Abstract
Introduction:
Molecular profiling of tumor tissue is the gold standard for treatment decision-making in advanced non-small cell lung cancer (NSCLC). Results may be delayed or unavailable due to insufficient tissue, prolonged wait times for biopsy, pathology assessment and testing. We piloted the use of plasma testing in the initial diagnostic workup for patients with suspected advanced lung cancer.
Methods:
Patients with ⩽15 pack-year smoking history and suspected advanced lung cancer referred to the lung cancer rapid diagnostic program underwent plasma circulating-tumor DNA testing using a DNA-based mutation panel. Tissue testing was performed per standard of care, including comprehensive next-generation sequencing (NGS). The primary endpoint was time from diagnostic program referral to cancer treatment in stage IV NSCLC patients (Cohort A) compared to a contemporary cohort not enrolled in the study (Cohort B) and an historical pre-COVID cohort referred to the program between 2018 and 2019 (Cohort C).
Results:
From January to June 2021, 20 patients were enrolled in Cohort A; median age was 70.5 years (range 33–87), 70% were female, 55% Caucasian, 85% never smokers, and 75% were diagnosed with NSCLC. Seven had actionable alterations detected in plasma or tissue (4/7 concordant). Fusions, not tested in plasma, were identified by immunohistochemistry for three patients. Mean result turnaround time was 17.8 days for plasma NGS and 23.6 days for tissue (
Conclusion:
Liquid biopsy in the initial diagnostic workup of patients with suspected advanced NSCLC can lead to faster molecular results and shorten time to treatment even with smaller DNA panels. An expansion study using comprehensive NGS plasma testing with faster turnaround time is ongoing (NCT04862924).
Background
In the current era of precision medicine, clinical management of newly diagnosed non-small cell lung cancer (NSCLC) patients requires knowledge of molecular alterations to guide treatment decisions.1,2 Targeted therapies have changed the management of molecularly-driven NSCLC and new treatments targeting actionable alterations are being continuously developed. Molecular testing of tumor tissue is the gold standard for diagnosis and genotyping. However, 15–40% of lung cancer patients do not have enough tissue for successful molecular testing.3–6 As such a significant proportion of lung cancer patients do not have test results available at the time of oncology consultation.7–9
The wait time for starting treatment is a period of uncertainty for patients that causes fear and anxiety. 10 Prolonged wait times may have a detrimental impact on patient outcomes, with fewer patients able to access effective therapy contributing to reduced survival and quality of life.11–13 Delayed tumor biopsy and molecular profiling results are associated with significant patient attrition, with up to 17% of lung cancer patients dying or becoming unfit for therapy before results are available. 13 In a previous report, nearly 20% of patients eligible for targeted therapy started chemotherapy instead because molecular results were not available at the time of treatment decision-making. 11
Multidisciplinary centralized referral programs can help shorten wait times for diagnosis and treatment of lung cancer. 14 However, many programs in managed care or publicly funded systems are impacted by limited resources for biopsy, pathology, and molecular testing. Wait times for patients to access diagnostic tests, including imaging and biopsies, have also increased significantly during the COVID-19 pandemic. 15
Liquid biopsies are minimally invasive tests that can detect circulating-tumor DNA (ctDNA) and identify targetable alterations in a proportion of lung cancer patients, with a sensitivity of 80% in plasma.16,17 Although the gold standard for molecular testing is tumor tissue genotyping, plasma ctDNA analysis has demonstrated clinical utility as an alternative or complementary tool in NSCLC, especially in clinical scenarios where tissue or time for genotyping is limited. 17 Liquid biopsies using plasma are more convenient and safer for patients than repeat tissue biopsies and their use for molecular profiling may lead to cost savings in some patient populations.16,18–21 Although considered a valid tool for genotyping in newly diagnosed patients with advanced pathologically proven NSCLC, 17 the clinical utility of liquid biopsies as an initial approach for biomarker evaluation prior to cancer diagnosis remains to be demonstrated in prospective studies. 22 Preliminary studies suggest that turnaround times (TATs) and time to treatment (TTT) may be shortened with a complementary approach.23,24 However, the role of liquid biopsy in the pre-diagnostic phase, a plasma-first approach, has not yet been established.
The ACCELERATE study (NCT04863924) aims to prospectively assess the utility of liquid biopsy to accelerate TTT for patients with radiographic evidence of advanced lung cancer. Herein we report the results from a pilot cohort using liquid biopsy in the pre-diagnostic phase in patients with suspected advanced lung cancer.
