Abstract
Background:
Human epidermal growth factor receptor-4 (HER4) and yes-associated protein-1 (YAP) are candidate therapeutic targets in oncology. YAP’s transcriptional coactivation function is modulated by the HER4 intracellular domain (HER4-ICD)
Methods:
We performed immuno-phenotypic profiling of pathway markers in a consecutive breast cancer series with 25 years of clinical follow up (
Results:
Membrane localisation of phospho-HER4 [pHER4(Y1162)] was infrequent in primary breast cancer, but very frequent in brain metastases (5.9%
Discussion:
Our findings suggest that the canonical-mechanism where Hippo pathway-mediated phosphorylation of YAP ostensibly excludes it from the nucleus is dysfunctional in breast cancer. The data are consistent with pYAP(S127) having independent transcriptional functions, which may include transducing neuregulin signals in brain metastases. Consistent with mechanistic studies implicating it as an ER co-factor, nuclear pYAP(S127) associations with breast cancer clinical outcomes were dependent on ER status.
Conclusion:
Preclinical studies investigating HER4 and nuclear YAP combination therapy strategies are warranted.
Introduction
Relapse of breast cancer in the brain leads to significant morbidity and premature death, generally within 2years of diagnosis. 1 Epidemiological data indicate that symptomatic brain metastases are diagnosed in approximately 15% of metastatic breast cancer patients, 2 but develop in as many as 40% with disseminated disease.1,3–5 Patients with HER2-positive (HER2+) and triple-negative breast cancer (TNBC) are more likely to develop brain metastases than those with oestrogen receptor-positive (ER+) breast cancer,6,7 indicating there are subtype-specific determinants of metastasis to the brain. Treatment can include surgical resection, focused or whole-brain radiotherapy and chemotherapy. 8 These interventions can improve quality-of-life and overall life expectancy but are rarely curative. Together with population ageing, better systemic cancer control and prolonged patient survival are likely to further elevate the incidence of brain-metastatic breast cancer in the future.9,10
There is a shortage of molecular-targeted therapeutics for effective treatment of brain metastases. Resolving this requires a deeper understanding of their vulnerabilities. One approach is to identify molecular features that set them apart from their parent primary cancers, which potentially represent brain-specific adaptations required for outgrowth. Another is to identify targetable molecular alterations that are frequent in brain metastases regardless of their status in the primary tumour, which may be more broadly applicable and amenable to drug repurposing. Analysis of patient-matched primary and metastatic tumours has been applied to identify candidates in both categories.11–14 For example, we and others showed that human epidermal growth factor receptors (HER) are pivotal to brain metastasis pathogenesis; particularly HER2 and HER3.11,13,15,16 These receptors promote tumour progression through ligand-dependent activation. Abundant in the brain, 17 neuregulin growth factors bind to HER3, resulting in dimerization, cross-phosphorylation and recruitment of relay proteins that bring about oncogenic cellular changes, notably including potent activation of PI3K.18,19 HER3 induction is one of many adaptations that occur as metastatic cells opportunistically exploit the brain microenvironment.20–23
HER4 is also responsive to neuregulins, can dimerise with other HERs and is frequently activated in brain metastases.16,24 There are currently no specific inhibitors of HER4, but several agents with pan-HER activity are under clinical development, including neratinib, afatinib, dacomitinib and poziotinib. Clinical evidence that HER4 activity could be an important mediator of brain metastasis came from the randomised phase III NALA trial, which compared lapatinib (standard-of-care inhibitor of EGFR and HER2) with neratinib (inhibitor of EGFR, HER2 and HER4), both in combination with capecitabine, for treatment of HER2+ metastatic breast cancer. 25 The neratinib regimen doubled 12-month progression-free survival (PFS) and significantly delayed the time to intervention for symptomatic intracranial disease. Based on these findings, the United States Food and Drug Administration (FDA) approved neratinib for patients with treatment-refractory, metastatic HER2+ breast cancer. 26 The potential for delaying intracranial disease in clinically HER2-negative breast cancer has not yet been established.
Apart from canonical receptor tyrosine kinase (RTK) activity, ligand-activated HER4 also undergoes juxta- and intra-membrane proteolysis, releasing a C-terminal intracellular domain (HER4-ICD) that can translocate to the nucleus. In the mammary gland, HER4-ICD transcriptional coactivation of STAT5A is essential for normal lobuloalveolar development, epithelial differentiation and milk production.27,28 In breast cancer, it is reportedly an ER cofactor associated with poor prognosis in ER+ disease,
29
but results have been mixed, with a literature meta-analysis finding no overall relationship with survival.
