Abstract
Background:
FOLFIRINOX, used in metastatic pancreatic cancer (MPC), is highly efficacious but also toxic. Various dose modifications for FOLFIRINOX have been introduced to reduce toxicity. However, these studies lack a unified pattern for ‘planned’ dose modification, and the ‘actually administered’ dose varied more.
Objective:
To map a 10-year trend for ‘planned’ and ‘actual’ doses of FOLFIRINOX and investigate the clinical outcomes according to dose modification.
Data sources and methods:
A comprehensive systematic literature search was conducted from January 2011 to September 2021. All studies for FOLFIRINOX as first-line treatment in MPC were considered. Selected studies were firstly classified according to prospective versus retrospective research, secondly standard versus modified FOLFIRINOX, and thirdly ‘planned’ versus ‘actual’ dose. For evidence-mapping for the trend of dose modification, we developed a web-based interactive bubble-plot program (www.RDI-map.com). Objective response rate (ORR) and hematologic toxicity were set as endpoints for the comparison of clinical outcomes according to dose modification.
Results:
A total of 37 studies were identified for evidence-mapping (11 prospective and 26 retrospective studies). There were 12 different types of ‘planned’ dose modification in FOLFIRINOX ranging 75–100% oxaliplatin, 75–100% irinotecan, 0–100% 5-fluorouracil (5-FU) bolus, and 75–133% 5-FU continuous injection. The ‘actual’ dose further decreased to 54–96%, 61–88%, 0–92%, and 63–98%, respectively. For the standard versus modified FOLFIRINOX, the ORR was 28.2% (95% CI: 22.5–33.9%) and 33.8% (95% CI: 30.3–37.3%), respectively (p = 0.100), and the incidence of febrile neutropenia was 11.6% (95% CI: 0–16.0%) and 5.5% (95% CI: 0–8.9%), respectively (p = 0.030).
Conclusions:
RDI-map.com enables multifactorial evidence-mapping for practical FOLFIRINOX dose reduction. The pattern of dose modification was not consistent across studies, and there was a significant gap between the ‘planned’ and ‘actual’ doses. Modified FOLFIRINOX showed similar efficacy to the standard regimen with reduced incidence of febrile neutropenia.
Keywords
Introduction
Pancreatic cancer (PC) is one of the most devastating cancers, with a 5-year survival rate of 10%. 1 The incidence and death rate associated with PC are increasing, and it is expected to account for the second most common cause of cancer-related death in the United States by 2040.1,2 Surgical resection is the only curative option for PC, but less than 20% of the patients are diagnosed at the resectable stage. 1 Therefore, most patients with PC are diagnosed at an advanced stage where chemotherapy plays an important role.
Over the past 10 years, two representative combination chemotherapy regimens – FOLFIRINOX (folinic acid, 5-fluorouracil (5-FU), irinotecan, and oxaliplatin) and gemcitabine with nab-paclitaxel – have been proven to improve survival of patients with metastatic PC (MPC) in phase III trials.3,4 However, FOLFIRINOX is limited for those with good performance status owing to its high toxicity rate.3,5 In an effort to reduce toxicity while maintaining efficacy, various dose modifications of FOLFIRINOX have been proposed, and several studies have reported that modified FOLFIRINOX was fit for the purpose.6–8 Despite the clinical potential of modified FOLFIRINOX, there are still indecisive problems. First, there are no randomized controlled trials (RCTs) directly comparing modified FOLFIRINOX to standard FOLFIRINOX, nor are there meta-analyses that include a sufficient number of trials for modified FOLFIRINOX.9,10 In addition, various types of dose modifications for FOLFIRINOX were attempted in previous trials, but an accurate trend analysis or consensus for an appropriate dose modification was not achieved.
Therefore, in this study, we aimed (1) to develop a web-based interactive bubble-plot program elucidating the pattern of the ‘planned’ and ‘actual’ dose of FOLFIRINOX administered in previous trials for MPC patients during the last 10 years and (2) to clarify the clinical potential of modified FOLFIRINOX and the need for modified regimen through a meta-analysis including a sufficient number of trials.
Methods
This study was conducted according to the Preferred Reporting Items for Systematic Reviews and Meta-analyses and the Meta-analysis of Observational Studies in Epidemiology reporting guidelines.11,12 A detailed protocol is registered at the International Prospective Register of Systematic Review (PROSPERO CRD 42021248796).