Methods
The pilot study was a prospective, single arm, nontherapeutic, minimally invasive study conducted at the University Health Network (UHN), Toronto, Canada. The study was registered on ClinicalTrials.gov on 14 January 2021 (ACCELERATE, NCT04863924). Conduct of this study was reviewed and approved by the institutional research ethics board (UHN Board C). All participants provided written informed consent before study enrolment. Between 14 January and 30 June 2021, eligible patients referred to the Lung Rapid Access Management Program (Lung RAMP) with suspected advanced lung cancer (stage IVA or B) based on imaging who were nonsmokers or with ⩽15 pack-year smoking history, were enrolled. Patients were enrolled after eligibility confirmation by the Lung RAMP Multidisciplinary Case Conference (MCC), including representation from Thoracic Surgery, Interventional Respirology, Radiology, Radiation, and Medical Oncology. Imaging tests to document stage IV disease included computed tomography (CT) of chest and abdomen and also positron emission tomography and total body bone scan as needed. Patients deemed eligible were approached to participate in the study, including liquid biopsy as part of their ongoing diagnostic workup. Other eligibility criteria included: (a) measurable disease with >1 cm of disease on CT in patients with solid lesions; (b) diagnostic tissue biopsy ordered or planned. Patients with pleural effusions and no measurable disease were eligible if the MCC favored a malignant diagnosis based on imaging. Patients were excluded if they had concurrent cancer, a cancer diagnosis other than lung cancer in the past 2 years, or if they were pregnant due to concerns of potentially confounding ctDNA results.
Patients underwent plasma ctDNA testing using a DNA-based mutation panel of single nucleotide variants (SNVs) and indels in 38 cancer-associated genes (Follow It®, Imagia Canexia Health, Vancouver, BC, Canada) through the Project Access to Cancer Testing and Treatment (supported by the Canadian Technology Digital Supercluster). The Follow It® assay uses a multiplexed polymerase chain reaction (PCR) DNA-based panel targeting hotspots in clinically actionable genes in lung cancer, allowing for the detection of single-base substitutions (SNVs), small deletions, and insertions (up to 24 bp). It evaluates the mutation status of tumor DNA at 337 hotspots and 26 exons in 38 known cancer-associated genes simultaneously, including clinically relevant variants in
Time from referral to treatment initiation was analyzed and compared between patients in the pilot cohort that underwent liquid biopsy (cohort A,
The primary endpoint was TTT, defined as the time from diagnostic program referral to treatment initiation for cohort A using plasma ctDNA, compared to cohorts B and C. Secondary endpoints included the identification of actionable targets, time to sample collection, and result TAT of plasma-based
Statistical methods
Patient characteristics were summarized using descriptive statistics. Two-sample
Results
From 14 January to 31 June 2021, 20 eligible patients were enrolled in cohort A (Supplemental Figure 1). The median age was 70.5 years (range 33–87). 70% were female, 55% Caucasian, 85% never smokers (see Table 1). All patients underwent testing of diagnostic tumor tissue as per standard of care. Twelve (60%) had lung adenocarcinoma upon final pathology reporting, five (25%) had lung non-adenocarcinoma histology (one each of large cell neuroendocrine, large cell, sarcomatoid, lymphoepithelioma-like carcinoma, and atypical carcinoid). Three (15%) had non-lung cancer diagnoses (one carcinoma of unknown primary, one gastric adenocarcinoma, one diffuse B-cell lymphoma).
Patient characteristics.

One patient was diagnosed with lung metastases from gastric adenocarcinoma, one patient with diffuse large-B lymphoma (JAK2, IDH1 gene alterations).
One patient underwent ctDNA plasma testing and CT-guided liver biopsy both negative for malignancy.
CT, computed tomography; ctDNA, circulating-tumor DNA; LCNEC, large cell neuroendocrine carcinoma; NSCLC, non-small cell lung cancer: NOS, not otherwise specified.
