Abstract
Background:
Docetaxel, cisplatin, and 5-fluorouracil (DCF) combination chemotherapy has been established as one of the standard neoadjuvant therapies for locally advanced esophageal squamous cell carcinoma (ESCC). However, little is known about prognostic factors in patients with residual pathological disease after neoadjuvant DCF followed by surgery for locally advanced ESCC who are candidates for adjuvant nivolumab.
Objectives:
This study aimed to investigate prognostic factors in patients with residual pathological disease after neoadjuvant DCF chemotherapy followed by surgery for locally advanced ESCC.
Design:
This was a retrospective cohort study.
Methods:
This retrospective cohort study included patients who received neoadjuvant DCF followed by surgery for locally advanced ESCC between June 2014 and January 2020 at the National Cancer Center Hospital East.
Results:
Among a total of 210 patients, 45 patients (21.4%) achieved a pathological complete response. The 3-year disease-free survival (DFS) rate was significantly lower in patients with residual pathological disease than in those with a pathological complete response [53.5% versus 74.5%; hazard ratio (HR): 2.09, 95% confidence interval (CI): 1.16–3.77, p = 0.01]. In patients with residual pathological disease (n = 165), multivariate analysis revealed that pathological node positivity (HR: 3.59, 95% CI: 1.92–6.71, p < 0.01), supraclavicular lymph node metastasis (HR: 2.15, 95% CI: 1.19–3.90, p = 0.01), and lymphovascular invasion (HR: 1.90, 95% CI: 1.14–3.17, p = 0.02) were significantly associated with poor DFS.
Conclusion:
In this largest-to-date cohort study, patients with residual pathological disease after neoadjuvant DCF followed by surgery for locally advanced ESCC had a poor prognosis. In these patients, pathological node positivity, including supraclavicular lymph node metastasis, and lymphovascular invasion were considered significant prognostic factors.
Keywords
Introduction
Esophageal cancer is the sixth leading cause of cancer-related deaths worldwide.1,2 The prevalence of esophageal adenocarcinoma is higher in Western countries, whereas more than 90% of cases in East Asia are esophageal squamous cell carcinoma (ESCC). 3 In patients with locally advanced ESCC, neoadjuvant chemoradiotherapy followed by surgery is the global standard of care.2,4–6 However, survival after surgery remains poor, particularly in the 60% of patients with the residual pathological disease after neoadjuvant chemoradiotherapy compared to the remaining 40% who achieved a pathological complete response.7–9 Checkmate 577 trial, 10 a randomized, placebo-controlled phase III trial, demonstrated that adjuvant treatment with nivolumab, a fully human monoclonal anti-programmed death-1 antibody, for up to 1 year significantly improved disease-free survival (DFS) after neoadjuvant chemoradiotherapy in patients with resected esophageal or gastroesophageal cancer and residual pathological disease, with a median DFS of 22.4 and 11.0 months with nivolumab and placebo, respectively [hazard ratio (HR): 0.69, 96.4% confidence interval (CI): 0.56–0.86, p < 0.01]. Consequently, the National Comprehensive Cancer Network guidelines for esophageal and esophagogastric cancers recommend adjuvant nivolumab as a preferred regimen after preoperative chemoradiation followed by R0 resection and residual pathological disease. 2
Meanwhile, docetaxel, cisplatin, and 5-fluorouracil (DCF) combination chemotherapy has been used as neoadjuvant chemotherapy to improve systemic control in patients with locally advanced ESCC.11–14 A three-arm phase III trial, the Japanese Clinical Oncology Group (JCOG) 1109, compared cisplatin plus 5-fluorouracil (CF), DCF, and CF with radiotherapy (CF-RT) as neoadjuvant therapy for locally advanced ESCC. It demonstrated that DCF improved overall survival (OS) compared to CF, with 3-year OS rates of 72.1% and 62.6% with DCF and CF, respectively (HR: 0.68, 95% CI: 0.50–0.92, p = 0.01), whereas CF-RT did not improve OS compared to CF, with 3-year OS rates of 68.3% and 62.6% with DCF and CF, respectively (HR: 0.84, 95% CI: 0.63–1.12, p = 0.12). 15 Based on the findings of the JCOG1109 NExT study, DCF has been added to standard neoadjuvant therapies for locally advanced ESCC. However, little is known about prognosis and prognostic factors in patients with residual pathological disease after neoadjuvant DCF chemotherapy.