30
A direct interaction between HER4-ICD and the oncogenic transcription coactivator, YAP (yes-associated protein), has been identified in breast tumour cell line nuclei: HER4-ICD directly induced YAP-regulated genes and pro-metastatic cell behaviour
Recently implicated in brain metastatic outgrowth in a mouse model of metastatic lung cancer as well as in human lung-brain metastasis,33,34 YAP is a transcriptional coactivator of the TEAD family of transcription factors. Collectively they regulate proliferation, cell fate and survival, with a large body of evidence now pointing to a role in microenvironment recognition.35,36 YAP-TEAD complexes are opposed by the Salvador-Warts-Hippo (‘Hippo’) pathway, which serves the evolutionarily conserved purpose of maintaining equilibrium according to tissue growth and cellular density cues. Hippo pathway kinases LATS1/2 phosphorylate YAP on five conserved HXRXXS motifs, including one at serine-127 [pYAP(S127)] that mechanistic studies indicate is necessary for 14-3-3 binding and cytosolic retention, ultimately leading to proteolysis. 37 YAP and its close paralog TAZ are dysregulated in a variety of cancers, with disconnection from Hippo pathway control critical for bypassing normal growth constraints.36,38 Secondary onco-protective actions of several agents used to treat congestive heart failure and inflammation may be partly mediated by impairing nuclear translocation of YAP (e.g. dasatinib, statins, pazopanib and several others directly modulate YAP’s interaction with its primary TEAD transcription factors [e.g. digitoxin and the COX-inhibitor, flufenamic acid)].
Functional and histopathology studies mostly concur that YAP expression is oncogenic, and that its activity in the nucleus plays a major role in mediating resistance to a number of different molecular-targeted agents.39–43 But paradoxically, YAP expression is reportedly a favourable prognostic indicator in oestrogen receptor-positive (ER+) breast cancers.41,44 Also still to be resolved is the observation that the phosphorylated YAP isoform typically associated with cytosolic retention [pYAP(S127)] has been detected in tumour cell nuclei of both primary and metastatic breast tumours, including brain metastases.40,45
The aim of this study was therefore to analyse the expression and phosphorylation status of HER4 and YAP in human breast cancer samples, with a view to developing a better understanding of their potential as therapeutic targets in early and/or metastatic breast cancer. From a biological point of view, we also wanted to explore the idea that expression and activation of HER4 are linked to YAP activation in brain metastases.
Methods
This study involved immunohistochemistry (IHC) analysis of: (1) a clinically annotated, consecutive series of primary breast tumours with >20 years follow up, herein referred to as the ‘general breast cancer cohort’ (described previously)46–52; and (2) patient-matched pairs of primary and brain-metastatic breast tumours, herein referred to as the ‘brain-metastatic breast cancer cohort’ (br.MBC) (Table 1). Both were sourced from the same diagnostic pathology centres in Queensland, Australia, and represent a similar patient population. Ethical approval for this study was obtained from human research ethics committees of the Royal Brisbane and Women’s Hospital (RBWH; 2005000785) and The University of Queensland (HREC/2005/022). Written consent to use tissue specimens and de-identified clinical data for research purposes is not a requirement under these approvals because the samples were sourced from pathology archives of the 1980s, and most patients are now deceased. Samples were de-identified for all the analyses performed in this study.
Cohort characteristics.

Of informative TMA samples this represents 30 matched pairs.
BC, breast cancer; BrM, brain metastases; br.MBC, brain metastatic breast cancers; ER, oestrogen receptor; HER, human epidermal growth factor receptor; NA, not applicable.
Tumours were sampled in tissue microarrays (TMAs) for IHC analysis of proteins in the HER4-YAP pathway, and other biomarkers of interest (Table 2). Maximum scores of duplicate cores were used for analysis.
Biomarkers analysed by IHC.
