Abstract
Background:
Thrombocytopenia is a common hematological toxicity in esophageal cancer (EC) patients receiving radiation therapy.
Objectives:
The purpose of this study was to construct and validate a nomogram for predicting the incidence of thrombocytopenia in EC patients receiving radiotherapy.
Design:
A retrospective study.
Methods:
All data were collected from 435 EC patients who received radiation therapy from a real-world cohort study from 2016 to 2021. Thrombocytopenia was assessed according to the toxicity criteria of the Radiation Therapy Oncology Group (RTOG). Univariate and multivariate logistic regression analyses were used to identify significant risk factors for thrombocytopenia. The result was presented in the form of a nomogram. The performance of the model was evaluated by the receiver operating characteristic (ROC) curves and calibration curves. External validation was conducted using an independent cohort of 149 EC patients from another cancer center. Propensity score matching was utilized to balance the covariates between the 1:1 matched two groups. Survival analysis was conducted using the Kaplan–Meier method and compared with the log-rank test.
Results:
A total of 104(23.91%) patients developed thrombocytopenia. Univariate and multivariate logistic regression analysis showed that age, body mass index, planning target volume, tumor location, tumor-node-metastasis stage, platelet count before radiotherapy, history of liver cirrhosis, cycles of induction chemotherapy, and concurrent chemotherapy regimen were the independent predictors of chemoradiotherapy-associated thrombocytopenia and were finally incorporated into our nomogram. The results of the ROC curve showed that the nomogram had high prediction accuracy. The sensitivity of the external validation set was 0.852 (95% CI: 0.663–0.958), and the specificity was 0.820 (95% CI: 0.740–0.883). The calibration curve presented good concordance (Hosmer-Lemeshow test, p = 0.825). An online nomogram-based prediction tool was developed to facilitate clinical implementation. No significant difference was observed in overall survival between patients who experienced severe thrombocytopenia and not (log-rank test, p = 0.390).
Conclusion:
This nomogram provides a prediction tool of chemoradiotherapy-associated thrombocytopenia in EC patients receiving radiotherapy, which may facilitate the potential candidate’s selection of high-risk EC patients for thrombocytopenia.
Plain language summary
Why was the study done? Patients with esophageal cancer often receive radiation therapy as part of their treatment. During this treatment, some patients develop a condition called thrombocytopenia – when blood platelet counts drop too low. Low platelet counts can lead to problems such as increased bleeding risk and treatment interruptions. Our study aimed to create a tool that could predict which patients might develop this side effect, allowing doctors to monitor these patients more closely. What did the researchers do? We collected data from 435 esophageal cancer patients who received radiation therapy between 2016 and 2021. We looked at many factors that might increase the risk of developing low platelet counts, such as age, body mass index (BMI), tumor characteristics, and treatment details. Using statistical methods, we identified which factors were most important and created a prediction tool called a nomogram – a visual calculator that doctors can use to estimate risk. What did the researchers find? About 1 in 4 patients (24%) developed low platelet counts during treatment. We found several factors that increased this risk, including older age, lower BMI, larger treatment area, tumor location, advanced cancer stage, lower platelet counts before starting treatment, history of liver cirrhosis, more cycles of initial chemotherapy, and certain chemotherapy combinations. Our prediction tool showed good accuracy, correctly identifying about 77% of patients who developed low platelet counts and 71% of those who did not. What do the findings mean? This study provides doctors with a tool to identify esophageal cancer patients who might be at higher risk for developing low platelet counts during radiation treatment. Since platelet recovery is typically slow and can significantly disrupt treatment schedules, early identification of high-risk patients is crucial. This prediction tool could help healthcare teams provide more personalized care.
Introduction
Esophageal cancer (EC) is the eleventh most common type of cancer and is ranked as the seventh leading cause of cancer death worldwide. 1 Radiation therapy (RT) is one of the main treatments for EC patients in different settings, including neoadjuvant, definitive, and palliative therapy. 2
Thrombocytopenia, a common complication, usually occurs during RT, especially when combined with chemotherapy, potentially causing delays and interruptions in treatment, leading to poorer clinical outcomes and incremental cost.3 –6 Prior studies reported that the risk of any grade thrombocytopenia in EC patients receiving chemoradiotherapy ranged from 21% to 64%, of which more than 10% were grade III or above.7,8 In the treatment of EC, thrombocytopenia is particularly worrisome due to the increased risk of fatal hemorrhage.9,10
However, the effective treatments for managing thrombocytopenia are still limited. Platelet transfusion and the application of recombinant human thrombopoietin (rhTPO) are two primary methods, along with several drawbacks. 11 Patients receiving platelet transfusions may develop fever, allergy, and acquired infectious diseases. 12 Although previous studies have revealed that rhTPO can shorten the chemotherapy-induced thrombocytopenia (CIT) duration, it still takes about 14 days to reach peak response.13,14 Therefore, accurately pretreatment screening high-risk thrombocytopenia EC patients and providing prophylactic treatment could be a potential strategy.