Therefore, this study investigated prognosis and prognostic factors in patients with residual pathological disease after neoadjuvant DCF chemotherapy followed by surgery for locally advanced ESCC.
Patients and methods
Patients
This retrospective observational study included data from patients who received neoadjuvant DCF chemotherapy followed by surgery for locally advanced ESCC at the National Cancer Center Hospital East in Kashiwa, Japan, between 1 June 2014 and 31 January 2020. The study period was designed to include patients with a minimum follow-up duration of 2 years. The study protocol was approved by the Institutional Review Board of the National Cancer Center Hospital East (2017-120). Informed consent was waived due to the retrospective observational study design, with an opt-out option provided on the institutional website.
The eligibility criteria were as follows: age of 18 years or older; locally advanced, histologically proven ESCC, with only supraclavicular lymph node metastasis allowed; Eastern Cooperative Oncology Group performance status (ECOG PS) scores of 0–2; adequate bone marrow and organ function; and surgery after neoadjuvant DCF chemotherapy. The exclusion criteria were as follows: known additional malignancy progressing or requiring active treatment within the previous 3 years and microscopically incomplete resection (R1) or macroscopically incomplete resection (R2) according to the Cancer Staging Manual of the American Joint Committee on Cancer 16 (Supplemental Figure 1).
Neoadjuvant DCF chemotherapy
The neoadjuvant DCF chemotherapy, including docetaxel (70 mg/m2, day 1), cisplatin (70 mg/m2, day 1), and 5-fluorouracil (750 mg/m2, days 1–5), was administered every 3 weeks. Prior to surgery, patients were scheduled to receive three cycles of neoadjuvant DCF chemotherapy. In patients with febrile neutropenia, Grade 4 neutropenia, or Grade 3 or higher thrombocytopenia, the dose of each drug was reduced by 20% in the subsequent cycle. Any additional dose modifications were made based on toxicities at the discretion of the treating clinician.
Surgical procedures
Patients with thoracic ESCC underwent subtotal esophagectomy with radical three-field lymphadenectomy, including cervical and upper mediastinal lymphadenectomy, via open thoracotomy, thoracoscopy, or robot-assisted thoracoscopy from the right side. Abdominal lymphadenectomy was performed using open laparotomy or laparoscopic surgery. Patients with cervical ESCC underwent cervical esophagectomy or pharyngo-laryngo-esophagectomy with radical cervical and upper mediastinal lymphadenectomy and reconstruction with a free jejunal graft.
Assessment
The efficacy endpoints were DFS, OS, and distant metastasis-free survival (DMFS). DFS was defined as the time interval between the date of surgery and recurrence, as clinically confirmed by endoscopy or computed tomography (CT) scans, or death from any cause. OS was defined as the time interval between the date of surgery and death from any cause. DMFS was defined as the time interval between the date of surgery and distant metastasis, as clinically confirmed by CT scans or death from any cause. CT scans were generally performed every 3 weeks during neoadjuvant DCF chemotherapy, every 4 months up to 2 years, and subsequently every 6 months up to 5 years after surgery.
Pathological staging was performed according to the 7th edition of the American Joint Committee on Cancer/Union for International Cancer Control staging system.16,17 The surgically resected specimens were pathologically diagnosed according to the Japanese Classification of Esophageal Cancer, and tumor regression was graded as follows: Grade 0: no recognizable cytological or histological therapeutic effect; Grade 1a: viable cancer cells accounting for two-thirds of the tumor; Grade 1b: viable cancer cells accounting for one-third or more but less than two-thirds of the tumor; Grade 2: viable cancer cells accounting for less than one-third of the tumor; and Grade 3: no evidence of viable cancer cells. 18 Adverse events during neoadjuvant DCF chemotherapy were assessed according to Common Terminology Criteria for Adverse Events version 5.0.