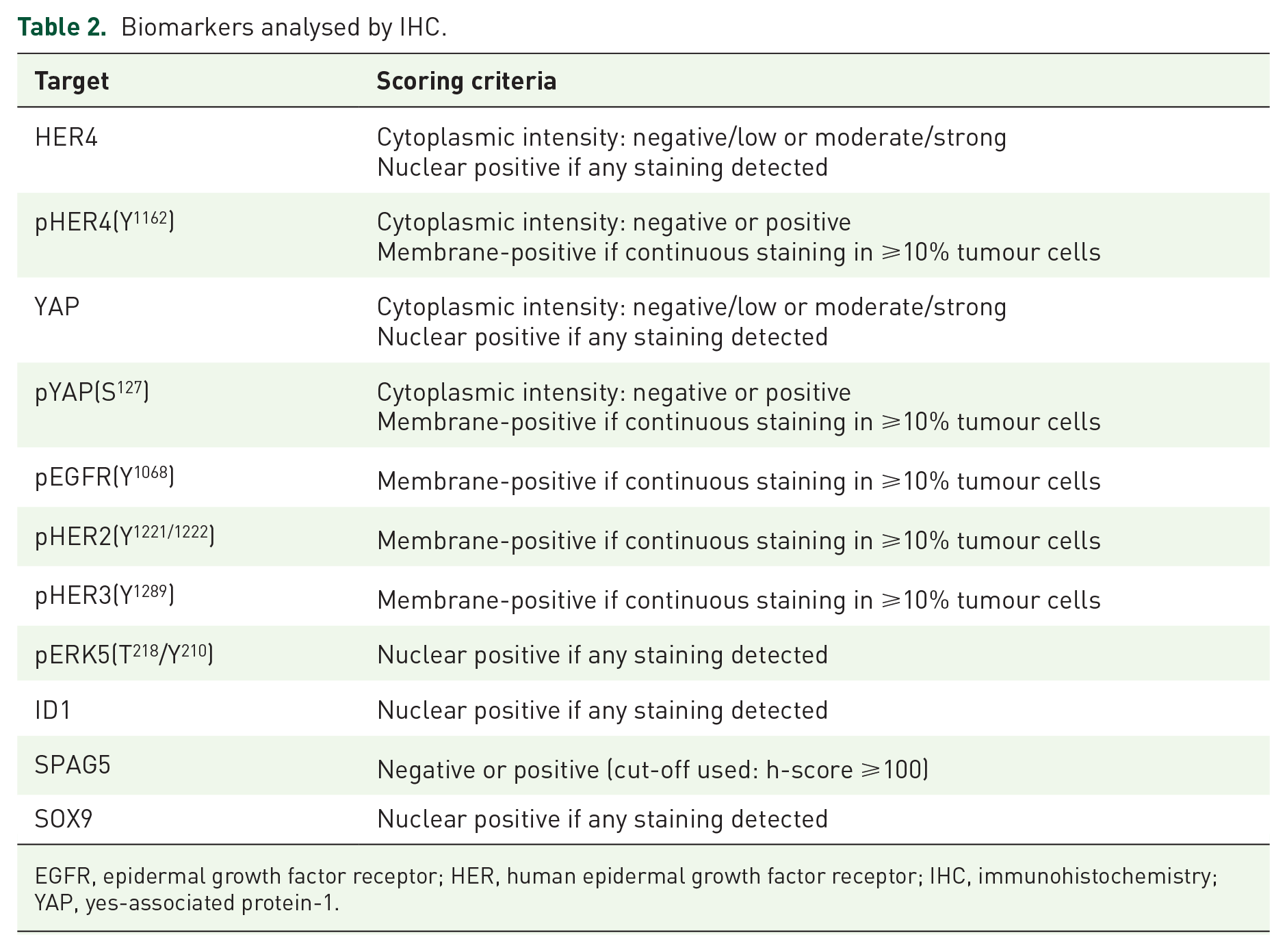
EGFR, epidermal growth factor receptor; HER, human epidermal growth factor receptor; IHC, immunohistochemistry; YAP, yes-associated protein-1.
Tumour tissue cohorts, histopathology and clinico-pathologic characterisation
A breast cancer-brain metastasis resource cohort was assembled from formalin-fixed paraffin-embedded (FFPE) primary breast cancer and patient-matched BrM samples of patients who had treatment in Queensland, Australia, between 2000 and 2018. Cases were identified on the basis of BrM tissue availability, then filtered based on availability of corresponding primary breast tumour tissue. Clinical diagnostic information and survival data were obtained from Pathology Queensland, Queensland Health and the Queensland Cancer Registry. Tumours were sampled in TMAs for IHC analysis (1 mm cores), with hematoxylin and eosin staining used to locate the tumour component of surgical specimens. A comprehensive histopathological review of all cases was conducted by experienced molecular pathologists (SRL, JMS). Clinico-pathologic information (e.g. histological type, grade and stage) and survival data were assembled in a database linked by the TMA position of each case. We assessed clinical biomarkers by IHC (see in the following) according to diagnostic reporting standards, as described previously 46 : (i) oestrogen and progesterone receptors (hormone receptors, HR); (ii) human epidermal growth factor receptor 2 (HER2).
