surgical cohorts were summarized in Table 1 and Supplemental Table 1, respectively. More detailed information on the radiotherapy and surgical groups of 3JECROG and SEER databases can be found in Supplemental Tables 2–5 (3JECROG/radiotherapy: Supplemental Table 2; 3JECROG/surgery: Supplemental Table 3; SEER/radiotherapy: Supplemental Table 4; SEER/surgery: Supplemental Table 5). The majority of the patients were male in both the radiotherapy and surgical cohorts (radiotherapy: 76.2%; surgery: 81.4%), and most of them had lower thoracic esophageal tumors (radiotherapy: 46.5%; surgery: 65.6%). The median age of patients in the radiotherapy cohort is 68 years (range: 18–100 years), while the median age of patients in the surgery cohort is 63 years (range: 14–100 years). The gender proportion and median age of patients in both databases were similar, but there were significant differences in tumor staging and pathological types. Patients in the 3JECROG database had significantly more advanced disease at diagnosis compared to those in the SEER database. As for pathological type, in 3JECROG database, 94.9% patients have SCC, while in the SEER database, AC accounts for 59.9% of all patients. The disease staging of patients in the surgical cohort is earlier than that of patients in the radiotherapy cohort. In the radiotherapy cohort, 48.9% of patients have stages III–IV diseases, while in the surgical group, the vast majority of patients (70.2%) have stages I–III diseases. Among the radiotherapy cohort, 6619 patients were from the 3JECROG database, and 28,333 patients were from the SEER database. Among the surgical cohort, 4027 patients were from the 3JECROG database, and 17,478 patients were from the SEER database. The screening flow charts for the two databases were illustrated in Supplemental Figure 1.
Baseline characteristics of younger, middle, and older age groups in the radiotherapy cohort.

AC, adenocarcinoma; SCC, squamous cell carcinoma.
Age–prognosis patterns in radiotherapy group
A U-shaped association with significant nonlinearity was observed between age and OS in the radiotherapy group of 3JECROG database [p < 0.001, Figure 1(a)]. The U-shaped association between CSS, PFS, and age was stronger, indicating that younger patients in the left extreme had higher risk of experiencing ECSM and DP. All three curves reached their nadir at approximately age 65 years, and patients aged 55–75 years had lower risks for all three outcomes compared with patients at both extremes.
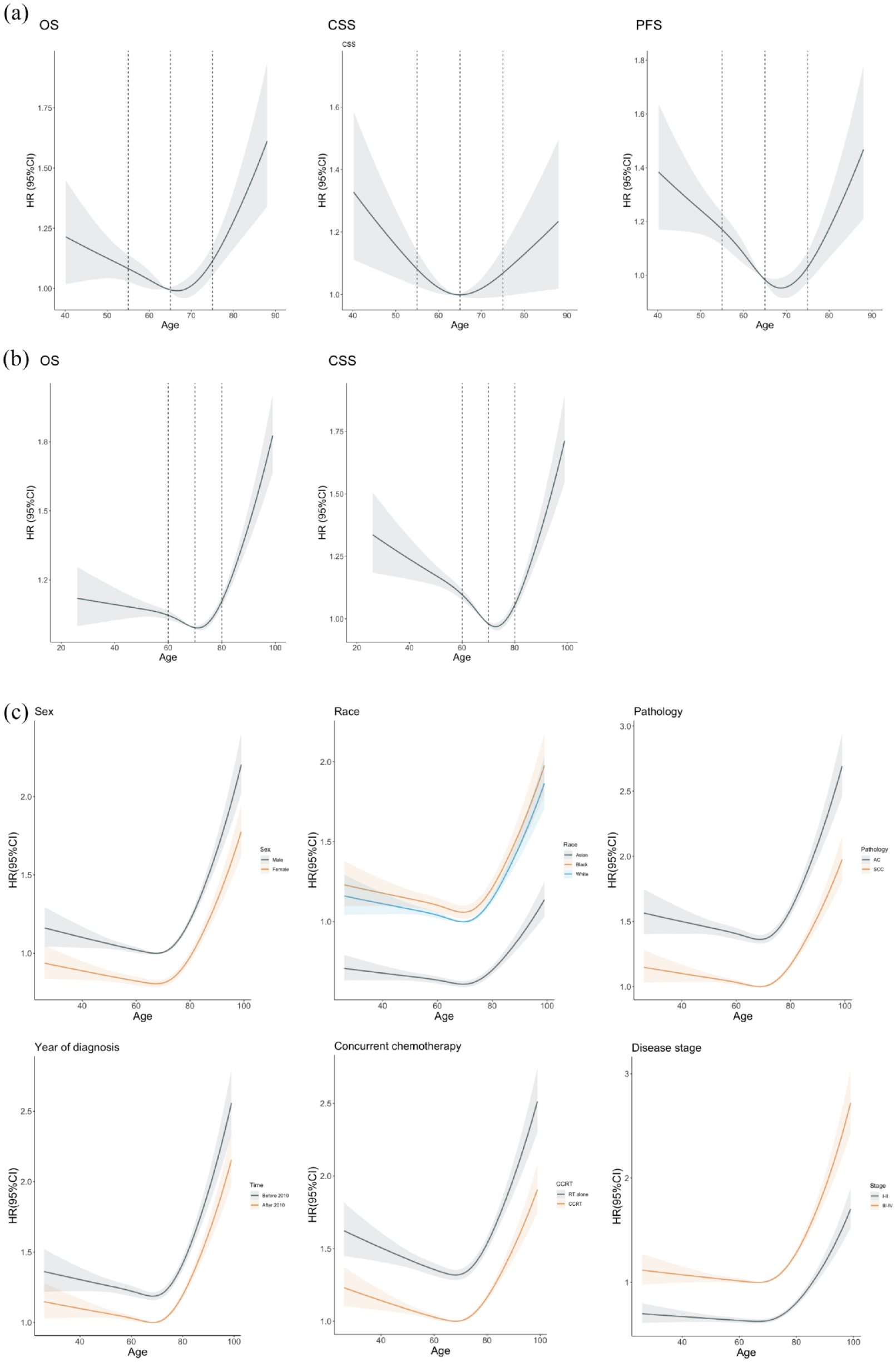
