Abstract
Background:
The efficacy of immunochemotherapy (ICT) remains poor in patients with polymetastases from esophageal squamous cell carcinoma (ESCC). While radiation therapy (RT) has shown promise in oligometastatic settings, its role when combined with ICT for polymetastatic ESCC (with >5 metastatic lesions) remains unclear.
Objective:
This study evaluated the efficacy and value of RT in patients with polymetastatic ESCC who received ICT as first-line treatment.
Design:
This multi-center cohort study was conducted at 20 hospitals in China.
Methods:
In total, 331 patients who received at least one cycle of first-line ICT between January 2019 and December 2021 were enrolled. Among them, 88 received ICT plus RT (RT group), and 243 received ICT alone (non-RT group). Propensity score matching (PSM) was performed to control for potential confounders (75 patients/group). Outcomes included overall survival (OS), progression-free survival, objective response rate (ORR), symptom control, and safety. This study was registered at the Clinicaltrials.gov registry (identification number NCT 05142709).
Results:
Both before and after PSM, no significant OS benefit was observed with RT group (median OS: 15.2 vs 12.2 months, hazard ratio (HR) 0.80 (0.60–1.07), p = 0.14; 15.0 vs 11.0 months, HR 0.80 (0.55–1.15), p = 0.23, respectively), though pre-PSM ORR favored RT (59.1% vs 40.3%, p = 0.003). RT demonstrated superior symptom control, with significantly higher rates of dysphagia improvement (63.3% vs 36.4%, p = 0.0006) and meaningful pain reduction (59.4% vs 40.0%, p = 0.007). Grade ⩾3 treatment-related adverse events were comparable between groups (38 vs 40 cases post-PSM, p = 0.74), with equivalent grade 5 toxicities (1.3% each).
Conclusion:
For polymetastatic ESCC, RT combined with ICT enhanced symptom control without severe toxicity, though it did not improve survival. This supports its personalized use for quality of life in symptomatic patients.
Introduction
In 2022 statistics, esophageal cancer ranked as the seventh most prevalent cancer and the fifth leading cause of cancer-related fatalities in China. 1 Esophageal squamous cell carcinoma (ESCC) is the dominant pathological type among esophageal cancers. ESCC with extensive metastases represents a devastating disease state, characterized by a median overall survival (OS) persistently below 12 months despite advances in systemic therapy. While first-line platinum-based chemotherapy combined with anti-programmed cell death-1 (PD-1) inhibitors (e.g., pembrolizumab in KEYNOTE-590) has improved response rates and survival outcomes compared to chemotherapy alone,2–6 intrinsic and acquired resistance frequently limit long-term efficacy, leaving 40%–60% of patients progressing within 6 months.
Local treatment was not allowed for all patients except palliative radiation for symptom relief in previous randomized phase III trials of first-line immunochemotherapy (ICT).2–6 While several phase II prospective randomized clinical trials have proved that patients with oligometastatic tumors have prolonged progression-free survival (PFS) and OS after adding radiation therapy (RT) for all visible lesions based on systemic treatment (mainly chemotherapy and targeted therapy),7,8 its value in patients with polymetastases is a subject of active investigation. Although radiotherapy is widely hypothesized to synergize with immunotherapy through mechanisms such as antigen release and abscopal effects,9–11 existing randomized trials have often only focused on oligometastatic diseases.12,13 Therefore, without conclusive evidence, extrapolating the benefits of RT to patients with polymetastases carries the risk of overtreatment in populations already burdened by systemic immune suppression and organ dysfunction.
Therefore, this real-world study challenges the “one-size-fits-all” paradigm by rigorously evaluating real-world outcomes of radiotherapy combined with ICT in polymetastatic ESCC (>5 metastases). We launched this study in China to evaluate the efficacy and value of RT for advanced ESCC patients with polymetastases who received ICT as first-line treatment.
Patients and methods
Patients
In this multicenter, retrospective, cohort study, we included advanced ESCC patients who received at least one cycle of PD-1 inhibitor plus chemotherapy between January 2019 and December 2021 from a working database across 20 hospitals in China (shown in Supplemental Table A1). The inclusion criteria were as follows: (1) aged 18 years or older, (2) an Eastern Cooperative Oncology Group (ECOG) performance status of 0–2; (3) histologically or cytologically confirmed stage IVB (TxNxM1, UICC/AJCC 8th edition) ESCC; (4) polymetastatic disease (PMD), which was defined as >5 metastatic lesions (the primary tumor and regional lymph nodes were not counted; and the number of metastatic extra-regional lymph lesions is determined by the involved regions of lymphatic drainage); (5) received at least one cycle of PD-1 inhibitor plus chemotherapy with or without RT; and (6) no prior systemic therapy. For the radiation group, the following criteria shall also be met: (1) received over 40 Gy dose of radiation to the primary lesion; (2) the interval between the last cycle of chemotherapy and radiation shall be within 12 weeks, and started RT before the progression of ICT. The exclusion criteria were as follows: (1) the presence of other active synchronous carcinomas; (2) receiving local therapy but not RT during the current treatment; (3) using PD-1 inhibitors alone for systemic therapy; (4) the occurrence of bleeding and perforation of the esophagus prior to treatment; or (5) insufficient clinical information.