Search strategy
A comprehensive literature search of databases (Embase, PubMed/Medline, and Scopus) was conducted between 1 January 2011 (the year of publication of the PRODIGE 4/ACCORD 11 trial, which is a representative study introducing standard FOLFIRINOX in MPC) and 30 September 2021 to select eligible studies. 3 The search was carried out using the following keywords: ‘pancreatic cancer’, ‘FOLFIRINOX’, and ‘FOLFOXIRI’. The bibliographies of all the selected studies and previous relevant meta-analysis were manually searched for potential eligible studies. Additional studies published during this systematic review period were also included.
Selection criteria
The inclusion criteria were as follows: (1) RCTs, non-randomized prospective studies, or retrospective studies; (2) studies with participants over 18 years of age; (3) studies including pathologically proven MPC; (4) studies in which FOLFIRINOX was used as first-line treatment for MPC; and (5) studies reporting at least one type of ‘planned’ or ‘actual’ dose information of ‘standard’ or ‘modified’ FOLFIRINOX. Dose information was categorized as follows: (A) ‘planned’ dose in the ‘standard’ FOLFIRINOX group; (B) ‘actually administered’ dose in the ‘standard’ FOLFIRINOX group; (C) ‘planned’ dose in the ‘modified’ FOLFIRINOX group; and (D) ‘actually administered’ dose in the ‘modified’ FOLFIRINOX group (Supplemental Figure S1).
The exclusion criteria were as follows: (1) case report, case series, and review articles; (2) articles not written in English; (3) non-human studies (animal or in vitro studies); (4) studies in which other treatments were administered along with FOLFIRINOX; and (5) studies reporting only the ‘planned’ dose in the ‘standard’ FOLFIRINOX group (only type A dose information).
Two independent reviewers (KJ and SC) screened the titles and/or abstracts of all the retrieved studies to exclude those that did not meet the eligibility criteria. Subsequently, full texts of the selected studies were assessed. For studies with overlapping patient cohorts, the most recent one was chosen. Disagreements were resolved by consensus between the two reviewers or with the help of a third reviewer (J-cL).
Data extraction
The following information was extracted from the selected studies: (1) study characteristics (first author, year of publication, country, study design, and sample size); (2) demographic characteristics (age, sex, and cancer stage); (3) dose information (‘planned’ or ‘actual’ dose of FOLFIRINOX (A, B, C, and D)); and (4) clinical outcomes and toxicity profile (objective response rate (ORR), rates of neutropenia and febrile neutropenia, hazard ratio (HR) for progression-free survival (PFS) and overall survival (OS), and odds ratio (OR) for neutropenia and febrile neutropenia in case of comparative studies).
All dose information was normalized to the relative dose intensity (RDI), with a reference standard dose of FOLFIRINOX (85 mg/m2 of oxaliplatin, 400 mg/m2 of leucovorin, 180 mg/m2 of irinotecan, and 400 mg/m2 of 5-fluorouracil (5-FU) bolus followed by 2400 mg/m2 of 5-FU (continuous infusion) for appropriate comparison. All this information was cross-checked by a second independent reviewer, and any disagreements were resolved by consensus.
Data presentation and synthesis
Evidence-mapping
We adopted the methodology ‘evidence-mapping’ to visualize the complex types of studies. Evidence-mapping helps to describe the trend, quantity, design, and characteristics of complex evidences and studies.13,14 Since there is still no representative automatic software for evidence-mapping, a new type of mapping program was needed.
Development of a web-based program for evidence-mapping of FOLFIRINOX dose
We developed an open-access, user-friendly, web-based interactive bubble-plot program (www.RDI-map.com) for evidence-mapping of the ‘planned’ and ‘actual’ doses of FOLFIRINOX reported in the included studies (Figure 1).