At present, several studies have demonstrated predictive factors of thrombocytopenia, including clinical and dosimetry parameters, primarily focusing on chemotherapy-induced myelosuppression and pelvic radiotherapy-associated thrombocytopenia.15 –18 However, an integrated and accessible risk prediction model for the incidence of thrombocytopenia in EC patients receiving radiotherapy is still lacking. Therefore, the aim of this study was to construct a clinical prediction model to enable personalized assessment of the risk of thrombocytopenia in EC patients receiving radiotherapy, so as to promote the development of preventive strategies for thrombocytopenia.
Materials and methods
Patients
With approval from the Institutional Ethics Committee, this study retrospectively analyzed 468 EC patients from a real-world cohort who received radiotherapy at the Cancer Hospital of Chinese Academy of Medical Sciences between 2016 and 2021. The external validation was conducted using an independent cohort of 149 esophageal cancer patients who received radiotherapy at Tianjin Medical University Cancer Institute and Hospital. This study was a retrospective analysis based on the real-world cohort study with the NCT registry (NCT05543057). The inclusion criteria included: (1) diagnosed and pathologically confirmed esophageal squamous cell carcinoma or adenocarcinoma; (2) received RT with or without chemotherapy; (3) availability of clinicopathology, radiotherapy dosimetry, and laboratory data; (4) regular hematology tests during and within the first month of RT. The exclusion criteria included: (1) missing hematological examination results; (2) lost to follow-up if patients could not be reached or contacted by the end of follow-up in October 2022. Finally, 435 patients were eligible. Clinical staging was conducted in accordance with the American Joint Committee on Cancer (AJCC) sixth edition of the TNM staging criteria. The reporting of this study conformed to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement. 19
Parameters for model building
Clinical data, imaging records, and laboratory test results were all obtained from the electronic medical records of the institution. Based on prior studies, various factors that may contribute to chemoradiotherapy-associated thrombocytopenia were selected, including age at diagnosis, gender, Karnofsky Performance Status (KPS), body mass index (BMI), tumor location, tumor-node-metastasis (TNM) stage, liver cirrhosis history, RT technique, PTV (planning target volume) volume, PTV dose, mean lung dose (MLD), cycles of induction chemotherapy, concurrent chemotherapy regimen, and platelet (PLT) count before RT. PLT counts were measured through routine hematological examination at different time intervals: within 1 month before RT (baseline) and weekly during and within the first month after RT.
Treatment
Patients received radiotherapy with intensity-modulated radiotherapy (IMRT) or volumetric modulated arc therapy (VMAT), with treatment goals including neoadjuvant radiotherapy, definitive radiotherapy, and palliative radiotherapy. The delineation of target volumes followed the National Comprehensive Cancer Network (NCCN) guidelines for EC patients. 20 The definitive radiotherapy involved a dosage of 50.4 Gy (1.8 Gy per fraction) to the PTV and 59.92 Gy (2.14 Gy per fraction) to the primary gross tumor volume (PGTV) over the course of 28 fractions. The neoadjuvant radiotherapy involved a dosage of 40–45 Gy, and palliative radiotherapy 50–50.4 Gy, with 1.8–2.0 Gy per fraction. Most patients receiving concurrent chemotherapy were treated with a three-week regimen of paclitaxel and a platinum-based doublet, while some elderly EC patients received S-1 (tegafur, 5-chloro-2,4-dihydroxypyridine, and potassium oxonate) monotherapy.
Definition for radiotherapy-associated thrombocytopenia and follow-up
Thrombocytopenia was categorized into four grades based on the toxicity criteria of the Radiation Therapy Oncology Group (RTOG). The cut-off values for the four grades were 100 × 109/l, 75 × 109/l, 50 × 109/l, and 25 × 109/l, respectively. The grade was determined by the lowest platelet count during RT and within the first month after completion. The occurrence of any grade chemoradiotherapy-associated thrombocytopenia was defined as the primary endpoint.
Follow-up began 1 month post-treatment and was performed at 3-month intervals for the first 2 years and at 6-month intervals thereafter. Evaluations included physical examinations, laboratory tests, and imaging studies to assess disease status. Data were collected from medical records.
Statistical analysis
The patient dataset was randomly divided, with 75% (n = 326) assigned to the training dataset and the remaining 25% (n = 109) to the validation dataset. Combined with Synthetic Minority Over-sampling Technique (SMOTE) simulation, the variables with a significance level of p < 0.1 in the univariate analysis were incorporated into the multivariate analyses, and the results were presented in the form of a nomogram. The receiver operating characteristic (ROC) curve of the model in the training and validation sets was drawn to evaluate the performance of the model. Propensity score matching (PSM) was employed to balance covariates between groups and mitigate differences in baseline characteristics. Survival analysis utilized the Kaplan–Meier method, and the survival curve comparisons were conducted through the log-rank test. Statistical analyses and data charts were performed using R software (version 4.3.3; R Foundation for Statistical Computing, Vienna, Austria) and IBM SPSS Statistics (version 26.0; IBM Corp., Armonk, NY, USA).
