Abstract
There are questions surrounding the real-world effectiveness of chemotherapeutic treatments for pancreatic ductal adenocarcinoma. This literature review compared the clinical characteristics and outcomes of available real-world evidence (RWE) for liposomal irinotecan in combination with 5-fluorouracil (5-FU) and leucovorin (LV), a treatment regimen indicated for patients with metastatic pancreatic ductal adenocarcinoma (mPDAC) who previously progressed on gemcitabine-based therapy.
A targeted literature search was conducted in the PubMed Central® and Embase® databases to identify available RWE regarding patients with mPDAC receiving liposomal irinotecan published within the last 5 years (January 2014–September 2019). Data were extracted for prior lines of therapy, performance status, overall survival (OS), progression-free survival (PFS), duration of exposure, and adverse events.
Six studies met inclusion criteria. A comparison of baseline patient characteristics and results with the included evidence reveals a clinically fragile, real-world patient population in terms of age (range: 61–68), prior lines of therapy with 34–61% of patients receiving ⩾2 lines of lines of prior therapy and performance status [49.8–100% of patients with Eastern Cooperative Oncology Group (ECOG) 0–1]. Studies observed wide OS (range: 5.3–9.4 months) and similar PFS (range: 2.3–4.1 months), with two studies measuring duration of exposure (7.3 weeks, 3.1 months). Patients analyzed by RWE studies tended to be older with significant disease progression, poor performance status, and more heavily pretreated compared with the phase III registrational trial (NAPOLI-1). Despite this, patients treated with liposomal irinotecan + 5-FU/LV therapy had similar outcomes as those in NAPOLI-1.
Background
Pancreatic ductal adenocarcinoma (PDAC), an exocrine subtype pancreatic malignancy affecting the pancreatic ductal epithelium, comprises approximately 85% of new pancreatic cancer cases in the United States (US) and is one of the most lethal cancers encountered.1–3 PDAC is characterized by diagnosis in the locally advanced or metastatic stages, with rapidly replicating cancer cells resulting in metastatic propensity and resistance to cytotoxic treatment due, in part, to a very dense stroma.1,2 PDAC tends to be diagnosed in later stages due to absence of symptomatology in the early stages. Late diagnosis generally eliminates the possibility for curative surgical intervention. The intrinsic cytotoxic resistance of the disease renders many chemotherapeutic treatments ineffective. 3
The combination of gemcitabine (gem) and nano-particle albumin bound paclitaxel (nab-pac) was approved by the US Food and Drug Administration (FDA) for first-line treatment of metastatic PDAC (mPDAC) in 2013, and other combinations, though not indicated for mPDAC, have shown comparable efficacy in clinical studies. 2 Clearly, patients with mPDAC could benefit from second line treatment options, but drug discovery has been challenging and incremental, with 12 phase III trial failures since 2015. 4 More recently, liposomal irinotecan (Onivyde®, historical names include nal-IRI, MM-398, or PEP02), an intravenous liposomal formulation that encapsulates the topoisomerase 1 inhibitor irinotecan in a lipid-bilayer vesicle, has emerged as the only second-line/post-gemcitabine treatment in mPDAC that is NCCN Category I, ASCO Preferred, and FDA-approved.5–7 On the basis of results from the NAPOLI-1 study, a pivotal phase III registrational trial, liposomal irinotecan in combination with 5-fluorouracil (5-FU) and leucovorin (LV) (liposomal irinotecan + 5-FU/LV) is indicated in the treatment of mPDAC as a second-line therapy following disease progression with gem-based treatment. 8
In the NAPOLI-1 study, combination therapy with liposomal irinotecan + 5-FU/LV significantly increased median overall survival (OS; primary endpoint) and progression-free survival (PFS; secondary endpoint) compared with 5-FU/LV alone. Median OS was 6.1 months [95% confidence interval (CI): 4.8–8.9] in patients randomized to liposomal irinotecan + 5-FU/LV, compared with 4.2 months (95% CI: 3.3–5.3) in patients randomized to 5-FU/LV alone [unstratified hazard ratio (HR): 0.67; 95% CI: 0.49–0.92;
The narrow eligibility criteria for patients enrolled in clinical trials can mean that the studied population is very different from the population treated in normal life, which can impact patient outcomes. This review compared patient demographics and clinical outcomes from existing real-world evidence (RWE) that assessed mPDAC patients treated with liposomal irinotecan + 5-FU/LV.