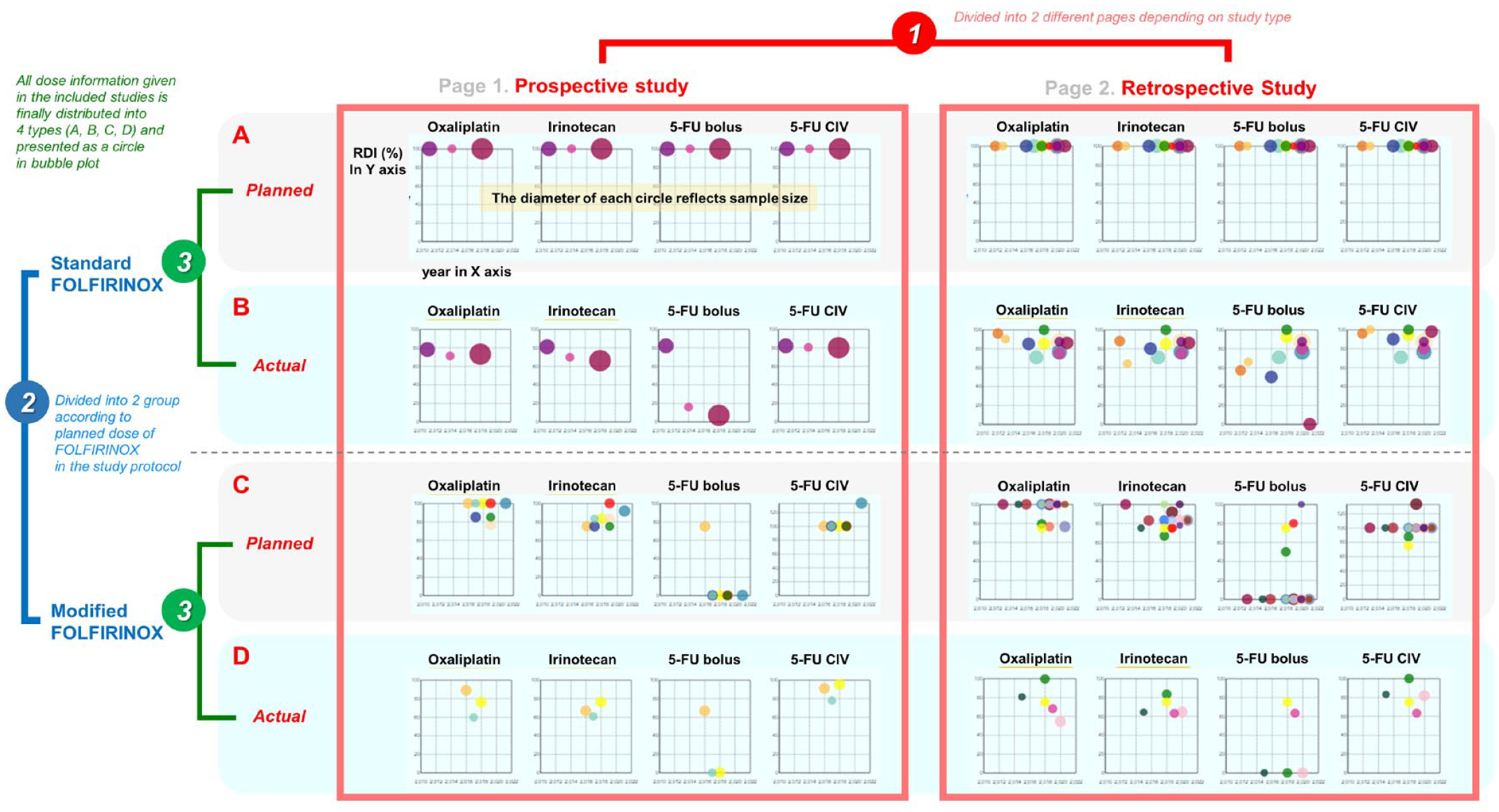
RDI-map.com for evidence-mapping of FOLFIRINOX dose.
RDI-map.com is mainly composed of two separate pages according to study type: prospective and retrospective. Each page consists of a total of 16 bubble-plots with four rows and four columns. Four bubble-plots for each agent of FOLFIRINOX (oxaliplatin, irinotecan, 5-FU bolus, and 5-FU CIV) were included in a row and distributed into four rows according to the type of dose information (A, B, C, and D). Below the bubble-plots, there are two bar graphs classified by country and year, and these graphs help users to sort and view the trend for a specific period or country. At the bottom, all the included studies are listed and more detailed information are provided as a table.