Results
Patients characteristics
A total of 435 patients were included, the majority of whom had stage III/IVA disease (344, 79%). Among the 51 patients diagnosed with stage IVB disease, 12 presented with organ metastases. For these patients, the median age at diagnosis was 63 years. A total of 72 patients (16.55%) received induction chemotherapy. Of the 240 patients who underwent concurrent chemotherapy, 48 (20%) received single-agent chemotherapy, while 192 (80%) received dual-agent chemotherapy. The mean PLT count before RT was 233.5 (±70.41). Eight patients had a history of liver cirrhosis. A total of 104 (23.91%) developed thrombocytopenia (G1+) during RT and within the first month after completion, of which 49 (47.12%) had grade 2 or above. Details are shown in Table 1. The demographic and clinical characteristics comparison between the development and external validation cohorts showed no significant differences in key variables. Baseline characteristics of the development and external validation datasets are shown in Supplemental Table E1.
Baseline characteristics of all patients.

BMI, Body Mass Index; IMRT, Intensity-Modulated Radiotherapy; KPS, Karnofsky Performance Status; PLT, Platelet; PTV, Planning Tumor Volume; RT, Radiotherapy; TNM stage, Tumor-Node-Metastasis stage; VMAT, Volumetric Modulated Arc Therapy.
Univariate and multivariate analyses
Univariate analysis revealed that age (p = 0.013), BMI (p = 0.001), PTV volume (p = 0.023), tumor location (p = 0.035), TNM stage (p < 0.001), PLT count before RT (p < 0.001), liver cirrhosis (p < 0.001), cycles of induction chemotherapy (p = 0.001), and concurrent chemotherapy regimen (p = 0.053) were statistically significant factors for thrombocytopenia during RT in the SMOTE cohort (Table 2). As shown in Table 3, multivariate analysis indicated that age (p = 0.005; OR, 0.555; 95% CI: 0.367–0.839), BMI, PTV volume (p = 0.001; OR, 