Statistical methods
DFS, DMFS, and OS were calculated using Kaplan–Meier estimates, and differences between groups were determined using the log-rank test with a two-sided p-value of 0.05. HRs and corresponding 95% CIs were determined using the Cox proportional hazards model. Follow-up time was defined as the time between surgery and the last follow-up for censored cases. The Cox regression model was used for multivariate analyses of DFS and OS, with factors with p-values less than 0.10 in the univariate analysis being forced into the model. Covariates included age, sex, ECOG PS score, tumor location, number of neoadjuvant DCF chemotherapy cycles, neoadjuvant DCF chemotherapy dose modification, histological differentiation, pathological T stage, pathological N stage, supraclavicular lymph node metastasis, intramural metastasis, lymphovascular invasion, perineural invasion, and tumor regression grade (TRG) on primary tumor tissue samples. All statistical analyses were performed using EZR (Saitama Medical Center, Jichi Medical University, Saitama, Japan), a graphical user interface for R (The R Foundation for Statistical Computing, Vienna, Austria).
Reporting guideline
The reporting of this study conforms to Strengthening the reporting of observational studies in epidemiology (STROBE) (Supplemental Table 1). 19
Results
Patient characteristics and outcomes
During the study period, 239 patients were treated with neoadjuvant DCF chemotherapy followed by surgery for locally advanced ESCC at the study institution. Patients who had R1 or R2 resection (n = 25), intraoperative death (n = 2), or additional malignancies progressing or requiring active treatment within the previous 3 years (n = 2) were excluded from the analysis.
Table 1 summarizes the baseline characteristics of the 210 patients included in the final analysis. The proportions of patients with clinical T3 and N1–3 stage ESCC were 84.3% (n = 177) and 87.1% (n = 183), respectively. In 128 patients (61.0%), dose modifications to neoadjuvant DCF chemotherapy were required. The neoadjuvant DCF chemotherapy was discontinued in 70 patients (33.3%) due to toxicities (n = 40), progression disease of DCF chemotherapy (n = 26), esophageal stenosis (n = 3), and patient preference (n = 1) (Supplemental Table 2). Most Grade 3 or worse treatment-related adverse events with neoadjuvant DCF chemotherapy were neutropenia (n = 163, 77.6%), leukopenia (n = 124, 59.0%), and febrile neutropenia (n = 55, 26.2%). In all, 129 patients (61.4%) received granulocyte-colony stimulating factor for febrile neutropenia or Grade 3 or higher neutropenia and 3 patients (1.4%) received prophylactic granulocyte colony-stimulating factor. In all, 45 patients (21.4%) achieved a pathological complete response, while the remaining 165 patients (78.6%) had a residual pathological disease. Ten patients (4.8%) had recorded complications, with Clavien–Dindo classification IIIb or higher [bleeding (n = 3), leakage suture (n = 2), cardiopulmonary arrest (n = 1), chylothorax (n = 1), pyothorax (n = 1), pyloric dysfunction (n = 1), and graft necrosis (n = 1)].
Patient characteristics.
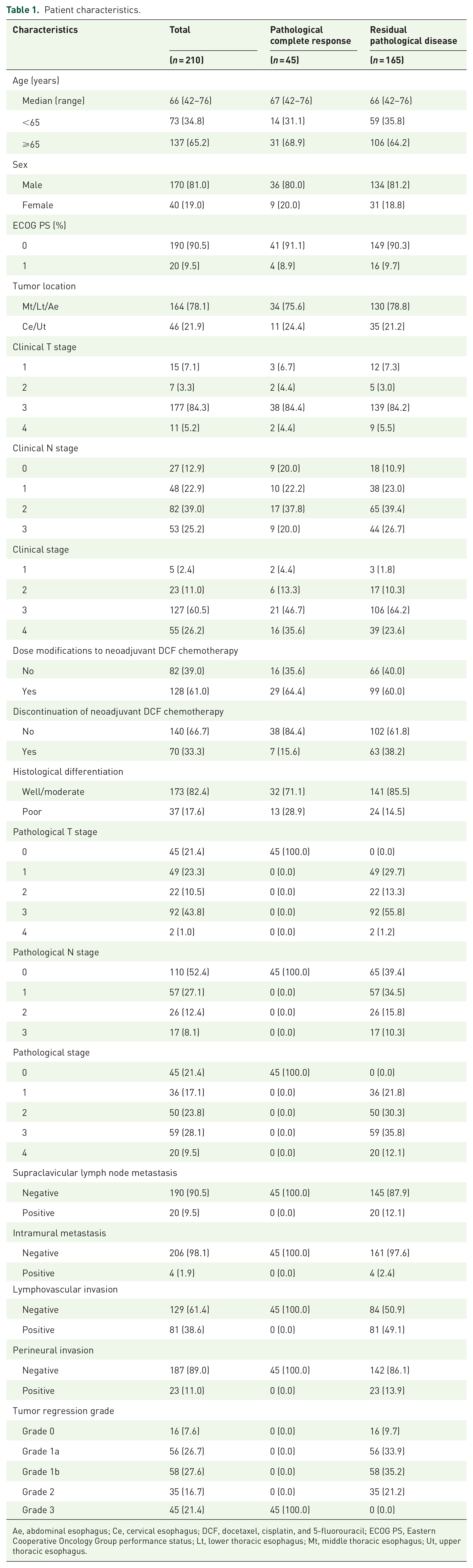
Ae, abdominal esophagus; Ce, cervical esophagus; DCF, docetaxel, cisplatin, and 5-fluorouracil; ECOG PS, Eastern Cooperative Oncology Group performance status; Lt, lower thoracic esophagus; Mt, middle thoracic esophagus; Ut, upper thoracic esophagus.