Meta-analysis of clinical outcomes according to the planned dose of FOLFIRINOX
To assess clinical outcomes according to dose modification of FOLFIRINOX, the ORR and hematological toxicity were selected as primary endpoints, as they were most commonly reported in the included studies. The ORR and the rate of hematologic toxicity (neutropenia and febrile neutropenia) were pooled and compared between the planned ‘standard’ FOLFIRINOX group and the planned ‘modified’ FOLFIRINOX group. Additional pooled analyses only with comparative studies (studies that were originally designed to compare standard FOLFIRINOX and modified FOLFIRINOX) as a sensitivity analysis was conducted and subgroup analyses according to study baseline characteristics such as study type, year of publication, or country were also proceeded.
Risk of bias assessment
The quality of included studies was evaluated using the Joanna Briggs Institute (JBI) Manual for Evidence synthesis. 15 This checklist includes 9 items for prospective experimental studies and 11 items for retrospective cohort studies, and the answers for the checklist can be rated as one of the four options: yes, no, unclear, or not applicable.
Statistical analysis
The DerSimonian-Laird random-effects model was applied for the pooled analysis, because high heterogeneity across studies was expected. All statistical tests were 2-sided and p values less than 0.05 were considered statistically significant. Statistical heterogeneity was evaluated using I 2 -statistics with a cutoff of 25%, 50%, and 75% for low, moderate, and high degrees of heterogeneity, respectively. Publication bias was assessed using visualized contour-enhanced funnel plots, and Egger’s test was performed to evaluate asymmetry in funnel plots. All statistical syntheses and analyses were conducted using R software, version 4.0.2 (The R foundation, Vienna, Austria).
Results
A total of 3538 potentially relevant articles (Embase, 1770; PubMed/Medline, 863; and Scopus 905) were identified (Figure 2). After the removal of duplicates, 2168 studies were included in the title and abstract assessment. Of these, 269 records were selected as relevant publications, and a full-text review was conducted. After the full-text review for eligibility, 232 records were excluded because they did not have enough dose information on FOLFIRINOX (N = 117), did not use FOLFIRINOX as first-line treatment (N = 47), did not include patients with MPC (N = 35), had an overlap of enrolled patients (N = 24), and were not related to PC (N = 9). Finally, 37 studies were included for evidence-mapping of the trend of ‘planned’ and ‘actual’ dose of FOLFIRINOX, and 27 studies were included in the meta-analysis for comparison of clinical outcome according to planned dose of FOLFIRINOX.3,6–8,16–48
