Abstract
Breast cancer (BC) is the most commonly diagnosed cancer worldwide. Advanced BC with brain metastasis (BM) is a major cause of mortality with no specific or effective treatment. Therefore, better knowledge of the cellular and molecular mechanisms underlying breast cancer brain metastasis (BCBM) is crucial for developing novel therapeutic strategies and improving clinical outcomes. In this review, we focused on the latest advances and discuss the contribution of the molecular subtype of BC, the brain microenvironment, exosomes, miRNAs/lncRNAs, and genetic background in BCBM. The blood–brain barrier and blood–tumor barrier create challenges to brain drug delivery, and we specifically review novel approaches to bypass these barriers. Furthermore, we discuss the potential application of immunotherapies and genetic editing techniques based on CRISPR/Cas9 technology in treating BCBM. Emerging techniques and research findings continuously shape our views of BCBM and contribute to improvements in precision therapies and clinical outcomes.
Background
Based on data released from the International Agency for Research on Cancer in 2021, breast cancer (BC) has surpassed lung cancer as the most common cancer in the world. 1 One of the greatest challenges to BC treatment is distant metastasis to different organs, such as the lung, brain, bone, and liver. 2 Brain metastasis (BM) affects approximately 30–50% of patients with metastatic BC and has become a major cause of morbidity and mortality.3,4 In terms of epidemiology, the incidence of BCBM has increased significantly over the past decade due to earlier diagnosis by imaging techniques and more effective treatment of primary BC. The incidence of BM varies depending on the BC subtype.5–7 In triple-negative breast cancer (TNBC), the incidence ranges from 25% to 45% and is approximately 50% in human epidermal growth factor receptor 2 (HER2)-positive BC.8,9 The prognosis of breast cancer brain metastasis (BCBM) patients is usually poor, with a median survival time ranging from 3 to 25 months.6,10 BCBM is also associated with serious cognitive and emotional impairment. Therefore, quality of life and life expectancy are significantly impacted in patients with BM. Young age, high histological grade, large tumor size, TNBC type, HER2 enrichment, and extracerebral metastasis are currently thought to be high-risk factors for BCBM. 11 However, the pathological and molecular mechanisms underlying the development of BCBM remain unclear. A better understanding of the underlying mechanisms and exploration into novel therapeutic strategies are urgently needed.
The metastasis cascade of BCBM involves multiple steps consisting of proliferation, extracellular matrix (ECM) invasion, vascular intravasation, survival in the bloodstream, vascular extravasation, and brain colonization.12,13 The activation of epithelial–mesenchymal transition (EMT) is considered to be an important step for primary cancer cell dissemination. BC cells enter the bloodstream through EMT and ultimately can implant into the brain. 13 To survive and proliferate in the brain microenvironment, BC cells must acquire several properties needed to pass through the blood–brain barrier (BBB), accumulate around blood vessels, and promote neoangiogenesis until the BBB is modified to be the blood–tumor barrier (BTB), which is typical in BM. Compared to breast microenvironments, the brain has a distinct anatomy, cell types, metabolic pathways, and local immune microenvironment. The reverse of the EMT, the mesenchymal–epithelial transition, occurs in a process of disseminated tumor cells, enabling them to metastasize from the breast to the brain. Metastatic BC cells can arrive in the cerebellum, cerebral cortex, and other areas of the brain, and are referred to as parenchymal metastasis, leptomeningeal metastasis, and choroid plexus metastasis, respectively. 14 The most common type of BM is parenchymal metastasis (90%), leptomeningeal metastasis accounts for 8%, and choroid plexus metastasis is rare.
The currently used multimodal treatment approaches for BCBM patients, such as surgery, radiation, chemotherapy, and targeted therapy, have led to poor outcomes for most patients. Evidence obtained over decades has shown that, due to low permeability and poor penetration through the BBB/BTB, systemically administered chemotherapy and targeted therapies are largely ineffective as BCBM treatments.15,16 No specific agents for BCBM therapy have been approved by the FDA.
17
Recent advances in clinical trials and preclinical research, however, have confirmed that systemic delivery of therapeutic agents
In this review, we highlight the up-to-date understanding of BCBM molecular mechanisms and explore novel therapeutic strategies. We first introduce the BC molecular subtypes, aiming to provide a significant clinical context, which requires deeper scientific investigation. We next discuss the permeability of the BBB and BTBs, as well as the role played by the distinct brain microenvironment in BCBM. We also discuss the roles of genes, exosomes, miRNAs, and lncRNAs in BCBM. Furthermore, we provide perspectives on recent research that may lead to novel approaches for the treatment of BCBM patients (Figure 1).