1.002; 95% CI: 1.001–1.003), tumor location, TNM stage, PLT count before RT (p < 0.001; OR, 0.981; 95% CI: 0.977–0.985), liver cirrhosis (p = 0.003; OR, 31.550; 95% CI: 3.365–295.797), cycles of induction chemotherapy (p = 0.004; OR, 2.026; 95% CI: 1.245–3.297), and concurrent chemotherapy regimen were identified as independent predictors of thrombocytopenia. Ultimately, these factors were employed in the construction of the nomogram.
Univariate analysis of clinicopathologic, dosimetric, and treatment parameters in predicting thrombocytopenia during RT in the SMOTE cohort.
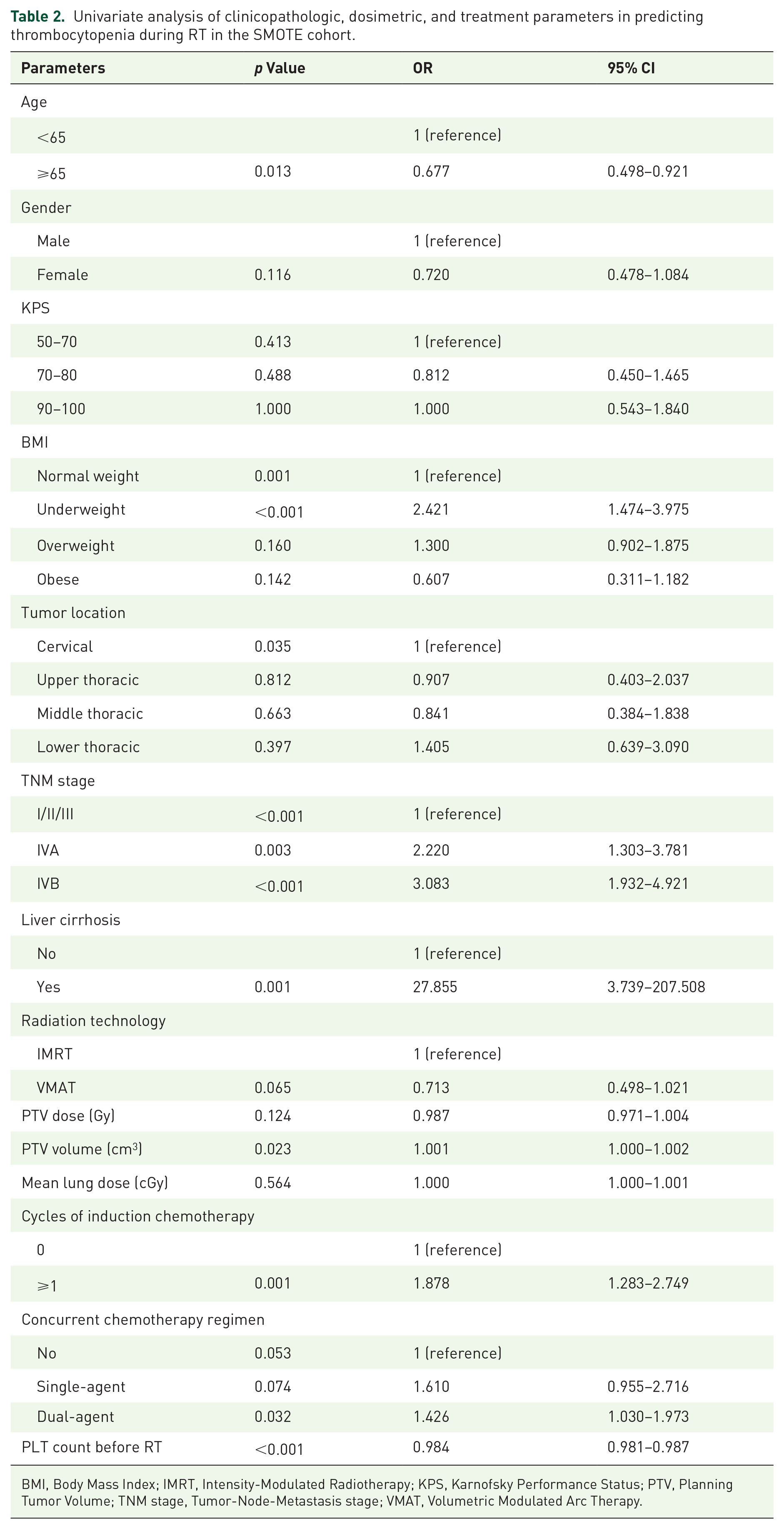
BMI, Body Mass Index; IMRT, Intensity-Modulated Radiotherapy; KPS, Karnofsky Performance Status; PTV, Planning Tumor Volume; TNM stage, Tumor-Node-Metastasis stage; VMAT, Volumetric Modulated Arc Therapy.
Multivariate analysis of clinicopathologic, dosimetric, and treatment parameters in predicting thrombocytopenia during RT.

BMI, Body Mass Index; IMRT, Intensity-Modulated Radiotherapy; KPS, Karnofsky Performance Status; PLT, Platelet; PTV, Planning Tumor Volume; RT, Radiotherapy; TNM stage, Tumor-Node-Metastasis stage; VMAT, Volumetric Modulated Arc Therapy.
Construction and validation of the nomogram
Based on the results of the multivariate analysis, a nomogram was developed incorporating nine independent risk factors (Figure 1). The area under the ROC curve (AUC) of the training, internal validation, and external validation sets was 0.759 (95% CI: 0.699–0.819), 0.757 (95% CI: 0.648–0.867), and 0.892 (95% CI: 0.830–0.954), respectively (Figure 2). The sensitivity of the external validation set was 0.852 (95% CI: 0.663–0.958), the specificity was 0.820 (95% CI: 0.740–0.883), and the accuracy was 0.826 (95% CI: 0.755–0.883). As depicted in Figure 3, the calibration plot demonstrates strong agreement between the predicted and actual probabilities of developing thrombocytopenia.