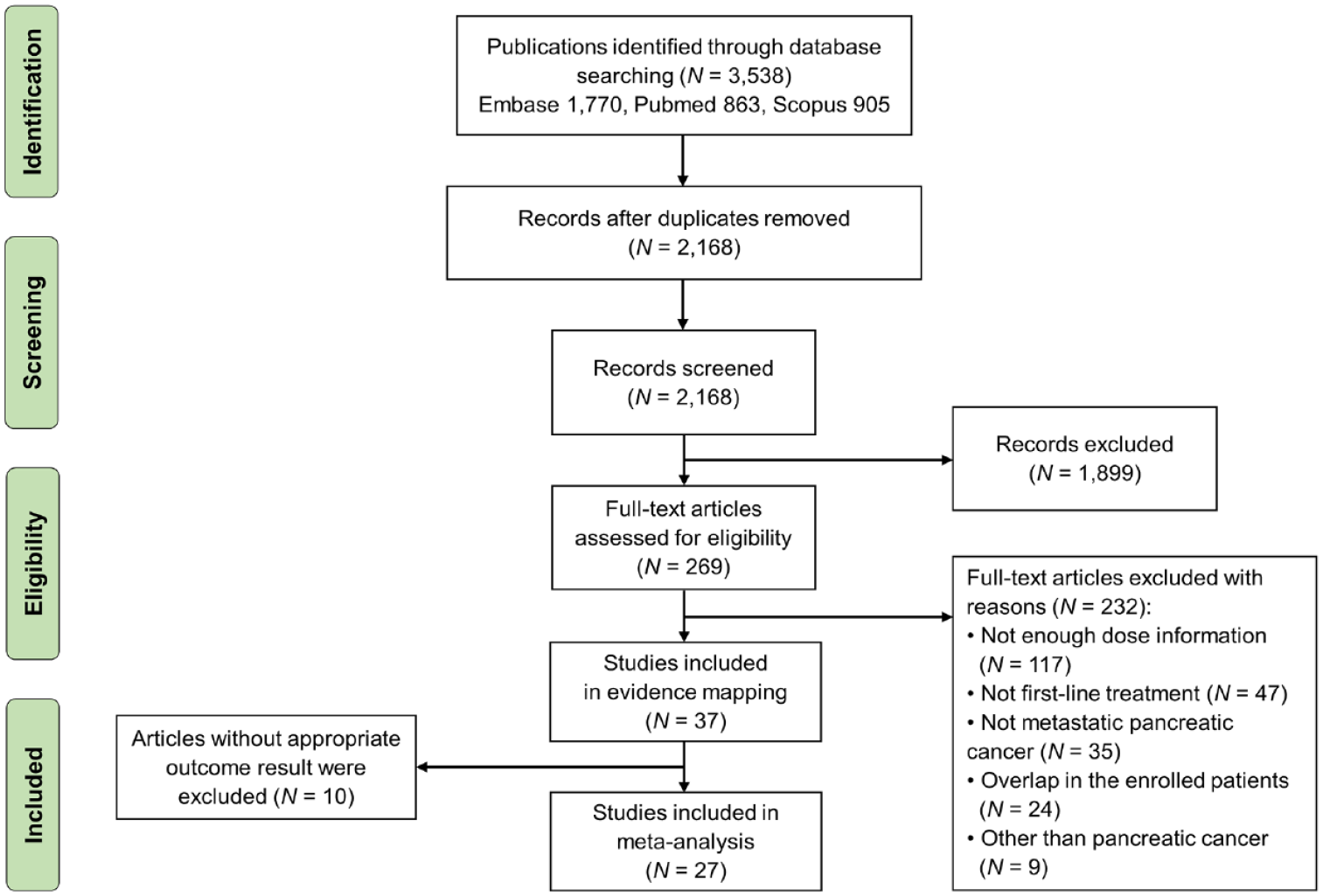
PRISMA flowchart of the systematic literature search.
Description of the included studies
Of the 37 studies, 11 and 26 were prospective studies3,16–25 and retrospective studies,6–8,26–48 respectively (Table 1). The country included the most was Japan (N = 10) followed by Korea (N = 6), USA (N = 5), and China (N = 5). Among the prospective studies, three studies provided both type A and B dose information, three studies provided type C and D information, and the remaining five studies provided only type C information. A total of six studies in the prospective group provided ‘actual’ dose information (type B or D). In the retrospective studies, two studies reported all four types of dose information, nine studies reported type A and B information, two studies reported type C and D information, one study reported type A and C information, 11 studies reported only type C information, and one study reported only type D information. In the retrospective group, 14 studies reported ‘actual’ dose information (type B or D).
Summary of included studies a (N = 37).

Each study was distributed by study type (prospective versus retrospective), study design (standard FOLFIRINOX versus modified FOLFIRINOX), and dose information provided (A, B, C, and D) and presented in the table in this order. Therefore, one study can occupy multiple rows in this table.
Dose information was mostly expressed as median RDI (range, if possible) compared to the dose of standard FOLFIRINOX regimen. For some exceptions, specific details were written in the comments.
In case of studies including patients with other than metastatic stage, the number of patients was expressed as the total number of patients (number of patients with metastatic stage).
5-FU, 5-fluorouracil; RDI, relative dose intensity.
Trend of dose modification of FOLFIRINOX in trials
In the evidence-mapping for dose of FOLFIRINOX, the ‘planned’ dose in all the studies (N = 37) was as follows: 75–100% oxaliplatin, 75–100% irinotecan, 0–100% 5-FU bolus, and 75–133% 5-FU CIV. A total of 24 studies included the ‘planned’ modified FOLFIRINOX group, and 12 different types of modified regimens were identified in these studies (Figure 3(a)). The most frequent ‘planned’ modified regimen was 100% oxaliplatin, 83.3% irinotecan, 0% 5-FU bolus, and 100% 5-FU CIV (N = 7, 29.2%). Most studies with ‘planned’ modified regimen were designed to maintain the dose of oxaliplatin (N = 17, 70.8%) and 5-FU CIV (N = 22, 91.7%).

Trend analysis of dose modification of FOLFIRINOX using RDI-map.com. (a) Various types of planned dose modification in FOLFIRINOX. (b) Gap between ‘planned’ and ‘actual’ dose of FOLFIRINOX.