Association between age and prognosis in patients with esophageal cancer receiving nonsurgical treatments (radiotherapy). Nonlinear relationships between age and OS, CSS, PFS were shown in nonsurgical patients of (a) 3JECROG and (b) SEER databases. The p values for nonlinearity were less than 0.05 for all outcomes in both datasets. (c) Subgroup analyses to study the effect modification by sex, race, histological type, year of diagnosis, concurrent chemotherapy, and disease stage. No statistically significant age-by-factor interactions were observed in all subgroup analyses.
Obvious nadir point and significant nonlinearity were also observed in the age–prognosis curves of radiotherapy cohort in SEER database, though age exhibited a J-shaped association with OS [Figure 1(b)]. The association between age and CSS, however, resembled that of 3JECROG databased and showed a U-shaped curve due to higher risk of ECSM in younger patients. Both curves reached their nadir points at approximately age 70 years, with a 5-year right-shift compared to that of the 3JECROG. Patients aged 60–80 years had lower risk for ACM and ECSM compared to patients at both extremes.
Validation of age–prognosis pattern and subgroup analyses
The above age–prognosis patterns were validated in the Cox proportional regression analysis and Kaplan-Meier (KM) analysis of survival after combining two datasets. In Cox regression analysis where 34,952 radiotherapy patients were divided equally into fifths by age, patients in the 2nd youngest fifth of age had a 4% (95% CI: 1–9%) decreased risk for ACM and a 8% (95% CI: 4–13%) decreased risk for ECSM, and patients in the 3rd youngest fifth of age had an 7% (95% CI: 4–12%) decreased risk for ACM and a 14% (95% CI: 10–19%) decreased risk for ECSM compared to the youngest fifth (Table 2). Moreover, such a difference in relative risk for ACM and ECSM between age groups remained persistent after exclusion of rare histological type, extremely short follow-up or survival time (<3 months) or with extreme age (>100 years) in subsequent sensitivity analyses.
Hazard ratio (95% CI) of OS and esophageal CSS to age in patients receiving radiotherapy in the setting of original analysis and sensitivity analyses.

Model 1: original analysis, without exclusion.
Model 2: sensitivity analysis, exclusion of extreme age (>100 years).
Model 3: sensitivity analysis, exclusion of deaths occurred in the early follow-up period (3 months).
Model 4: sensitivity analysis, exclusion of patients with rare/unknown histological type.
CI, confidence interval; CSS, cancer-specific survival; OS, overall survival.