Methods
A targeted search of PubMed Central® and Embase® databases for available RWE in pancreatic adenocarcinoma patients receiving liposomal irinotecan was conducted by two experienced reviewers in late September 2019. The search strategy contained the following terms and their associated MeSH® and Emtree® terms: irinotecan, nanoliposomal, nal-IRI, Onivyde®, database, registry, cohort, cohort analysis, retrospective, pragmatic, and pragmatic trial. The full search string can be found in Supplemental Table S1. Publications meeting the following criteria were included: patient population with mPDAC, use of liposomal irinotecan + 5-FU/LV, RWE study designs (database studies, registries, cohort analyses, and pragmatic clinical trials), study populations from January 2014 to September 2019. Manuscripts and congress materials were eligible for inclusion in the targeted review. Studies of primary indications other than mPDAC, non-real-world data or study designs (e.g., randomized clinical trial, preclinical, or formulation studies), pre-clinical studies, and studies published before 2014 were excluded.
A descriptive comparison of RWE studies was performed on study population demographics, treatment information, and outcome measures. The following data were extracted for comparison: median age at treatment onset (years), number of prior lines of therapy, duration of therapy exposure (DOE, weeks), adverse events (AEs)/toxicity experienced (%), ECOG performance status, OS (months), and PFS (months). Statistical analysis information reported for each outcome measure was extracted. 9
Results
The search strategy yielded 81 studies, 75 of which were excluded according to the criteria presented in Figure 1. The remaining six publications – Glassman

Integrated database flow diagram.
Selected baseline patient characteristics of RWE studies.

ECOG, Eastern Cooperative Oncology Group; IQR, interquartile range; RWE, real-world evidence.
Selected treatment effectiveness results of RWE studies.
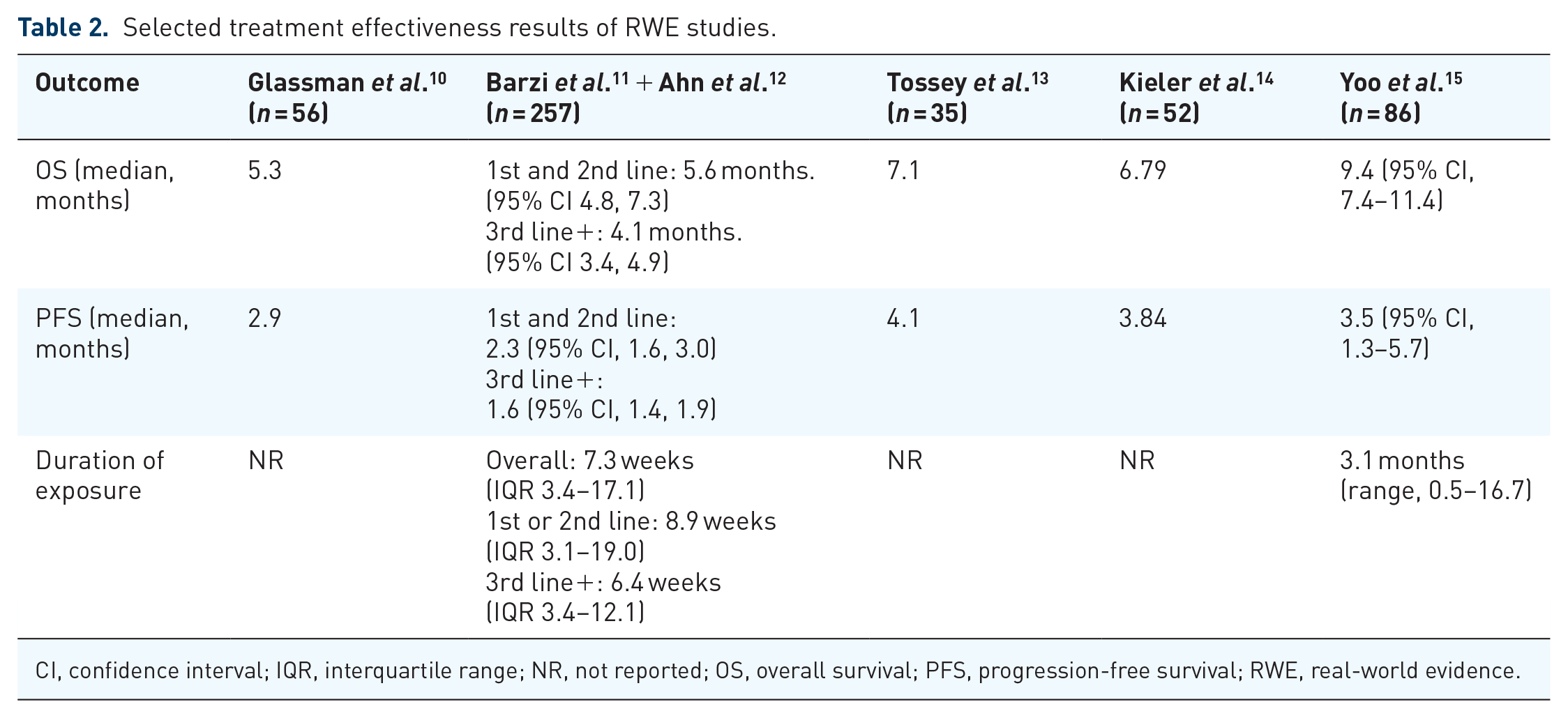
CI, confidence interval; IQR, interquartile range; NR, not reported; OS, overall survival; PFS, progression-free survival; RWE, real-world evidence.