The ‘actually administered’ dose was reported in 20 studies (6 prospective and 14 retrospective studies). The ‘actual’ dose reduced to 54–96% for oxaliplatin, 61–88% for irinotecan, 0–92% for 5-FU bolus, and 63–98% for 5-FU CIV (Figure 3(b)). The pattern of dose reduction varied from study to study. However, the gap between the ‘planned dose’ and the ‘actual dose’ was consistently high regardless of study type (prospective or retrospective design) or the planned regimen (standard FOLFRINOX or modified FOLFIRINOX).
Clinical outcomes according to dose modification
To assess clinical outcomes according to the ‘planned dose’ of FOLFIRINOX, 27 studies were included. Including three comparative studies in both groups, 14 and 16 studies were included in the planned ‘standard’ FOLFIRINOX group and the planned ‘modified’ FOLFIRINOX group, respectively. The pooled overall ORR was 30.9% (95% CI: 27.4–34.4%), and the heterogeneity across studies was moderate (I2 = 62%, p < 0.010) (Figure 4). The ORRs for the planned ‘standard’ group and planned ‘modified’ group were 28.2% (95% CI: 22.5–33.9%) and 33.8% (95% CI: 30.3–37.3%), respectively (P for subgroup difference = 0.100). However, the heterogeneity of the planned ‘standard’ group was high (I2 = 78%, p < 0.010), whereas that of the planned ‘modified’ group was low (I2 = 0%, p = 0.800).

Forest plot to compare ORR according to planned dose of FOLFIRINOX.
Regarding hematological toxicity, 22 and 20 studies were included to analyze the rate of neutropenia and febrile neutropenia, respectively. The results for hematological toxicity in the included studies were highly heterogeneous. The pooled rate of neutropenia for all studies was 43.8% (95% CI: 33.2–54.3%) (Figure 5(a)). The rate for the planned ‘standard’ FOLFIRINOX group and the planned ‘modified’ FOLFIRINOX group were 50.4% (95% CI: 0–100%) and 35.3% (95% CI: 0–100%), respectively (P for subgroup difference = 0.160). The pooled rate of febrile neutropenia for all studies was 9.4% (6.3–12.4%) (Figure 5(b)). The rate was 11.6% (95% CI: 0–16.0%) and 5.5% (95% CI: 0–8.9%) for the planned ‘standard’ group and the planned ‘modified’ group, respectively (P for subgroup difference = 0.030).

Forest plots to compare hematological toxicity according to planned dose of FOLFIRINOX. (a) Neutropenia. (b) Febrile neutropenia.
Among these studies, three comparative studies on planned ‘standard’ FOLFIRINOX versus planned ‘modified’ FOLFIRINOX groups were identified. A pooled analysis of clinical outcomes of these studies was conducted as a sensitivity analysis (Supplemental Figure S2). The pooled HR values of the planned ‘modified’ group versus the planned ‘standard’ group for PFS and OS were 0.98 (95% CI: 0.73–1.30) and 0.96 (95% CI: 0.74–1.25), respectively. The pooled OR values for neutropenia and febrile neutropenia events were 0.45 (95% CI: 0.18–1.10) and 0.90 (95% CI: 0.43–1.92), respectively.
Finally, subgroup analyses of the ORR and toxicity according to age, sex (female versus male dominant study), study type (prospective versus retrospective), year of publication, or country (Asian countries versus non-Asian countries) were conducted to determine the factors associated with heterogeneity among studies (Supplemental Figure S3). In the subgroup analysis according to age divided by the median of 62 years, the heterogeneity of ORR in the ‘standard’ FOLFIRINOX group decreased (I2 = 53%, p = 0.090 in the older subgroup and I2 = 68%, p < 0.001 in the younger subgroup). Additionally, in the older subgroup analysis, the ORR of ‘standard’ FOLFIRINOX was significantly inferior to that of modified FOLFIRINOX (18.2% [95% CI: 11.2–25.2%] versus 36.1% [95% CI: 31.2–41.0%]; p < 0.001). However, other subgroup analyses failed to identify factors that could explain high heterogeneity among studies.