The study received approval from the institutional ethics boards of the authors’ institutions. This study was registered at the Clinicaltrials.gov registry (identification number NCT 05142709). The requirement for written informed consent was waived because this study retrospectively analyzed anonymized patient data.
Procedures
All patients included in this study received anti-PD-1 therapy plus chemotherapy with or without RT as their first-line treatment for metastatic ESCC. Anti-PD-1 antibodies (pembrolizumab, nivolumab, toripalimab, sintilimab, tislelizumab, and camrelizumab) were administered every 3 weeks until disease progression or unacceptable toxicity, or up to 1 or 2 years.
Radiotherapy administration was guided by multidisciplinary evaluation and physician-assessed clinical indications. Treatment modalities included concurrent delivery with ICT or sequential application post-induction systemic therapy. Simulation involved contrast-enhanced CT scans (5 mm slice thickness) in treatment position, with planning executed through Eclipse or Pinnacle treatment systems. Target delineation incorporated endoscopic and cross-sectional imaging findings: gross tumor volume (GTV) comprised primary esophageal lesions and radiographically confirmed regional node metastases (short-axis ⩾1 cm), while clinical target volume extended 0–0.5 cm radially and 3.0 cm longitudinally beyond esophageal GTV. Planning target volumes (PTV) incorporated 5–10 mm margins accounting for setup variability. Metastatic non-regional nodes (supraclavicular, para-aortic, retroperitoneal) were included if located within the primary lesion’s anatomical drainage territory. Irradiation to the metastatic sites included extra-regional nodes and visceral metastases, the PTV of which was created by expanding the GTV by 0.5–1 cm. For lesions near critical structures such as the spinal cord or bowel, a risk-adapted radiotherapy technique was employed.
Demographic information and disease characteristics of patients at the commencement of anti-PD-1 therapy, with or without radiotherapy, were systematically gathered and analyzed. This information was obtained through a specifically designed, anonymized case reporting form utilizing patient electronic medical records and follow-up via telephone. The clinical data encompassed various patient characteristics prior to commencing treatment with PD-1 inhibitors. Furthermore, the regimen of ICT, the dose, fractionation, and technique of combined RT, any interruptions in treatment, treatment-related adverse events (TRAEs), the date and location of disease progression, subsequent salvage treatments, and the date of patient death were meticulously recorded. Patients attended regular follow-up physical examinations, blood tests, chest and abdominal CT scans, barium esophagography, and ultrasonography at 2- or 3-month intervals for the first 2 years, every 6 months for the next 3 years, and then annually. Any further treatment after disease progression was at the discretion of the treating physicians and patients.
Efficacy assessment
The primary endpoints of this study included OS and PFS. These were defined as the duration from the initiation of treatment (the first dose of immunotherapy) until the radiological progression of any tumor (PFS), death from any cause, or the last follow-up (PFS, OS). The best overall responses were assessed by board-certified radiologists or independent investigators at each institution in accordance with the Response Evaluation Criteria in Solid Tumors (version 1.1). The objective response rate (ORR) was quantified as the percentage of patients exhibiting a partial response (PR) or complete response (CR) to the treatment. Meanwhile, the disease control rate (DCR) was determined as the proportion of patients achieving the best overall response (CR or PR) or those with stable disease. Patients were assigned a best overall response of progressive disease (PD) if disease progression was documented at the first on-treatment tumor assessment. Symptom assessment was performed at baseline and after the completion of treatment to compare the symptom relief rates between the RT and non-RT groups. Improvement was defined by the presence of any of the following criteria: documented symptom resolution, a categorical reduction in severity, or the absence of any subsequent dysphagia-related complaint in the medical records. Safety evaluations were continuously conducted for all systemic therapies commencing from the initiation of immunotherapy until the predefined data cutoff point was reached. The severity of TRAEs was classified according to the Common Terminology Criteria for Adverse Events (version 5.0).