The metastasis cascade of BCBM. The steps in the formation of BCBM consist of proliferation, ECM invasion, vascular intravasation, hematogenous dissemination, vascular extravasation, and brain colonization. BC cells acquire several properties needed to cross the BBB, accumulate around blood vessels and promote neoangiogenesis until the BBB is disrupted to be the BTB.
BC subtypes and their association with BM
In 2000, Perou
Although the brain is not the most easily accessible site for HER2-positive BC metastasis, some factors in these patients may lead to preferential BCBM. For example, after trastuzumab therapy, survival rates are significantly improved, but drug penetration across the BBB is limited, and HER2-positive BC cells tend to spread to the brain. From 30% to 55% of patients develop BM. 9 TNBC patients have a 25–46% chance of developing BM. 9 The formation of BM in TNBC is frequently accompanied by the progression of extracranial metastasis, but BM is an early manifestation, which may be due to the lack of effective medications and the high aggressiveness of these tumor cells. When TNBC patients develop BM, the prognosis is poor, with an average survival time of less than 6 months.
Using the SEER database, Martin
According to a recent systematic review published by the Jerzak group, the incidence of BCBM was 31%, 32%, and 15% for HER2-positive, TNBC, and HR+/HER2− patients, respectively. Patients with HER2-positive BC and TNBC had a high incidence of BM.
7
Tseng
Clinically relevant BC subtypes and their association with BM.4,26,27
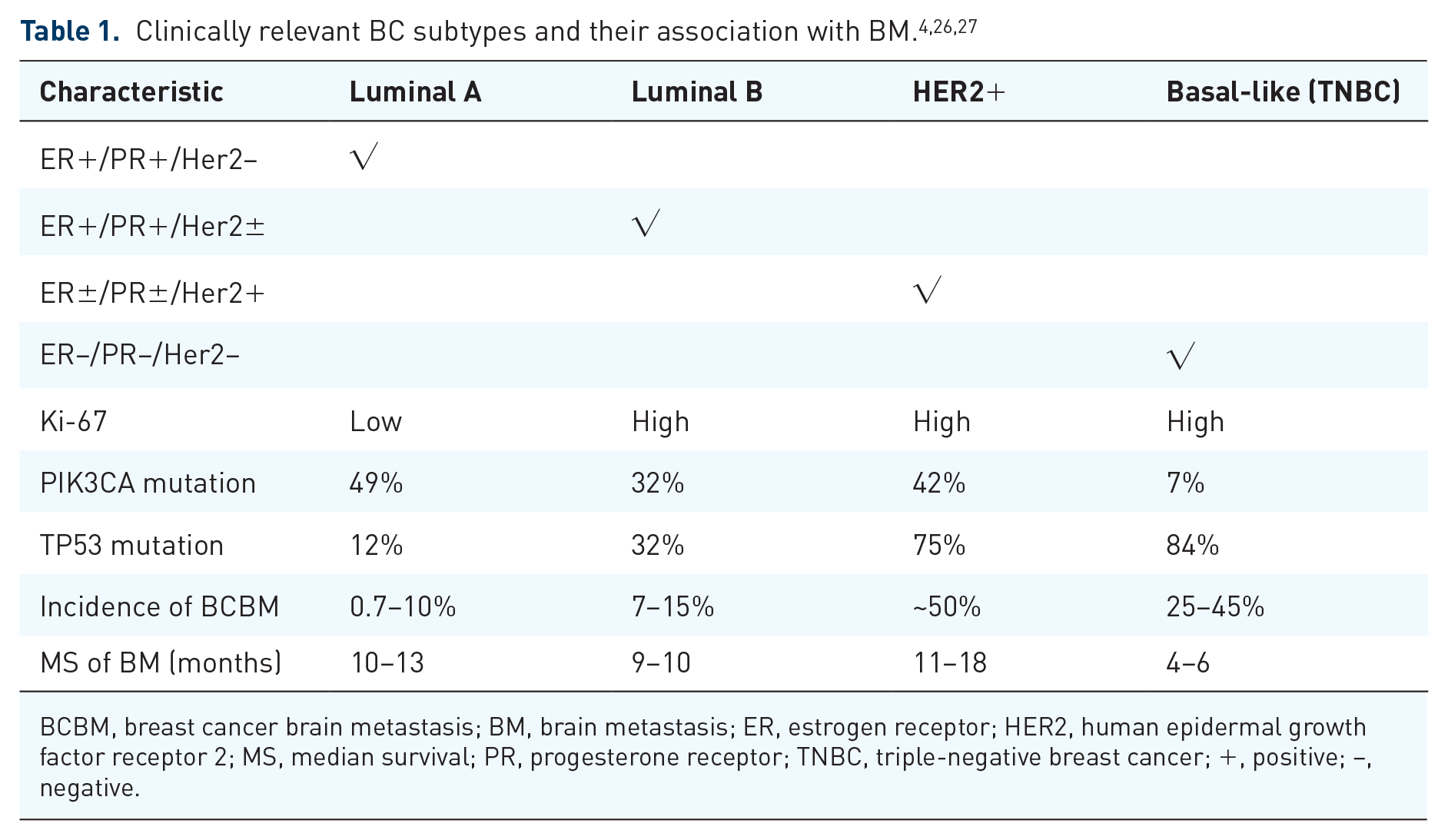
BCBM, breast cancer brain metastasis; BM, brain metastasis; ER, estrogen receptor; HER2, human epidermal growth factor receptor 2; MS, median survival; PR, progesterone receptor; TNBC, triple-negative breast cancer; +, positive; −, negative.
Brain microenvironment and BCBM
The tumor microenvironment is made up of cellular, noncellular, and ECM components. The tumor and its surrounding microenvironment meet specific criteria to initiate cancer cell invasion, colonization, and growth in the distant brain. 28 A growing body of evidence shows that the brain microenvironment is closely related to the development of BCBM. Analyzing how tumor cells cross the BBB and interact with various components of the brain microenvironment can help in understanding BCBM development.