Risk of bias assessment
Risk of bias of the included studies was evaluated in accordance with the JBI manual according to study type (Supplemental Tables S1 and S2). When the answer ‘yes’ in each question in the checklist was regarded as one point, almost all included studies were evaluated as appropriate with a score of five or higher. The funnel plot and Egger test of ORR for the assessment of publication bias showed no significant asymmetry (P for Egger test = 0.221) (Supplemental Figure S4).
Discussion
In the present study, we aimed to determine a 10-year trend for ‘planned’ and ‘actual’ doses of FOLFIRINOX as first-line treatment for MPC and investigated the clinical outcomes according to dose modification. Using evidence-mapping methodology, it was possible to recognize the trends in FOLFIRINOX dose used in previous trials and we found that there were various different types of ‘planned’ dose modification in FOLFIRINOX and a significant gap between the ‘planned’ and ‘actual’ doses, regardless of study type or planned dose. In addition, our meta-analysis revealed similar efficacy and lower rate of febrile neutropenia with the planned ‘modified’ regimen than with the planned ‘standard’ regimen.
We developed an open-access, user-friendly, web-based interactive bubble-plot program (RDI-map.com), which allowed us to map the entire chronological trend for FOLFIRINOX dose. The most well-known study on ‘modified FOLFIRINOX’ dose in patients with PC is the PRODIGE 24/ACCORD 24 trial, in which 100% oxaliplatin, 83.3% irinotecan, 0% 5-FU bolus, and 100% 5-FU CIV were used as adjuvant treatment. 49 Although most researchers consider this dose modification as the standard ‘modified FOLFIRINOX’ dose, only seven studies (29.2%) used the same dose modification and 11 other types of dose modifications were observed in the mapping. In addition, the ‘actual’ dose differed significantly from the ‘planned’ dose, even in the prospective studies using the ‘planned’ modified dose of FOLFIRINOX. Since there has been no consensus on the appropriate dose of ‘modified FOLFIRINOX’ so far, further trials to determine the appropriate dose of ‘modified FOLFIRINOX’ are necessary.
In the meta-analysis, the pooled ORR in the planned ‘standard’ FOLFIRINOX group was 28.2% and it was comparable with the ORR of 31.6% in the PRODIGE 4/ACCORD 11 trial. 3 The ORR of planned ‘modified’ FOLFIRINOX group in this meta-analysis showed highly consistent results and the pooled ORR of modified regimen was not lower than that of ‘standard’ regimen (33.8% versus 28.2%; p = 0.100). In addition, the rates of neutropenia and febrile neutropenia in the ‘modified’ group were lower than those in the ‘standard’ group, as reported in the PRODIGE 24/ACCORD 24 trial. 49 Although there were no RCTs directly comparing ‘modified’ FOLFIRINOX to ‘standard’ FOLFIRINOX, there were a few meta-analyses for this issue previously. Previous meta-analyses have consistently shown non-inferior clinical efficacy and lower toxicity of modified regimen, but these meta-analyses had a limitation in which the number of trials for modified FOLFIRINOX was small. Therefore, the present meta-analysis, which included a lot more trials for modified regimen, could clarify the clinical potential of modified FOLFIRINOX and justify the need for modified regimen. In addition, a recent phase II randomized trial named as PANOPTIMOX-PRODIGE 35 trial also reported that a new dose modification strategy of 4 months of standard FOLFIRINOX followed by 5-FU maintenance treatment showed similar clinical efficacy and longer median survival without deterioration in quality-of-life scores, further emphasizing the need for dose modification. 50
However, the heterogeneity of clinical outcomes among studies in this meta-analysis may confound the interpretation of the results. Despite various subgroup analyses, factors related with high heterogeneity were not clear, except for age in the standard FOLFIRINOX group. One of the potential factors for heterogeneity, not included in the subgroup analyses, may be the actually administered dose. The relationship between actually administered dose intensity and treatment response in FOLFIRINOX is still controversial. A previous study by Lee et al. suggested that the ORR was maintained with the threshold of the cumulative RDI of 70%, but the other study by Vary et al. reported that they could not find any cutoff of dose reduction for disease control rate.32,51 Therefore, future studies for dose reduction in FOLFIRINOX should elucidate the relationship between actually administered dose intensity, or the pattern of dose reduction, and treatment response.