Immunohistochemistry
IHC antibodies and staining conditions are detailed in Table 3. Primary antibodies were selected on basis of prior use in published reports (preferably for IHC applications) and a manufacturer specificity guarantee. Before applying the antibodies to test sample cohorts, we also validated their specificity experimentally, first by confirming substantially reduced IHC signal in cell lines depleted of their targets by siRNA (section
Antibodies and conditions used for IHC analysis.
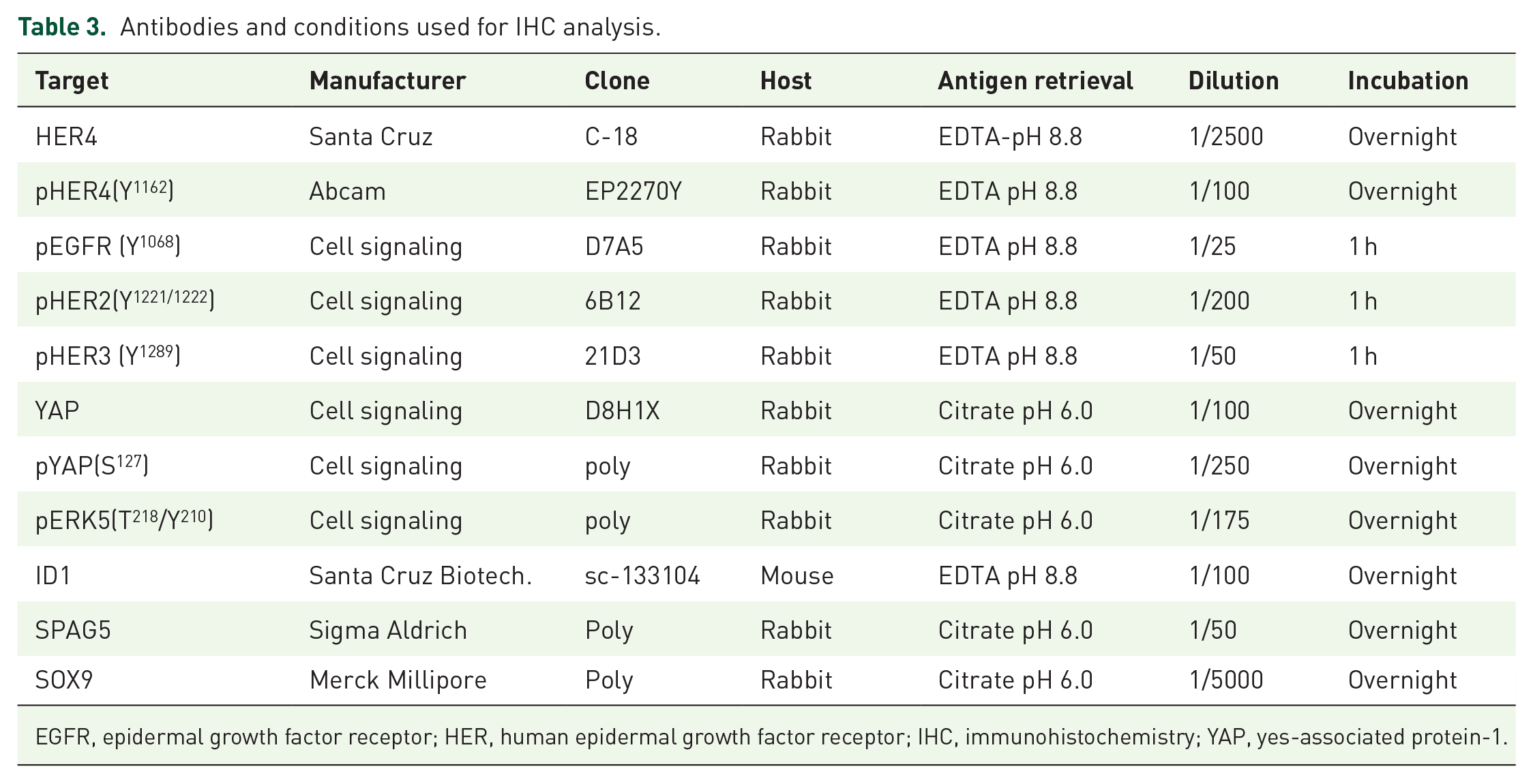
EGFR, epidermal growth factor receptor; HER, human epidermal growth factor receptor; IHC, immunohistochemistry; YAP, yes-associated protein-1.