The median follow-up period was 36.2 months (95% CI: 30.3–40.7 months). During the study period, 82 patients (39.0%) had tumor recurrence, including locoregional recurrence (n = 21), distant metastasis (n = 51), and locoregional and distant metastases (n = 10). The treatments for recurrent disease are summarized in Supplemental Table 3. Among those with recurrence, 82 patients received subsequent therapy for recurrent disease, including palliative chemotherapy (n = 33), salvage chemoradiotherapy (n = 25), surgery (n = 10), radiotherapy (n = 1), and the best supportive care alone (n = 13). During the study period, the leading causes of death were ESCC recurrence (n = 41), other malignancies (n = 5), and other causes (n = 4).
Prognostic impact of pathological complete response in patients receiving neoadjuvant DCF chemotherapy followed by surgery
There were no clear differences in baseline characteristics, including age, sex, ECOG PS score, and clinical T and N stages, between patients with pathological complete response and those with residual pathological disease (Table 1). The rate of tumors with histologically poor differentiation was significantly higher in the pathological complete response group than in the residual pathological disease group (28.9% versus 14.5%, p = 0.05). The 3-year DFS and OS rates were significantly lower in the residual pathological disease group than in the pathological complete response group (53.5% versus 74.5%; HR: 2.09, 95% CI: 1.16–3.77, p = 0.01; and 71.4% versus 91.2%, HR: 2.94, 95% CI: 1.18–7.69, p = 0.02, respectively; Figures 1 and 2).

Kaplan–Meier curves for disease-free survival according to residual pathological disease.

Kaplan–Meier curves for overall survival according to residual pathological disease.
Prognosis in patients with residual pathological disease
Univariate analysis (Table 2) revealed that pathological T Stages 3–4 (HR: 2.49, 95% CI: 1.53–4.06), pathological N Stages 1–3 (HR: 4.82, 95% CI: 2.65–8.77), supraclavicular lymph node metastasis (HR: 3.10, 95% CI: 1.78–5.39), lymphovascular invasion (HR: 3.44, 95% CI: 2.12–5.58), perineural invasion (HR: 2.01, 95% CI: 1.13–3.60), and TRG Grade 0–1b (HR: 4.38, 95% CI: 1.91–10.03) were all associated with significantly shorter DFS. However, multivariate analysis revealed that pathological N Stages 1–3 (HR: 3.59, 95% CI: 1.92–6.71, p < 0.01), supraclavicular lymph node metastasis (HR: 2.15, 95% CI: 1.19–3.90, p = 0.01), and lymphovascular invasion (HR: 1.90, 95% CI: 1.14–3.17, p = 0.02) were independently associated with poor DFS.
Univariate and multivariate analyses of disease-free survival in patients with residual pathological disease.

Ae, abdominal esophagus; Ce, cervical esophagus; CI, confidence interval; DCF, docetaxel, cisplatin, and 5-fluorouracil; ECOG PS, Eastern Cooperative Oncology Group performance status; HR, hazard ratio; Lt, lower thoracic esophagus; Mt, middle thoracic esophagus; Ut, upper thoracic esophagus.