Although this study showed the need for a dose modification of FOLFIRINOX and the absence of a standardized dose modification protocol, it is not easy to make a new protocol for an already commercialized chemotherapy regimen. Moreover, a single-dose modification protocol may not be sufficient because several studies have already reported that there could be various confounding factors in the response and metabolism of chemotherapy agents such as race or genetic predispositions.25,52,53 Therefore, until the best dose modification protocol or prediction model for appropriate dose modification of FOLFIRINOX is made, a tailored approach that combines the information provided in this evidence-mapping, such as trend, range of dose modification, and specific research results classified according to geographical or chronological context, with patient characteristics and oncologist’s personal experience on chemotherapy, should be considered.
This study has some limitations. First, most articles included in this study provided comparable quantitative results on clinical outcomes, but only insufficient and heterogeneous information about dose of FOLFIRINOX. For example, the statistical expressions of dose information were different across studies, including the median dose, mean dose, or dose in range. It was difficult to conduct a conventional statistical analysis on dose information; therefore, we adopted a descriptive evidence-mapping method and developed RDI-map.com, which can be used to show trends on dose information intuitively. Second, many retrospective studies were included in this analysis; thus, the potential bias related to study type was inevitable. However, there were enough prospective studies (N = 11), and similar trends even in the prospective studies, such as the diversity in the ‘planned’ modified FOLFIRINOX doses and the gap between the ‘planned’ dose and ‘actual’ dose, were identified. Third, the included studies were missing key information such as the reason for dose reduction or detailed dose reduction protocol that might explain the reason of high heterogeneity among studies or help finding appropriate dose modification strategy. Finally, most studies included in the meta-analysis were single-arm studies or comparative studies that used regimens other than FOLFIRINOX, and thus, the ORR was used as the primary endpoint for clinical efficacy assessment instead of PFS or OS. However, similar results were obtained in the sensitivity analysis of comparative studies of standard FOLFIRINOX versus modified FOLFIRINOX. Despite these limitations, this study has sufficient clinical significance because this is the first study to summarize and illuminate the practical pattern of FOLFIRINOX dose for MPC, to the best of our knowledge. In addition, we clearly demonstrated the limitations and considerations required in the current chemotherapy regimen for MPC using a web-based interactive mapping program.
Conclusion
RDI-map.com, a multifaceted evidence-mapping program for dose modification of FOLFIRINOX, showed that there was still no consensus on the appropriate dose of ‘modified FOLFIRINOX’ and that there was a significant gap between the ‘planned’ and ‘actual’ dose. In addition, our study again showed that the planned ‘modified’ FOLFIRINOX had similar efficacy and reduced toxicity compared to the planned ‘standard’ regimen. Therefore, these data rationalize the need to standardize modified FOLFIRINOX dose for patients with MPC and provide a basis for both clinicians and researchers in determining the appropriate FOLFIRINOX dose.
Supplemental Material
sj-docx-1-tam-10.1177_17588359231175441 – Supplemental material for Real-world dose reduction of standard and modified FOLFIRINOX in metastatic pancreatic cancer: a systematic review, evidence-mapping, and meta-analysis
Supplemental material, sj-docx-1-tam-10.1177_17588359231175441 for Real-world dose reduction of standard and modified FOLFIRINOX in metastatic pancreatic cancer: a systematic review, evidence-mapping, and meta-analysis by Kwangrok Jung, Suhyun Choi, Hyunjoo Song, Kyuhan Kwak, Soyeon Anh, Jae Hyup Jung, Bomi Kim, Jinwoo Ahn, Jaihwan Kim, Jin-Hyeok Hwang and Jong-chan Lee in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-2-tam-10.1177_17588359231175441 – Supplemental material for Real-world dose reduction of standard and modified FOLFIRINOX in metastatic pancreatic cancer: a systematic review, evidence-mapping, and meta-analysis
Supplemental material, sj-docx-2-tam-10.1177_17588359231175441 for Real-world dose reduction of standard and modified FOLFIRINOX in metastatic pancreatic cancer: a systematic review, evidence-mapping, and meta-analysis by Kwangrok Jung, Suhyun Choi, Hyunjoo Song, Kyuhan Kwak, Soyeon Anh, Jae Hyup Jung, Bomi Kim, Jinwoo Ahn, Jaihwan Kim, Jin-Hyeok Hwang and Jong-chan Lee in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