Statistical analysis
Continuous variables are reported as medians along with interquartile ranges (IQRs) and were analyzed using one-way analysis of variance. Categorical variables are summarized through frequencies and percentages, with comparisons executed using Chi-square analysis. Fisher’s exact test was used to compare categorical variables when the expected count per cell was less than 5. The median follow-up duration was determined using the reverse Kaplan–Meier method. Kaplan–Meier analysis facilitated the estimation of PFS and OS, with the results expressed as median values accompanied by 95% confidence intervals (CIs). To assess survival differences between groups, the log-rank test was applied. ORR, DCR, symptom relief rates, and safety outcomes were presented as proportions for each treatment group, with differences analyzed through Pearson’s χ² test (applying Yates’ correction when necessary) or Fisher’s exact test. Univariate and multivariate Cox regression analyses were conducted to assess the independent impacts of clinical and pathological features on OS and PFS. All covariates significantly linked to OS and PFS in univariate analysis were subsequently incorporated into the multivariate analysis. Multiple imputation was deemed unnecessary as efficacy endpoints were accessible for all patients, and the baseline variables exhibited minimal missing data.
Subgroup analyses were conducted to compare PFS and OS between the RT cohort and the non-RT cohort. Hazard ratios (HRs) were determined using univariable Cox regression analysis, and an interaction test (Wald test) was conducted for each variable in relation to treatment. The results were illustrated using forest plots.
To adjust for the possible bias induced by the retrospective design of this study, 1:1 nearest-neighbor propensity score matching (PSM) with a caliper of 0.1 was used, accounting for age, sex, ECOG score, baseline supportive care, eating obstruction, pathological differentiation, metastatic sites, anti-PD-1 regimens, number of cycles of anti-PD-1 treatment, chemotherapy regimens, and cycles of chemotherapy. We evaluated the balance of covariates between arms using the standardized mean difference (SMD). After matching, we performed a comparison of clinical outcomes between the two groups. All statistical analyses were performed via R software (version 4.2.2) and SPSS® statistical software (version 22.0; IBM Corp., Armonk, NY, USA). Statistical significance was indicated by p ⩽ 0.05. The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement. 14
Results
Patient characteristics of the entire cohort
A total of 331 patients were initially diagnosed with polymetastatic ESCC and were therefore included in our analysis. Among these patients, 88 (26.6%) received ICT combined with RT (ICRT), while 243 (73.4%) received ICT alone (Figure 1). Patient demographics, tumor characteristics, and treatment-related information before and after PSM are shown in Table 1.

Flow chart of patient enrollment. A total of 1197 patients were screened from 20 cancer centers, and finally, 331 eligible patients were included.
Baseline covariates before and after matching.

Data are presented as a number (percentage) of patients.
Other doublet regimens, paclitaxel plus fluorouracil, gemcitabine plus cisplatin.
ECOG, Eastern Cooperative Oncology Group; FP, fluorouracil (5-fluorouracil /capecitabine/S-1) plus platinum; PD-1, programmed cell death-1; RT, radiation therapy; SMD, standardized mean difference; TP, taxanes (paclitaxel/abraxane) plus platinum.
Treatment details
In the RT group (n = 88), 68 patients received RT to only the primary tumor (esophageal lesions and regional nodes), and 20 patients received RT to all tumor sites (the primary tumor combined with the metastatic lesions). All irradiated patients were treated with conventionally fractionated radiotherapy (typically 1.8–2.0 Gy per fraction) using 6 MeV X-rays via intensity-modulated radiotherapy or volumetric-modulated arc therapy techniques, with the exception of one patient who received stereotactic ablative radiotherapy (SABR) for lung metastases. The median radiation dose to the primary tumor was 50.4 Gy (IQR 50.0–60.0). The RT group exhibited a significantly higher proportion of nonregional lymph node involvement compared to the non-RT group (27.3% vs 14.4%, p = 0.007), suggesting potential clinical implications for treatment stratification. A total of 83 patients (94.3%) completed the course of RT, and 5 patients (5.7%) discontinued RT because of poor tolerance, TRAEs, or an unwillingness to complete the treatment. The radiotherapy characteristics of the RT group are reported in Supplemental Table A2.
The median number of anti-PD-1 antibody cycles administered was identical in both the RT and non-RT groups: 4 cycles (range: 1–35), with no significant statistical difference (F = 1.254, p = 0.264). Similarly, the median chemotherapy cycles remained consistent across groups (RT: 4 cycles, range 1–11; non-RT: 4 cycles, range 1–20), showing comparable non-significance (F = 1.726, p = 0.190). Specific treatment regimen distributions are detailed in Table 1, and the main reasons for systemic treatment discontinuation are reported in Supplemental Table A3.
Treatment effectiveness
At a median follow-up of 26.1 months (IQR 19.7–31.0) from the start of anti-PD-1 treatment, the median OS duration was 12.7 months (95% CI: 11.2–14.2), and the median PFS duration was 6.8 months (95% CI: 6.0–7.6). The best ORR and DCR of the 331 patients were 45.3% and 81.3%, respectively. At the time of analysis, 236 (71.3%) of 331 patients experienced disease progression, and 254 (76.7%) deaths were observed (Supplemental Table A4).