According to the ‘seed and soil’ hypothesis, tumor dissemination is closely related to the interactions between cancer cells and the microenvironment. 29 When tumor cells metastasize to the brain, they face a complex microenvironment that is significantly different from that of the primary site. In the primary tumor environment, various cellular components, such as cancer cells, immune cells, mesenchymal stem cells (MSCs), and endothelial cells, can promote tumor growth. 30 The brain is known to consist of parenchyma and leptomeninges.31,32 The brain parenchyma is composed of dense neurons and glial cells. The crosstalk between tumor cells and these brain-specific cells is a distinguishing feature of BM. The leptomeninges (pia and arachnoid) are brain-specific structures. In addition, the brain metabolic environment differs from that of the primary tumor site. In comparison to the breast microenvironment, the brain microenvironment differs in terms of anatomy, cells, pathways, and immune environment, suggesting that tumor cells that metastasize to the brain must also possess unique features to cross the BBB and grow efficiently in this distinct environment. 33 The dynamic interaction between cancer cells and the brain microenvironment, which is mediated by stromal cells, immune cells and the ECM, is important for cancer development after cell implantation in the brain. 34
Molecular crosstalk between astrocytes and cancer cells during BCBM development has been extensively documented.
35
Astrocytes are highly abundant in the brain and are involved in forming the BBB and maintaining tissue homeostasis. To colonize and proliferate in the brain, BC cells must pass through the BBB.
36
The crucial roles played by tumor-killing or tumor-promoting astrocytes in BM have been well characterized. A1 and A2 are two kinds of reactive astrocytes. The A2 subtype accounts for the majority of astrocytes associated with tumor growth, and these cells express the phosphorylated form of STAT3 at a high level. During the early stages of BC, the STAT3 signaling pathway in BC cells is activated by cytokines and growth factor receptors, creating an immunosuppressive microenvironment that promotes tumor growth and tumor cell metastasis.
37
The evidence has indicated that STAT3 signaling plays a crucial role in the molecular crosstalk between BC cells and astrocytes.
34
For example, Yu
The brain has long been thought to be an immune-privileged organ with circulating cells excluded from the brain parenchyma. However, this concept has recently been challenged. Functional lymphatic vessels have been discovered and characterized in the meninges of the dorsal and basal skull.