Univariate analysis (Table 3) revealed that pathological T Stages 3–4 (HR: 2.41, 95% CI: 1.26–4.60), pathological N Stages 1–3 (HR: 3.04, 95% CI: 1.50–6.16), supraclavicular lymph node metastasis (HR: 2.75, 95% CI: 1.36–5.57), lymphovascular invasion (HR: 4.05, 95% CI: 2.09–7.87), and TRG Grade 0–1b (HR: 3.53, 95% CI: 1.27–9.78) were all associated with significantly shorter OS times. However, in multivariate analysis, the lymphovascular invasion was found to be significantly associated with poor prognosis (HR: 2.52, 95% CI: 1.23–5.17, p = 0.01), whereas pathological N Stages 1–3 (HR: 1.96, 95% CI: 0.94–4.12, p = 0.07) and supraclavicular lymph node metastasis (HR: 1.84, 95% CI: 0.89–3.77, p = 0.10) were found to be associated with poor OS, albeit without statistical significance.
Univariate and multivariate analyses of overall survival in patients with residual pathological disease.
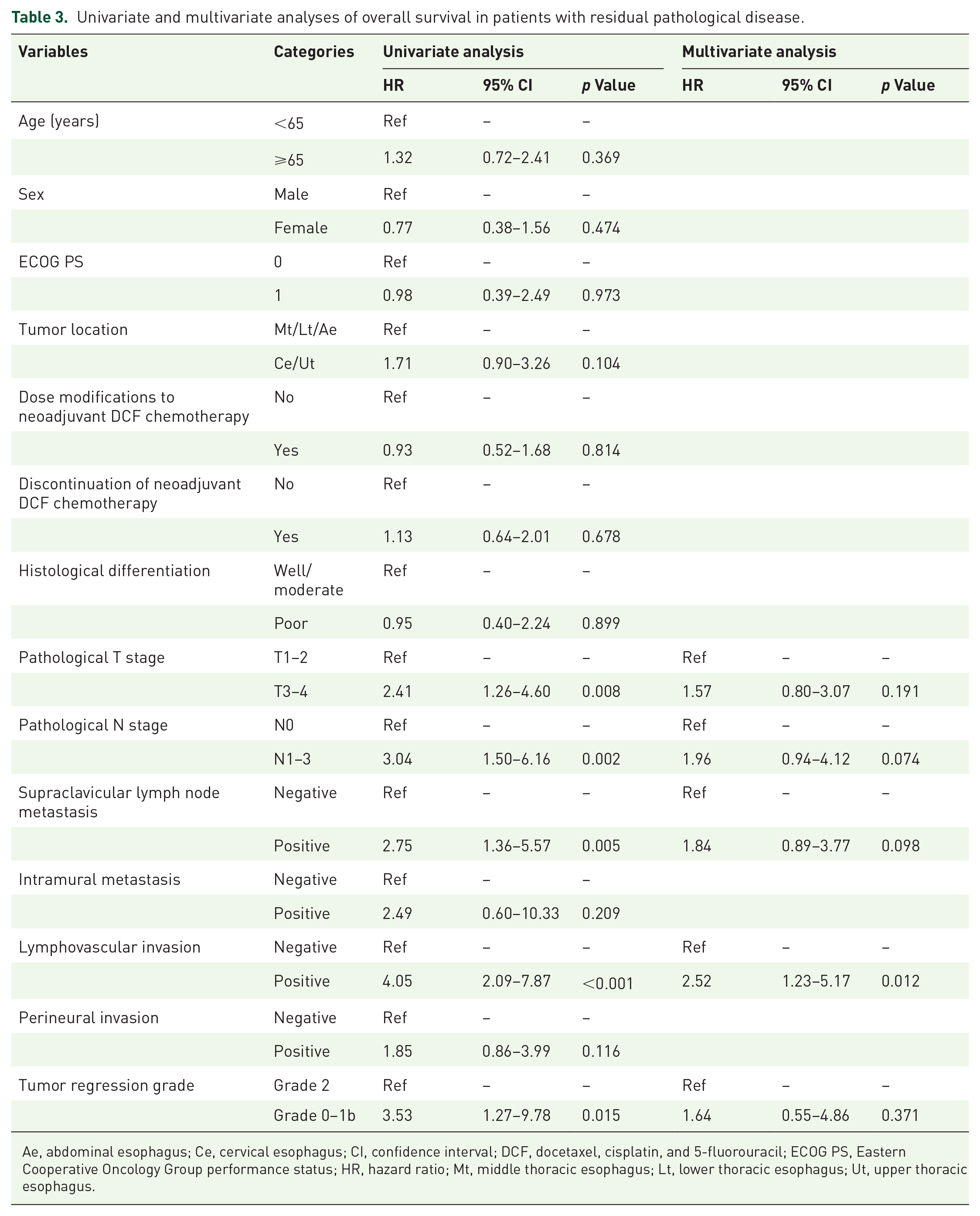
Ae, abdominal esophagus; Ce, cervical esophagus; CI, confidence interval; DCF, docetaxel, cisplatin, and 5-fluorouracil; ECOG PS, Eastern Cooperative Oncology Group performance status; HR, hazard ratio; Mt, middle thoracic esophagus; Lt, lower thoracic esophagus; Ut, upper thoracic esophagus.