Before PSM, significant difference was not observed in both OS and PFS between the RT group and non-RT group (median OS, 15.2 (95% CI: 11.1–19.3) vs 12.2 (95% CI: 10.6–13.8) months, HR 0.80 (0.60–1.07), p = 0.14, Figure 2(a); median PFS, 6.7 (95% CI: 5.6–7.8) vs 6.9 (95% CI: 6.0–7.8) months, HR 1.02 (0.78–1.34), p = 0.89, Figure 2(b)). ORR was higher in patients who received RT than those did not (59.1% vs 40.3%, OR 2.14, 95% CI: 1.30–3.51, p = 0.003), but DCR was not significantly different between RT group and non-RT group (84.1% vs 80.2%, OR 1.30, 95% CI: 0.68–2.50, p = 0.43).
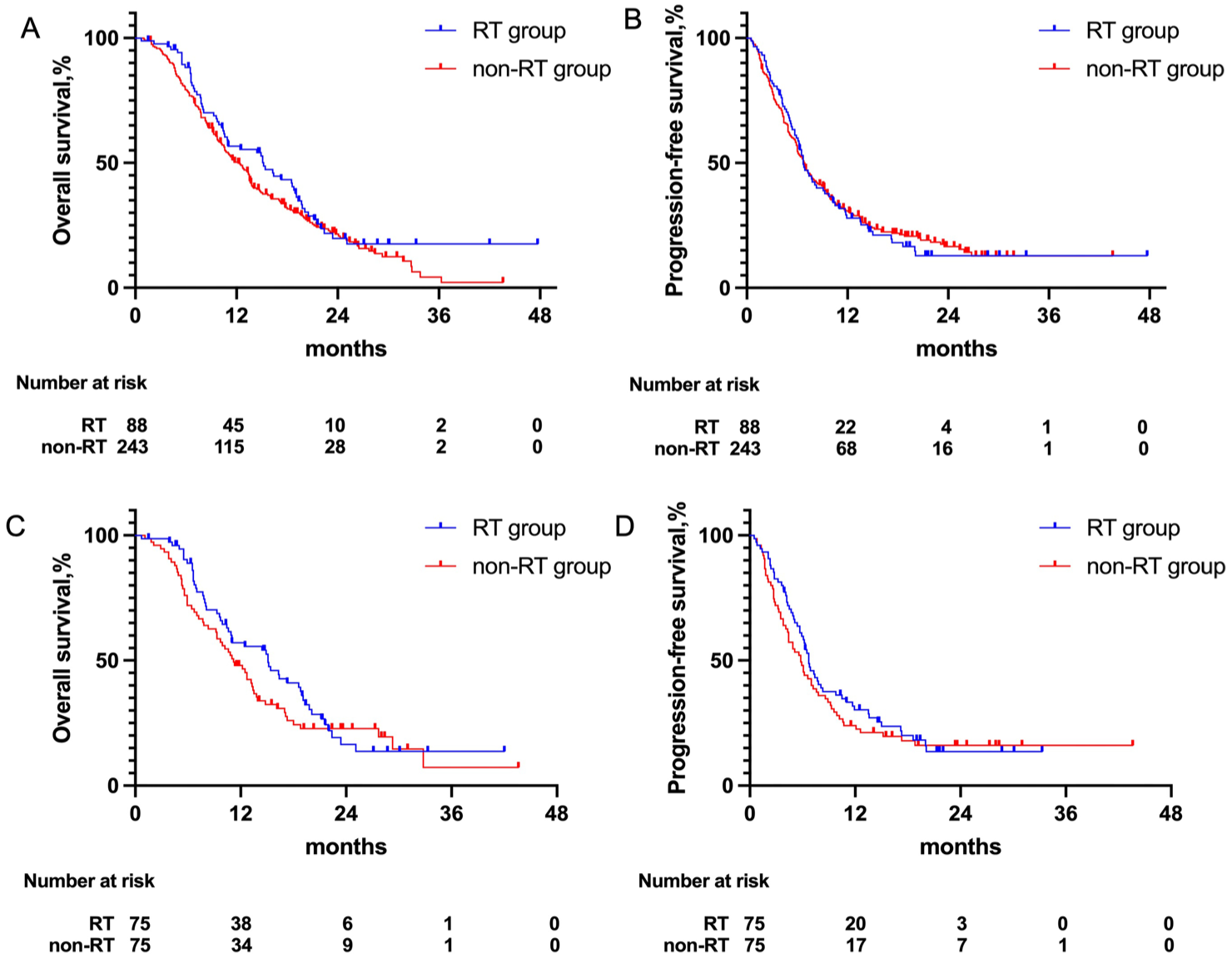
(a) OS curves for patients treated with RT and without RT before PSM. (b) PFS curves for patients treated with RT and without RT before PSM. (c) OS curves for patients treated with RT and without RT after PSM. (d) PFS curves for patients treated with RT and without RT after PSM.