39
These structures can transport cerebrospinal fluid (CSF) and immune cells into deep cervical lymph nodes.39,40 Further research confirmed that they play crucial roles in regulating brain tumor immunity.41,42 The discovery of brain lymphatic pathways prompted scientists to investigate the neuroimmunology hypothesis, and provided an important breakthrough for understanding the mechanism and developing a treatment strategy for BM. Although the types of immune cells in the brain differ from those in peripheral organs, the brain is no longer considered an immune privileged organ, which increases the possibility that immunotherapy for BM can be developed.
43
Brain lymphocytes interact with the systemic lymphatic system through the meningeal lymphatic pathway in deep cervical lymph nodes, and reinvigorated T cells enter the brain to exert antitumor effects. This interaction may be the reason that, in previous immune checkpoint studies, scientists found immunotherapy to be effective against BM. Studies have shown encouraging results for the use of immune checkpoint inhibitors in BM patients. Notably, Ogiya
However, to date, immunotherapies have provided little benefit in treating BM due to low BBB permeability and poor penetration of the BBB. In addition, BC is one of the major causes of leptomeningeal metastasis, and little is known about the role that the brain microenvironment plays in leptomeningeal metastasis. How do BC cells influence the subarachnoid microenvironment? They secrete complement component 3, which opens BBB tight junctions, allowing growth factors to enter the CSF to promote metastatic growth 46 (Figure 2).

Crosstalk among BC cells, glial cells, and the brain microenvironment. Exosomes, miRNAs, and lncRNAs derived from BC cells can increase BBB permeability and promote BC cell crossing. In addition, the higher BTB permeability was due to upregulated astrocytic secretion of IL-6 and CCL2. The STAT3 signaling pathway in BC cells establishes an immunosuppressive microenvironment to promote tumor growth and metastasis. In addition, crosstalk between microglia and reactive astrocytes has the same effect. Astrocyte-released exosomes transfer
Bypassing the BBB and BTB
Scientists have made significant advances in their understanding of the biology of the BBB in recent decades. 47 The BBB is a neurovascular unit (NVU) that separates circulating blood and brain tissue and is composed of endothelium capillaries, a vascular basement membrane, pericytes, and astrocytic endfeet.48,49 Endothelial cells form the impermeable BBB by surrounding astrocytes and the basement membrane. The BBB is both a physiological and biochemical obstacle that protects the brain from potentially hazardous chemicals while also maintaining homeostasis. 50 By regulating ions, the BBB creates a stable microenvironment for neuronal function. However, the BBB also limits the transport of therapeutic agents to the brain, making systemic therapies less effective in treating BM than in treating other types of cancer. As BM develops from BC or other cancer cells, the BBB is disrupted, and a BTB is formed. 51 Although the structure is damaged, a BTB can impede the transport of anticancer medicines to brain tumors, leading to poor treatment results. Recent advances in scRNA-seq of NVU cells have provided distinct cell-specific transcriptional profiles of the BBB that have been previously unknown. 15 Undoubtedly, single-cell analysis of the BTB formed during the process of BM development will reveal more unique characteristics of the NVU and produce new treatment strategies.
Drugs cross the BBB/BTB through different mechanisms, which mainly include paracellular and transcellular pathways; AMT, RMT, CMT, and efflux transporters.
52
The dysregulation of transcellular movement involving heterogeneous changes has been observed in BTB transport pathways. Disruption of BBB tight junctions is most common in the BTB endothelium. The tumor vasculature becomes progressively heterogeneous as BCBM progresses. Both preclinical and clinical studies have shown the heterogeneity of BTBs. The permeability of the BTB is affected by its structural changes. BTB permeability was found to be significantly variable in more than 2000 BCBM cases.
53
Gril
Exosomes and BCBM
Exosomes are small extracellular vesicles (30–150 nm in diameter) secreted by many different cell types, including BC cells. Exosomes are composed of a lipid bilayer and carry various functional molecules, such as lncRNAs, miRNAs, and proteins, which mediate complex cell–cell communication. 64 Intercellular communication is a key feature of tumor development and metastasis. Tumor cells are known to release more exosomes than normal cells, and tumor-derived exosomes have shown a profound ability to change microenvironments. 65 The unique characteristics of exosomes, such as their nanosize, ability to transport cargo to various cells, high biocompatibility and affinity, and ability to cross the BBB, make them suitable for a wide range of applications, such as anticancer therapy and drug delivery systems.66–68 Exosomes are important in the bilateral communication between tumors and nonmalignant cells in BC. 69 Exosomal miRNAs and lncRNAs have been shown to affect the transcriptome of receptor cells and promote BC metastasis. 70 They can be used as biomarkers for tumor prognosis and diagnosis and for targeted therapeutics. Exosomes are involved in the entire BCBM process, including disruption of the BBB and establishment of a brain metastatic microenvironment.