Of the 12 patients with tissue biopsy-proven adenocarcinoma of the lung, 10 (83%) had actionable oncogenic driver alterations detected in plasma or tissue (Figure 1). Using the plasma DNA mutation hotspot panel, five patients had actionable alterations detected in
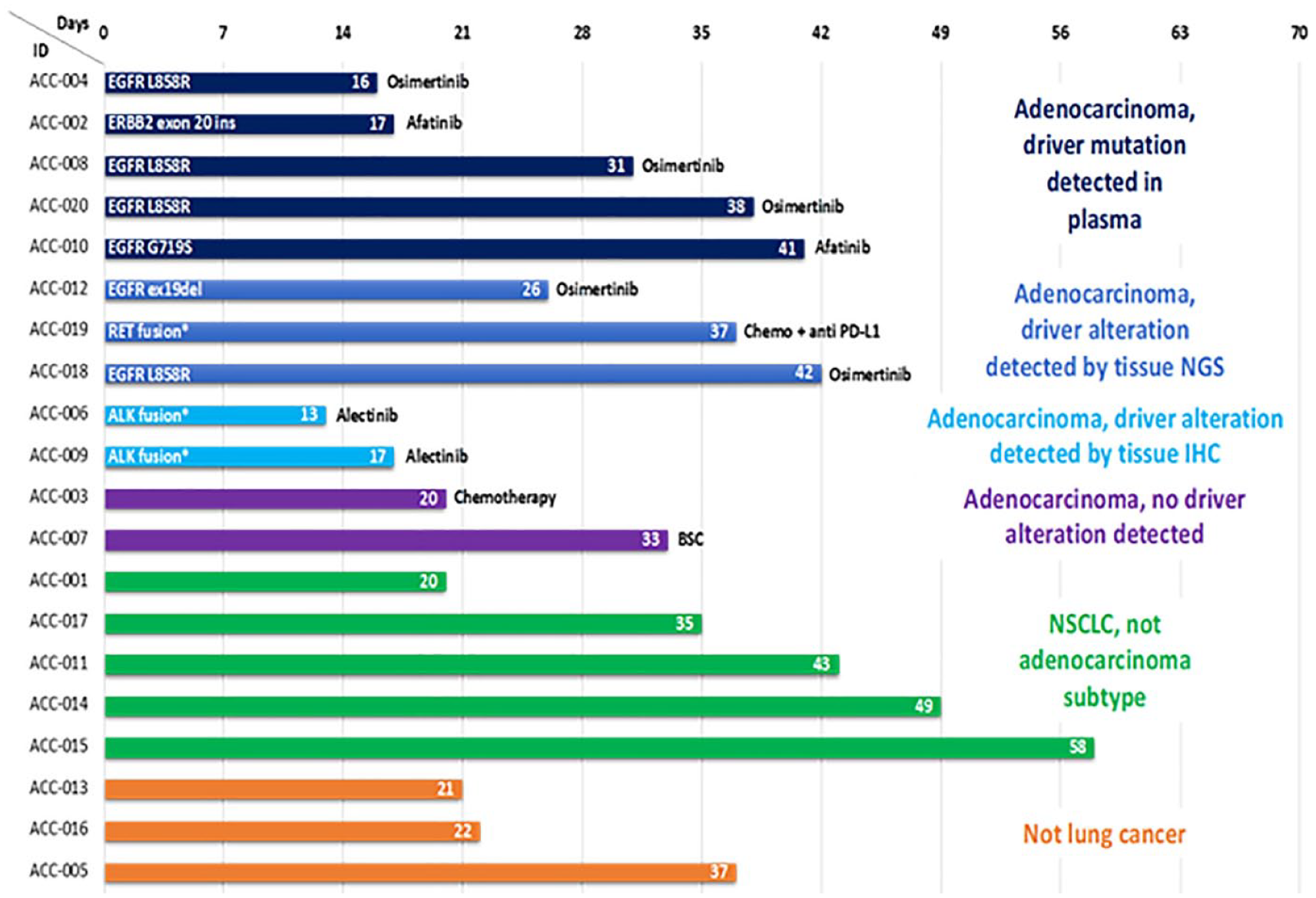
Cohort A swimmer plot of time from referral to the program to treatment initiation.
Overall concordance was 71% between plasma and tissue for gene alterations that were assayed using both methods, and sensitivity ranged between 60 and 100% (Supplemental Table 2).
Comparison cohort characteristics
Cohort B included all patients referred to Lung RAMP that were not enrolled in the study (
Molecular testing method and results.

CT, computed tomography; EBUS, endobronchial ultrasound; IHC, immunohistochemistry; TBNA, transbronchial needle aspiration.
Cohort C included patients with advanced NSCLC harboring an actionable alteration that were light or never smokers referred to Lung RAMP during 2018, prior to the COVID-19 pandemic (
Turnaround times
TATs are displayed in Figure 2(a). Mean time from program referral to plasma testing was 7.1 days (SD 6.9) in cohort A. Tissue biopsy for study participants was performed a mean of 11.5 days (SD 9.8) after referral. The mean TAT for plasma molecular results was 17.8 days (SD 5.6) and 23.6 days (SD 9.0) for tissue results using a larger comprehensive NGS panel (OCAv3,

Mean wait times (days) in cohort A NSCLC (
In the contemporaneous cohort B, the mean time from referral to biopsy was 18.6 days (SD 10.5) and the mean TAT for tissue NGS results was 24.4 days (SD 7.2). In the pre-COVID-19 cohort C, the mean time from referral to biopsy was 25 days (SD 16.7) and the mean TAT for tissue results using a 15-gene NGS panel was 17.1 days (SD 5.5).