After PSM, 75 patients who received ICR and 75 patients who underwent ICT alone were matched. A notable reduction in SMDs after matching can be observed, indicating that the PSM effectively improved the balance of covariates between the two groups. The baseline characteristics of the patients were well balanced in all covariates (Table 1). The median OS was 15.0 (95% CI: 11.0–19.0) months for the RT group and 11.0 (95% CI: 9.3–14.0) months for the non-RT group (HR 0.80 (0.55–1.15), p = 0.23; Figure 2(c)). The median PFS was 6.7 (95% CI: 5.9–9.8) months in the RT group and 5.8 (95% CI: 4.4–7.8) months in the non-RT group (HR 0.86 (0.60–1.22), p = 0.39; Figure 2(d)). Both ORR (41 of 75 (54.7%) vs 32 of 75 (42.6%); p = 0.14) and DCR (61 of 75 (81.3%) vs 57 of 75 (76.0%); p = 0.43) were not significantly different between the RT group and the non-RT group. Subgroup analysis demonstrated that radiotherapy did not show superior treatment efficacy over non-RT regimens in most patient subsets except in four specific cohorts: patients without eating obstruction, those with extra-regional lymph nodes only, individuals receiving <5 cycles of PD-1 inhibitors, and cases exhibiting PD as best overall response (Figure 3).

(a) Forest plot of subgroup analysis of overall survival after PSM. (b) Forest plot of subgroup analysis of progression-free survival after PSM. A total of two patients in the RT group and one in the non-RT group were excluded due to missing relevant baseline characteristic data.
Multivariate Cox regression analysis identified several significant prognostic factors for OS, including age, supportive nutritional care, and cycles of PD-1 inhibitors (Supplemental Table A5). Individuals aged ⩾65 years (adjusted HR 0.54, 95% CI: 0.35–0.83, p = 0.005), the absence of supportive nutritional care (adjusted HR 0.29, 95% CI: 0.16–0.55, p < 0.001), and receiving ⩾5 cycles of PD-1 inhibitors (adjusted HR 0.52, 95% CI: 0.33–0.82, p = 0.005) were associated with a longer OS. For PFS, multivariate analysis revealed significant associations with ECOG performance status (adjusted HR 0.50, 95% CI: 0.27–0.91, p = 0.024), nutritional support (adjusted HR 0.44, 95% CI: 0.25–0.78, p = 0.005), and PD-1 inhibitor cycles (adjusted HR 0.48, 95% CI: 0.31–0.74, p < 0.001; Supplemental Table A6).
The 88 patients treated with RT were stratified by dose to assess its prognostic significance: ⩾50 Gy (n = 71) versus 40–50 Gy (n = 17). Comparison of the subgroups revealed that a higher radiation dose (⩾50 Gy) was not associated with a significant survival benefit (median OS: 15.2 vs 15.1 months, HR 0.66, 95% CI: 0.33–1.33, p = 0.84; median PFS: 6.9 vs 6.3 months; HR 0.71, 95% CI: 0.39–1.32, p = 0.23; Supplemental Figure A1). An exploratory analysis of irradiation site selection revealed comparable survival outcomes between patients receiving primary-only irradiation (n = 68) and those receiving multi-site irradiation (n = 20), with no significant benefit observed for either strategy (HR 0.97, 95% CI: 0.54–1.75, p = 0.92 for OS; HR 1.10, 95% CI: 0.64–1.89, p = 0.74 for PFS, Supplemental Figure A2).
This study revealed that incorporating local radiotherapy into ICT significantly enhanced symptom control outcomes when compared to ICT alone. Notably, patients in the RT group exhibited superior symptom relief, with 63.3% (38 of 60 patients) achieving substantial improvement in dysphagia clinically significant and functionally impairing symptom in advanced esophageal cancer, compared to only 36.4% (55 of 151 patients) in the non-RT group (p = 0.0006). Furthermore, the radiotherapy cohort demonstrated better pain management outcomes, with 59.4% (41 of 69 patients) of patients in the RT group reporting clinically meaningful pain reduction, in contrast to 40.0% (79 of 195 patients) in the non-RT group (p = 0.007).
Adverse events
The majority of adverse events were classified as grade 1 or 2. Therefore, we investigated grade 3–5 TRAEs in all patients. The results showed that there were no differences between the two groups with or without RT for grade 3 or higher TRAEs (38 cases of 75 patients vs 40 cases of 75 patients, p = 0.74, Table 2) after PSM. One patient (1.3%) with RT and 1 patient (1.3%) without RT had grade 5 TRAEs. Deaths caused by TRAEs were thrombocytopenia in the RT group (one case (1.3%)) and pneumonitis in the non-RT group (one case (1.3%)).
Grade 3–5 treatment-related adverse events.