Adverse events of RWE study results.
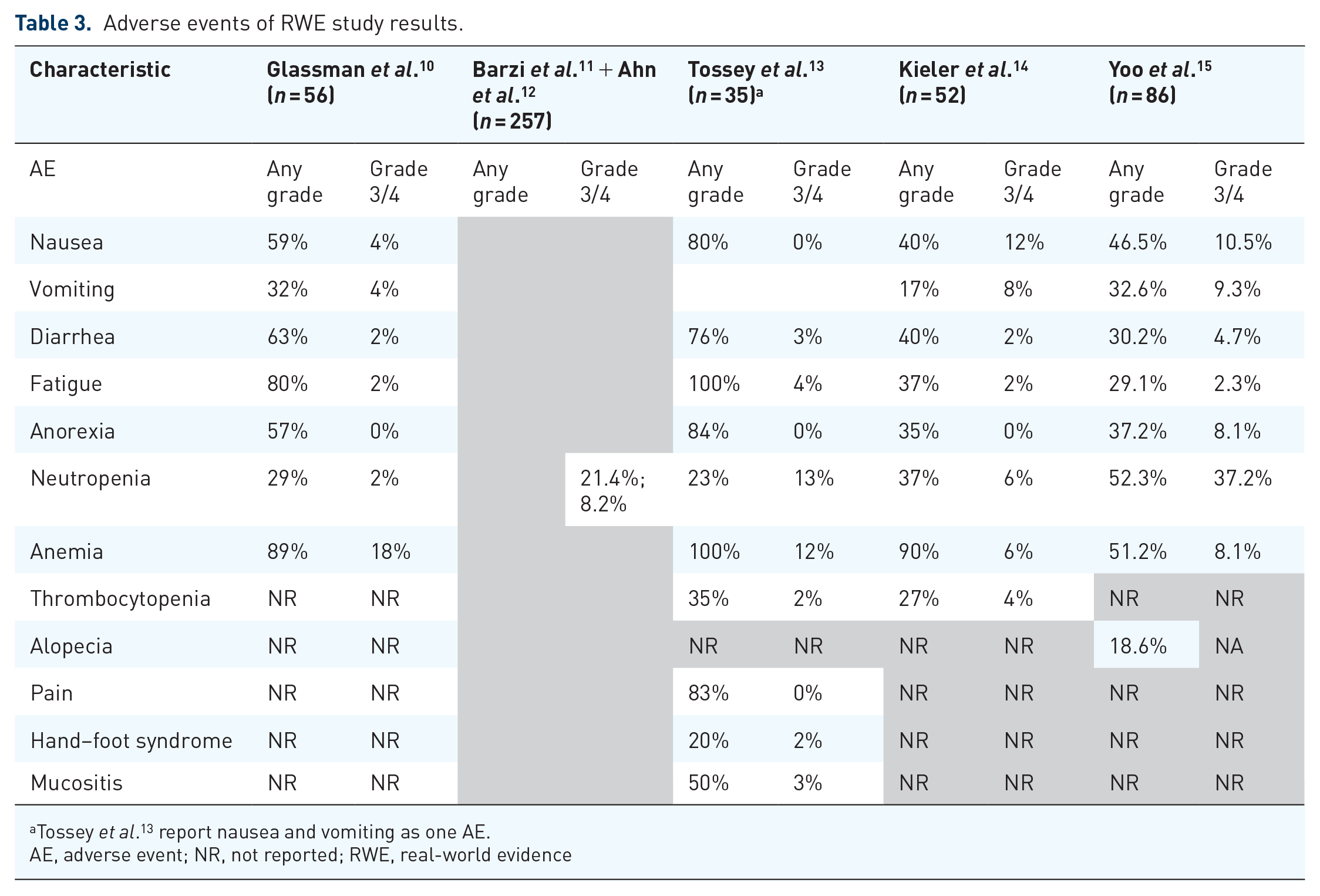
Tossey
AE, adverse event; NR, not reported; RWE, real-world evidence
Eligible studies
Glassman
Barzi
Tossey
Kieler
Yoo
Patient characteristics
A total of 56 patients (median age: 68 years) were included in the Glassman
Overall survival
The OS reported in Glassman
Progression-free survival
Glassman
Duration of exposure
The median DOE for the population in Barzi
Adverse events/toxicity
In Glassman
The frequencies of AEs observed are summarized in Table 3.