Chen
CEMIP (a Wnt-related protein) was found to be abundant in BC-derived exosomes and to promote BM. A proinflammatory vascular niche was induced by BM-promoting cytokines and chemokines in secreted CEMIP-carrying exosomes. CEMIP was identified in brain tissue-derived exosomes from BCBM patients. CEMIP overexpression in primary and metastatic tumors has been linked to BM progression and poor prognosis. These findings indicate that targeting exosomal CEMIP may be a promising approach for BM treatment.
74
The highly dynamic feature of the cytoskeleton in cancer cells during metastasis makes cytoskeletal proteins attractive targets for cancer treatment. Fazakas
Camacho
Watabe
MiRNAs and lncRNAs in BCBM
MiRNA
As noted above, exosomal noncoding RNAs (ncRNAs) may be crucial markers for diagnosis, prognosis, and therapy prediction. The most studied exosomal ncRNAs to date are miRNAs and lncRNAs. MiRNAs are small ncRNA molecules that negatively modulate gene expression by inhibiting target mRNA translation or degradation. Almost 50,000 papers on miRNA and cancer have been published to date. As upstream of oncogenes and tumor suppressor genes, miRNAs are involved in the regulation of all steps of BCBM formation, from EMT to invasion, intravasation, extravasation, and colonization in the brain. In the past decade, BMs have attracted much attention with the discovery of miRNAs inducing tumor metastasis. MiRNAs are thought to be promising therapeutic targets for BCBM 86 (Figure 3).

The role of miRNAs and lncRNAs in the formation of BCBM. For example, (a) miR-1258 decreased MMP9, cyclooxygenase-2 and EFGR by inhibiting heparanase, resulting in decreased permeability; (b) downregulation of miR-202-3p increased MMP-1 and disrupted tight junctions; (c) downregulation of miR-509 increased RhoC and TNF-α, resulting in decreased permeability; (d) downregulation of XIST activated EMT and MET, resulting in stemness, evasion, and migration, increased miR-503 levels and induced microglial polarization to promote BCBM. (e) Lnc-BM interacts with JAK2 to activate STAT3, leading to the activation of ICAM1 and CCL2 and inducing CCL2-dependent macrophage recruitment to BCBM.
Many studies have shown that miRNAs are specifically implicated in the development of BCBM. For example, the level of miRNA-7 is significantly downregulated in BC cell-derived CSCs that metastasize to the brain. Specifically, Okuda
MiR-802-5p and miR-194-5p have been identified as promising markers for the early detection of BM, indicating that their detection in liquid biopsy samples may be a novel approach for predicting the risk of BCBM.
91
MiR-211 was found to be a prognostic marker of BM both in TNBC mice and patients.
92
High levels of miR-211 promote tumor cell colonization in the brain by increasing BBB permeability and tumor cell stemness. MiR-211 drives brain colonization
Wyss
Although these pioneering studies suggest important roles for miRNAs in BCBM, there are still gaps in our understanding. Further research is needed to clarify the roles played by miRNAs to establish them as early biomarkers and potential targets in BCBM therapy. 95
LncRNA
LncRNAs, which are longer than 200 nucleotides in length, were discovered relatively recently. Extensive research has shown that lncRNAs are key regulators of cancer cell signaling pathways. Cancer cell invasion of tissues is the first step of tumor metastasis. Many lncRNAs have been identified as modulators of invasion and EMT; they regulate EMT
Molecular genetic of BCBM
The biological mechanisms underlying BM of HER2-positive and TNBC are largely unknown. The lncRNA XIST has been shown to be significantly downregulated in BM; XIST expression was lower in 48% of TNBC and 28% of HER2-positive BC samples compared with 19% of luminal A BC subtype samples. This finding suggests that EMT was suppressed and that the metastasis rate was reduced by XIST expression. 84 Both EGFR and HER-2 receptors are members of the ErbB family. In BCBM patients, EGFR mutations and overexpression of HER2/HER3 are common. 98 The HER2/HER3 association appears to be particularly important in BCBM, as it may drive BM by releasing MMPs that can damage the BBB. 99
HER2 can heterodimerize with TrkB and be activated by BDNF, implying that paracrine signaling promotes the survival of HER2-positive BCBM.