Figure 1(c) showed subgroup analyses after grouping patients from both datasets by sex, race, histological type (SCC versus AC), receiving of concurrent chemotherapy or not, year of diagnosis (before and after 2010), and stage of disease (stage I–II versus stage III–IV) at diagnosis. Although certain degree of heterogeneity could be observed, the relationship between age and the log-scale HR of OS remained J-shaped in all subgroups. No statistically significant age-by-factor interactions were observed in all subgroup analyses.
Differences in baseline characteristics among different age groups
As shown in Supplemental Tables 2 and 3, significant imbalances were seen in baseline characteristics between different age groups in both cohorts. Namely, younger group (age < 55 years in 3JECROG, <60 years in SEER) had more male patients, more advanced primary disease, higher proportion of lymph node metastasis, more advanced TNM stages, and higher proportion of patients receiving concurrent chemotherapy (p < 0.001). In addition, the proportion of black patients was higher in the younger group in the SEER database, while more patients in the older group lived in metropolitan areas (p < 0.001).
Age–prognosis patterns in surgical group
Significantly different age–prognosis associations were seen in surgical cohorts compared to that of the radiotherapy group. In 3JECROG, relative log-scale HR for ACM, ECSM, and ACM ± DP all increased with age [Figure 2(a)]. Such patterns were validated in SEER database, though the relative risk for ACM did not soar until 70 years and increased rather mildly with age before that [Figure 2(b)]. In terms of CSS, relative risk for ECSM also increased among younger patients at the left extreme in the surgical group, making age present an association approximated to J-shaped with CSS in SEER database. Cox proportional regression analysis and KM analysis for survival also confirmed that relative risk for mortality increased with increasing age among surgical patients. Patients in the 2nd, 3rd, 4th, and 5th youngest fifth of age had a 7% (95% CI: 1–11%), 13% (95% CI: 5–19%), 19% (95% CI: 12–25%), 43% (95% CI: 30–57%) increased risk for ACM compared to the youngest fifth, respectively (Supplemental Table 6). Sensitivity analyses made little difference to the estimated associations between age and OS or CSS. As no obvious nadir was observed in age–prognosis curves of surgical cohorts, we divided surgical patients into younger, middle, and older groups using the criteria derived from the radiotherapy cohorts.
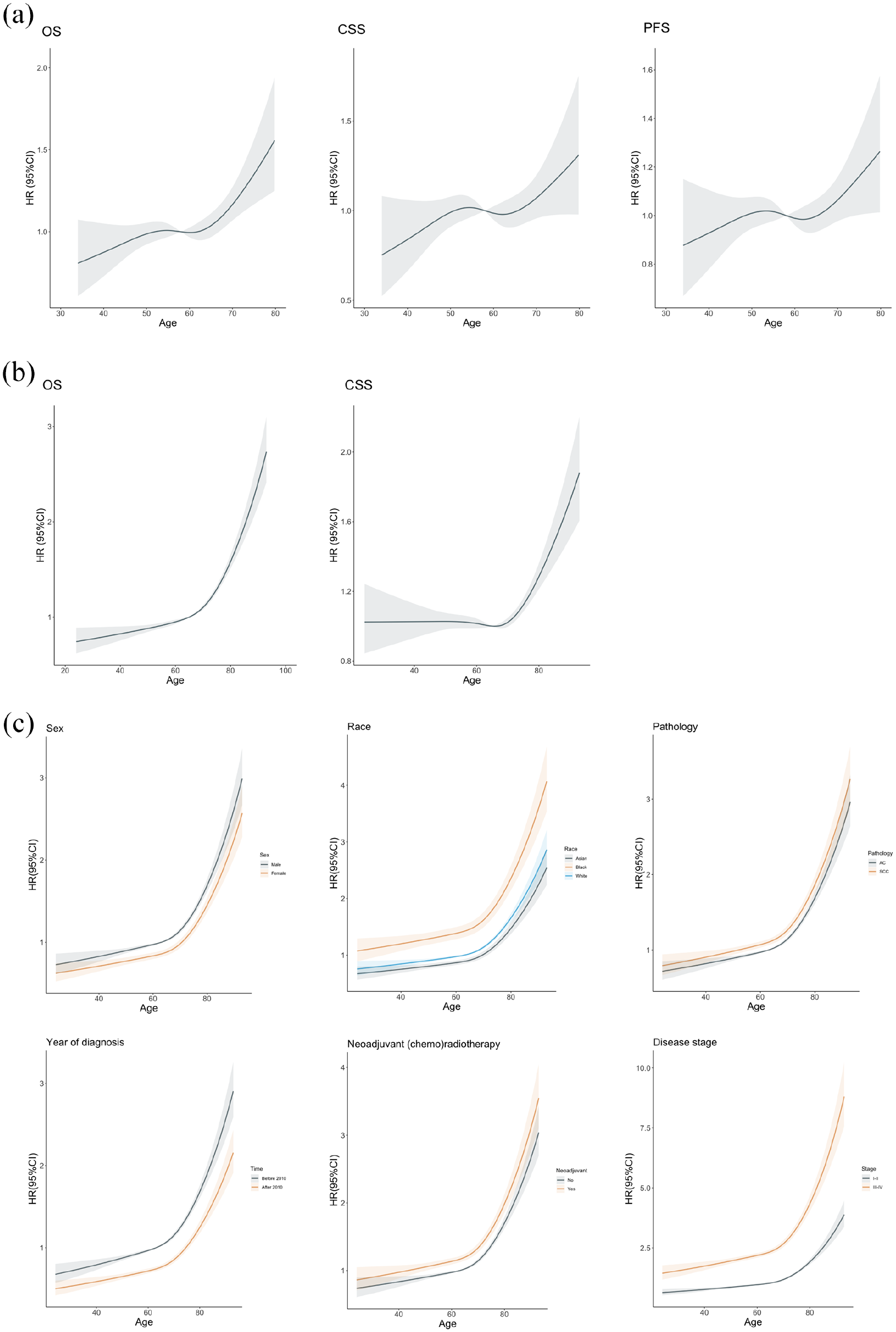
Association between age and prognosis in patients with esophageal cancer receiving surgical treatments. Nonlinear relationships between age and OS, CSS, PFS were shown in surgical cohorts of (a) 3JECROG and (b) SEER databases. The p values for nonlinearity were less than 0.05 for all outcomes in both datasets. (c) Subgroup analyses to study the effect modification by sex, race, histological type, year of diagnosis, concurrent chemotherapy, and disease stage. No statistically significant age-by-factor interactions were observed in all subgroup analyses.