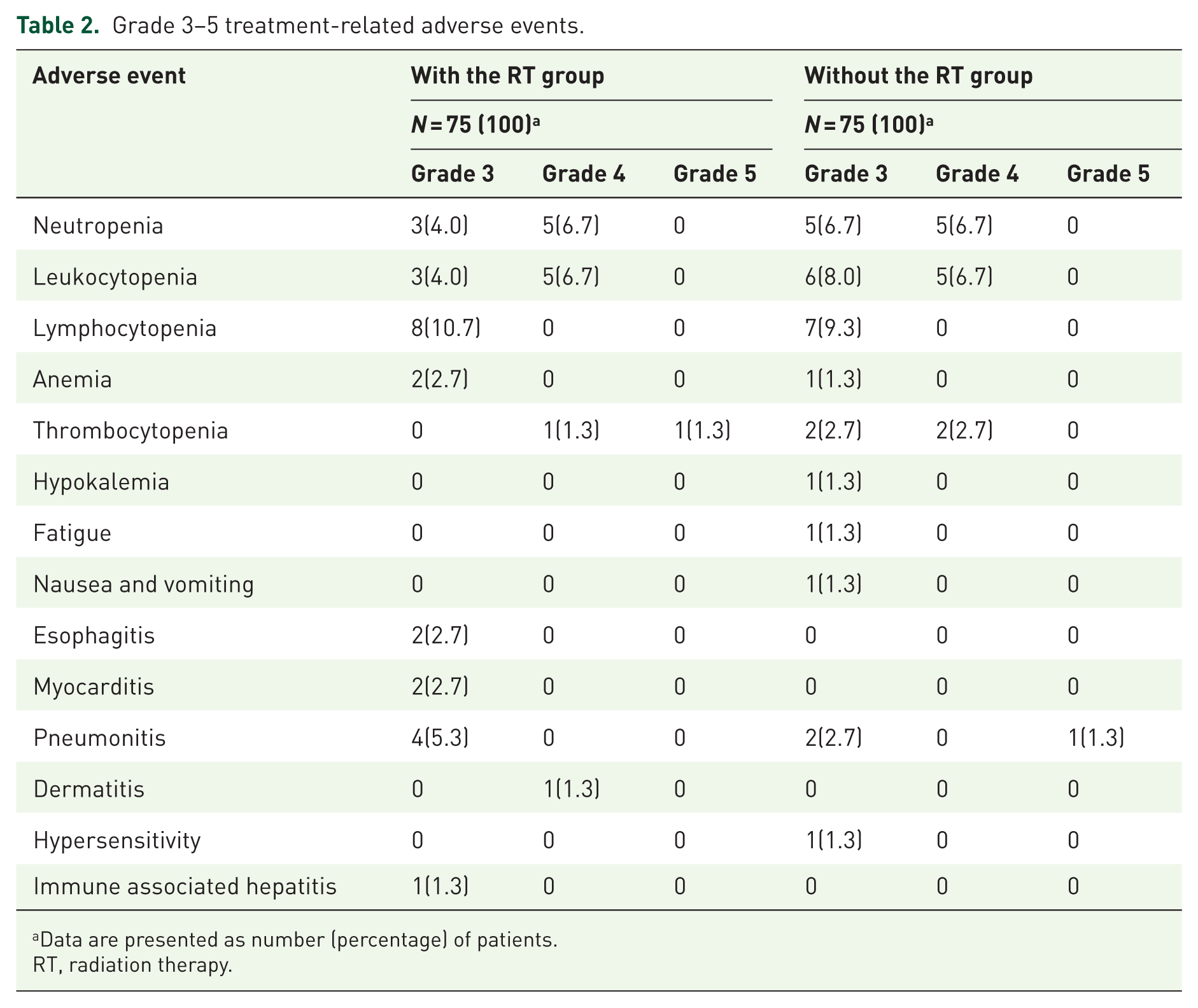
Data are presented as number (percentage) of patients.
RT, radiation therapy.
Discussion
This comprehensive analysis of 331 polymetastatic ESCC patients provides critical insights into the role of RT in combination with ICT, revealing nuanced therapeutic trade-offs between survival outcomes, symptom control, and safety profiles. While our PSM analysis demonstrated no statistically significant survival advantage for ICRT combination therapy over ICT alone (median OS: 15.0 vs 11.0 months, HR 0.80, p = 0.23), the study uncovered several clinically meaningful observations that warrant careful interpretation.
This study demonstrated a median OS of 12.2 months in the non-RT group before PSM, showing a numerically superior outcome compared to the 10.5-month median OS observed in the KEYNOTE-590 trial’s Chinese cohort receiving pembrolizumab combined with cisplatin and 5-fluorouracil. 3 However, this result remained shorter than the reported median OS ranges of 15.3–16.7 months from the Asian ESCORT-1st 2 and ORIENT-15 trials. 4 Importantly, such cross-trial comparisons require cautious interpretation due to substantial heterogeneity in patient populations and treatment protocols between real-world studies and randomized controlled trials. Notably, while the ESCORT-1st trial 2 included 19.8% of patients with regional recurrences only, the current real-world analysis exclusively enrolled patients with polymetastatic ESCC, representing a distinct clinical population with potentially worse prognosis.