100
In BCBM, HER2 and EGFR can influence cell proliferation by modulating DNA topoisomerase I
The unique molecular features of HER2-positive and TNBC tumor cells contribute to BM. Changes in the EGFR and HER2 signaling pathways are more frequent in HER2-positive and TNBC patients, and these changes have been related to a higher BM risk as well as a poorer prognosis. 12 In these two BC subtypes, XIST downregulation is more prevalent, promoting EMT and BC cell migration and specifically increasing the proclivity of circulating tumor cells to invade the brain. 84 Extravasation is more frequent in HER2-positive tumors because cells of this BC subtype bind to β4 integrin, allowing them to cross the BBB. Among BC subtype cells, TNBC cells exhibit the most CSC-like features. TNBC cells frequently overexpress genes involved in the stem cell pluripotency pathway, such as SOX2. A high level of SOX2 expression is related to a shorter survival time for patients with BCBM. In addition, the PI3k/Akt/mTOR pathway and the TP53 gene have been associated with increased BM in HER2-positive and TNBC patients. The impact of BC molecular subtypes, such as HER2-positive BC and TNBC, on BCBM development and prognosis is undeniable; however, many questions remain unanswered. Single-cell RNA sequencing (scRNA-seq) allows a better understanding of cell heterogeneity in BC and has great potential for clarifying the complex mechanisms of BM, potentially leading to new strategies for individualized treatment. 105
BM has been reported to involve a significant genetic component, with the expression of related genes either upregulated or downregulated. Many genes with increased expression and associated with BM have been identified. For example, HBEGF, COX2, and ST6GALNAC5 are mediators of cancer cell penetration of the BBB and regulation of the formation of BCBM.
106
The drivers of primary BC development, such as TP53, MLH1, PIK3CA, and KIT, are frequently mutated in BCBM.
107
Lee
The activation of metastatic genes or the silencing of metastatic suppressor genes frequently leads to progression to BM. Mutation of metastatic genes such as MMP2 and CXCR4 may lead to abnormal protein products, which can promote tumor cell metastasis to distal organs. The expression of the COX-2 gene, which controls tumor invasion, extravasation, and metastasis, was downregulated in metastatic tissues. The RRM2, MMP1, FOXM1, BRCA1, and E-cadherin genes have also been implicated in BCBM.
109
The gene expression of nestin, prominin-1, CK5, and aSMA in BC has also been linked to BM. The patient survival rate after the development of BM has been reported to be as low as 2 months.
110
Villodre
Cancer-related genes are typically classified as proto-oncogenes or tumor suppressor genes. Gene mutations at the nucleotide, transcriptional, or epigenetic level can cause abnormal or inhibited gene expression. CRISPR/Cas9 can be used to generate targeted mutations at every level. The CRISPR/Cas9 editing system is a powerful genome-altering tool that has shown great promise for use in cancer therapy, including BCBM. It is widely used to suppress the expression of various genes and repair mutations in genetic disorders.109,112 CRISPR/Cas9 can be used to knockout oncogenes and thus attenuate cancer development and metastasis. Scientists are using liposome-templated hydrogel nanoparticles that can penetrate the BBB to deliver CRISPR/Cas9
Novel therapeutic strategies for BCBM
To date, the preferred treatment option for BCBM treatment is based on localized strategies, such as surgery and/or radiotherapy. Treating BCBM with the same systemic therapy used to treat primary BC has been less effective in the past decade. However, the rapid development of anti-HER2 drugs has given new hope to BCBM patients.
117
Scientists have developed many anti-HER2 drugs, such as tyrosine kinase inhibitors, monoclonal antibodies, and antibody–drug conjugates (ADCs). Lin
Although the results of early clinical studies on therapies targeting BTB permeability-related pathways were disappointing, promising results have been obtained in recent clinical trials. Notably, in a recent clinical trial, tucatinib, a small-molecule HER2 kinase inhibitor, was the first targeted agent discovered to enter the brain. 122 In the phase II HER2CLIMB trial, trastuzumab and capecitabine plus tucatinib were administered to treat HER2-positive metastatic BC (48% of the patients had BCBM). The median progression-free survival (PFS) was 2.2 months longer with a median OS duration increased by 4.5 months in the Tucatinib treatment group compared with the placebo group. 123 Tucatinib increased the median intracranial PFS duration by 4.7 months and the median OS duration by 6.1 months in 291 BCBM patients. Tucatinib also increased the median PFS by 5.0 months and the median OS duration by 5.3 months in 66 BCBM patients who chose delayed radiotherapy. 118 The role played by the BBB and BTB in this therapy, however, remains unknown. The mechanisms of drug entry into brain tumors are poorly understood, and additional well-designed preclinical and clinical imaging studies are needed.