Discussion
The RWE in mPDAC reviewed here demonstrates that patients tend to be older, have experienced more lines of therapy (47.3% with two or more prior lines), with poor performance status (69.1% with ECOG 0-1) in practice settings, compared with prior clinical trial research (NAPOLI-1, [ClinicalTrials.gov identifier: NCT01494506]). 9 Only one of the included studies had a median age below that of the patients treated with liposomal irinotecan + 5-FU/LV in the NAPOLI-1 trial. Additionally, patients tended to be heavily pre-treated, with some patients receiving three or more chemotherapy regimens before being treated with liposomal irinotecan combination therapy. Despite evidence of clinical fragility in this real-world patient population, liposomal irinotecan combination therapy had a similar effectiveness in terms of OS and PFS as reported in NAPOLI-1.
AEs in NAPOLI-1 were identified in 56 (48%) patients assigned to the liposomal irinotecan + 5-FU/LV arm. AEs that resulted in dose reduction occurred in 39 (33%) patients in the liposomal irinotecan + 5-FU/LV arm. The most common AEs that precipitated dose reduction were neutropenia (27%), diarrhea (13%), vomiting (11%), and fatigue (14%). Similar distributions of AEs were observed in analyzed RWE, suggesting that the safety profile of liposomal irinotecan combination is comparable across clinical practice settings.
This comparison of available RWE for patients with mPDAC undergoing liposomal irinotecan + 5-FU/LV treatment indicates that outcomes of patients treated with liposomal irinotecan + 5-FU/LV, including survival and progression outcomes, are consistent across multiple real-world settings and are similar to those reported in NAPOLI-1. Safety profile and frequencies of AEs reported in the observed real-world studies were comparable. Taken together, the RWE suggests that liposomal irinotecan + 5-FU/LV is effective in routine clinical practice settings, with a similar adverse event profile to a randomized controlled trial, while being administered to clinically fragile populations. These characteristics likely define the real-world status of mPDAC patients outside of clinical trial settings.
RWE has several notable limitations. Data elements may be missing or incorrect due to entry error. Moreover, the characteristics of each RWE study should be considered. Glassman
Conclusion
There is a pressing need for more effective chemotherapeutic treatments for mPDAC. RWE provides a supplemental source of information for more comprehensive understanding of patient outcomes for this less controlled patient population in real-world clinical practice settings. The RWE studies examined here provide evidence supports the real-world effectiveness of liposomal irinotecan treatment across different settings of care and patient types. Unlike in randomized controlled trials, in routine clinical practice, patients tended to be older with more prior lines of therapy and poor performance status. Additionally, some real-world patients were heavily pre-treated before being treated with liposomal irinotecan combination therapy. Despite this, patients treated with liposomal irinotecan combination therapy had similar OS, PFS, and AE outcomes as those in NAPOLI-1.
Supplemental Material
Supplemental_Table_1 – Supplemental material for Comparing real-world evidence among patients with metastatic pancreatic ductal adenocarcinoma treated with liposomal irinotecan
Supplemental material, Supplemental_Table_1 for Comparing real-world evidence among patients with metastatic pancreatic ductal adenocarcinoma treated with liposomal irinotecan by Jim Koeller, Andy Surinach, Steven R. Arikian, Marko Zivkovic, Patrick Janeczko, Paul Cockrum and George Kim in Therapeutic Advances in Medical Oncology
Footnotes
Acknowledgements
Conflict of interest statement
PC is an employee of Ipsen. AS, SRA, MZ, PJ are employees of Genesis Research and report an advisory/consulting role with Ipsen. JK reports a consulting advisory/consulting role with Ipsen, Coherus, Daiichi, and Tocagen. GK reports no conflict of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was sponsored by Ipsen Biopharmaceuticals, Inc. The sponsor was involved in the design of the study, analysis, and interpretation as well as final approval of the manuscript.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