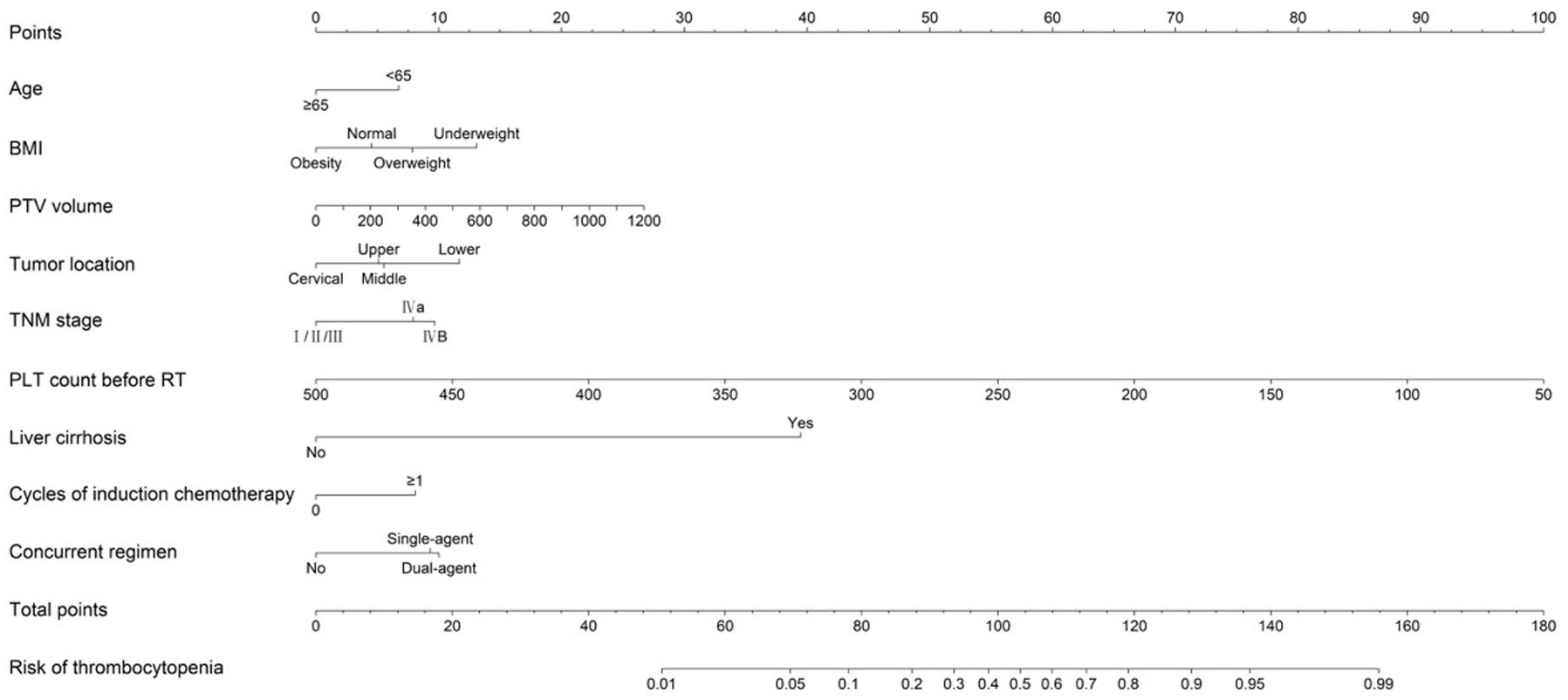
The nomogram model for chemoradiotherapy-associated thrombocytopenia probability prediction in EC patients based on clinical and chemoradiotherapy-associated parameters.

The area under the ROC curve (AUC) of the internal validation set was 0.757 (95% CI: 0.648–0.867), indicating that the prediction model has good prediction accuracy.
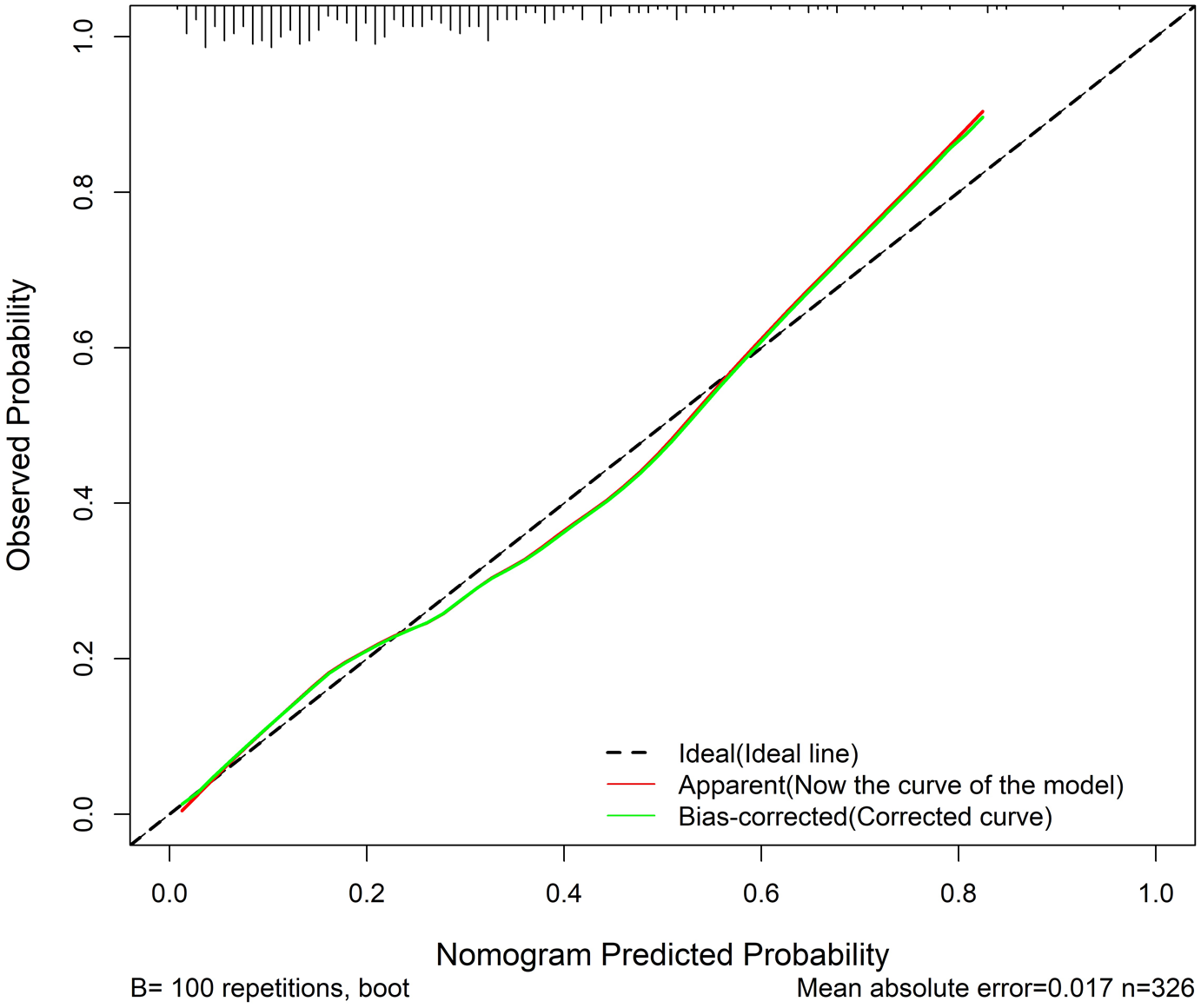
The calibration curve for predicting chemoradiotherapy-associated thrombocytopenia in the validation set.
Clinical application of the nomogram
Nomogram application steps: Collect the above nine parameters before initiating radiotherapy. Each of these parameters is assigned a score on the point scale axis. By summing each individual score, a total score can be easily calculated, and by projecting the total score onto a lower total score scale and drawing a vertical line down to the “Risk of thrombocytopenia” line, we can estimate the probability of thrombocytopenia. An online nomogram-based prediction tool was developed to facilitate clinical implementation, available at https://yjx1301.shinyapps.io/Thrombocytopenia_Nomogram/.
Here is a hypothetical case: a 62-year-old male with middle thoracic esophageal cancer (stage IVA), BMI 18.5 kg/m2, PTV volume 550 cm3, baseline platelet count 170 × 109/l, with liver cirrhosis, one cycle of induction chemotherapy, and planned dual-agent concurrent chemotherapy. After calculating his total score of 180.6, the risk of developing thrombocytopenia is 99%.
Correlation between thrombocytopenia and survival
The median follow-up period was 24.0 months (range 21.9–26.1 months). Patients with thrombocytopenia grade ⩾2 were categorized into group 1, while those with a grade 1 or no platelet reduction were categorized into group 0. PSM was employed to balance covariates between the two groups, with the matching variables including age, sex, BMI, TNM stage, PTV volume, surgical intervention, perforation, mean lung dose, KPS, induction chemotherapy, and concurrent chemotherapy regimen, with a 1:1 ratio and a caliper of 0.05. PSM produced 45 patient pairs, and the patient characteristics of both groups before and after propensity matching are shown in Supplemental Table E2. All matching variables were balanced between the two groups. As illustrated in Figure 4, no significant difference in overall survival (OS) was observed between the two groups (p = 0.390). Similarly, cancer-specific survival showed no significant difference between the two groups (Supplemental Figure E1). In addition, no significant differences were observed in progression-free survival and distant metastasis between the two groups after PSM (Supplemental Figure E2). Univariate Cox regression analyses of OS indicated that thrombocytopenia was not a significant factor affecting survival outcomes in this cohort (Supplemental Table E3). The impact of severe thrombocytopenia on treatment course and supportive care requirements is presented in Supplemental Table E4.