Contrasting our results with the landmark SABR-COMET trial, which demonstrated significant survival benefits with RT in oligometastatic disease, 8 highlights the critical heterogeneity in metastatic tumor biology and treatment responsiveness across disease burden subgroups. In real-world management of polymetastatic ESCC, local radiation is often selectively applied to a limited number of lesions for palliative purposes (administered to 38.1% of 331 patients in our cohort). Furthermore, the primary indications for metastatic site irradiation were consolidation after a favorable response to systemic therapy and management of oligoprogressive diseases. In previous prospective studies, despite the use of the SABR technique being applied to partial metastatic lesions (such as a single lesion), the combination of RT and immunotherapy did not improve the response rate and outcome of out-field lesions. 15 Our results align with those reported by Li et al., 16 who observed no significant survival differences between ICRT and matched IC-alone subgroups in non-oligometastatic ESCC patients, paradoxically despite observing survival benefits with first-line ICRT in their overall cohort. Wu et al. 17 retrospectively found that the combination of RT could only significantly prolong OS in patients with locoregional recurrence but not the entire cohort of metastatic ESCC compared with ICT alone. 17 These findings provide critical insights into the nuanced role of RT in managing polymetastatic ESCC. The lack of survival benefit observed in patients with PMD may be attributed to the immunosuppressive tumor microenvironment associated with high metastatic burden, which could counteract the potential synergistic effects of RT and systemic therapies. This aligns with emerging evidence that systemic immunosuppression in advanced cancer stages limits the abscopal effects of RT and diminishes its capacity to enhance the efficacy of immunotherapy. Clinically, these findings advocate for a stratified approach to RT utilization. In patients with extensive metastases, clinicians should prioritize systemic treatment optimization while avoiding indiscriminate addition of RT, which may delay effective treatment cycles or exacerbate toxicity without meaningful survival gains.
The primary objective of RT for metastatic esophageal cancer is often to effectively control symptoms (e.g., improving dysphagia, pain, bleeding). Reported survival outcomes for patients treated with conventional palliative-dose regimens (e.g., 30 Gy in 10 fractions, 35 Gy in 14 fractions) are generally not associated with improved survival, with median OS typically around 9 months.18–20 Low-dose RT has been demonstrated to significantly enhance the abscopal response to RT plus a PD-1 inhibitor. 21 However, results from a large national observational cohort study of 12,683 patients (ESCC account for 24%) found primary tumor-directed RT with definitive dose (e.g., ⩾50.4 Gy) was associated with better survival (median OS of 11.3 vs 7.6 vs 8.4 months) and long-term relief of dysphagia when compared with low-dose RT or chemotherapy alone, 22 suggesting that local control may be important to prognosis. In these studies, the treatment approach for the lower-dose cohort was characterized by larger fraction sizes delivering a low total dose, consistent with a pure palliative intent. This strategy prioritizes a shorter treatment course and reduced acute toxicity, with less expectation of local control. However, the survival outcomes in both of our subgroups (40–50 Gy cohort and ⩾50 Gy cohort) appear notably more favorable (approximately 15 months) than the typically reported median OS of around 9 months with palliative RT. This discrepancy can likely be attributed to patient selection factors in our retrospective study. Patients selected for any radiotherapy (including the 40–50 Gy cohort) may have had a better baseline performance status and a more indolent disease course compared to the broader population of metastatic patients included in palliative RT studies.
The survival neutrality observed despite improved ORR (59.1% vs 40.3%, p = 0.003 pre-PSM) suggests potential decoupling of tumor response metrics from survival endpoints in advanced ESCC. This phenomenon may reflect the aggressive biology of PMD, where transient local control fails to alter systemic progression patterns. The discordance aligns with recent reports in checkpoint inhibitor trials where response rates did not consistently translate to survival benefits in gastrointestinal malignancies.23,24 Although radiotherapy failed to demonstrate survival advantages in this study, it served as an essential therapeutic modality for symptom palliation. For patients with symptomatic metastatic ESCC, radiotherapy targeting the primary site should be considered a primary strategy to improve quality of life. This is strongly supported by our findings: dysphagia improved after RT in 63.3% of patients in the RT group, which was consistent with the results of previous studies (45%–66%).17–19,25 The symptom benefit occurred without significant exacerbation of high-grade toxicities (grade ⩾3 AEs: 38 vs 40 cases post-PSM, p = 0.74), supporting RT’s safety profile when integrated with modern IC regimens. It has been proposed that comprehensive irradiation of metastatic sites may serve as a viable strategy to overcome tumor heterogeneity, eradicate resistant subclones, and thereby potentiate a systemic antitumor immune response. 26 However, a phase II study 27 found that targeting all lesions failed to significantly improve survival (median PFS: 9.8 months; median OS: 16.5 months) in ESCC patients with multiple lymph node metastases or distant oligometastases, contrasting with the positive results of major phase III randomized trials like ORIENT-15 4 and RATIONALE-306 6 trials. For patients with PMD, various circumstances, including disease progression and treatment side effects, may prevent the administration of radiotherapy and affect the overall efficacy. Therefore, in the palliative setting, radiotherapy to the primary site represents an efficient and patient-centric approach to achieve meaningful clinical benefit.