As discussed above, the BBB and BTB are impediments to therapeutic intervention. Therefore, additional new BBB disruption techniques are needed to improve the permeability of drugs to metastatic tumors in the brain. Invasive and noninvasive approaches for improving antitumor agent delivery across the physical, cellular, and molecular barriers of the BBB and BTB are being developed as discussed in this review.
Given the poor prognosis of BCBM in patients receiving currently available treatments, the development of novel and more effective therapies is urgently needed. Notably, no FDA-approved treatments are specifically available for the treatment of BCBM. 17 Surgery and radiotherapy are the conventional cancer therapies applied for BCBM patients. Systemic therapy, such as chemotherapy and targeted therapy, is a challenging to administer in BCBM treatment because of the BBB. To address the challenges associated with BCBM, basic and clinical research has been directed to developing novel drug targeting and delivery approaches to enhance drug efficacy in the brain. 50
Focused ultrasound
Focused ultrasound (FUS) is a special noninvasive approach for safely and reversibly opening the BBB/BTB and increasing the accumulation of antitumor agents in metastatic brain tumors. FUS can be used to specifically target brain areas and damage the BBB/BTB.
56
Low-intensity FUS can cause microbubbles to oscillate, which increases vessel pressure, expands tight junctions, and increases membrane permeability.
124
The degree of BBB/BTB opening is proportional to the degree of drug permeation. Numerous preclinical studies have shown that FUS can be used to increase the amounts of several systemically administered drugs that penetrate the BBB/BTB and are thus delivered to brain tumors by more than fourfold.
125
Applying FUS in a preclinical model, Wu

Novel approaches to overcome the BBB/BTB. (a) FUS; (b) (MTH) for BBB transport; (c) nanocarriers. The details of the techniques are shown in the text.
Molecular Trojan horse
The MTH approach depends on monoclonal antibodies or engineered peptides bound to endogenous receptors for RMT delivery of biologics through the BBB. 52 Most chemotherapeutic and targeted drugs are large molecules and cannot penetrate the BBB in adequate therapeutic quantities. By attaching these drugs to MTHs, the so-called bifunctional molecules are formed, which can promote antitumor drug penetration of the BBB by binding to receptors on endothelial cells. 128 Different classes of high-molecular-weight biologics have been fused with different MTHs and delivered into the brain. 52 For example, antitumor agents fused to IgG monoclonal antibodies can cross the BBB mediated by a BBB receptor, such as the transferrin receptor (TfR); in this case, an anti-TfR monoclonal antibody is the MTH, and the antitumor agent-anti-TfR fusion molecule can be delivered into the brain. 129
Schackert
Antibody–drug conjugates
Trastuzumab emtansine (T-DM1) is a HER2-targeting ADC that consists of the cytotoxic agent emtansine (DM1) conjugated to trastuzumab that delivers DM1 to cancer cells, leading to inhibited tubulin polymerization and cell death.
117
Trastuzumab deruxtecan (T-DXd) was the second ADC to be approved for use in metastatic BC patients who had at least two types of anti-HER2 treatment.
135
Endocytosis of a biparatopic anti-HER2 ADC into endothelial cells
Nanomedicines
The enhanced permeation and retention effect is the main hypothesized benefit of using nanocarriers for cancer therapy.
136
Cancer therapy has been remarkably improved over the past few years because of the development of different antitumor agents at the nanoscale.
137
More recently, biomaterial-based nanodrugs have been developed and applied in preclinical studies and clinical trials as cancer immunotherapies.
138
Nanoparticles used in targeted drug delivery systems exhibit unique advantages, such as reliable delivery, sufficient loading capacity, nanosized, and drug-loading plasticity.
137
Nanodrugs have increased the efficacy and reduced the toxicity of parent drugs in clinical trials of BC treatment.