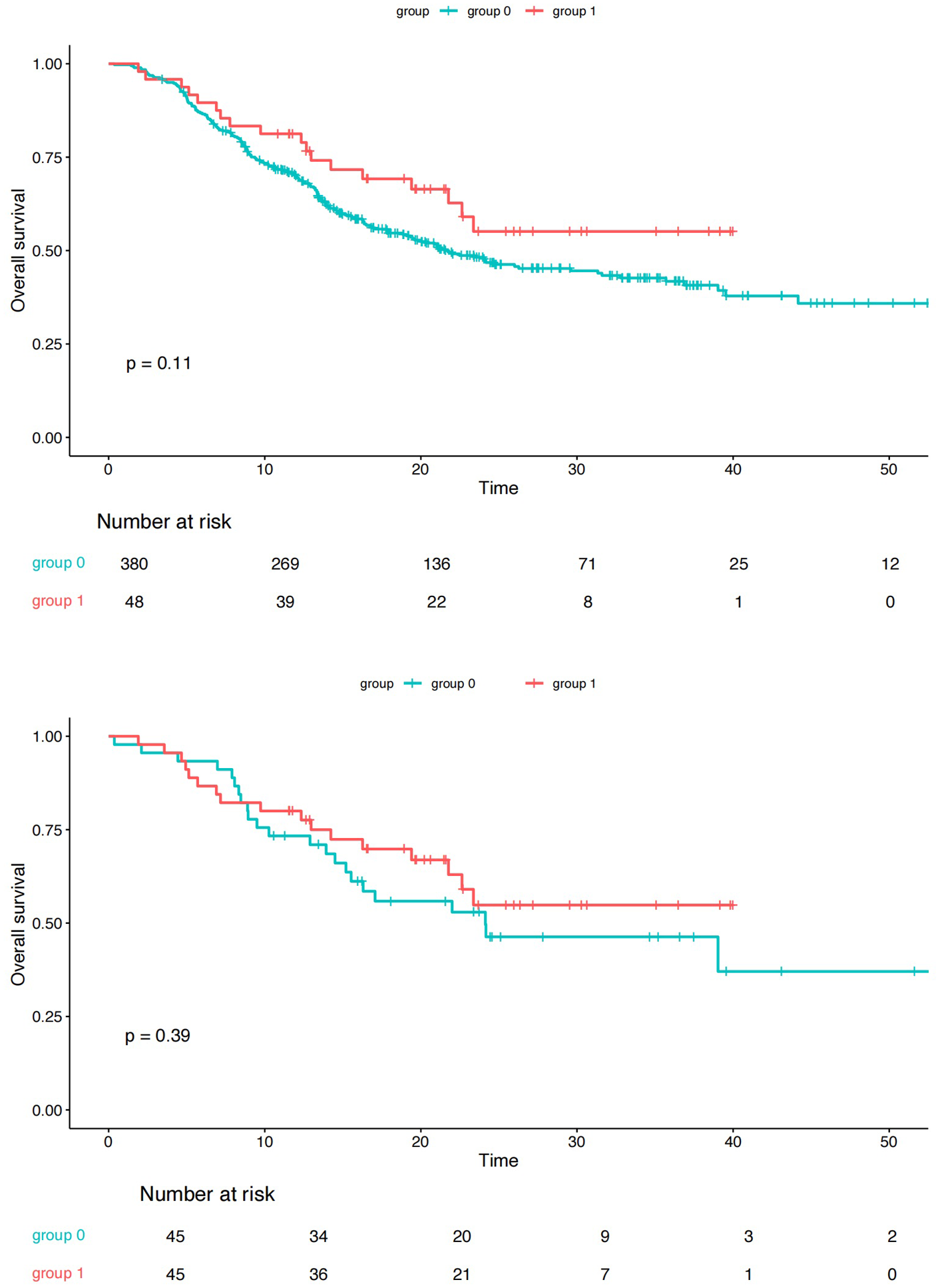
Kaplan–Meier curves showing overall survival between group 0 (blue line) and group 1 (red line) before and after PSM.
Discussion
Thrombocytopenia can be a significant issue in EC patients receiving RT, but the associated risk factors remain unknown. To the best of our knowledge, this is the first nomogram model for forecasting the occurrence of thrombocytopenia in EC patients receiving radiotherapy based on clinical and chemoradiotherapy-associated parameters.
In this cohort, the incidence rate of thrombocytopenia in patients with EC during radiotherapy was 23.91%, close to that in previous studies. 21 Since the patients included in this study did not fully receive concurrent chemotherapy or induction chemotherapy, the incidence rate of thrombocytopenia was at the lower end of the range.
The findings of this study indicated that compared with mono-chemotherapy, combined therapy could contribute to the increased occurrence of thrombocytopenia. Furthermore, multiple cycles of induction chemotherapy significantly raised the occurrence of thrombocytopenia. This is in line with expectations, due to cytotoxic chemotherapy can affect platelet counts through several pathophysiological mechanisms, such as affecting the megakaryocyte and platelet production pathways, causing hematopoietic cell apoptosis and reducing the self-renewal capacity of Hematopoietic Stem Cell (HSC), increasing platelet destruction rate, and enhancing platelet clearance by immune mechanisms.21,22 Likewise, an observational study involving 320 patients revealed a notable increase in CIT incidence with the accumulation of treatment cycles. 23
In our study, PTV volume was found to be associated with the increased incidence of thrombocytopenia during radiotherapy. Several studies have documented a substantial link between radiation to the thoracic vertebral body and the occurrence of acute hematologic toxicities in patients with EC,24,25 which can be explained by the effect of ionizing radiation in inducing senescence of HSC. 26 Notably, most studies primarily focused on the mean vertebral dose (MVD).18,27 The correlation analysis was conducted between PTV volume and thoracic vertebral dosimetric parameters using a representative patient subset, which revealed strong positive correlations between them, including mean vertebral dose (r = 0.736) and vertebral V10–V40 (r > 0.71) (Supplemental Table E5). Given these high correlations and to avoid multicollinearity in the model, we opted to exclusively include PTV volume as our predictor due to its universal availability in treatment planning systems and greater clinical practicality for nomogram implementation.