We also compared the baseline demographic and clinicopathological characteristics between different age groups in both surgical cohorts, and results showed that significant imbalances were also detected (Supplemental Tables 4 and 5). Similar to that of the radiotherapy group, younger patients in the surgical group also had more advanced primary disease, higher proportion of lymph node metastasis, and more advanced TNM stages. Besides, a higher proportion of younger patients received neoadjuvant (chemo)radiotherapy (p < 0.001).
In subgroup analyses, the association between age and OS resembled that of the SEER database in most subgroups, with no statistically significant interactions detected between age and subgroup factors [Figure 2(c)].
Discussion
In this study, we identified two distinct age–prognosis association patterns in patients with EC treated with curative surgery and radiotherapy. In the radiotherapy group, similar to the findings of Lieu et al. 1 in metastatic colorectal cancer, age showed a U-/J-shaped relationship with survival. In patients receiving surgical treatments, however, the U-shaped curve disappeared and was replaced by a near-linear relationship between age and survival. That is to say, the risk for ACM increased with age in patients with EC receiving surgical treatments. The abovementioned associations were verified separately in two large national databases, partly validating the reliability of our findings. Our findings were also validated by Cox regression analysis for survival after combining the two databases together.
Although few previous studies had investigated the age–prognosis relationships of EC from a nonlinear perspective, our findings could get some indirect support from several large-scale retrospective researches. In a Taiwan study including 14,393 EC patients, the majority of patients received nonsurgical radiotherapy and the study-defined middle-age group (45–64 years) showed significantly better OS compared to that of the younger group (<45 years, HR: 0.86, 95% CI: 0.78–0.96, p = 0.0067) in the multivariate Cox regression analysis. 12 Moreover, no significant difference was detected between the OS of younger (<45 years) and older (⩾65 years) groups, which also correlates with the age–prognosis pattern found in our analysis. On the other hand, in a Dutch population-based cohort study involving 59,584 EC patients, most potentially curable patients received surgical treatment, and the results showed that younger group (defined as patients <50 years in this study) had superior survival outcomes compared to that of the middle-age group (50–74 years) and older group (⩾75 years). 13 Although the definition and division of different age groups may have slight variance, the results of the above studies were consistent with the two distinct age–prognosis patterns found in our study.
As compared to younger patients, older patients had higher risk of dying from concomitant diseases, we specifically investigated the association between age and ECSM in both treatment-modality groups. As we expected, relative risk increased in younger patients when the outcome was switched from ACM to ECSM, yet it did not change the overall trend of age–prognosis pattern in both treatment cohorts.