IHC was performed on 4 µm FFPE TMA sections using the Mach1 Universal HRP-Polymer Detection Kit (BioCare Medical, Pacheco, CA, USA). Briefly, sections were deparaffinized with xylene and hydrated in a graded ethanol series (95–70%). Heat-induced antigen retrieval was performed using a decloaking chamber (BioCare Medical) with sodium citrate buffer (0.01 M, pH 6.0) for 5 min at 125°C, or EDTA buffer (0.001 M, pH 8.8) for 30 min at 95°C. Antigen retrieval was performed using α-chymotrypsin (Sigma-Aldrich, St. Louis, MO, USA) for 10 min at 37°C. Sections were rinsed with tris-buffered saline (TBS), then treated with 0.3% hydrogen peroxide for 10 min. Non-specific staining was blocked with MACH1 Sniper blocking reagent (BioCare Medical). Primary antibody in TBS was applied to the slide for 1 h at room temperature or overnight at 4°C in a humidified slide chamber. For rabbit primary antibodies, MACH1 anti-rabbit secondary antibody conjugated to horseradish peroxidase was applied for 30 min at room temperature. Diaminobenzidine chromogen substrate was applied for 1–5 min. Lastly, slides were counterstained with hematoxylin for 30 s and cover-slipped with DPX mountant (Sigma-Aldrich). For analysis, stained slides were scanned at 40× magnification using an Aperio AT Turbo (Leica Biosystems, Wetzlar, Germany).
IHC scoring and analysis
High resolution digital IHC light microscopic images were used to assess IHC staining. Scoring of the tumour compartment of TMA cores was performed by at least two independent observers, according to criteria determined during a combined preliminary review. Individual TMA core images were linked to a database containing other clinicopathologic data by a TMA position code, then reviewed in a blinded manner. Statistical testing was done using GraphPad PRISM Software (v8.2; GraphPad Software, San Diego, CA, USA), with specific tests indicated accordingly in the main text and figure legends.
siRNA-mediated depletion of IHC antibody targets and antibody validation studies
Cell lines (T47D and MDA-MB-468) were purchased from the American Type Culture Collection (Manassas, VA, USA), maintained in recommended culture conditions and regularly screened for Mycoplasma. For transient knockdown studies, cells were transfected with 100 nM siRNAs comprising of a mixture of three different sequences for each gene (Gene Pharma, Shanghai, China) using FugeneHD (Promega, Madison, WI, USA): YAP-homo-1858, CUGCCACCAAGCUAGAUAATT; YAP-homo-1517, GACGACCAAUAGCUCAGAUTT; YAP-homo-862, GCAUCUUCGACAGUCUUCUTT; ERBB4-homo-1815, GGUCCUGACAACUGUACAATT; ERBB4-homo-2474, CCAGCUGGUUACUCAACUUTT; ERBB4-homo-3213, ACUGAGCUCUCUCUCUGACTT; negative control, UUCUCCGAACGUGUCACGUTT. After 24 h, cells were fixed in 10% neutral buffered formalin (Sigma) and paraffin embedded. Cell pellet sections (6 μm) were heat-retrieved in antigen retrieval buffers as indicated in Table 3 using a Decloaking Chamber in Background Sniper blocking solution (Biocare Medical). Slides were incubated with primary antibodies as indicated in Table 3. Detection was performed using the Mach1 kit (Biocare Medical). Slides were then scanned and imaged on an Aperio AT Turbo slide scanner (Leica Biosystems).
Datasets and statistics
The following datasets were analysed in this study: TCGA breast cancer RNASeq (V2 RSEM) dataset (mRNA expression
Results
Expression and activation of HER4 in major breast cancer subtypes
We used a HER4 antibody that recognises a C-terminal epitope in all known isoforms to perform IHC analysis in the general breast cancer series. This antibody was validated for specificity using siRNA mediated gene knockdown (Supplemental Figure S1A). In line with published data,57–59 we considered nuclear staining to represent nuclear HER4-ICD, and cytoplasmic staining as a mixture of isoforms, including mitochondrial HER4-ICD and a range of trafficking intermediates. IHC analysis revealed expression of HER4 in tumour cell nuclei as well as cytoplasm (Figure 1A) and approximately half of all cases were positive (Supplemental Table S1). There was a direct relationship between nuclear and cytoplasmic expression, particularly in TNBC (Figure 1B;

Expression and activation of HER4 in early breast cancer. (A) Representative breast tumour cores stained for HER4. (B) Chi-square analysis of the proportions of nuclear and cytoplasmic HER4 staining in major breast cancer subtypes. (C) Chi-square analysis of the proportions of cases exhibiting compartment specific HER4 staining. (D) KM analysis of the relationships between HER4 compartment categories (blue and green) and BCSS. Pie charts indicate proportion of cohort in each category. (E) Representative tumour cores stained for pHER4(Y1162). (F) Chi-square analysis of the proportions of cases with pHER4(M+) across molecular subtypes (i) and proliferative status of HER2+ cases (ii). (G) Kaplan–Meier analysis of relationships between pHER4(M) staining and BCSS.