Survival outcomes in patients with pathological node positivity and/or lymphovascular invasion
Among the 165 patients with residual pathological disease, 120 (72.7%) had at least one prognostic factor of pathological node positivity, supraclavicular lymph node metastasis, and lymphovascular invasion. The 3-year DFS rate was significantly lower in patients with pathological node positivity, supraclavicular lymph node metastasis, and/or lymphovascular invasion than in those without pathological node positivity or lymphovascular invasion (42.0% versus 84.1%; HR: 5.47, 95% CI: 2.51–11.91, p < 0.01; Figure 3). In addition, distant recurrence was more frequent in patients with pathological node positivity, supraclavicular lymph node metastasis, and/or lymphovascular invasion (52/120, 43.3%) than in those without pathological node positivity or lymphovascular invasion (4/45, 8.9%). The 3-year DMFS rates in patients with and without pathological node positivity, supraclavicular lymph node metastasis, and/or lymphovascular invasion were 53.5% and 90.7%, respectively [HR: 6.87, 95% CI: 2.48–19.02, p < 0.01; Supplemental Figure 2(a)]. Similarly, the 3-year OS rate was significantly lower in patients with pathological node positivity, supraclavicular lymph node metastasis, and/or lymphovascular invasion than in those without pathological node positivity or lymphovascular invasion [63.5% versus 90.7%; HR: 5.13, 95% CI: 1.84–14.35, p < 0.01; Supplemental Figure 2(b)].
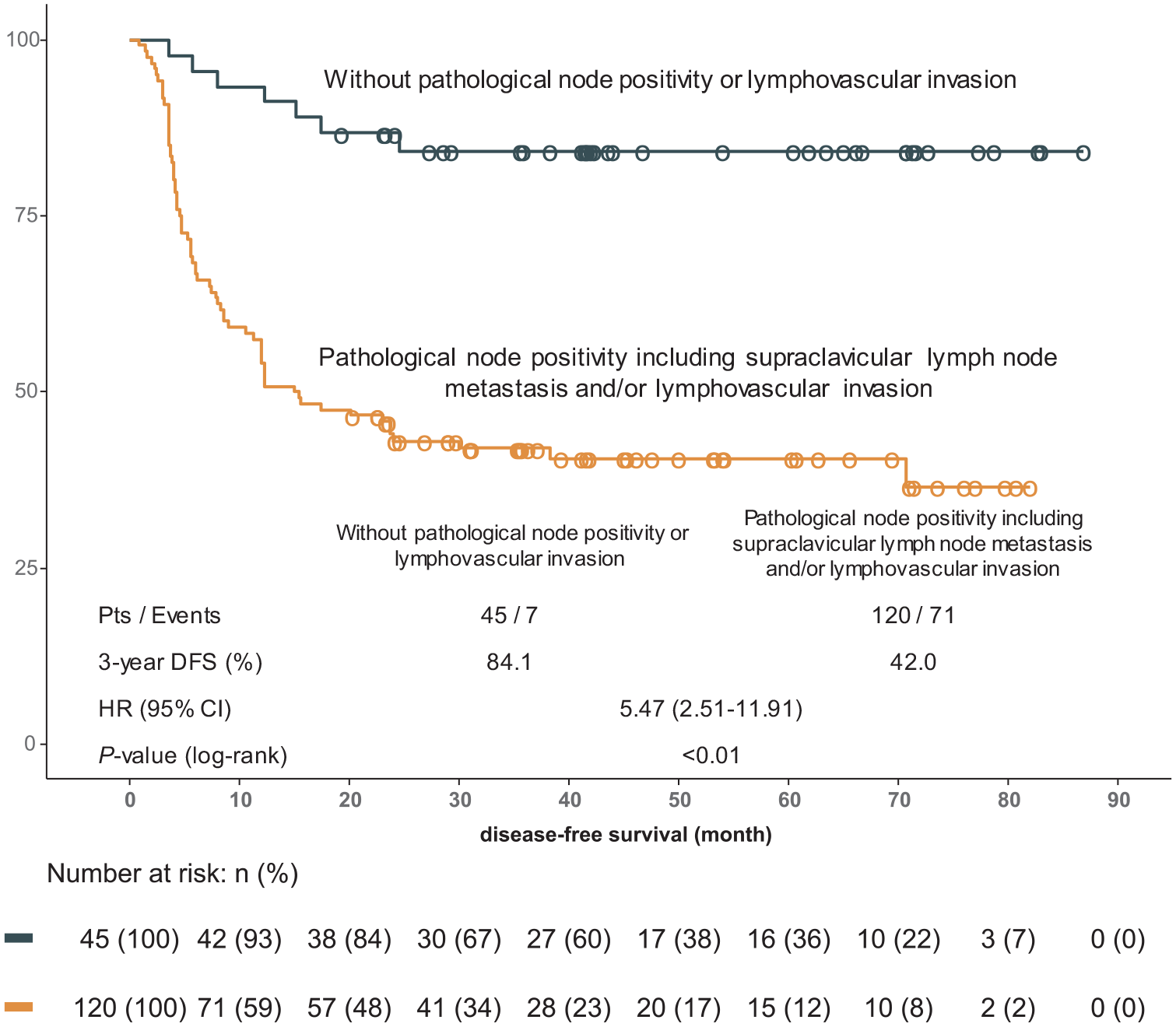