Many previous studies have found the phenomenon that younger EC patients presented with more advanced disease at diagnosis compared to their older counterparts.5,14 This phenomenon has been validated in our study and considered to be the leading cause for inferior survival of younger patients in the radiotherapy group. Both lower tumor awareness and higher tumor invasiveness among younger population may contribute to this phenomenon, though specific explanations remained to be revealed by further studies. Nevertheless, although younger patients in both treatment cohorts presented with more advanced disease at diagnosis, it seems that only the survival of those in the radiotherapy cohort was affected. We believe that this may be ascribed to the following reasons. First, younger patients in the radiotherapy group had far more advanced disease than those of the surgical group, which may affect their response to radical radiotherapy. Second, neoadjuvant chemo(radio)therapy is significantly more frequently applied among younger patients in the surgical group and may bring them survival benefits. Whatever the reason was, our finding suggested that it was meaningful to raise awareness of EC among young adults for early detection of tumors. As for survival comparison between different treatment groups, although significantly longer OS and CSS were observed in all age subgroups in the surgical cohort compared to that of the radiotherapy cohort, we do not think it could lead to the conclusion that surgery is superior to radical (chemo)radiotherapy in the treatment of EC due to imbalanced baseline tumor stages among two treatment cohorts. Nevertheless, we do believe our results support more aggressive tri-modality treatment among younger patients, as younger patients in the surgical cohort showed superior survival in spite of more advanced disease.
Among all the results, it particularly caught our interest that the age–prognosis curves of the radiotherapy cohort in SEER dataset shifted rightward by approximately 5 years compared to that of the 3JECROG. The reason for this right-shift remained unknown, but we did observe a much higher proportion of patients receiving concurrent chemotherapy in the SEER database compared to that in the 3JECROG. In fact, the relatively low application of concurrent chemotherapy among EC patients receiving definitive radiotherapy in China has long been discovered by our previous series of 3JECROG studies, 15 and the above phenomenon is particularly prominent among elderly patients in their 70s. In this study, the proportion of patients age between 75 and 80 years receiving CCRT was 77.1% and 18.2% in SEER and 3JECROG databases, respectively. We assume this huge disparity in CCRT rates may be the leading cause for survival difference among elderly patients between two datasets and the rightward shift of age–prognosis curves in SEER database.
As a combined analysis of two retrospective databases, our study had several limitations. First, since the detailed dose and site of radiotherapy was not described in SEER database, we cannot guarantee that all radiotherapy patients included in the SEER database received full-dose definitive radiotherapy. Nevertheless, as not all radiotherapy patients enrolled in 3JECROG database completed full-course radiotherapy, the mismatch of patients from two databases caused by lack of information may not be as significant as imagined. Second, due to the retrospective nature of the study, a considerable number of variables were incomplete after combining the two databases and thus could not be adjusted in the multivariable Cox model. The reliability of the results of this study still needs further confirmation through prospective or large-scale retrospective studies. Last, patients in the SEER database did not have accurate AJCC/UICC TNM classifications, which affected our further in-depth analysis on this aspect.
Conclusion
To conclude, our study for the first time explored the association between age and prognosis in patients with EC receiving different definitive treatment modalities from a nonlinear perspective. Patients in surgical and radiotherapy cohorts showed distinct age–prognosis association patterns, and explanations for this difference were explored by analyzing baseline characteristics between different age groups in both cohorts. Further translational studies are warranted to reveal potential biologic or genetic difference between different age groups in patients with EC.
Supplemental Material
sj-docx-1-tam-10.1177_17588359241261009 – Supplemental material for Two distinct age-prognosis patterns in patients with esophageal cancer undergoing surgical and radiotherapy treatments: a combined analysis of 3JECROG and SEER databases
Supplemental material, sj-docx-1-tam-10.1177_17588359241261009 for Two distinct age-prognosis patterns in patients with esophageal cancer undergoing surgical and radiotherapy treatments: a combined analysis of 3JECROG and SEER databases by Chen Li, Xiao Chang, Qifeng Wang, Qingsong Pang, Zefen Xiao, Wencheng Zhang and Zhiyong Yuan in Therapeutic Advances in Medical Oncology
Footnotes
Acknowledgements
We thank the Jing-Jin-Ji Esophageal and Esophagogastric Cancer Radiotherapy Oncology Group (3JECROG) for its effort in collecting data from participating institutions.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