Phospho-HER4 (pHER4) functions at the plasma membrane and has separate functions to HER4-ICD. We performed IHC analysis of the general breast cancer cohort using an antibody against phospho-tyrosine 1162 [pHER4(Y1162)]. Tumour cell membrane staining was detected in 5.9% of cases (
HER4 is induced and activated in breast cancer brain metastases
We next analysed HER4 and pHER4 in patient-matched primary breast cancers and brain metastases. Compared with the general breast cancer cohort, where the incidence of brain metastases is estimated to be 5–10%,2,60 there were no significant differences in HER4 or pHER4 in brain metastatic breast cancers (Figure 2A; BC

Expression and activation of HER4 in brain-metastatic breast cancers and brain metastases. (A) Chi-square analysis of cyto.HER4, nu.4ICD and m.pHER4 in major breast cancer subtypes. *
Nuclear pYAP(S127) is an ER-dependent prognostic indicator in breast cancer
Previous studies have demonstrated that nucleo-cytoplasmic shuttling and phosphorylation are important modulators of YAP’s transcriptional activity,35,36,61 but the clinical relevance in breast cancer has not been clearly established. We analysed the expression of YAP and pYAP(S127) in the general breast cancer cohort using validated IHC antibodies (Supplemental Figure S1B). Staining was homogeneous in breast tumour cell cytoplasm and/or nuclei (Figure 3A), and neither YAP nor pYAP(S127) was associated with histological type, grade, HER2 or ER status (Supplemental Tables S2 and S3). There was a direct association between nuclear and cytoplasmic YAP (Figure 3Bi;

Expression and activation of YAP in early breast cancer, and relationship with patient survival.
We explored the clinical significance of pYAP(S127) being localised to tumour cell cytoplasm or nuclei by performing Kaplan–Meier analysis. Cytoplasmic pYAP(S127) was generally protective in this cohort, but phosphorylation of the nuclear pool stratified survival oppositely depending on ER status (Figure 3C). Strikingly, nuclear pYAP(S127) was a favourable prognostic indicator in ER+ cases but marked poor prognosis in TNBC (Figure 3C). In the ER+ cases (Figure 3Ciii) the hazard-ratio associated with nuclear pYAP(S127) was 0.53 (log-rank test; 95% CI: 0.35–0.81;
Others have reported that
Nuclear pYAP(S127) has ER-dependent relationships with breast tumour size, ERK5 and HER4
Since YAP promotes the phosphorylation and nuclear translocation of ERK5 in myogenesis,
63
we performed IHC analysis of nuclear pERK5 as candidate marker of YAP activity in breast cancer. There were no marked changes in the frequency of unphosphorylated YAP expression according to nuclear pERK5 status, but pYAP(S127), particularly the nuclear pool, was directly associated with nuclear pERK5 (Figure 4A). The working model of YAP function denotes that LATS-mediated phosphorylation at serine-127

Clinico-pathologic correlates of cytoplasmic- or nuclear-localised pYAP(S127).
In normal tissues, hippo pathway-mediated phosphorylation and cytoplasmic sequestration of YAP limit tissue growth. We found associations between nuclear pYAP(S127) and breast tumour size that were consistent with survival data, with nuclear pYAP(S127) inversely associated with ER+ tumour size, but directly associated with ER-negative tumour size. Subcellular distribution analysis confirmed that not only is the phosphorylated isoform more frequent overall, but pYAP(S127) is preferentially localised to the nucleus in larger ER-negative tumours (Figure 4B).
As YAP activity is regulated by HER4-ICD
Taken together, these data suggest that ER is a key determinant of YAP function, that nuclear pYAP(S127) may be an active isoform in TNBC and that the previously identified transcriptional interaction with nuclear HER4-ICD32,66 could be relevant in human disease.