Recently, numerous reports have provided evidence that the lungs play a significant role in platelet biogenesis, indicating that megakaryocytes (Mks) are typically present in the venous blood returning to the lungs.28 –30 Hence, irradiation of the lungs during radiotherapy may directly influence the functionality of megakaryocytes within the pulmonary circulation. Nevertheless, our study findings did not reveal a significant correlation between the mean lung dose and the development of thrombocytopenia. A possible explanation is that the irradiated area of the lung during EC radiotherapy is limited. In our study, the median mean lung dose among all patients was 10.48 Gy. Thus, small-area, low-dose lung irradiation might not have a significant impact on the reduction of platelets.
Notably, our results indicate that individuals with low baseline PLT counts and a history of liver cirrhosis are at an elevated risk of developing chemoradiotherapy-associated thrombocytopenia. Several previous studies have also reported a substantial correlation between low baseline platelet counts and treatment-associated thrombocytopenia.15,16 For liver cirrhosis patients, a reduction of TPO mainly contributes to a high incidence of reduced platelets. 31 Previous research has indicated that the incidence of thrombocytopenia in patients with severe liver cirrhosis can be as high as 78%. 32 Within our investigation, six out of these eight patients experienced thrombocytopenia during radiotherapy. Thus, for patients with liver cirrhosis, it is necessary to strengthen clinical monitoring for PLT count.
Other factors, including age, BMI, TNM stage, and tumor location, were also included in our model. It is worth noting that age more than 65 years is a protective factor for the development of thrombocytopenia. This may be due to in actual clinical decision-making, more conservative treatment strategies and technical details, which are difficult to quantify, are usually used for older patients. Previous studies also observed the correlation between low BMI and hematologic complications induced by chemotherapy.33 –35
In this study, we analyzed the relationship between thrombocytopenia and prognosis and found that the occurrence of G2 or above thrombocytopenia during radiotherapy did not significantly affect patient prognosis. In general, the impact of thrombocytopenia during concurrent chemorediotherapy (CCRT) on the survival of esophageal cancer patients is still a matter of debate. There have been limited reports that have delved into the connection between CCRT-induced myelosuppression and the treatment outcomes in EC patients. A study involving 65 esophageal cancer patients demonstrated that individuals who developed grade 3 or more severe thrombocytopenia received a reduced dosage of chemotherapy, faced more interruptions in their radiotherapy, and had notably lower 1-year (p = 0.004) and 2-year (p = 0.003) survival rates. 36 Another study demonstrated that platelet count < 150 × 109/l at diagnosis is an independent unfavorable prognostic indicator in EC patients. 37 Possible explanations for this result may be the advances in therapeutic drugs in the last two decades; in our study, thrombocytopenia was managed promptly by rhTPO with good efficacy.
However, several limitations should be mentioned. First, it was a retrospective analysis, and the selective biases could not be avoided. Second, further validation across multiple institutions with larger and more diverse patient populations is needed to strengthen the generalizability of the nomogram. Third, there was a possibility that certain confounding factors were not taken into account in our study.
Conclusion
This nomogram provides a prediction tool for chemoradiotherapy-associated thrombocytopenia in EC patients receiving radiotherapy, which may facilitate the potential candidate’s selection of high-risk EC patients for thrombocytopenia.
Supplemental Material
sj-docx-1-tam-10.1177_17588359251363894 – Supplemental material for A nomogram for predicting the risk of chemoradiotherapy-associated thrombocytopenia in patients with esophageal cancer: a real-world cohort study
Supplemental material, sj-docx-1-tam-10.1177_17588359251363894 for A nomogram for predicting the risk of chemoradiotherapy-associated thrombocytopenia in patients with esophageal cancer: a real-world cohort study by Jia-Xuan Yang, Si-Yu Zuo, Jie Yang, Lin-Rui Gao, Zong-Mei Zhou, Nan Bi, Qin-Fu Feng, Ji-Ma Lv, Lin Sun, Wen-Qing Wang, Lei Deng, Xin Wang, Tao Zhang, Jian-Yang Wang, Ze-Fen Xiao, Wen-Cheng Zhang and Wen-Yang Liu in Therapeutic Advances in Medical Oncology
Footnotes
Acknowledgements
We would like to express our sincere gratitude to Fei-Ze Wu, Wen-Peng Jiao, Ju-Ping Wang, and Hui-Qin Chen for their valuable assistance in data collection.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