139
Nanoparticle-mediated targeted delivery of chemotherapeutic drugs to BM is currently among the most frequently used approaches. Different nanoparticles, such as polymeric nanoparticles, liposomes, and micelles, have been investigated for their potential as drug delivery systems because they can cross the BBB.
16
These new drug delivery methods can be used to gain spatial and temporal control over BCBM treatment. Several nanoparticle-based strategies have been evaluated for their ability to enhance drug transport across the BBB.
47
However, studies have shown very low levels of nanoparticle accumulation in the brain. Although various promising nanoparticles are continuously being developed, the rules for designing BBB-/BTB-penetrable nanoparticles must first be established.
140
Li
Nanoparticles can reestablish the effectiveness of conventional chemotherapy by improving permeation and retention, thereby reducing side effects, extending treatment windows, and shortening drug delivery intervals. However, the translation of nanomedicines has not been as successful as originally anticipated. Moreover, although clinical benefits have been demonstrated, the number of nanomedicines available for BCBM treatment remains limited. 146
Immunotherapies
Various lines of evidence have shown that tumor cells hijack immune regulatory mechanisms in the brain to perpetrate their survival and development. 147 The development of immunotherapy with immune checkpoint inhibitors and targeted therapy has greatly improved the prognosis of various cancers. Immunotherapy is one of the most promising advances in BM therapy because it stimulates the immune system to specifically attack cancer cells. Although the brain was previously considered an immune-privileged organ, BM is now thought to be a potential target of various immunotherapeutic drugs. 148 Immunotherapies have shown promise in preclinical research and produced significant clinical outcomes in BM patients. Checkpoint inhibitors are the most frequently studied immunotherapeutic agents for BM. Although checkpoint immunotherapy has been shown to be less effective in BM, some novel targeted therapies have been approved, and they have increased the median 5-year survival from 18.4% to 32.6%. 149
As previously stated,44,45 BM occurs in an environment with fewer immune cells than in primary BC tissues; therefore, a high TIL content in the BCBM is associated with better outcomes. Changing the brain immune microenvironment may be a novel strategy for treating BCBM. 150
T-cell immunotherapy with chimeric antigen receptors (CARs) is being studied as a treatment for solid tumors and metastatic brain tumors.151,152 Priceman
Conclusions
For the past decade, effective treatment has significantly increased the life expectancy of BC patients. However, the incidence of BM has been steadily increasing as well. Unfortunately, despite improved systemic treatments, the prognosis for BCBM patients remains poor. Understanding the molecular characteristics of BCBM, as well as the development of new and more effective therapies, is urgently needed. In this review, we focused on the latest advances in understanding the molecular mechanism of BCBM and novel therapeutic strategies.
BC is molecularly heterogeneous and can be classified into several subtypes. The TNBC and HER2-positive subtypes are undeniably linked to an increased risk of BM and a worsened prognosis. To date, local management, such as surgery or radiotherapy, has been the preferred treatment option for BM. Developing effective drug delivery and systemic therapies for BCBM remains pharmacological challenges due to the highly selective permeability and protective function of the BBB. Various approaches for overcoming BBB/BTB limitations have been investigated, including FUS, nanodrugs, and MTHs, to improve drug delivery and distribution into the brain. The brain microenvironment and region-specific genomic alterations in the brain are emerging as potential targets for BCBM therapy. Furthermore, the application of novel genome-altering approaches as well as new engineering strategies such as CRISPR/Cas9 editing has resulted in an unprecedented shift in our understanding of the heterogeneity of BCBM and has provided novel insight into more effective therapeutics. New immunotherapy-based treatment strategies, such as immune checkpoint inhibitors, targeted therapies, and nanomedicines that enhance BBB penetration, may improve efficacy while limiting side effects. Furthermore, ongoing trials and prospective treatments are expected to improve outcomes for BCBM patients. Understanding the mechanism of BCBM will help us to develop additional more effective antimetastatic strategies. However, more research is needed to validate the identified gene and molecular mechanism targets in potential clinical applications. For example, CRISPR screening could be used to further link genetic mutations with phenotypic changes and ATAC-seq could be used to detect epigenetic changes in BCBM. 155 The discovery of the molecular mechanisms underlying BCBM has opened up new avenues for future research aimed at the development of novel targeted therapies. Hopefully, ongoing studies and novel research approaches will be beneficial in addressing current challenges and improving treatment strategies for BCBM patients.
Footnotes
Acknowledgements
None.