Kaplan–Meier curves for disease-free survival in patients with residual pathological disease according to pathological node positivity, supraclavicular lymph node metastasis, and/or lymphovascular invasion.
Discussion
This is the largest cohort study investigating prognostic factors in patients with locally advanced ESCC receiving neoadjuvant DCF chemotherapy followed by surgery. In this study, we found that pathological node positivity, supraclavicular lymph node metastasis, and/or lymphovascular invasion were strongly associated with poor DFS and OS in patients with residual pathological disease.
While previous studies have reported a poor prognosis for patients with the residual pathological disease after neoadjuvant chemoradiotherapy followed by surgery for locally advanced ESCC,7,20–22 few studies have investigated the prognosis of patients with ESCC who received neoadjuvant DCF chemotherapy followed by surgery.14,23–26 In the present study, 21.4% of patients achieved a pathological complete response, which is consistent with the reported rate of 18.6% in patients with no residual tumors in the JCOG1109 NExT study. 15 Similar findings were observed in patients with residual pathological disease after neoadjuvant chemoradiotherapy followed by surgery in the Checkmate-577 trial. Meanwhile, the findings of the JCOG1109 study suggested that the pathological complete response rate may not be useful as a surrogate marker for OS in patients treated with different treatment modalities, such as chemotherapy and chemoradiotherapy. However, our findings suggest that a pathological complete response may still be considered a prognostic factor for certain neoadjuvant treatment modalities.