Shifts in YAP expression and phosphorylation during brain metastatic progression are dependent on ER status and linked to activation of HER4’s tyrosine kinase activity
Next, we compared YAP expression and phosphorylation in the general breast cancer cohort, brain-metastatic breast cancers and matched brain metastases, and found significant changes across this progression series. YAP phosphorylation was significantly less frequent in brain metastatic, ER+ breast cancers than unselected ER+ cases (Figure 5A). The frequency of pYAP(S127) increased slightly in matching brain metastases, typically in both nuclei and cytoplasm, but overall the most prominent phenotype was unphosphorylated nuclear YAP (Figure 5A). Conversely, in ER-negative disease, brain metastatic primary breast cancers had less cytoplasmic pYAP(S127), and the nuclear pYAP(S127) phenotype was unchanged. Comparison of brain metastases with matching ER-negative primary cancers indicated that YAP is more frequently expressed and phosphorylated overall (Figure 5A; top/middle panel). To account for the fact that pYAP(S127) is often present in cytoplasmic and nuclear compartments at the same time, we performed the analysis after categorising cases into subgroups with: (1) negative/low pYAP expression (light grey); (2) cytoplasm-predominant pYAP(S127) (cyto>>nucleus; orange); (3) nuclear-predominant pYAP(S127) (nucleus>>cyto; purple); or (4) high levels of pYAP(S127) in both compartments, (uniformly high; dark grey). We observed that the pYAP(S127) pool shifts to the nucleus during brain metastatic progression (Figure 5A).

Expression and phosphorylation of YAP in breast cancers and matching brain metastases.
Finally, we assessed the proportions of brain-metastatic breast cancers and matched brain metastases that could be amenable to therapeutics that target HER4’s RTK activity or reduce nuclear translocation of YAP. The proportion of cases exhibiting membrane-associated pHER4 or nuclear YAP/pYAP(S127) was similar for ER+ and -negative breast tumours (55–60%; Figure 5B). The proportion exhibiting both phenotypes (potentially amenable to combination therapy) was very high for ER-negative brain metastases (~70%; Figure 5B), due mostly to very frequent induction of pHER4 (Figure 2). In addition to highlighting possible therapeutic opportunities, these findings suggest that nuclear pYAP(S127) may be involved in transducing neuregulin signals in brain metastases, particularly those with an ER-negative phenotype.
Discussion
We previously found a high frequency of HER4 phosphorylation in brain metastases from various cancers. 16 Given that nuclear HER4-ICD activity has been linked to YAP signalling31,32 and YAP is an important player in metastatic breast cancer, 45 we undertook this study to investigate whether HER4 activation could be linked to YAP signalling in breast cancer brain metastases. We integrated subcellular localisation, phosphorylation and ER status in our analyses, and found that all three variables were essential to tease out specific clinicopathologic and biological correlates of HER4 and YAP, uncovering relationships that would not have been identified in analyses based on expression alone. For example, Cao and colleagues found that nuclear YAP is a favourable prognostic indicator in ER+ breast cancer, 41 while our approach clarified that the subgroup of ER+ cases with better outcome is specifically those where the nuclear YAP pool is phosphorylated.
The high frequency of nuclear pYAP(S127) in primary and metastatic breast tumours was somewhat surprising, since this isoform apparently represents Hippo pathway-mediated negative feedback once YAP is marked for sequestration in the cytoplasm. We found significant differences in the frequency of nuclear pYAP(S127) in good
Mechanistically, it remains to be determined why pYAP(S127) would be present in the nucleus, but this has been observed previously.40,45,72 It is possible that YAP is expressed at a level that overwhelms 14-3-3-mediated sequestration and/or ubiquitin-mediated proteolysis, particularly for metabolically active tumours that have a high endoplasmic reticulum stress burden. 73 Assuming that serine-127 phosphorylation marks negative feedback on YAP and that its presence in the nucleus is due to ‘overflow’, this implies that YAP inactivation is linked to less aggressive forms of ER+ breast cancer. TNBC aside, our findings in ER+ breast cancer (Figure 3C) are consistent with this, and with a recent report identifying YAP as a novel ERα co-factor required for oestrogen-mediated transcriptional regulation of the enhancer landscape in MCF7 cells. 74 Zhu and colleagues found that, in addition to its canonical Hippo pathway target elements, YAP co-occupies ERα-active enhancers, recruits enhancer activation machinery and may be critical for the modulation of genome architecture by ERα.
The identification of genome-wide enhancer reprogramming as a novel mechanism of oestrogen action adds to the rationale backing efforts to develop YAP inhibitors in molecular oncology.