The OS benefit observed specifically in the subgroup without eating obstruction indicates that patients with a lower tumor burden are more likely to derive survival advantages from local control. Notably, our subgroup analysis identified four cohorts potentially benefiting from RT—patients without eating obstruction, those with extra-regional lymph node-only, individuals receiving <5 cycles of PD-1 inhibitors, and cases with PD as best response. These findings propose a precision radiotherapy paradigm, where selective application based on metastatic distribution and treatment response may optimize outcomes. Our multivariate analysis identified modifiable prognostic factors with direct clinical implications. The survival benefit associated with ⩾5 PD-1 inhibitor cycles (HR 0.52, p = 0.005) and nutritional support (HR 0.29, p < 0.001) highlights the importance of treatment adherence and supportive care optimization. These findings align with emerging concepts of “precise palliation therapy” where nutritional optimization may potentiate treatment efficacy.28,29
Several limitations warrant consideration. First, the retrospective design introduces inherent selection bias, particularly evident in the higher baseline nonregional lymph node involvement in the RT group (27.3% vs 14.4%, p = 0.007). While PSM mitigated confounding, residual bias from unmeasured variables (e.g., tumor mutational burden, PD-L1 status) persists. Second, the absence of serial biomarker monitoring precluded analysis of dynamic changes in immune profiles or tumor microenvironment characteristics during treatment. Future prospective studies incorporating standardized RT protocols, longitudinal biospecimen collection, and patient-reported outcome metrics will be essential to validate these observations and identify biomarkers predictive of RT benefit in specific metastatic contexts. Third, the sample size of some subgroups is too small, which may lead to biased results in the subgroup and exploratory analyses. Therefore, the findings from these underpowered analyses should be interpreted with caution and are considered hypothesis-generating rather than conclusive.
Conclusion
In conclusion, our findings demonstrate that the combination of radiotherapy with ICT failed to confer survival advantages in polymetastatic ESCC. Clinically, our findings advocate for a stratified management approach. In polymetastatic ESCC patients without imminent symptomatic threats, prioritizing systemic therapy optimization over immediate RT appears prudent. Conversely, for those with debilitating symptoms like dysphagia or pain, early RT integration provides meaningful palliation without compromising safety. The identified predictive subgroups warrant validation in prospective trials to establish RT candidate selection criteria.
Supplemental Material
sj-docx-1-tam-10.1177_17588359261419635 – Supplemental material for The role of thoracic radiotherapy in the setting of immunotherapy in polymetastatic esophageal squamous cell carcinoma: a multi-center cohort study
Supplemental material, sj-docx-1-tam-10.1177_17588359261419635 for The role of thoracic radiotherapy in the setting of immunotherapy in polymetastatic esophageal squamous cell carcinoma: a multi-center cohort study by Qi Liu, Junqiang Chen, Wenbin Shen, Jinjun Ye, Honglei Luo, Xizhi Zhang, Wenyang Liu, Yujin Xu, Yingying Zhang, Anwen Liu, Xiaopeng Li, Hong Ge, Qifeng Wang, Hui Luo, Lina Zhao, Conghua Xie, Qin Lin, Congying Xie, Luhua Wang and Kuaile Zhao in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-2-tam-10.1177_17588359261419635 – Supplemental material for The role of thoracic radiotherapy in the setting of immunotherapy in polymetastatic esophageal squamous cell carcinoma: a multi-center cohort study
Supplemental material, sj-docx-2-tam-10.1177_17588359261419635 for The role of thoracic radiotherapy in the setting of immunotherapy in polymetastatic esophageal squamous cell carcinoma: a multi-center cohort study by Qi Liu, Junqiang Chen, Wenbin Shen, Jinjun Ye, Honglei Luo, Xizhi Zhang, Wenyang Liu, Yujin Xu, Yingying Zhang, Anwen Liu, Xiaopeng Li, Hong Ge, Qifeng Wang, Hui Luo, Lina Zhao, Conghua Xie, Qin Lin, Congying Xie, Luhua Wang and Kuaile Zhao in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