In this study, we also assessed the prognosis of patients with residual pathological disease and found that pathological node positivity, supraclavicular lymph node metastasis, and lymphovascular invasion were independent poor prognostic factors for DFS in multivariate analysis. In fact, patients with pathological node positivity, supraclavicular lymph node metastasis, and/or lymphovascular invasion had poor outcomes, with 3-year DFS and OS rates of 42.0% and 63.5%, respectively. Furthermore, 66 of the 71 events (93.0%) in the DFS analysis were distant recurrences or deaths. The mainstay of treatment for distant recurrence is palliative chemotherapy or best supportive care alone, whereas salvage chemoradiotherapy may be considered for treating patients with locoregional recurrence.2–4 Accordingly, patients with pathological node positivity or lymphovascular invasion populations, which comprised 72.7% of patients with residual pathological disease in the current study, should be strongly considered candidates for adjuvant nivolumab therapy to reduce the risk of recurrence. Conversely, patients without pathological node positivity, supraclavicular lymph node metastasis, or lymphovascular invasion had favorable outcomes, with high 3-year DFS and OS rates comparable to those observed in patients with a pathological complete response. Considering the physical burden of intensive neoadjuvant DCF chemotherapy and invasive surgery for ESCC, patients without pathological node positivity, supraclavicular lymph node metastasis, or lymphovascular invasion may not require additional treatment, including adjuvant nivolumab therapy, if patients or physicians are concerned about immune-related adverse events, which occur in approximately 20–30% of the treated patients. 10
However, no clinical trial has yet demonstrated the utility of adjuvant nivolumab after neoadjuvant DCF therapy followed by surgery. The Checkmate-577 trial enrolled patients who underwent R0 resection for esophageal or gastroesophageal cancer and had residual pathological disease after neoadjuvant chemoradiotherapy but excluded those who had received neoadjuvant chemotherapy. The post hoc analysis of the Checkmate-577 trial revealed a consistent efficacy of adjuvant nivolumab therapy for DFS, irrespective of radiotherapy dosage ranging from less than 41.4 Gy to more than 50.4 Gy. 10 Furthermore, the ATTRACTION-3 trial, which demonstrated the superiority of nivolumab monotherapy over chemotherapy in OS for unresectable advanced or metastatic ESCC, revealed a consistent efficacy of nivolumab irrespective of radiotherapy history. 27 These findings support the utility of adjuvant nivolumab therapy after neoadjuvant chemotherapy.
The present study has several limitations. First, this was a non-randomized retrospective study with a limited sample size conducted at a single institution. However, this is the largest cohort of patients receiving a newly established standard treatment of neoadjuvant DCF chemotherapy followed by surgery for ESCC, and the rates of pathological complete response and 3-year DFS were comparable to those reported in the JCOG1109 NExT study. 15 Second, the median follow-up period of 36.2 months was relatively short, resulting in an incomplete analysis for OS. Although OS is certainly the most important endpoint, adjuvant nivolumab therapy has been approved in many countries, including the United States, European countries, and Japan, based on the improved DFS observed in the Checkmate-577 trial, which was conducted over a median follow-up of 24.4 months. Therefore, we believe that the present study provides important information for identifying adjuvant nivolumab therapy as a candidate therapy for patients with locally advanced ESCC. Finally, the study included only Japanese patients. However, standard surgical procedures for lymphadenectomy differ between Japan and other countries. Therefore, our study findings should be interpreted with caution for the utility of adjuvant nivolumab therapy in Western patient populations.
In conclusion, in this largest-to-date cohort study, we found that patients with residual pathological disease after neoadjuvant DCF chemotherapy followed by surgery had a poor prognosis compared to those with a pathological complete response. Furthermore, pathological node positivity, supraclavicular lymph node metastasis, and lymphovascular invasion were strongly associated with a poor prognosis in patients with residual pathological disease.
Supplemental Material
sj-docx-1-tam-10.1177_17588359241229432 – Supplemental material for Prognosis of patients with residual pathological disease after neoadjuvant docetaxel, cisplatin, and 5-fluorouracil therapy and surgery for esophageal squamous cell carcinoma: a retrospective cohort study
Supplemental material, sj-docx-1-tam-10.1177_17588359241229432 for Prognosis of patients with residual pathological disease after neoadjuvant docetaxel, cisplatin, and 5-fluorouracil therapy and surgery for esophageal squamous cell carcinoma: a retrospective cohort study by Mashiro Okunaka, Daisuke Kotani, Hisashi Fujiwara, Kazuma Sato, Naoto Fujiwara, Saori Mishima, Shingo Sakashita, Takayuki Yoshino, Takeo Fujita and Takashi Kojima in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