Time to systemic treatment initiation
Time to treatment initiation is showed in Figure 2(b), with individual treatment data detailed in Figure 1. In the cohort A, using liquid biopsy, mean time to systemic treatment initiation (TTT), was 32.6 days (
In the cohort A, patients with alterations identified in plasma (
In the contemporaneous cohort B, mean time from referral to targeted therapy treatment in the subgroup of patients with actionable alterations detected by tissue NGS was 48.7 days (
In the pre-COVID cohort C, three patients had an
Discussion
In this pilot study of never or light smokers (⩽15 pack-years) with suspected advanced lung cancer, we explored the feasibility of liquid biopsy for molecular genotyping as part of the pre-diagnostic workup for lung cancer. In our study, this approach was easily integrated and accepted by patients and providers. Time to treatment initiation in patients with lung cancer was significantly improved with a ‘plasma first’ approach compared to both contemporary and historical cohorts that did not undergo plasma testing as part of their diagnostic workup (mean 32.6 days
Our pilot study is limited by small numbers. Also, despite the selection of never or light smokers with radiological evidence of advanced lung cancer confirmed by a multidisciplinary committee, only 12 (60%) had lung adenocarcinoma, while 3 had a non-lung cancer diagnosis. These results highlight the need for tissue biopsy for lung cancer diagnosis and pathologic subtyping: ‘plasma first’ does not mean ‘plasma only’. For this pilot study, patients underwent plasma ctDNA testing using a DNA-based mutation panel which detected SNVs, and indels in 38 cancer-associated genes, but not fusions, with a mean TAT for molecular results of 17.8 days. The concordance between plasma and tissue for identification of actionable mutations (excluding fusions) was 71%. Plasma NGS before tissue NGS can increase detection of therapeutically targetable mutations, especially when tissue DNA is insufficient or unavailable. However, despite ctDNA sensitivity continues to improve, it is not perfect. Some tumors may not shed ctDNA, likely explaining why two patients with NSCLC harboring an
In our study, we also found that time from referral to targeted therapy initiation was shorter in cohort B
Aggarwal
Our study showed similar results in a more selected population of never or light smokers, and also highlighted the impact of multidisciplinary diagnostic assessment programs. However, our pilot study also highlights potential limitations of a plasma-first approach, including the need for tissue to confirm histological diagnosis and the importance of using large panel comprehensive NGS assays with rapid TAT to maximize benefit.
In summary, a ‘plasma-first’ approach appears to accelerate molecular genotyping and TTT in selected patients with advanced NSCLC. However, the impact on clinically meaningful outcomes for patients such as quality of life, survival, and cost still needs to be demonstrated in prospective trials. An expansion study (NCT04863924) is ongoing, with a target accrual of 150 patients regardless of smoking history. A comprehensive DNA-based assay that also detects fusions with a 7-day TAT will be used (SNV, indels, fusions, CNV; InVisionFirst™, Inivata). Clinical outcomes including quality of life and cost effectiveness of a plasma-first approach will be collected.
Conclusion
A plasma-first approach in the diagnostic algorithm for patients with suspected advanced lung cancer may increase TTT, access to precision medicine, and potentially even improve patient outcomes.
Supplemental Material
sj-docx-1-tam-10.1177_17588359221126151 – Supplemental material for Plasma-first: accelerating lung cancer diagnosis and molecular profiling through liquid biopsy
Supplemental material, sj-docx-1-tam-10.1177_17588359221126151 for Plasma-first: accelerating lung cancer diagnosis and molecular profiling through liquid biopsy by Miguel Garcia-Pardo, Kasia Czarnecka, Jennifer H. Law, Alexandra Salvarrey, Roxanne Fernandes, Jason Fan, Lucy Corke, Thomas K. Waddell, Kazuhiro Yasufuku, Laura L. Donahoe, Andrew Pierre, Lisa W. Le, Noor Ghumman, Geoffrey Liu, Frances A. Shepherd, Penelope Bradbury, Adrian Sacher, Tracy Stockley, Prodipto Pal, Patrik Rogalla, Ming Sound Tsao and Natasha B. Leighl in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