36
But the fact that we found high levels of nuclear pYAP(S127) were related to large tumour size and poor prognosis in TNBC indicates that identifying appropriate patient populations for treatment will require consideration of context-specific activity. If nuclear pYAP(S127) is solely the pool in excess of sequestration capabilities, these findings would suggest that YAP activity is somehow protective in ER-negative tumours, which is at odds with a body of literature suggesting otherwise,38,75,76 and with the contrasting survival curves for cytoplasmic and nuclear pYAP(S127) pools in our TNBC cohort (Figure 3C). Alternative possibilities are that nuclear pYAP(S127) has undescribed function(s), or that Hippo pathway feedback is simply not a dominant influence on YAP activity in TNBC. Chen and colleagues mutated the equivalent phosphorylation site [YAP(S112)] in the mouse, and found that despite major defects in cytoplasmic translocation, there were no visible developmental consequences because of a compensatory decrease in YAP protein levels.
61
The authors cautioned against extrapolating results from early mechanistic experiments (largely,
Concerning HER4, we found that phosphorylated, membrane-associated protein is infrequent in primary breast cancers, though enriched in HER2+ cases, with Ki67 data suggesting it promotes proliferation in this context. Conversely, membrane-associated pHER4 was abundant in brain metastases. The fact that pHER4 was induced concomitantly with pHER3 indicates this is likely an adaptation to the neuregulin-rich brain microenvironment, which may lead to increased phosphorylation of YAP (Figure 5). To our knowledge, this is the first study to report these changes in a matched series of breast tumours and brain metastases. Nuclear HER4-ICD was not associated with nuclear YAP levels in any of the cohorts we analysed, but disease-specific survival of HER4-ICD subgroups was further stratified by nuclear pYAP(S127) status (Figure 4D), and the overall frequency of nuclear co-expression increased to ~50% in brain metastases (Figure 5). In principle, these findings provide support for pan-HER inhibitor clinical trials in metastatic breast cancer, with inclusion of intracranial response assessment criteria that measure clinically meaningful endpoints for patients with brain involvement.25,77 In addition, building site-specific relapse assessment into early breast cancer trial protocols would enable assessment of any reduction in the risk of brain relapse. Combining pan-HER inhibitors with agents known to oppose YAP activity (e.g. Verteporfin, Pazopanib) would also be worthwhile exploring in a preclinical setting.
In summary, this study finds that in ER+ breast cancer, pYAP(S127) is a favourable prognostic indicator. Nuclear translocation of un-phosphorylated YAP is common in brain-metastatic primary breast cancers and brain metastases with ER+ phenotypes. Conversely in ER-negative breast cancer, nuclear-localised pYAP(S127) is associated with markers of aggressive clinical behaviour and relatively short survival. Nuclear pYAP(S127) levels increase further when disease relapses in the brain. This co-occurs with HER4 activation in as many as 70% of ER-negative brain metastases, suggesting that combination targeting strategies could be effective.
Supplemental Material
Kalita-de_Croft-Suppl_tables – Supplemental material for Clinicopathologic significance of nuclear HER4 and phospho-YAP(S127) in human breast cancers and matching brain metastases
Supplemental material, Kalita-de_Croft-Suppl_tables for Clinicopathologic significance of nuclear HER4 and phospho-YAP(S127) in human breast cancers and matching brain metastases by Priyakshi Kalita-de Croft, Malcolm Lim, Haarika Chittoory, Xavier M. de Luca, Jamie R. Kutasovic, Bryan W. Day, Fares Al-Ejeh, Peter T. Simpson, Amy E. McCart Reed, Sunil R. Lakhani and Jodi M. Saunus in Therapeutic Advances in Medical Oncology
Footnotes
Acknowledgements
The authors thank Dominique Ezra for helpful discussions and critical manuscript review. We are grateful for support from Metro North Hospital and Health Services, the Brisbane Breast Bank, Colleen Niland and Kaltin Ferguson for sample and data collection, and to patients past and present who donate tissue and clinical information for research.
Author contributions
Conceptualization: PK-dC, JMS; methodology/experiments: PK-dC, ML, HC, XdL, JRK, BWD, FA, PTS, AEMR; drafting, review and editing: all authors; supervision and funding: SRL, JMS; project administration: JMS.
Conflict of interest statement
The authors declare that there is no conflict of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by grants from the Australian National Health and Medical Research Council (APP1017028) and Cancer Council Queensland (APP1106310).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
